1. Introduction
Nature has long been recognized as a valuable repository of compounds possessing unique biological activities relevant to human health (1, 2). Among the vast array of secondary metabolites found in plants, sesquiterpene lactones (SL) have garnered significant attention due to their presence in Asteraceae species and their diverse array of active principles found in traditional phytomedicines (3–7). With a structural foundation of 15 carbons and a fused α-methylene-γ-lactone ring, SL consists of over 8000 compounds, each offering a variety of modifications and structures (4, 8, 9). These compounds exhibit a broad spectrum of potential health benefits, encompassing anti-inflammatory (10), antitumoral (3, 6), antioxidant (11), neuroprotective (12), hepatoprotective (13), immune-stimulating (4, 7), antimicrobial (14), and antiparasitic properties (8).
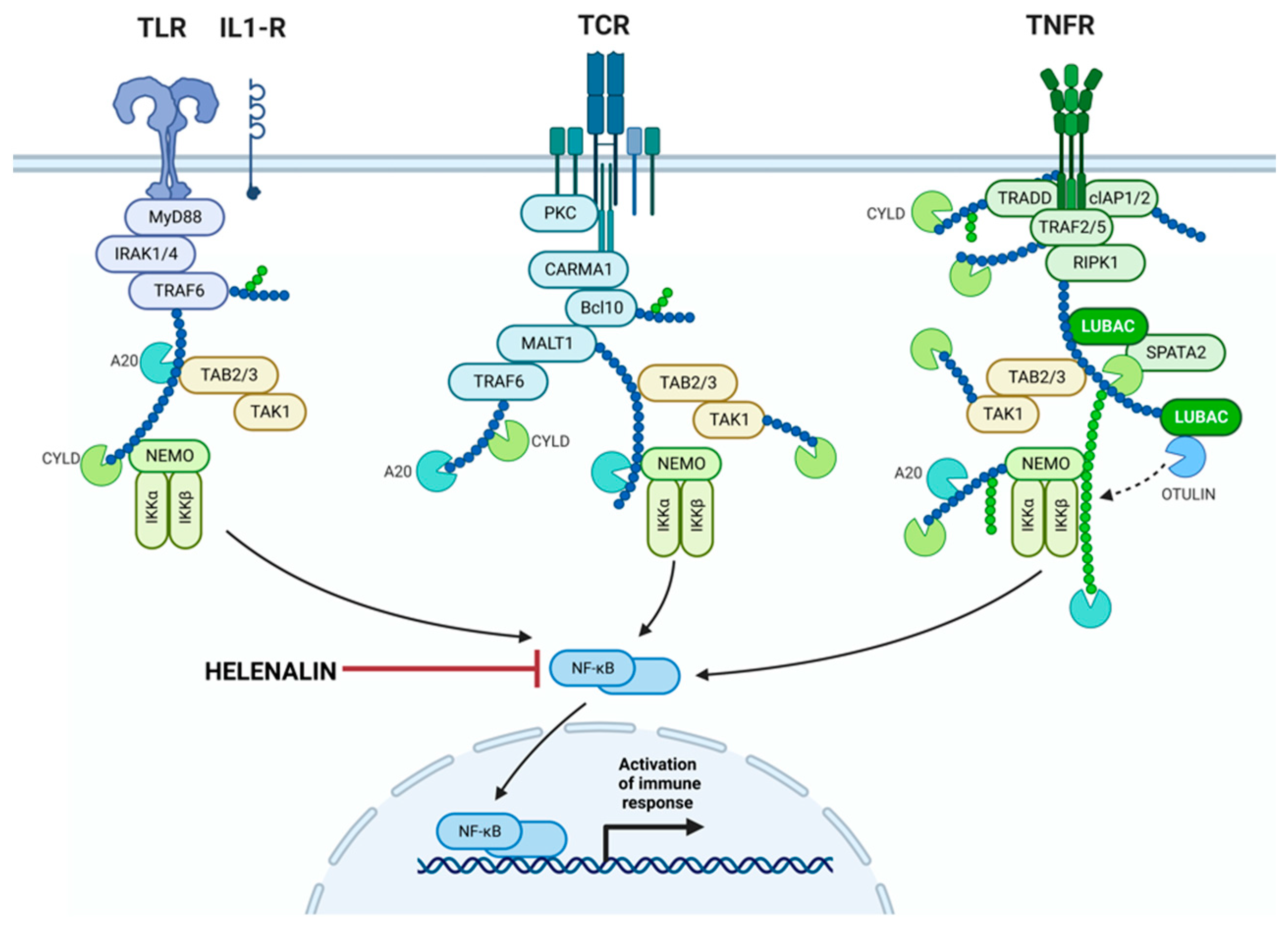
Given their capability to modulate inflammation pathways induced by oxidative stress, SL emerge as promising candidates for addressing chronic inflammatory diseases (4, 11, 15). Notably, helenalin, a pseudoguaianolide sesquiterpene lactone derived from Arnica Montana and Arnica chamissonis ssp. foliosa, has recently regained attention as a lead compound for inflammation treatment (15-19). Its anti-inflammatory mechanism diverges from that of non-steroidal anti-inflammatory drugs (NSAIDs), attributed mainly to its potent inhibition of nuclear transcriptional factor-𝛋ß (NF-𝛋ß) signaling (6, 16). Helenalin-exposed TCD4 cells have shown reduced interleukin-2 (IL-2) production and CD25 expression, suggesting helenalin's potential as an anti-inflammatory therapy (19-21). This is particularly significant in chronic inflammation diseases, where conventional treatments carry risks of adverse events and nonadherence (7, 9, 10, 22-24)
