Introduction
Drug abuse is the excessive use of psychoactive, prescription or over-the-counter drugs, for purpose other than those for which the drug is meant for. The abuse of drugs causes a high-risk individual to engage in violent and aggressive behaviour by altering brain activities. There is a complex relationship between drug abuse and violence, with intoxication, neurotoxic and withdrawal symptoms often being mixed up and misinterpreted (Bouchatta et al. 2016). Nigeria being the most populous country in Africa, has evolved to be a centre for usage and trafficking of drugs commonly among the population of youth (Jatau et al. 2021). Due to the regulation of drug of abuse by the United Nations Office on Drugs and Crimes (UNODC), Nation Drug Law Enforcement Agency (NDLEA) and other Drug law enforcement agencies, a lot of youth and teenagers have resulted to the use of unconventional substances to get high. There is an estimated report that 18 in 20 people aged 12 years or older depend on psychoactive substance (Danjuma et al. 2015).
These substances can be classified based on their algological composition or based on their effects on the substance users (Ugbedeojo et al. 2021). Psychoactive substances have been identified to be abused in Nigeria and include volatile solvents, plant based psychoactive substances, lizard dung/excretes, cocoa paste mixed with tobacco, glue, soak away/pit toilet fumes (bio-generic gas) and other psychoactive faunas. Inhalation of volatile substances for their euphoric effects is known as inhalant abuse. Glues and other adhesives have been abused for years, other solvents like toluene and petroleum are also sniffed, hence the term “solvent abuse” is given to this dangerous habit, huffing, bagging, dusting, and sniffing are the different ways of inhalation abuse (Jayanth et al. 2017).
Glue is a sticky non-metallic substance used to join two separate objects together by application on both surfaces and allowed to dry, often used for the repair of broken things. The inhalation of this substance is said to be a bedrock of the abuse of more addictive drugs. Glue inhalation has been reported to be a common problem among street children of countries in continents like Asia, America, and Africa. (Egbuonu et al. 2016; Kazeem and Chutel 2018; Munawar et al. 2019; Ramlagan et al. 2010). Abusers who seek its euphoric effect are typically teenagers and younger adults (Vitale and Gutovitz 2018) who inhale glue to get “high” also young adults of low-income population seeking for alternative means of getting high from unconventional substances (Danjuma et al. 2015).
Glue inhalation seems to be the most commonly unconventional substance abuse due to easy accessibility, usage in day to day activities and its potential danger of abuse through inhalation by both children and adults glue inhalation has become one of the rising major public health and socioeconomic problem worldwide (Danjuma et al. 2015; UNODC 2021) a rising health concern in Nigeria with Northern Nigeria taking the lead (Danjuma et al. 2015 ; Abasiubong et al. 2014). The main active constituent in glue is toluene (Munawar et al. 2019). Toluene exposure may result in chronic respiratory system, cardiac malfunction, euphoria, hallucinations, depression, cognitive dysfunction, ataxia, sedation, mental confusion, unconsciousness, insomnia, headaches, renal malfunction, liver and peripheral nerves damage, hypoxia, and hearing loss (Cruz and Bowen 2021; Kuranchie et al. 2019; Lee and Valentino 2021). Toluene is suspected to be an anxiolytic agent with CNS depressant activity (López-Rubalcava et al. 2000; Páez-Martı́inez et al. 2003). Sniffing of glue can also lead to abdominal pain in children (Munawar et al. 2019; Ugbedeojo et al. 2021). Jayanth et al. (2017) reported a case of a 22-year-old male who died on the way to hospital after sniffing several tubes of “Fevibond” glue.
Despite the high prevalence of glue inhalation or sniffing, and serious consequences recorded in so many studies, not enough is known about the behavioural effects of abusing glue (Bouchatta et al. 2016; Cruz and Bowen 2021). The primary solvents contained in rubber solution have been broadly studied individually but not as a mixture (glue) and its withdrawal effects is not well known, hence the necessity for further studies with animal models properly designed to explore the effect of the solvents and to evaluate the CNS activities and toxicity profile of glue inhalation. We, thus, evaluated the anxiolytic and depressant effects of glue inhalation, including the withdrawal effects of glue inhalation after two weeks of abstinence from glue in Wistar rats.
Methods
Experimental animals: Seventy-two (72) male Wistar rats weighing between 145-160g were obtained from the animal house, Faculty of Pharmaceutical sciences, Amadu Bello University, Zaria. These animals were maintained in the animal care facilities of Usmanu Danfodiyo University Sokoto state, Nigeria under standard conditions.
Glue (Adhesive): The inhalant used in this study is the ‘Diamond rubber solution’ by Anhui Morshine International Cp., Ltd., Hefei, Anhui China available in tubes of 15-20mls. It is used for patching up tires and based on verbal communications, it is the commonly abused type of glue (adhesive) in Sokoto state, Nigeria.
Inhalation protocol: Each animal was placed in semi-hermetic plastic boxes (26× 17 × 15cm) with corncob bedding and wire tops, a hole in the cover for aeration, and the airflow around the exposure chamber was consistent throughout the entire experiment. The glue was put into a small glass dish covered by a grid (to prevent direct contact between the animals and the glue) and set at one side of the inhalation chamber, and an empty dish was placed for the control groups on the other end of the inhalation chamber. Immediately after the exposure sessions (4hours for acute toxicity study, and 30 minutes for sub chronic study), the rat was removed, and returned to their habitual cages (Bouchatta et al. 2016).
Toxicity Study
Acute toxicity study: The LD50 of the glue inhalation was determined using the modified Lorke’s method (Lorke 1983) and the OECD guideline for inhalation toxicity (Arts et al. 2008). This test was carried out in two phases.
Phase 1: Three groups of three rats each (n=3) were exposed to (0.5 mL, 1 mL, and 2 mL of glue) for 4hours respectively, after which the rats were immediately removed, returned to their cages, and observed for 24 hours for signs of acute toxicities and possible mortality.
Phase 2: Three (3) groups of rats (n=1 for each) was exposed to (5 mL, 10 mL, and 20 mL of glue), for 4hours respectively. Vital organs of the animal with mortality were extracted and taken for histological studies, while the other animals were returned to their habitual cages and observed for 24 hours and 14 days, for any sign of toxicity and possible mortality. The LD50 was identified by determining the geometric mean of the highest dose survived by the rat and the lowest dose that led to mortality.
Formula; LD50 = √ D0 × D100, where D0 is the highest dose that the animal survived and D100 is the lowest dose that caused the death of the animal. The findings from the acute toxicity study were used to determine the duration of exposure and quantity of glue that was used in the sub-acute and behavioural studies.
Sub-Acute Toxicity Study: Using the guideline of the Organisation for Economic Co-operation and Development (OECD TG 412) (Arts et al. 2008), Twenty-four male wistar rats were randomly distributed using decision analyst, into four groups of six rats each (n=6): control and experimental group, a dish containing glue was placed in the inhalational chamber of the experimental group, while an empty dish was placed in the centre of the control inhalation chamber. The first group was exposed to air, while the second, third and fourth group inhaled graded concentrations of (2 mL, 4 mL, and 8 mL) glue, 30 minutes daily for 28 days, with daily observation on general signs of toxicity and mortality being carried out. The animals were weighed and recorded weekly, and on the 29th day the animals were euthanized under general anaesthesia, using diethyl ether. Blood samples were collected via cardiac puncture, and whole blood was collected in EDTA bottles for haematological evaluations and the serum was collected in plain bottles for biochemical analysis, while histopathological examination was done on the lungs, livers, kidneys, hearts, brains, and eyes tissues.
Biochemical Study
The following parameters were determined from the blood serum; Albumin, Total Protein, Alkaline phosphatase (ALP) test, Alanine aminotransferase (ALT) test, Aspartate Aminotransferase test (AST), Total Bilirubin, Direct Bilirubin, Glucose, Serum Electrolyte, Urea, and Creatinine, at the Biochemistry Laboratory of Usmanu Danfodiyo University Teaching Hospital, Sokoto state, using methods by (Abdullahi et al. 2020; Kharchoufa et al. 2020) also using an Automatic Chemistry Analyser, and adhering to the manufacturers manual (Randox).
Histological Study
A qualified histopathologist, unaware of the experimental groups to which each animal belongs to was employed for all histological examinations of the lungs, livers, kidneys, hearts, brains, and eyes tissues.
Slide preparation: After sacrifice, the organs were extracted, rinsed with tap water and stored in 10% formalin, except for the eye and brain tissues that was stored in burn solution. Each organ tissue was cut transversely and put into separate cells, after which the tissue was processed using tissue processing machine for internal support. It was embedded with paraffin for external support, trimmed, and then cut into tiny sections of 3-5microns and then placed on a frosted glass Slade. It was dewaxed and hydrated, and then stained with Hematoxylin and Eosin stain (H and E). It was mounted with Dibutyl phthalate Polystyrene Xylene (DPX) and viewed with an X40 objective lens.
Haematological Study: The following haematological tests was carried out on blood samples, collected from all animals in each group and placed in an EDTA bottles: white blood cells (WBC), red blood cell (RBC), granulocyte (GRA), lymphocyte (LYM), haemoglobin (Hb), haematocrit (HCT), mean corpuscular volume (MCV), mean corpuscular haemoglobin (MCH), mean corpuscular haemoglobin concentration (MCHC), red cell distribution width (RDW), platelets (PLT), and mean platelet volume (MPV). Using the method by (Kharchoufa et al. 2020), haematological study was performed using an automatic haematology analyser (AHA) available in Haematology laboratory of Medistop diagnostic centre, Mabera, Sokoto state.
Experimental design to measure CNS activity
The design for this experiment was adopted from (Saha and Banerjee 2013). Thirty-six (36) male Wistar rats were randomly distributed using decision analyst into six (6) groups with six (6) animals in each group:
Group I: Control group received only air.
Group II: low concentration of glue inhalation (2 mL).
Group III: Intermediate concentration of glue inhalation (4 mL).