Dry eye disease (DED), characterized by ocular surface inflammation due to hyperosmolarity, is associated with excessive reactive oxygen species (ROS) production, oxidative stress, and inflammation mediated by lymphocytes. This chronic inflammation stems from various ocular factors such as uncorrected refractive errors, prolonged screen usage, systemic conditions, hormonal changes, poor blinking, environmental factors, and aging. The convergence of these factors triggers persistent tear film degradation, leading to microanatomical changes on the ocular surface. In this context, patients experience inflammation affecting both cornea and conjunctiva, marked by T-cell infiltration, increased pro-inflammatory cytokines, and matrix metalloproteinases (MMPs), including MMP-9 (25-27). Pro-inflammatory interleukins (IL-1, IL-6, IL-8) and tumor necrosis factor (TNF) α further contribute to chronic ocular surface damage (25, 27-29). NF-𝛋ß emerges as a key regulator of ocular surface inflammation, responding to diverse stimuli involved in DED (25, 28-30).
Currently, lubricant eye drops represent the standard approach for DED management (28). Although treatments like topical cyclosporine, corticosteroids, and NSAIDs exist, a definitive curative anti-inflammatory therapy for DED remains elusive (28). Despite their efficacy, these treatments are associated with potential adverse effects such as ocular pain, irritation, cataract formation, and ocular hypertension (29, 31, 32). Patient compliance with DED treatment is notably low, with some studies reporting compliance rates as low as 10.2% (27, 28, 33). Up to 60% of patients use treatment on an as-needed basis, highlighting the need for approaches that enhance treatment compliance, alleviate symptoms, and reduce adverse events (33, 34).
To explore a novel anti-inflammatory approach, this clinical study evaluates the, tolerability, safety and efficacy of a patented ophthalmic topical formulation containing Helenalin. The aim is to assess potential clinical effects in patients diagnosed with DED, aiming to enhance treatment compliance, improve patient comfort, and alleviate symptoms while minimizing adverse events (33, 34). Through this investigation, we seek to contribute to the growing body of knowledge surrounding alternative therapies for chronic ocular surface inflammation and dry eye disease.
2.1. Materials and Methods
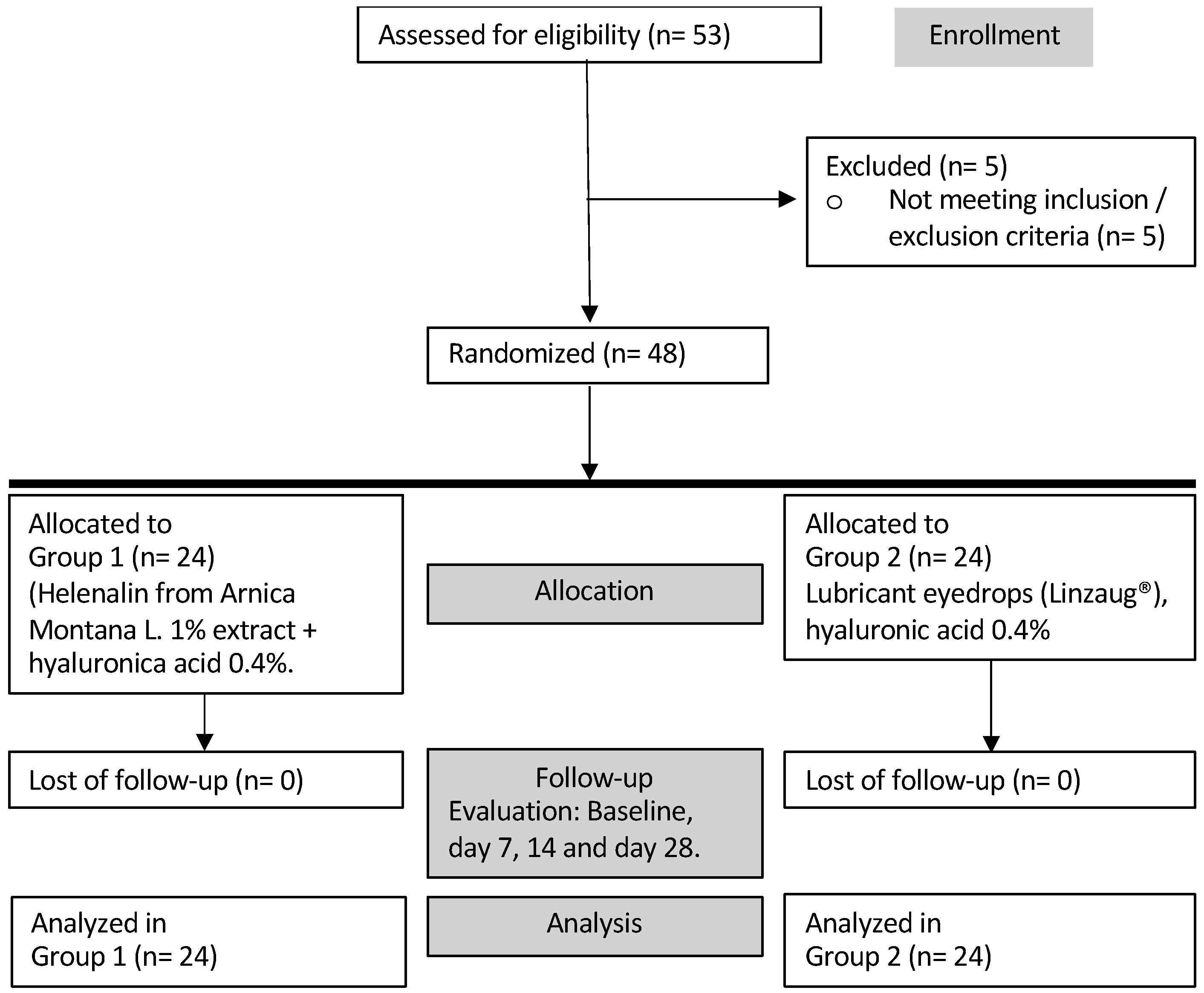
In pursuit of evaluating the tolerability, safety, and efficacy of an innovative ophthalmic topical formulation, a comprehensive single-center, prospective, interventional, double-blinded, randomized, controlled clinical trial was meticulously executed. This clinical investigation engaged the participation of both healthy subjects' eyes (pertaining to the safety and tolerability clinical study) and patients grappling with mild to moderate dry eye disease (DED) (pertaining to the efficacy clinical study), including a confirmation of the presence of Matrix Metalloproteinase 9 (MMP-9) through a positive InflammaDry© test result exceeding 40 ng/ml. The trial, which was undertaken from January to May 2023, adhered to the ethical principles encapsulated in the Declaration of Helsinki and took place in the ISO 9001:2015 certified ASG Clinical Retinal Research S.C., an ophthalmological research center situated in Zapopan, Mexico. Prior to patient enrollment, the study received approval from the internal review board and the CRMQ Ethics and Research Committee (ID: CRMQ-OFT-001-2022), in full alignment with the International Conference on Harmonization on Good Clinical Practices, accompanied by strict adherence to all pertinent local regulatory requisites and laws. Informed written consent was obtained from all participants and their accompanying witnesses, after ensuring a comprehensive understanding of the study's nature and potential adverse events.