Group IV: High concentration of glue inhalation (8 mL)
Group V: Diazepam 1 mg/kg p.o or Imipramine 10 mg/kg p.o dissolved in vehicle depending on the test
Animals were exposed for at least three weeks before the behavioural study, and the test was carried out between 6:00 pm -12:00 am.
Anxiolytic activity using elevated plus maze (EPM): The plus maze apparatus consisted of two open arms (without walls), (50 × 10 cm) and two enclosed arms (50 × 10 cm) with a high wall of 40cm arranged in a way that similar arms are directly opposite to each other, with a central square of 10 cm which looks like a plus sign (Kumar et al. 2013). The apparatus was elevated by a support, 50 cm above floor level. The method was adopted from (Sofidiya et al. 2022). Rats from groups I, II, III, IV, and V as stated in the experimental design above, were included in this study. The group V received 1 mg/kg of diazepam orally 30 minutes prior to the test, while groups I, II, III and IV were exposed to air, 2mL, 4mL and 8mL of glue respectively, 30 minutes prior to the test. Each rat was placed individually at the centre of the elevated plus maze with its head facing toward an open arm, and away from the observer. The test lasted for 5 min, and the following parameters were observed during the period with the help of a camera: (a) Time spent in open arm, (b) Number of entries into open arm, and (c) Time spent in closed arm.
Anxiolytic activity of glue inhalation using ‘hole-board’ test (HBT): Rats from groups I, II, III, IV, and V as stated in the experimental design above, were included in this study. This method was adopted from (Sofidiya et al. 2022). Group V received 1 mg/kg of diazepam orally 30 minutes prior to the test, while groups I, II, III and IV were exposed to air, 2mL, 4mL and 8mL of glue respectively, 30 minutes prior to the test. Animals from each group were individually placed in the centre of the hole-board and away from the observer, the test lasted for 5mins, and the following parameters were observed during the period with the help of a camera: (a) Number of head dips, (b) Number of rearing, (c) The latency of the first head dip, and (d) Spontaneous movements (number of squares crossed with all four paws).
Anxiolytic activity using Open Field Test (OFT): The open field area was made of plain wood and consists of a square area (72×72×35cm). The floor had a square sheet of wood (72×72cm) with the surface divided into sixteen squares (18×18cm). One side of the wall was made of plexiglass material for visibility of the animal inside the box. The apparatus was illuminated by a 60 W bulb placed at a height of 100cm. The method was adopted from (Sofidiya et al. 2022). Rats from groups I, II, III, IV, and V as stated in the experimental design above, were included in this study. Group V received 1 mg/kg of diazepam orally 30 minutes prior to the test, while groups I, II, III and IV were exposed to air, 2mL, 4mL and 8mL of glue respectively, 30 minutes prior to the test. Each rat was placed in the centre square of the apparatus, away from the observer and the following parameters were recorded; (a) Number of lines crossed, (b) Number of centre square entries, (c) time spent in centre square, (d) time spent in the periphery (e) Number of rearing, (f) Number of grooming, (g) Number of assisted rearing (Kumar et al. 2013). Locomotion was also measured by counting the number of quadrants each animal crossed with all four paws (Taiwo et al. 2012).
CNS depressant activity using Forced Swim Test (FST): Using a vertical transparent cylinder tank (height: 40 cm; diameter: 18 cm, containing water to a height of 15 cm and maintained at a temperature of 25°C as adopted from Can et al. (2012). Rats from groups I, II, III, IV, and V as stated in the experimental design above, were included in this study. Group V received 10 mg/kg of imipramine orally, one hour prior to the test, while groups I, II, III and IV were exposed to air, 2mL, 4mL and 8mL of glue respectively, 30 minutes prior to the test. Each rat was placed gently by the tail into the water tank with no possible escape route and forced to swim. The total time spent floating or immobile was recorded with help of a camera. The test was performed under normal illumination, although the rats were allowed to acclimatize with the test environment during the first 60 seconds off the six (6) minutes test session, leaving only the last five (5) minutes to be analysed. The rats were closely monitored throughout the test period in case of drowning. On completion of the test, the rats were removed from the tank and wiped dry with a towel before placing them back in their home cages. Immobility time was considered a measure of learned helplessness or behavioural despair.
CNS Depressant Activity Using Tail Suspension Test (TST): Rats from groups I, II, III, IV, and V as stated in the experimental design above, were included in this study. Group V received 10 mg/kg of imipramine orally, one hour prior to the test, while groups I, II, III and IV were exposed to air, 2mL, 4mL and 8mL of glue respectively, 30 minutes prior to the test. The rats were suspended over the edge of a shelf, with its nose approximate 20-25 cm from the floor, using a piece of adhesive tape, about 17cm long, off which 2cm was used to stick the rat (1cm from the tip of the tail) and the other 15cm is used for the suspension. The duration of immobility was recorded for a period of 6 minutes, immobility time was recorded during the last 5 minutes. Immobility can also be recorded for the entire duration of suspension as long as it’s consistent with all rats and groups. The total immobility time was recorded for each rat with the help of a camera. Animal was considered immobile when they hang passively and completely motionless.
Withdrawal effect following glue inhalation exposure.
After 28 days of exposure to glue inhalation using the same inhalation protocol described in the sub-acute study above, the Wistar rats were abstained from glue exposure.
Conditioned Place Preference (CPP): Before withdrawal effects could be studied, dependence was tested using a modified ‘conditioned place preference’ method adopted from (Gerasimov et al. 2003; Lynch et al. 2010). The animals in group I, II, III and IV from the experimental design above were subjected to the CPP test, consisting of three distinct compartments separated by two guillotine doors. The walls of the middle chamber were grey with a smooth floor, while the conditioned compartment (21 × 21 × 27.5 cm, internal volume of 12L) had black walls with a smooth floor, the unconditioned compartment had white walls with a smooth floor. The lids of the three compartments were made of transparent plexiglass and the compartmental barriers were guillotine doors. The wire gauze in the brown box was used to introduce glue while the openings on the top of the white and brown compartments were used to introduce air respectively under positive pressure. Animals were introduced into the chambers by opening the lid which was immediately closed afterwards and left to move freely for 15 minutes, the procedure was monitored with the help of a video. The test was carried out in three phases; the day 1 was the baseline, the day 2 was the conditioning (drug) and the day 3 was the main test. Animals showing preference to a particular box were exempted from the study.
Withdrawal effect: After the conditioned placed preference test, the rats were abstained from air and glue inhalation, for 14 days. The rats involved were observed for any physical sign of withdrawal like anxiety, seizures and behavioural changes that are potentially life-threatening (Lynch et al. 2010). On the 15th day the animals were euthanized, and blood samples were collected for biochemical and haematological, for any sign of recovery.
Statistical Analysis
Data were stored in Microsoft excel, while data analysis was performed using Statistical Package for Social Science (SPSS) Version 19. Results were presented in percentages, tables, and figures. The results were summarized as mean ± Standard Error of Mean. One-way ANOVA was done to test the statistical difference with Post-hoc test using Tukey multiple- comparison test for the normally distributed data. Test analysis was considered significant at p<0.05.
Results
Inhalational Acute Toxicity studies of glue inhalation: During the acute toxicity test, mortality was observed in phase II of acute toxicity study, at the highest concentration of 20 mL during the last minutes of a 4-hour exposure to glue with a median lethal concentration of 14.14ml (
Table 1). Additionally, the animals exhibited a series of exploratory behaviour like rearing, grooming (rubbing of the forelimbs against the nose), seizure, salivation, and then followed by sedative effects and significantly abnormal gait.
Table 1.
(a): Inhalational acute toxicity studies of glue.
Table 1.
(a): Inhalational acute toxicity studies of glue.
| Dose (mL) |
Number of Animals dead/Used |
% Mortality |
| Phase I |
|
|
| 0.5 |
0/3 |
0 |
| 1.0 |
0/3 |
0 |
| 2.0 |
0/3 |
0 |
| Phase II |
|
|
| 5.0 |
0/1 |
0 |
| 10 |
0/1 |
0 |
| 20 |
1/1 |
1 |
Table 1.
(b): Histopathological analysis of organs of the animal with mortality.
Table 1.
(b): Histopathological analysis of organs of the animal with mortality.
| |
Organs |
Observation |
| 1 |
Lung |
Diffuse alveolar wall damage with moderate interstitial inflammation |
| 2 |
Heart |
Regular cardiac myocytes |
| 3 |
Kidney |
Normal glomeruli, acute tubular necrosis |
| 4 |
Liver |
Regular hepatocyte with moderate portal triaditis and regular central vein |
Effect of Glue Inhalation on Liver Function Test, following 28 days Sub-acute Exposure in Wistar rats.
The result of the liver function analysis showed that there was significant increase (P˂0.05) in some of the liver function parameters at various concentrations including total protein, total bilirubin, direct bilirubin, ALP, ALT, and a significant decrease in cholesterol following 28 days of glue inhalation (
Table 2)
Effects of Glue Inhalation on Renal Function Test following 28 days Sub-acute toxicity study: Result from the renal function analysis showed significant changes (p˂0.05) in some of the renal function parameters, with increased urea concentration at 8ml and increased serum electrolyte level in particular potassium and bicarbonate at 2mL, 4mL, and 8mL, following 28 days of glue inhalation (
Table 3).