2.2. Study Formulation
In congruence with the principles of Good Manufacturing Practice and the Mexican Pharmacopoeia 2020 (FEUM) (35), the ophthalmic formulation under scrutiny was meticulously prepared. This sterile, translucent ophthalmic suspension, ensconced within a 15 ml container, was crafted to incorporate helenalin (0.008% to 0.015%) and dehydroalanine tiglate (5% to 7%), deriving their origins from a 1% extract of
Arnica montana, which is included in the Mexican Herbal Pharmacopoeia (36). The Drug to Extract Ratio (DER) falling within the gamut of 1:5 to 1:7 was achieved. Moreover, this formulation incorporated hyaluronic acid, presenting a concentration of 0.4%. The formulation's pH was set at 7.4, complemented by a viscosity of 20-cPs. Noteworthy additions included ethylene diamine tetra-acetic acid (0.0005 g), benzalkonium chloride as a preservative (0.0002 g), hydroxypropylmethylcellulose (0.01 g), monobasic sodium phosphate (0.0003 g), dibasic sodium phosphate (0.0012 g), sodium chloride (0.009 g), sodium hydroxide (0.0001 g), and grade 2 purified water (0.9647 g). This formulation adhered to Mexican regulations. The full list of compounds can be appreciated in
Table 1.
2.3. Evaluation of Tolerability and Safety in Healthy Volunteers (Phase I clinical study)
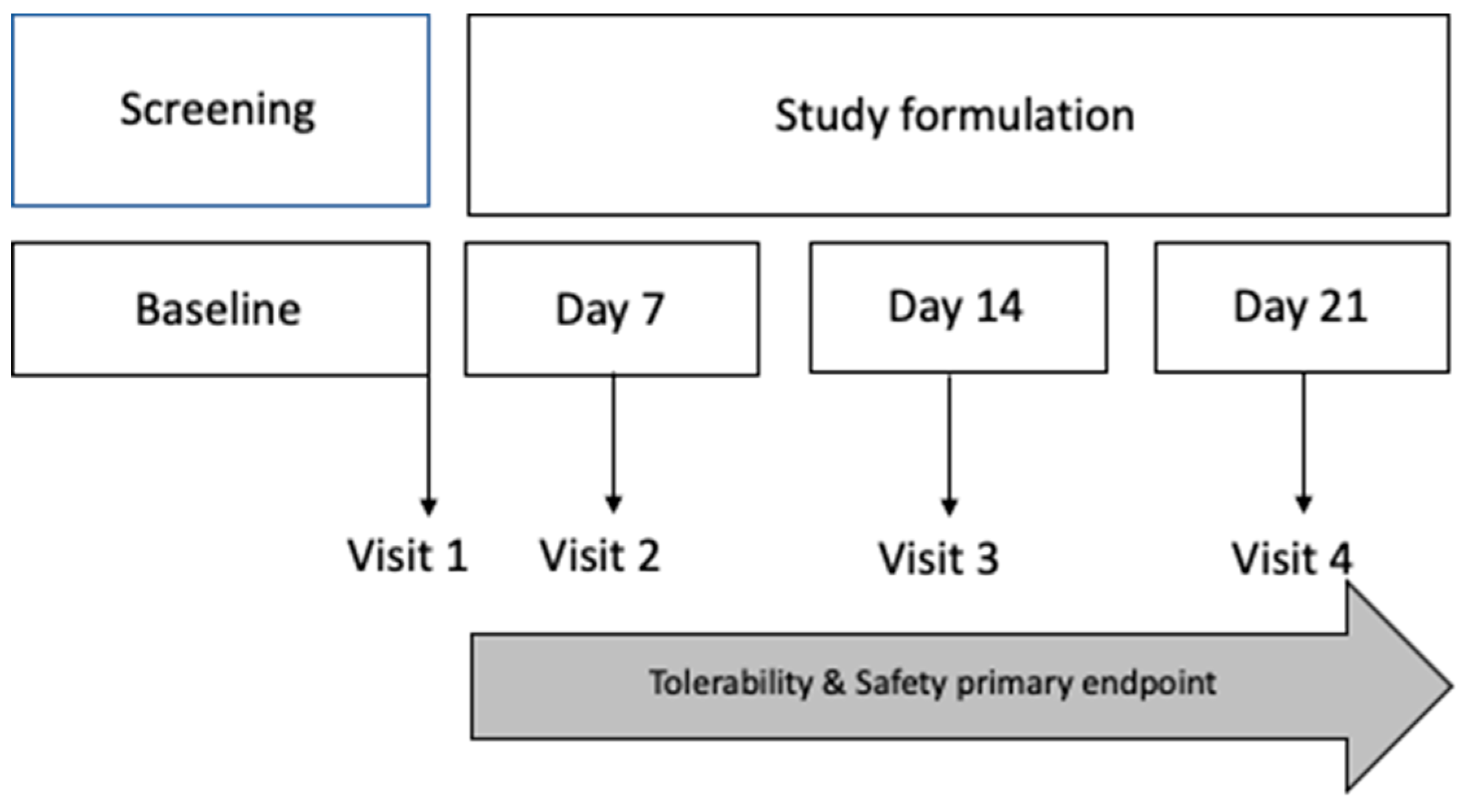
A phase I clinical study was designed to assess the safety and tolerability of this ophthalmic formulation. Healthy individuals ranging from 18 to 65 years of age, were selected to participate in this phase I study. Good health was defined as the absence of any medical or surgical history (except for intraocular lens surgery performed more than 12 months prior). A stipulated baseline best-corrected visual acuity (BCVA) of no less than 0.1 in both eyes, an Ocular Surface Disease Index (OSDI) <13 (37, 38), a non-invasive film tear breakup time (NIF-BUT) >10 seconds, absent corneal and conjunctival staining, and a negative result on the InflammaDry® MMP-9 test were part of the inclusion criteria in this phase. Additionally, candidates were scrutinized for any ocular pathologic conditions, which, if present, led to exclusion of the study. It was mandatory for participants to refrain from using any form of eyedrops, particularly ocular lubricants, in both eyes for a minimum duration of six months prior to their enrollment in the study. Key exclusion criteria included systemic diseases associated with DED, history of recurrent ocular inflammation, ocular-lid trauma, active ocular-lid infection, use of systemic trigger-dry eye drugs, and corneal abnormalities that could interfere with the study evaluation tests (such as ocular staining, allergy to fluorescein sodium or lissamine green dyes). Participants´ demographic and baseline clinical data were systematically compiled five days before the onset of the study formulation application. After enrollment, participants were instructed to routinely apply one drop of the formulation three times a day in the study eye, for a period of two weeks. Follow-up was conducted the week after the application period, gauging any potential adverse events in alignment with tolerability and safety guidelines endorsed by FEUM (35, 36) and Comisión para la Protección contra Riesgos Sanitarios-COFEPRIS (
Figure 1). Compliance was also monitored; any value below 90% was tantamount to non-compliance and, thereby, excluded from statistical analysis.
Follow-up ocular evaluations included: the measurement of BCVA adhering to the ETDRS protocol, the determination of intraocular pressure (IOP) employing a tonometer (iCare® TA01i Tonometer), and the quantification of corneal endothelial cell density (cECD) through the utilization of specular microscopy (Perseus endothelial microscope from Costruzione Strumenti Oftalmici, Firenze, Italy). Additionally, a slit lamp examination with ocular surface staining leveraging fluorescein and lissamine green reagents was performed (AK-Fluor®, Akorn, Lake Forest, IL, USA and Rose Stone Enterprises, Alta Loma, CA, USA, respectively). Ocular AEs were reported concurring with NOM-220-SSA1-2016, which contains the Mexican regulatory guidelines for the instillation and handling of commercial and research drugs, herbal medicines, medical devices, and their potential adverse events (including ophthalmic products) (39). To reduce interobserver discrepancies, a single certified technician assessed safety and efficacy.