Effects of Glue Inhalation on Haematological indices following 28 days Sub-acute toxicity study: There was a marked reduction in granulocyte level and increased platelet and lymphocyte levels although not statistically significant at all concentrations. (
Table 4)
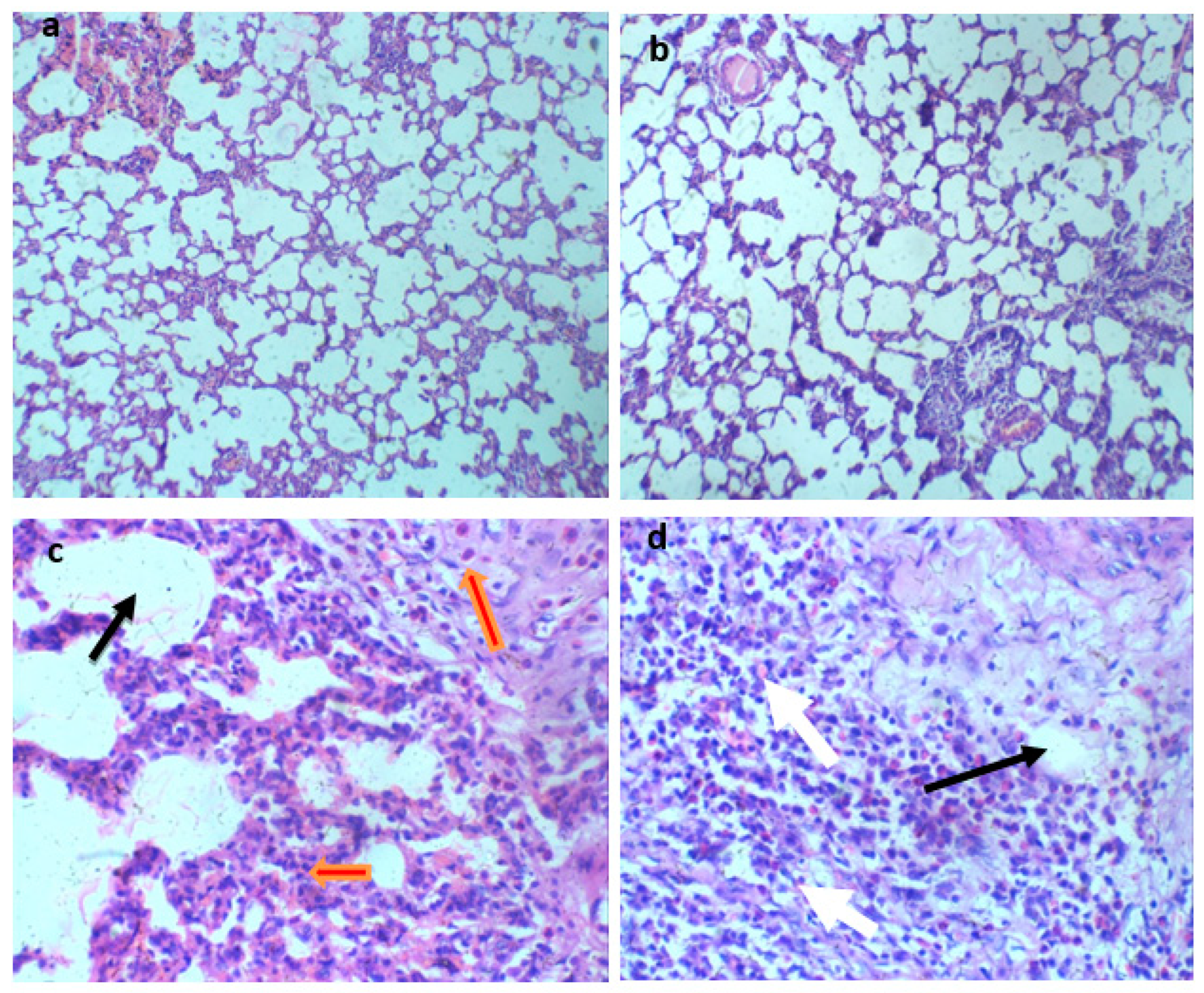
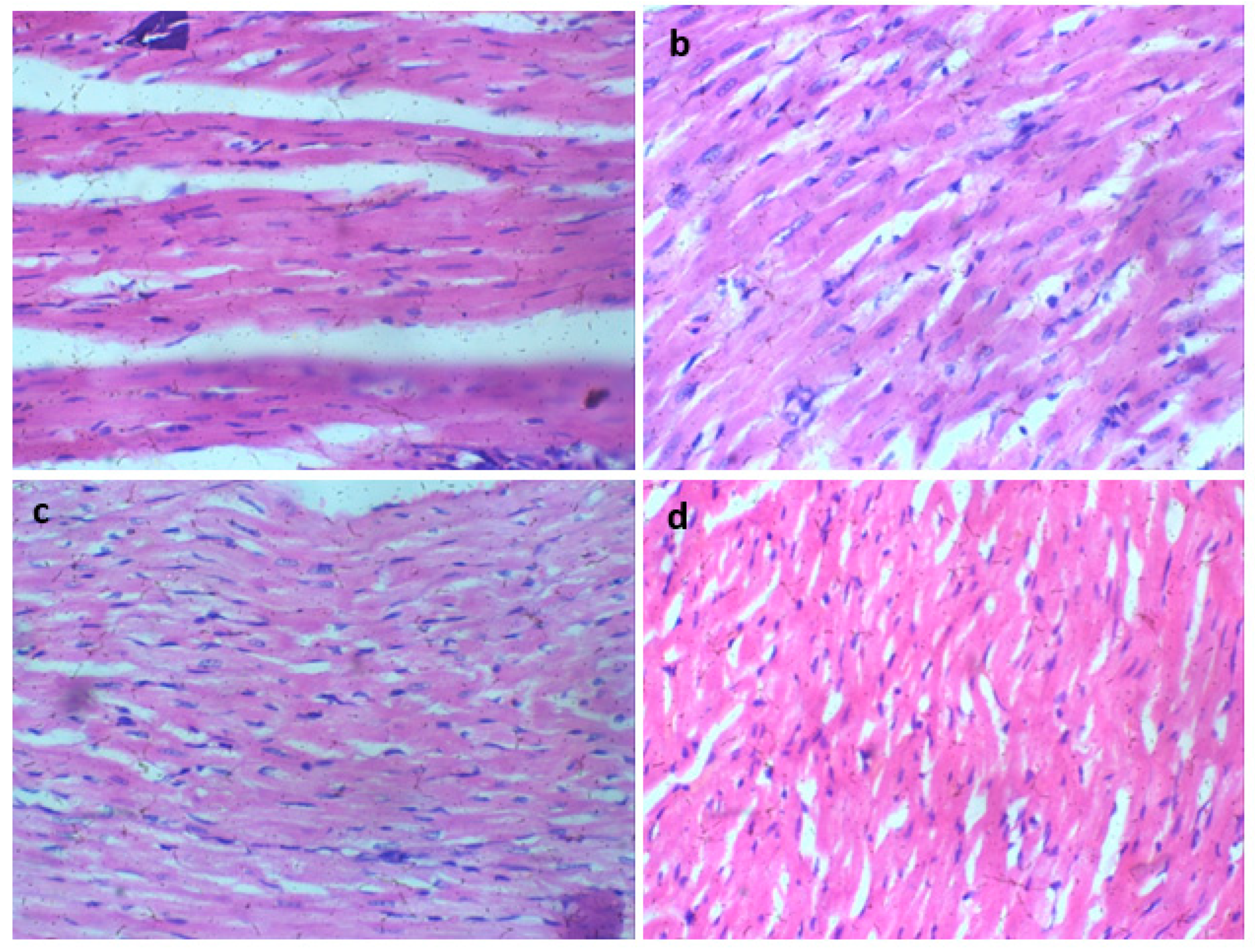
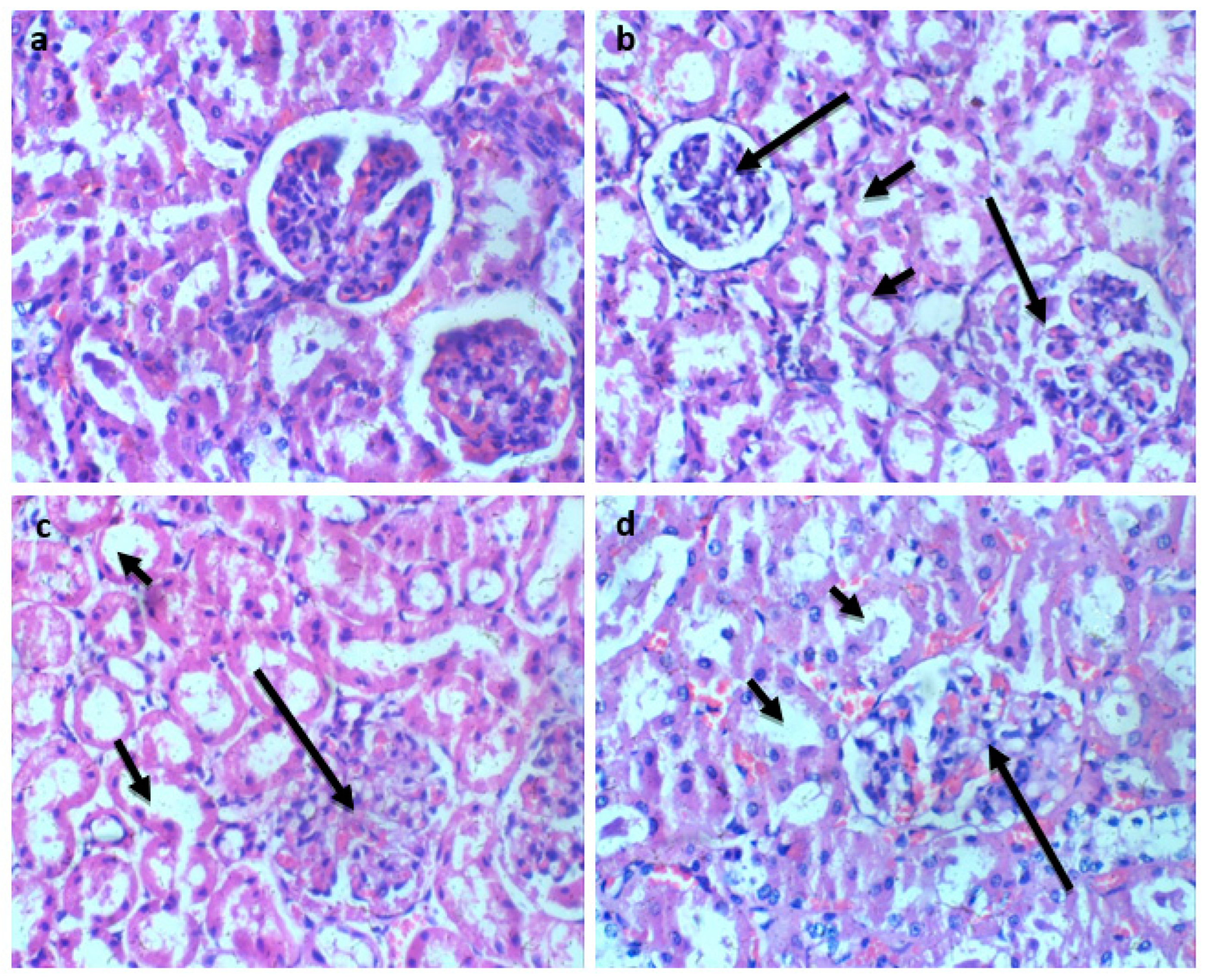
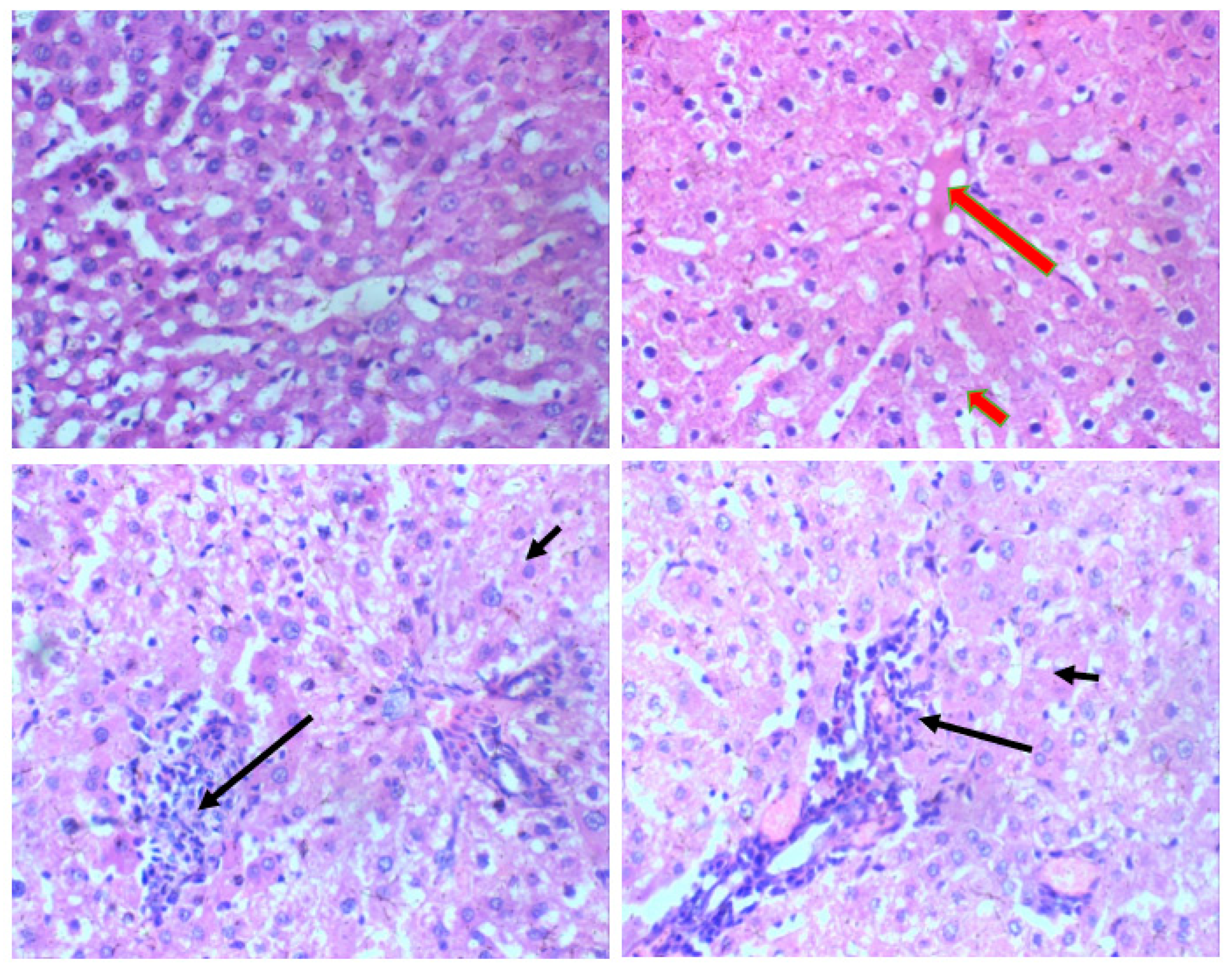
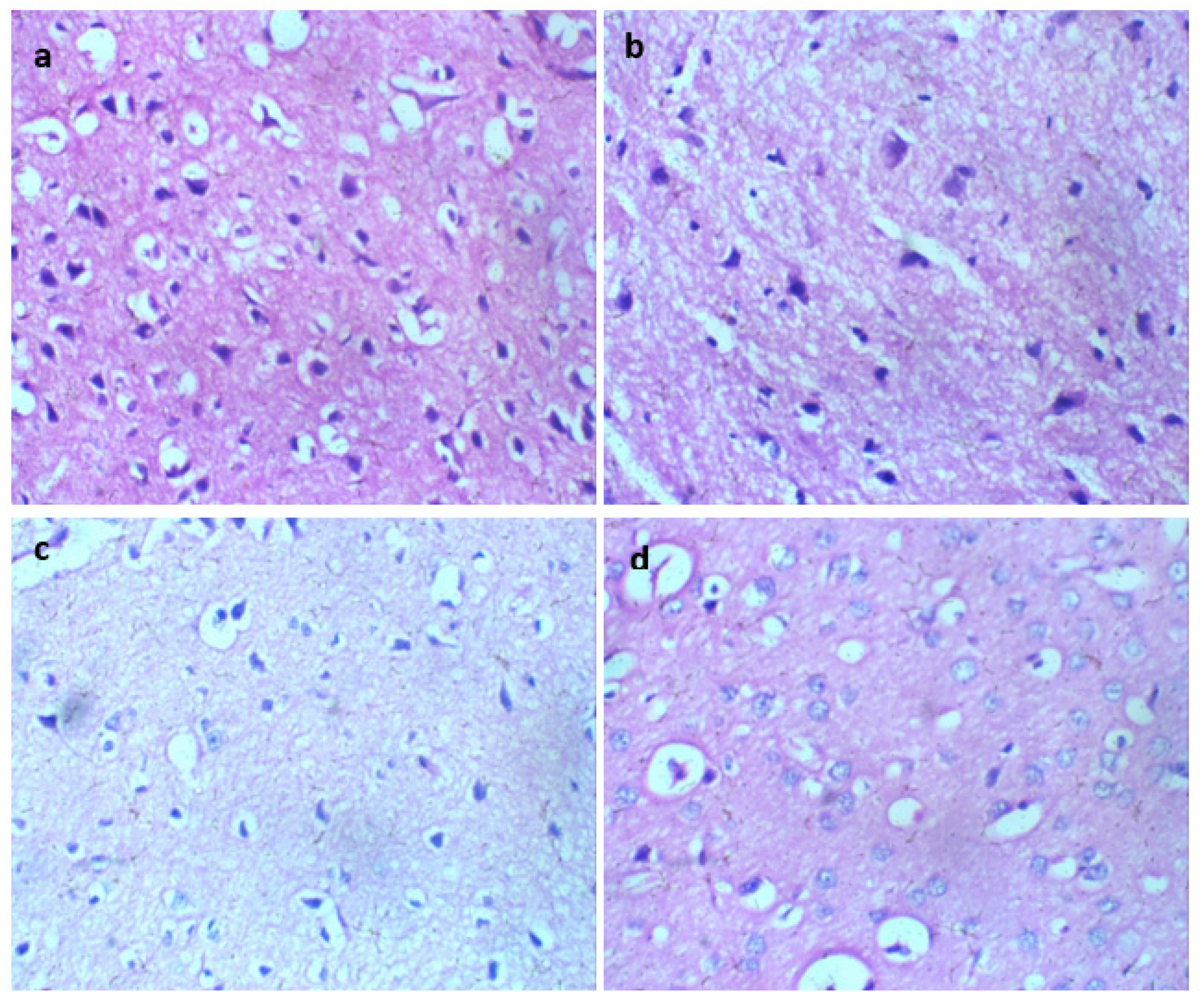
Histological analysis of Wistar rats following 28 days of Glue Inhalation: Histological analysis of the sample tissues showed no significant damage observed in Wistar rats’ kidney, heart, brain, and eye tissues of exposed rat in all group, except for the liver tissue which showed mild and moderate portal triaditis at concentrations of 4mL and 8ml respectively, also the lung tissue presented with mild interstitial inflammation at concentration of 2mL and moderate interstitial inflammation at concentrations 4mL and 8mL (
Table 5;
Figure 1,
Figure 2,
Figure 3,
Figure 4 and
Figure 5).