2.4. Evaluation of Efficacy in Patients with Mild to Moderate DED (Phase II clinical study)
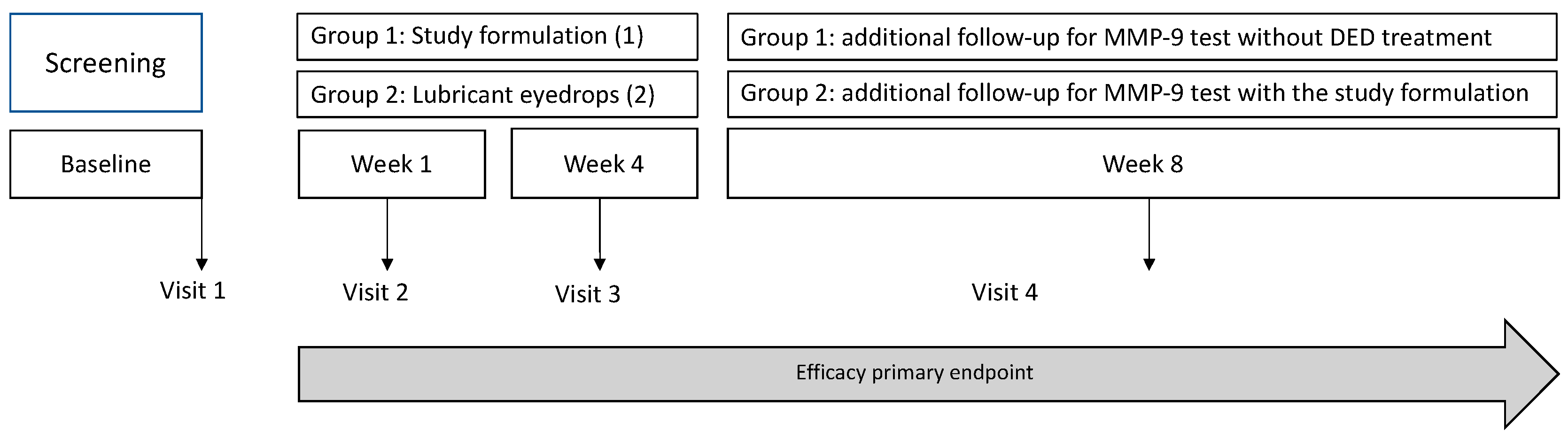
We designed a phase II clinical study to evaluate the efficacy of the ophthalmic formulation. This was a prospective, randomized, double-blinded, crossover, interventional study comparing the study formulation with lubricant eye drops (HA 0.04%). The inclusion criteria for the study were mild to moderate DED, based on OSDI score (37, 38), a non-invasive film tear breakup time (NIF-BUT) <10 seconds, corneal and conjunctival staining, the presence of visual strain, and a daily screen interaction of less than 8 hours. Patients required a positive test result in the InflammaDry© test, as confirmation of elevated levels of MMP-9 (positive test result in levels >40ng/ml). After meeting these criteria, participants were randomly assigned into one of two efficacy study groups, and they were subsequently assigned to either receive the trial ophthalmic formulation (group 1) or a commercially available lubricant eye drop containing hyaluronic acid (group 2) (Linzaug® Laboratorios Opko, México) as control. Both eye drops were administered according to pre-specified protocols and frequencies (1 drop three times a day (TID) in both eyes).
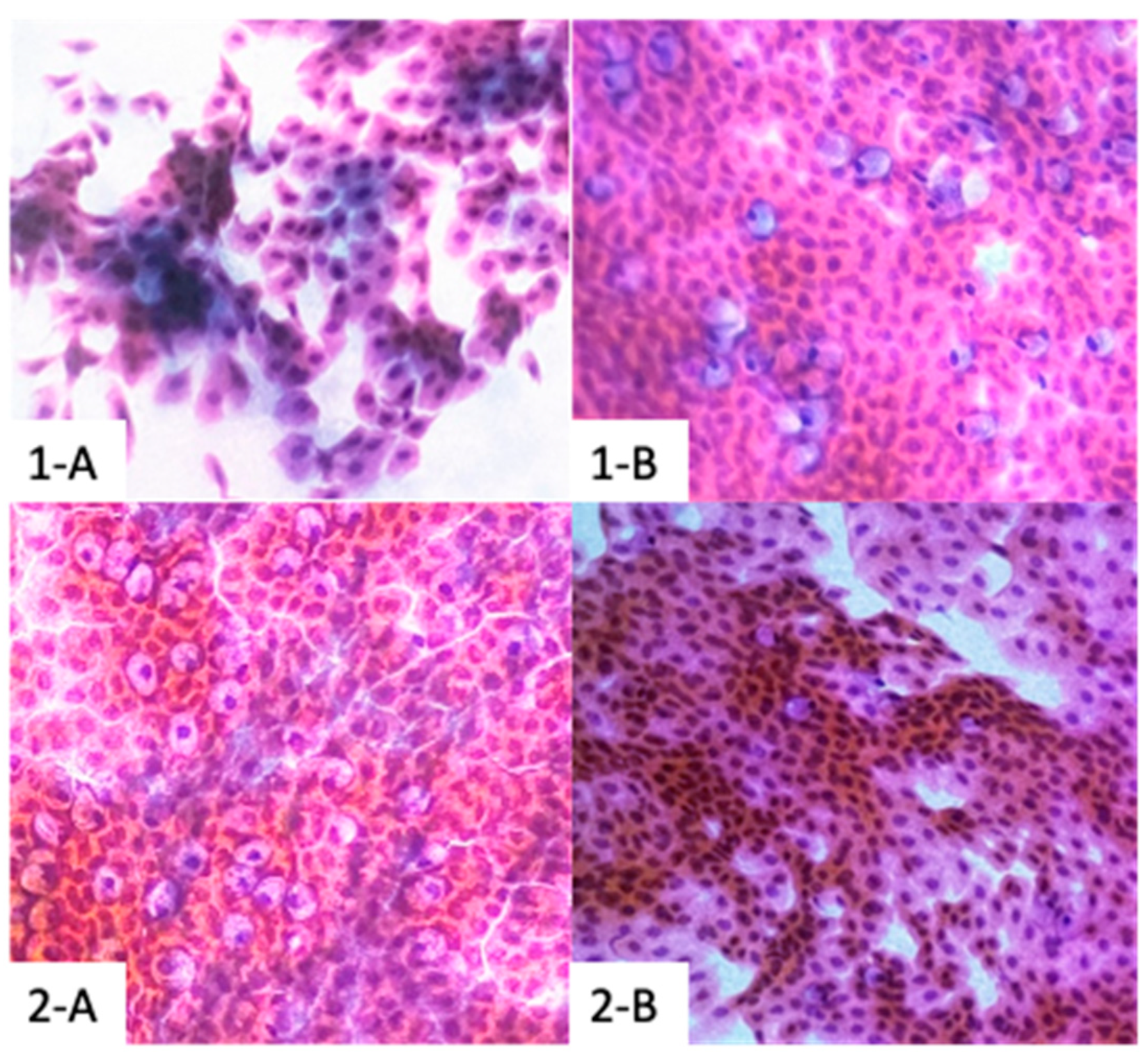
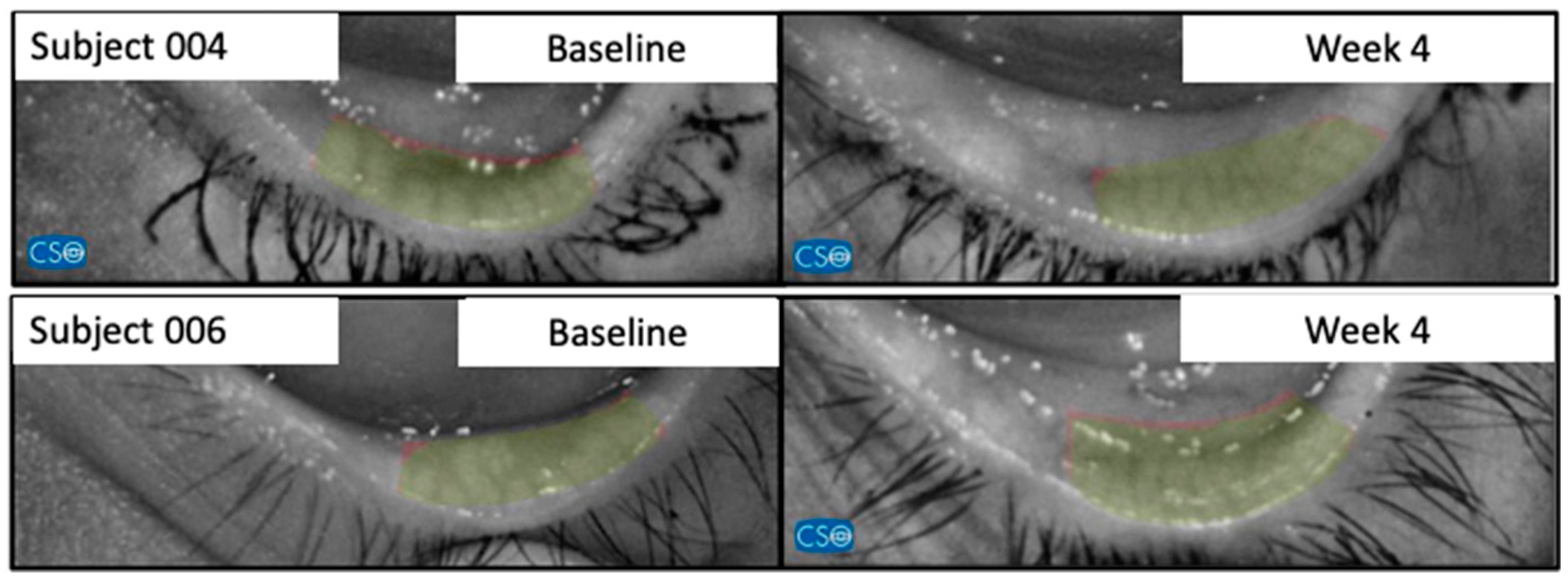
To evaluate the formulation's efficacy, an evaluation strategy was devised, incorporating an array of ophthalmic parameters, tested at day 7, day 14, and day 28. These test included the assessment of best-corrected visual acuity (BCVA), the probing of non-invasive film tear breakup time (NIF-BUT) (using a Schwind Sirius+ topographer) (CSO SRL, Italy), and non-invasive average breakup time (NIAvg-BUT), the evaluation of meibomian gland changes (using the Schwind Sirius® Meibomiography) and the detection of MMP-9 using the InflammaDry® MMP-9 Test. Additional evaluations included conjunctival impression cytology (CIC), Schirmer's test 1, ocular surface staining, and the ocular irritability test (for techniques and technical details, consult Appendix A). To reduce interobserver discrepancies, a single certified technician performed the BCVA measurement. Safety and efficacy assessments were performed by a single, blinded, clinical investigator at each visit.
For the crossover, an additional comprehensive 4-week follow-up period was implemented for MMP-9 testing exclusively, encompassing both study groups. Participants in the treatment group (group 1) were directed to discontinue the application of the study formulation, while those in the control group (group 2) initiated the administration of the study formulation according to the established protocol, employing a crossover design without a washout period.
2.5. Data Analysis and Statistical Methods