Effect of glue inhalation on behaviour of male Wistar rats using elevated plus maze: Inhalational administration of glue at the dose of 8mL showed significant (p<0.05) increase in the duration time spent in the open arms compared to the control group (air), and even so when compared to diazepam at a dose of 1mg/kg a standard anxiolytic agent. Although not statistically significant, it also decreased the number of entries into the closed arm at concentrations of 2mL and 8mL group compared to the control group, but not as much as the diazepam at a dose of 1mg/kg (
Table 6)
Table 6.
(a): Effect of glue inhalation on the behaviour in Wistar rats using elevated plus maze (open Arm).
Table 6.
(a): Effect of glue inhalation on the behaviour in Wistar rats using elevated plus maze (open Arm).
| GROUPS |
NUMBER OF ENTRY |
TIME SPENT (Sec.) |
| Control (Air) |
0.17 ± 0.17 |
12.0 ± 5.71 |
| Glue (2mL) |
0.33 ± 0.21 |
10.0 ± 7.28 |
| Glue (4mL) |
0.17 ± 0.17 |
43.33 ± 11.46 |
| Glue (8mL) |
0.33 ± 0.21 |
143.17 ± 46.63ab
|
| Diazepam (1.0mg/kg) |
0.33 ± 0.33 |
128.50 ± 50.93 |
Table 6.
(b): Effect of glue inhalation on elevated plus maze (Close Arm).
Table 6.
(b): Effect of glue inhalation on elevated plus maze (Close Arm).
| GROUPS |
NUMBER OF ENTRY |
TIME SPENT |
| Control (Air) |
2.67 ± 1.05 |
205.83 ± 47.97 |
| Glue (2mL) |
1.17 ± 0.17 |
239.83 ± 26.60 |
| Glue (4mL) |
3.67 ± 0.71 |
249.67 ± 9.67 |
| Glue (8mL) |
1.67 ± 0.61 |
107.67 ± 45.21 |
| Diazepam (1.0mg/kg) |
1.0 ± 0.37 |
95.17 ± 51.86 |
Effect of Glue Inhalation on the exploratory capability of Wistar rats using Hole Board Test: The result of hole board test conducted to evaluate the anxiolytic-like effect of glue inhalation on Wistar rats, showed significant increase (p<0.05) in number of head dips on hole board test compared to control group (air) (glue 4mL vs control (air)), and more than the standard anxiolytic (diazepam) at a dose of 1mg/kg. (
Table 7)
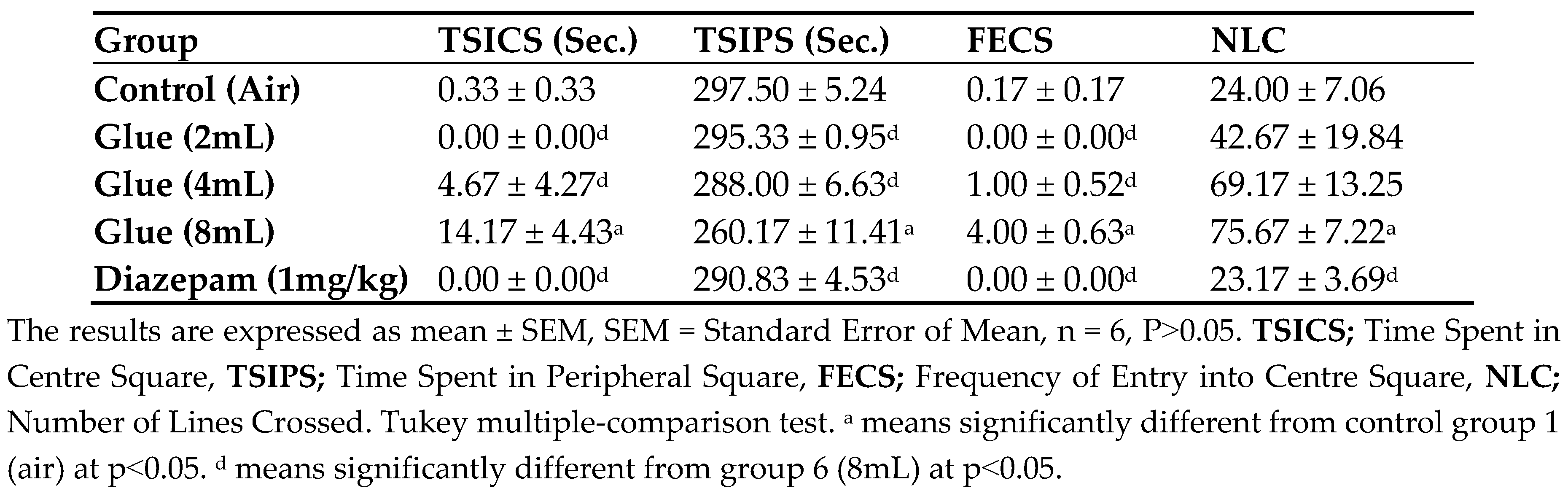
Effect of Glue Inhalation on the behaviour in Wistar rats using Open Field Test (OFTs): The inhalational administration of glue at a dose of 8mL showed significant increase (p<0.05) in time spent in centre square, a significant decreased time spent in the periphery, also a significant increase in frequency of entry into centre square and the number of lines crossed in open field test compared to control group, even more than the standard anxiolytic (diazepam) at a dose of 1mg/kg (
Table 8)
Effect of Glue Inhalation on the duration of immobility in Forced Swim Test in Wistar rats: The inhalational administration of glue at a dose of 4mL showed significant (p<0.05) increase in the duration of immobility in forced swim test compared to the standard antidepressant, Imipramine 10mg/kg and relatively higher than the control (air). Significantly (p˂0.05), at dose glue 8ml, there was a decrease in the duration of immobility in forced swim test compared to the control (air), more so compared to the standard antidepressant (imipramine) at a dose of 10mg/kg. (
Table 9)
Effect of Glue Inhalation on dependence of Wistar rats to glue inhalation using Conditioned Place Preference test (CPP): The inhalational administration of glue at all concentrations showed no significant (p<0.05) difference between the baseline and the main test of the treated groups compared to the control group (Air) following 28 days of glue inhalation, essentially inhalation of glue did not show any dependence property on wistar rats following 28 days sub chronic exposure (
Table 10).
Effect of 2 weeks withdrawal from Glue Inhalation on Liver Function following 28 days of exposure to glue: There were significant increases (p˂0.05) in some of the liver function parameters (total and direct bilirubin, total protein, ALT, ALP, AST, and albumin) following a 2week abstinence from 28 days of glue inhalation at concentrations of 2mL, 4mL, and 8mL in Wistar rats (
Table 11).
Effect of 2 weeks withdrawal from Glue Inhalation on renal function parameter of Wistar rats following 28 days of exposure to glue: There were significant increases in some of the haematological parameters (creatinine, sodium, potassium, and bicarbonate) especially at concentrations of 2mL and 4mL, and a significant decrease in the potassium level at 8mL compared to the control group, following a 2week abstinence from 28 days of glue inhalation (
Table 12)
Effect of 2 weeks of withdrawal from Glue Inhalation on Haematological parameters of Wistar rats following 28 days of exposure to glue: There were significant increases (p<0.05) in haematocrit, platelet count, and granulocyte count, especially at 8mL of glue, why there was a and a significant decrease in lymphocyte count of the haematological parameters of the wistar rats following a 2week abstinence from 28 days of glue inhalation at concentrations of 2mL, 4mL, and 8mL compared to the control group (
Table 13).
Discussion
This study evaluated the effect of abusing a volatile substance in Wister rats. The exploratory behaviour and sedation observed during the acute toxicity study are similar to the findings of Bouchatta et al. (2016) in addition, the animal showed signs of recovery between 20 minutes after exposure, this implied that acute glue sniffing has a short duration of action. The histological analysis of the animal with mortality showed diffuse alveolar wall damage with moderate interstitial inflammation of the lung, acute tubular necrosis in the kidney, and a regular hepatocyte with moderate portal triaditis of the liver.
The most widely accepted and validated animal models were used in this study for both anxiety and depression, they were models used to evaluate the anxiolytic property of diazepam and the antidepressant property of antidepressants (Haller and Alicki 2012; Yan et al. 2010). When a rodent is placed in a new environment there is a conflict between initially hiding from an unknown risk and the tendency to explore the new environment. Rodents naturally avoid, open, bright, and elevated spaces, hence the movement of rats in the EPM is because of two main motivations; the exploratory drive and fear drive which are evoked by new stimuli, and so assessing anxiety in rodent is done by using the ratio of time spent in the open arms to the time spent in the enclosed arm.
The Central Nervous system effect of glue inhalation has demonstrated that glue inhalation exhibits significant anxiolytic activity, which is believed to be concentration dependent, the reason glue (8mL) group did not show increased immobility time compared with the control group (air) is unknown, however the glue (2mL) group showed reduced immobility time compared to the control and may be presented as an antidepressant effect at this dose.
The EMP is sensitive to both anxiolytic and anxiogenic agents, and basically staying in the open arm caused physiological stress which can manifest as increased defecation and/or urination, but exposure to typical anxiolytics like diazepam or substances with anxiolytic property increase exploration of these open arms (Campos et al. 2013). Therefore, in the EPM, increase in the time spent in the open arm as against the time spent in the closed arm by the glue is an indication of anxiolytic activity, although glue did not increase the number of entries into the open space. Similarly, in HBT which follows similar principles of fear drive and exploratory drive, the anxiety state of the rat is presumed to be inversely proportional to the number of head dips (Campos et al. 2013). The increase in the number of head dips by rats exposed to glue inhalation is suggested an index of anxiolytic activity.
The Open field test shows a higher number of lines crossed which indicates increase in locomotive activity and/ or a lower level of anxiety, more so, increased entry and time spent in the centre square expresses decreased anxiety altered by the anxiolytic activity of the substance, therefore in the OFT the increase in number of lines crossed, frequency of entries into centre square and time spent in centre square suggested an index of locomotive activity and anxiolytic activity, with increased in the glue treated group compared to the control group and even the diazepam (positive control). The increased locomotive activity is contrarily to the findings of Pierre et al. (2016) who discovered decrease of motor activity at a high concentration (Pierre et al. 2016).
The FST and TST are model that are based on behavioural despair or helplessness, Rodents initially try to escape or save themselves in both tests, but eventually exhibit despair (immobility). The observed decrease in the duration of immobility for glue 2ml groups compared to the control group may suggest an antidepressant activity at this concentration. This study showed an increase in the duration of immobility of the imipramine group contrarily to other studies, this finding may be as result of the differences in the strain of rats (Vaugeois et al. 1997), or delayed onset of action of imipramine.
The active constituent(s) responsible for the anxiolytic and CNS depressant activity of glue may be toluene which has been reported to have anxiolytic and CNS depressant activity, as it significantly inhibit the NMDA subtype of glutamate-activated ion channels but had little effect on AMPA subtype of ionotropic glutamate receptor even though they are closely related, some researchers also found that toluene increases dopamine release and dopaminergic neurons activity, as well as act via inhibitory GABAA receptor (Cruz et al. 2014; López-Rubalcava et al. 2000; Páez-Martı́inez et al. 2003). However, the CNS action of benzene is not entirely clear, but some studies have proposed that benzene has a CNS depressant activity. The consumption of volatile substances in an extensive and excessive manner is quite dangerous to the users (Pierre et al. 2016).