The sample size was calculated using a formula for the difference between two independent means where nc is the sample size for the reference group (group 2) and ne is the one for the study group (group 1). D = (Mc-Me) where Mc is the mean of the first group and Me is the mean of the second one, S2 is the variance of both distributions, which are assumed to be equal, Zß is the value of the abscissa axis of the standard normal function where the probability (1 -ß) accumulates; assuming a mean difference in the OSDI value between the treatment group (group 1) and the reference group (group 2) of 22.4 OSDI units and a standard deviation of 15 units (l); the study would require a sample size of 19 subjects for each group, in order to achieve a power of 80% and a significance level of 5%, considering a 10-unit margin of superiority. Considering a loss of 20%, it is concluded that the final sample per group will be a minimum of 23 subjects: nc = ne = 2*S2 / D2 * (Z α/2 *Zß)2
Data was analyzed using the SPSS 22.0 software (IBM SPSS Statistics for Macintosh, version 22.0, IBM Corp, Armonk, NY, USA). Quantitative variables were described using means and standard deviation. Qualitative variables were described using frequencies and percentages. Intra-group analysis: they will be determined through the Wilcoxon rank test, for the quantitative variables. Analysis between groups: the differences between groups will be analyzed utilizing the Student’s T-test or the Mann–Whitney U statistic if applicable. Significance was defined as a p-value < 0.05.