During the 28-days Sub-acute sessions the observed concentration dependent effect are classical signs of Central Nervous System activity that can result in CNS functional damage. Continues seizure for instance, can result in impairment or paralysis of the affected region, abnormal respiration and respiratory depression which are the major causes of mortality in inhalation abuse can result to death of a glue sniffer. However, we also observed that the unconscious animals recovered between 5-20 minutes of discontinued exposure, which may explain why glue sniffers need to keep inhaling the glue to maintain the state of euphoria which makes it even dangerous.
At the end of the 28 days Sub-acute test, glue inhalation changed some haematological parameters indicative of infection (Ferreira et al. 2017), there was no decrease WBC, RBC or HB contrarily to the finding by Pierre et al. who discovered a decrease in WBC, RBC and HB at doses 320mg/L and 640mg/L, following exposure to ‘‘Dia’’ glue for 28days (Pierre et al. 2016). Similarly, the liver function parameters present findings consistent with Pierre et al. (2016) who used ‘‘Dia’’ glue on wistar rats at 320mg/L and 640mg/L for 28 days and (Pierre et al. 2016). Apparently, inhalation of glue caused elevated serum potassium, bicarbonate and urea concentrations in treated animal which can alter the kidney function and may result to kidney disease or failure. The major organ involved in the regulation of electrolytes levels is the kidney, thereby maintaining homeostasis, which makes urea and serum electrolyte the commonly requested biochemical tests for renal function assessment (Nwaji et al. 2022). The stable histological findings may be as a result of the time limit, a significant damage to the organs may occur following chronic abuse of glue.
This study showed that glue inhalation did not cause addiction in wistar rats, after 28 days of exposure to glue. However, we observed the effect of withdrawal after Sub-acute abuse of glue inhalation, and the result showed more intense hostility among rats in group glue 4mL and 8mL, one week after cessation of glue exposure, although the aggressive behaviour subsided by the end of the second week of abstinence. This finding is in tandem with the result of Bouchatta et al. (2016) More so, hostility has been associated with benzodiazepine withdrawal in both humans (Wallace and Taylor 2009) and animals (Nath et al. 2000; Saxon et al. 2010), and previous studies have shown that toluene has GABAA agonistic modulatory effects (Cruz et al. 2014; López-Rubalcava et al. 2000), it is otherwise safe to say that the behavioural control can be likened to the activation GABAergic neurons which in turn, synapse with other neurons in control of aggressive behaviours (Bouchatta et al. 2016; Nelson and Trainor 2007).
Conclusion
Result from this study showed that glue sniffing has anxiolytic-like properties, reason why some young adults would want to abuse this substance to maintain a state of calmness. Furthermore, glue inhalation significantly caused elevated liver enzymes and increased serum electrolyte, which may result in hepatotoxicity or renal malfunction. Finally, this study in connection with previous behavioural studies suggested that from all indications glue inhalation has CNS depressant effect, as well as hepatotoxic, and hematopoietic effect which makes it highly toxic.
Recommendations
Further studies on the molecular targets or site of action of glue inhalation and evaluation of changes in neurotransmitters in the brain as a result of glue inhalation especially (GABA, dopamine, and serotonin) through western blot are expected to provide insights into the potential mechanism of action of glue inhalation on the CNS.
Availability and curiosity seem to be propelling the abuse of these harmful substances, therefore we recommend that the government and regulatory agencies in different countries where glue inhalation is prevalent, should create awareness among the youth population on the negative impact of abusing this substance, and apply preventative measures targeting producers and suppliers, to limit access to this substance.
Author Contributions
AOJ conceived and designed the study. OEI performed literature search, conducted the experiment and prepared the manuscript. BA, MLU, UMT, and AY supervised the research and performed data analysis. UM and MSH performed histopathological examination of the harvested organs. KAA critically reviewed and edited the manuscript. All authors read and approved the final version of the manuscript.
Funding
This study received no grants from any funding bodies or authorities. It was self-sponsored.
Data Availability Statement
Data are available upon reasonable request from the corresponding author.
Acknowledgement
We sincerely appreciate all the staff (Academics and Non-academics) of the Department of Pharmacology and Therapeutics as well as Dr. Aluefua Fidelis including the staff at the animal house, Faculty of Pharmaceutical sciences, Usmanu Danfodiyo University Sokoto, Nigeria for their support especially during the animal husbandry and behaviour studies, we could not have undertaken this journey without you all.
Conflicts of Interest
The authors declare that they have no relevant financial, non-financial or personal relationships that could have influenced the work reported in this paper.
Ethical Approval
The study was conducted in accordance with the ARRIVE guidelines (Kilkenny et al. 2013). The animals were cared for in accordance with the approved institutional protocols and the provisions for animal care and use. The Departmental Research and Ethics Committee approved the research protocol and assigned it a reference number UDUS/DREC/2022/017.
References
- Abdullahi, S. N., Dahiru, M. M., Rabiu, D. H., and Mohammed, J. M. (2020). Toxicological effect of inhaled mosquito incense sticks smoke on the histology and biochemical responses in experimental rats. Fudma journal of sciences, 4(2), 260–268. [CrossRef]
- Arts, J. H. E., Muijser, H., Jonker, D., van de Sandt, J. J. M., Bos, P. M. J., and Feron, V. J. (2008). Inhalation toxicity studies: OECD guidelines in relation to reach and scientific developments. Experimental and Toxicologic Pathology, 60(2–3), 125–133. [CrossRef]
- Bouchatta, O., Ouhaz, Z., Ba-Mhamed, S., Kerekes, N., and Bennis, M. (2016). Acute and chronic glue sniffing effects and consequences of withdrawal on aggressive behavior. Life Sciences, 152, 14–20. [CrossRef]
- Campos, A. C., Fogaça, M. V., Aguiar, D. C., and Guimarães, F. S. (2013). Animal models of anxiety disorders and stress. Revista Brasileira de Psiquiatria, 35(SUPPL.2). [CrossRef]
- Can, A., Dao, D. T., Arad, M., Terrillion, C. E., Piantadosi, S. C., and Gould, T. D. (2012). The Mouse Forced Swim Test. JoVE, 59, e3638. [CrossRef]
- Cruz, S. L., and Bowen, S. E. (2021). The last two decades on preclinical and clinical research on inhalant effects. Neurotoxicology and Teratology, 87, 106999. [CrossRef]
- Cruz, S. L., Rivera-García, M. T., and Woodward, J. J. (2014). Review of toluene action: clinical evidence, animal studies and molecular targets. Journal of Drug and Alcohol Research, 3, 1–8. [CrossRef]
- Danjuma, A., Ibrahim Taiwo, A., and Omoniyi, S. O. (2015). Nonconventional Use of Substances among Youth in Nigeria: Viewpoints of Students in a Nigerian Tertiary Institution. Journal of Nursing and Care, 04(06). [CrossRef]
- Egbuonu, A. C. C., Egbuonu, O. N. C., and Samuel, E. S. (2016). The prevalence of some psychoactive substances use among secondary school adolescents in Bosso Local Government Area, Nigeria. Journal of Environmental Toxicology and Public Health, 1, 6–10. [CrossRef]
- Ferreira, L. L., Gomes-Filho, J., Sumida, D., Bonfim, S., Sivieri-Araujo, G., Guimarães, G., and Cintra, L. (2017). Diabetic Rats Present High Mean Platelet Count in the Presence of Oral Infections. Brazilian Dental Journal, 28, 548–551. [CrossRef]
- Abasiubong F., Udobang J. A., Idung A. U., Udoh S. B., and Jombo H. E. (2014). Pattern of psychoactive substance use in the northern region of Nigeria. African Journal of Drug and Alcohol Studies, 13(2), 107–115.
- Gerasimov, M. R., Collier, L., Ferrieri, A., Alexoff, D., Lee, D., Gifford, A. N., and Balster, R. L. (2003). Toluene inhalation produces a conditioned place preference in rats. European Journal of Pharmacology, 477(1), 45–52. [CrossRef]
- Haller, J., and Alicki, M. (2012). Current animal models of anxiety, anxiety disorders, and anxiolytic drugs. In Current Opinion in Psychiatry (Vol. 25, Issue 1, pp. 59–64). [CrossRef]
- Jatau, A. I., Sha’aban, A., Gulma, K. A., Shitu, Z., Khalid, G. M., Isa, A., Wada, A. S., and Mustapha, M. (2021). The Burden of Drug Abuse in Nigeria: A Scoping Review of Epidemiological Studies and Drug Laws. Public Health Reviews, 42, 1603960. [CrossRef]
- Jayanth, S. H., Hugar, B. S., Praveen, S., and Girish Chandra, Y. P. (2017). Glue sniffing. The Medico-Legal Journal, 85(1), 38–42. [CrossRef]
- Kazeem, Y., and Chutel, L. (2018, May 17). Africa’s desperate youth are getting high on opioids. Blueprint Newspapers Limited. https://www.blueprint.ng/africas-desperate-youth-getting-high-opioids-yomi-kazeem-lynsey-chutel/. 17 May.
- Kharchoufa, L., Bouhrim, M., Bencheikh, N., el Assri, S., Amirou, A., Yamani, A., Choukri, M., Mekhfi, H., and Elachouri, M. (2020). Acute and Subacute Toxicity Studies of the Aqueous Extract from Haloxylon scoparium Pomel (Hammada scoparia (Pomel)) by Oral Administration in Rodents. BioMed Research International, 2020. 2020. [CrossRef]
- Kilkenny C, Browne WJ, Cuthill IC et al (2013) Improving bioscienceresearch reporting: the arrive guidelines for reporting animal research. Animals 4:35–44. [CrossRef]
- Kumar, V. , Bhat, Z. A., and Kumar, D. (2013). Animal models of anxiety: A comprehensive review. In Journal of Pharmacological and Toxicological Methods (Vol. 68, Issue 2, pp. 175–183). [CrossRef]
- Kuranchie, F. A., Angnunavuri, P. N., Attiogbe, F., and Nerquaye-Tetteh, E. N. (2019). Occupational exposure of benzene, toluene, ethylbenzene and xylene (BTEX) to pump attendants in Ghana: Implications for policy guidance. Http://Www.Editorialmanager.Com/Cogentenv, 5(1). [CrossRef]
- LEE, R., and VALENTINO, D. (2021). TOLUENE-INDUCED ACUTE LUNG INJURY: CASE REPORT. Chest, 160(4), A719. [CrossRef]
- López-Rubalcava, C., Hen, R., and Cruz, S. L. (2000). Anxiolytic-like actions of toluene in the burying behavior and plus-maze tests: differences in sensitivity between 5-HT1B knockout and wild-type mice. Behavioural Brain Research, 115(1), 85–94. [CrossRef]
- Lorke, D. (1983). A New Approach to Practical Acute Toxicity Testing. Arch. Toxicol, 54(4), 275–287.
- Lynch, W. J., Nicholson, K. L., Dance, M. E., Morgan, R. W., and Foley, P. L. (2010). Animal Models of Substance Abuse and Addiction: Implications for Science, Animal Welfare, and Society. Comparative Medicine, 60(3), 177.
- Munawar, K., Choudhry, F. R., Hadi, M. A., and Khan, T. M. (2019). Prevalence of and Factors Contributing to Glue Sniffing in the South Asian Association for Regional Cooperation (SAARC) Region: A Scoping Review and Meta-analysis. Substance Use and Abuse, 55(5), 752–762. [CrossRef]
- Nath, C., Saxena, R., and Gupta, M. (2000). Effect Of Dopamine Agonists And Antagonists On The Lorazepam Withdrawal Syndrome In Rats. Clinical and Experimental Pharmacology and Physiology, 27(3), 167–171. [CrossRef]
- Nelson, R. J., and Trainor, B. C. (2007). Neural mechanisms of aggression. Nature Reviews Neuroscience, 8(7), 536–546. [CrossRef]
- Nwaji, A. R., Ante, I. A., Nwoke, F. K., and Inwang, U. (2022). Changes in Serum Electrolytes, Urea and Creatinine in Nicotiana tabacum-treated Rats. Nigerian Journal of Physiological Sciences, 37(1), 153–156. [CrossRef]
- Páez-Martı́inez, N., Cruz, S. L., and López-Rubalcava, C. (2003). Comparative study of the effects of toluene, benzene, 1,1,1-trichloroethane, diethyl ether, and flurothyl on anxiety and nociception in mice. Toxicology and Applied Pharmacology, 193(1), 9–16. [CrossRef]
- Ramlagan, S., Peltzer, K., and Matseke, G. (2010). Epidemiology of drug abuse treatment in South Africa. South African Journal of Psychiatry, 16(2), 40–49. [CrossRef]
- Saha, S., and Banerjee, S. (2013). Central nervous system stimulant actions of Alpinia galanga (L.) rhizome: A preliminary study. Indian Journal of Experimental Biology, 51, 828–832.
- Sofidiya, M. O., Alokun, A. M., Fageyinbo, M. S., and Akindele, A. J. (2022). Central nervous system depressant activity of ethanol extract of Motandra guineensis (Thonn) AD. aerial parts in mice. Phytomedicine Plus, 2(1). [CrossRef]
- Taiwo, A. E., Leite, F. B., Lucena, G. M., Barros, M., Silveira, D., Silva, M. V., and Ferreira, V. M. (2012). Anxiolytic and antidepressant-like effects of Melissa officinalis (lemon balm) extract in rats: Influence of administration and gender. Indian Journal of Pharmacology, 44(2), 189–192. [CrossRef]
- Ugbedeojo A. A., Terungwa, L. T., and Terfa, T.J. (2021). Intake and Abuse of Psychoactive Substances and Its Relative Consequences: A Review. Science Journal of Analytical Chemistry, 9(2), 39. [CrossRef]
- UNODC. (2013). The challenge of new psychoactive substances Global SMART Programme 2013 Acknowledgements.
- UNODC. (2021). UNODC Early Warning Advisory (EWA) on New Psychoactive Substances (NPS). https://www.unodc.org/LSS/Home/NPS.
- Vaugeois, J.-M., Passera, G., Zuccaro, F., and Costentin, J. (1997). Individual differences in response to imipramine in the mouse tail suspension test. Psychopharmacology, 134(4), 387–391. [CrossRef]
- Vitale, C. M., and Gutovitz, S. (2018). Aromatic Toxicity. StatPearls. http://europepmc.org/books/NBK532257.
- Wallace, P. S., and Taylor, S. P. (2009). Reduction of appeasement-related affect as a concomitant of diazepam-induced aggression: evidence for a link between aggression and the expression of self-conscious emotions. Aggressive Behavior, 35(2), 203–212. [CrossRef]
- Yan, H. C., Cao, X., Das, M., Zhu, X. H., and Gao, T. M. (2010). Behavioral animal models of depression. In Neuroscience Bulletin (Vol. 26, Issue 4, pp. 327–337). [CrossRef]
Figure 1.
Lung section H and E X 100 (a-d). a (control): Normal regular alveoli space and interstitium. b (2 mL of glue): Normal regular alveoli space (black arrow), mild interstitium inflammatory infiltrate (white arrow) c (4mL of glue) and d (8mL of glue ): Normal regular alveoli space (black arrow) and with moderate interstitium inflammatory infiltrate (white arrow).
Figure 1.
Lung section H and E X 100 (a-d). a (control): Normal regular alveoli space and interstitium. b (2 mL of glue): Normal regular alveoli space (black arrow), mild interstitium inflammatory infiltrate (white arrow) c (4mL of glue) and d (8mL of glue ): Normal regular alveoli space (black arrow) and with moderate interstitium inflammatory infiltrate (white arrow).
Figure 2.
Heart session H and E X 100 (a-d).a (control), b (2mL of glue), c (4mL of glue), d (8mL of glue): Heart sections show regular cardiac myocytes.
Figure 2.
Heart session H and E X 100 (a-d).a (control), b (2mL of glue), c (4mL of glue), d (8mL of glue): Heart sections show regular cardiac myocytes.
Figure 3.
Kidney section H and E X 100. a (control): Kidney section shows regular glomeruli and renal tubules. b (2mL of glue), c (4mL of glue) d (8mL of glue): Kidney sections show regular glomeruli (Long arrow) and renal tubules (short arrow).
Figure 3.
Kidney section H and E X 100. a (control): Kidney section shows regular glomeruli and renal tubules. b (2mL of glue), c (4mL of glue) d (8mL of glue): Kidney sections show regular glomeruli (Long arrow) and renal tubules (short arrow).
Figure 4.
Liver section H and E X 100. a (control) and b (2mL of glue): Normal central vein (long arrow) and hepatocytes arrange in cords (short arrow). c (4mL of glue): Milde porta triaditis (long arrow) and hepatocytes arrange in cords (short arrow). d (8mL of glue): Moderate porta triaditis (long arrow) and hepatocytes arrange in cords (short arrow).
Figure 4.
Liver section H and E X 100. a (control) and b (2mL of glue): Normal central vein (long arrow) and hepatocytes arrange in cords (short arrow). c (4mL of glue): Milde porta triaditis (long arrow) and hepatocytes arrange in cords (short arrow). d (8mL of glue): Moderate porta triaditis (long arrow) and hepatocytes arrange in cords (short arrow).
Figure 5.
Brain session H and E 100. a (control), b (2mL of glue), c (4mL of glue), d (8mL of glue): A brain sections show regular neuropils.
Figure 5.
Brain session H and E 100. a (control), b (2mL of glue), c (4mL of glue), d (8mL of glue): A brain sections show regular neuropils.
Table 2.
(a): Effect of glue inhalation on liver function indices of Wistar rat.
Table 2.
(a): Effect of glue inhalation on liver function indices of Wistar rat.
| Parameters |
Control (Air) |
Glue (2ml) |
Glue (4ml) |
Glue (8ml) |
| Total Bilirubin (mg/dL) |
3.15 ± 0.23 |
3.61± 0.16 |
5.25 ± 0.51ab
|
6.62 ± 0.39ab
|
| Direct Bilirubin (mg/dL) |
1.64 ± 0.14 |
1.55 ± 0.08d
|
2.02 ± 0.34d
|
3.00 ± 0.14a
|
| ALP (u/L) |
42.63 ± 2.49 |
71.79 ± 6.35a
|
102.49 ± 6.06ab
|
115.07 ± 4.66ab
|
| AST (u/L) |
63.51 ± 8.57 |
72.92 ± 18.54 |
81.65 ± 17.32 |
80.13 ± 19.50 |
| ALT (u/L) |
22.48 ± 0.68 |
72.20± 8.61ac
|
96.60 ± 2.19ad
|
67.63 ± 0.64ac
|
| Total Protein (g/dL) |
6.00 ± 0.13 |
6.45 ± 0.22 |
6.95 ± 0.78 |
9.07 ± 0.86ab
|
| Cholesterol (mg/dL) |
216.64 ± 8.99 |
136.22 ± 12.47ac
|
198.97 ± 10.35 |
153.46 ±10.40ac
|
| Glucose (mmol/L) |
6.36 ± 0.25 |
5.96 ± 0.34 |
5.56 ± 0.30 |
6.72 ± 0.55 |
| Albumin (g/L) |
3.56 ± 0.17 |
3.56 ± 0.17 |
3.94 ± 0.11 |
3.65 ± 0.18 |
Table 3.
Effect of glue inhalation on renal function parameters in Wistar rats.
Table 3.
Effect of glue inhalation on renal function parameters in Wistar rats.
| Parameter |
Control (Air) |
Glue (2ml) |
Glue (4ml) |
Glue (8ml) |
| Urea (mmol/L) |
7.36 ± 0.58 |
9.06 ± 0.58 |
9.06 ± 0.55 |
10.20 ± 0.82a
|
| Creatinine (µmol/L) |
198.54 ± 20.60 |
218.82 ± 18.59 |
145.99 ± 10.88b
|
172.38 ± 12.23 |
| Na+ (mmol/L) |
141.08 ± 1.37 |
145.05 ± 1.99 |
145.67 ± 2.18 |
145.55 ± 2.58 |
| K+ (mmol/L) |
4.63 ± 0.14 |
4.55 ± 0.13a
|
4.73 ± 0.18a
|
5.00 ± 0.10a
|
| HCO3 (mmol/L) |
17.53 ± 0.28 |
20.50 ± 0.41a
|
19.67 ± 0.42a
|
21.10 ± 0.52a
|
Table 4.
Effect of glue inhalation on haematological indices following 28 days sub-acute study.
Table 4.
Effect of glue inhalation on haematological indices following 28 days sub-acute study.
| Parameters |
Control (Air) |
Glue (2mL) |
Glue (4mL) |
Glue (8mL) |
| White Blood Cell (10^9/L) |
21.65 ± 1.49 |
21.96 ± 1.43 |
22.60 ± 1.77 |
22.26 ± 1.74 |
| Red Blood Cell (10^12/L) |
6.79 ± 0.65 |
7.28 ± 0.23 |
7.03 ± 0.22 |
7.34 ± 0.24 |
| Haemoglobin (g/dL) |
14.58 ± 0.35 |
15.20 ± 0.36 |
14.67 ± 0.46 |
15.00 ± 0.37 |
| Haematocrit (%) |
37.28 ±1.10 |
37.87 ± 1.05 |
36.42 ± 1.23 |
37.62 ± 1.44 |
| Platelet (10^9/L) |
625.33 ± 33.89 |
614.00± 24.03 |
663.33 ± 44.17 |
699.00 ± 43.42 |
| Granulocyte (%) |
32.87 ± 3.82 |
24.85 ± 1.53 |
25.58 ± 2.20 |
23.35 ± 1.88 |
| Lymphocyte (%) |
67.78 ± 1.67 |
70.23 ± 0.86 |
71.80 ± 3.61 |
70.25 ± 1.44 |
Table 5.
Histological analysis of Wistar rats following 28 days exposure.
Table 5.
Histological analysis of Wistar rats following 28 days exposure.
| GROUPS |
ORGANS |
MICROSCOPIC OBSERVATION |
Group I
Control (air) |
Lungs |
Normal regular alveoli space and interstitium. |
| Liver |
Normal, portal triad, central vein, and hepatocytes |
| Kidney |
Normal glomeruli, tubules and interstitium. |
| Heart |
Regular cardiac myocytes |
| Brain |
Normal neurophils |
| Eye |
Normal conjunctiva, cornea, and retina |
Group II
Glue (2mL) |
Lungs |
Regular alveolar spaces with mild interstitial inflammation |
| Liver |
Normal, portal triad, central vein, and hepatocytes |
| Kidney |
Normal glomeruli, tubules and interstitium |
| Heart |
Regular cardiac myocytes |
| Brain |
Regular neurophils |
| Eye |
Normal conjunctiva, cornea, and retina |
Group III
Glue (4mL) |
Lungs |
Regular alveolar spaces with moderate interstitial inflammation |
| Liver |
Regular hepatocyte with mild portal triaditis and regular central vein |
| Kidney |
Normal glomeruli, tubules and interstitium |
| Heart |
Regular cardiac myocytes |
| Brain |
Regular neurophils |
| Eye |
Normal conjunctiva, cornea, and retina |
Group IV
Glue (8mL) |
Lungs |
Regular alveolar spaces with moderate interstitial inflammation |
| Liver |
Regular hepatocyte with moderate portal triaditis and regular central vein |
| Kidney |
Normal glomeruli, tubules and interstitium |
| Heart |
Regular cardiac myocytes |
| Brain |
Regular neurophils |
| Eye |
Normal conjunctiva, cornea, and retina |
Table 7.
Effect of glue inhalation on the behaviour in Wistar rats using hole board test.
Table 7.
Effect of glue inhalation on the behaviour in Wistar rats using hole board test.
| GROUPS |
NUMBER OF HEAD DIPS |
| Control (Air) |
8.0 ± 2.57 |
| Glue (2mls) |
12.0 ± 3.11c
|
| Glue (4mls) |
25.5 ± 3.27a
|
| Glue (8mls) |
12.3 ± 2.64c
|
| Diazepam 1.0mg/kg |
9.0 ± 3.82c
|
Table 8.
Effect of glue inhalation on the behaviour in Wistar rats using open field test.
Table 8.
Effect of glue inhalation on the behaviour in Wistar rats using open field test.
Table 9.
Effect of glue inhalation on the duration of immobility in Wistar rats using forced swim test (FST).
Table 9.
Effect of glue inhalation on the duration of immobility in Wistar rats using forced swim test (FST).
| GROUPS |
DURATION OF IMMOBILITY (Sec.) |
| Control (Air) |
135.83 ± 14.24 |
| Glue (2mL) |
87.17 ± 26.21c
|
| Glue (4mL) |
173.33 ± 9.77f
|
| Glue (8mL) |
32.50 ± 14.51ac
|
| Imipramine (10mg/kg) |
45.50 ± 9.11a
|
Table 10.
Conditioned placed preference test (CPP).
Table 10.
Conditioned placed preference test (CPP).
| Groups (mL) |
Base Line |
Main Test |
| TSDC (Sec.) |
TSWC (Sec.) |
TSDC (Sec.) |
TSWC (Sec.) |
| Control (Air) |
507.50 ± 118.00 |
331.83 ± 110.62 |
507.50 ± 118.00 |
331.83 ± 110.62 |
| Glue (2mL) |
721.33 ± 75.23 |
152.00 ± 65.23 |
650.33 ± 150.09 |
237.17 ± 150.59 |
| Glue (4mL) |
663.17 ± 36.52 |
164.67 ± 20.75 |
719.33 ± 25.22 |
140.17 ± 22.95 |
| Glue (8mL) |
583.50 ± 125.80 |
156.50 ± 113.96 |
612.17 ± 130.81 |
227.50 ± 129.67 |
Table 11.
Effect of 2 weeks withdrawal from glue inhalation on liver function indices.
Table 11.
Effect of 2 weeks withdrawal from glue inhalation on liver function indices.
| Parameters |
Control (Air) |
Glue (2ml) |
Glue (4ml) |
Glue (8ml) |
| Total Bilirubin (mg/dL) |
0.55 ± 0.01 |
1.34± 0.07acd
|
0.58 ± 0.01d
|
2.52 ± 0.11a
|
| Direct Bilirubin (mg/dL) |
0.31 ± 0.02 |
0.76 ± 0.01acd
|
0.42 ± 0.01ad
|
1.54 ± 0.01d
|
| ALP (u/L) |
55.66 ± 1.81 |
74.06 ± 1.32ad
|
66.24 ± 1.89ad
|
86.02 ± 3.14a
|
| AST (u/L) |
32.02 ± 1.56 |
46.67 ± 0.16ad
|
48.33 ± 0.07ad
|
65.45 ± 0.09a
|
| ALT (u/L) |
43.13 ± 0.33 |
68.65± 0.41acd
|
75.25 ± 0.07ad
|
26.35 ± 0.11a
|
| Total Protein (g/dL) |
6.64 ± 0.03 |
7.08 ± 0.01acd
|
5.59 ± 0.11ad
|
7.76 ± 0.02a
|
| Albumin (g/L) |
1.18 ± 0.00 |
2.11 ± 0.01acd
|
1.66 ± 0.01ad
|
1.38 ± 0.00a
|
Table 12.
Effect of 2 weeks withdrawal from glue inhalation on renal function parameter.
Table 12.
Effect of 2 weeks withdrawal from glue inhalation on renal function parameter.
| Parameter |
Control (Air) |
Glue (2ml) |
Glue (4ml) |
Glue (8ml) |
| Urea (mmol/L) |
17.77 ± 0.09 |
21.05 ± 1.01 |
24.62 ± 3.90 |
18.73 ± 0.06 |
| Creatinine (µmol/L) |
110.27 ± 0.31 |
167.34 ± 0.54ad
|
181.71 ± 9.78ad
|
136.41 ± 0.31a
|
| Na+ (mmol/L) |
132.00 ± 1.34 |
138.67 ± 1.30ad
|
140.83 ± 0.48ad
|
133.33 ± 1.28 |
| K+ (mmol/L) |
3.80 ± 0.07 |
4.33 ± 0.10ad
|
4.60 ± 0.08a
|
3.32 ± 0.10ac
|
| HCO3 (mmol/L) |
17.53 ± 0.28 |
23.22 ± 0.15ad
|
23.25 ± 0.29ad
|
20.75 ± 0.23 |
Table 13.
Effect of 2 weeks withdrawal from glue inhalation on haematological parameters.
Table 13.
Effect of 2 weeks withdrawal from glue inhalation on haematological parameters.
| Parameters |
Control (Air) |
Glue (2ml) |
Glue (4ml) |
Glue (8ml) |
| White Blood Cell (10^9/L) |
17.13 ± 0.57 |
12.14 ± 1.02a
|
18.73 ± 1.94b
|
14.43 ± 1.14 |
| Red Blood Cell (10^12/L) |
7.69 ± 0.25 |
7.92 ± 0.29 |
7.40 ± 0.17 |
7.86 ± 0.11 |
| Haemoglobin (g/dL) |
14.83 ± 0.28 |
15.27 ± 0.43 |
14.45 ± 0.27 |
15.05 ± 0.23 |
| Haematocrit (%) |
42.55 ±0.48 |
45.67 ± 0.99a
|
41.22 ± 0.52b
|
42.05 ± 0.83b
|
| Platelet (10^9/L) |
494.67 ± 12.40 |
691.83 ± 22.97 |
598.83 ± 114.50 |
813.67 ± 41.17a
|
| Granulocyte (%) |
22.18 ± 0.98 |
28.93 ± 2.21 |
24.37 ± 0.97 |
32.95 ± 4.56a |
| Lymphocyte (%) |
74.73 ± 1.34 |
68.82 ± 2.48 |
66.57 ± 2.10 |
62.18 ± 2.62 |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).