Submitted:
19 August 2023
Posted:
22 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mayer, A.M.; Staples, R.C. Laccase: New functions for an old enzyme. Phytochemistry 2002, 60, 551–565. [Google Scholar] [CrossRef]
- Mot, A.C.; Silaghi-Dumitrescu, R. Laccases: complex architectures for one-electron oxidations. Biochem. Biokhimii͡a 2012, 77, 1395–407. [Google Scholar] [CrossRef]
- Hüttermann, A.; Mai, C.; Kharazipour, A. Modification of lignin for the production of new compounded materials. Appl. Microbiol. Biotechnol. 2001, 55, 387–384. [Google Scholar] [CrossRef]
- Baldrian, P. Fungal laccases – occurrence and properties. FEMS Microbiol. Rev. 2006, 30, 215–242. [Google Scholar] [CrossRef]
- Morsi, R.; Bilal, M.; Iqbal, H.M.N.; Ashraf, S.S. Laccases and peroxidases: The smart, greener and futuristic biocatalytic tools to mitigate recalcitrant emerging pollutants. Sci. Total Environ. 2020, 714, 136572. [Google Scholar] [CrossRef]
- Majeau, J.-A.; Brar, S.K.; Tyagi, R.D. Laccases for removal of recalcitrant and emerging pollutants. Bioresour. Technol. 2010, 101, 2331–2350. [Google Scholar] [CrossRef]
- Sterjiades, R.; Dean, J.F.D.; Eriksson, K.-E.L. Laccase from Sycamore Maple ( Acer pseudoplatanus ) Polymerizes Monolignols. Plant Physiol. 1992, 99, 1162–1168. [Google Scholar] [CrossRef]
- Dean, J.F.D.; LaFayette, P.R.; Rugh, C.; Tristram, A.H.; Hoopes, J.T.; Eriksson, K.-E.L.; Merkle, S.A. Laccases Associated with Lignifying Vascular Tissues. In ACS Symposium Series; 1998; Vol. 697, pp. 96–108. [CrossRef]
- Crestini, C.; Perazzini, R.; Saladino, R. Oxidative functionalisation of lignin by layer-by-layer immobilised laccases and laccase microcapsules. Appl. Catal. A Gen. 2010, 372, 115–123. [Google Scholar] [CrossRef]
- Bar Nun, N.; Tal Lev, A.; Harel, E.; Mayer, A.M. Repression of laccase formation in Botrytis cinerea and its possible relation to phytopathogenicity. Phytochemistry 1988, 27, 2505–2509. [Google Scholar] [CrossRef]
- Worrall, J.J.; Chet, I.; Hutterman, A. Association of Rhizomorph Formation with Laccase Activity in Armillaria spp. Microbiology 1986, 132, 2527–2533. [Google Scholar] [CrossRef]
- Lupan, A.; Matyas, C.; Mot, A.; Silaghi-Dumitrescu, R. Can geometrical distortions make a laccase change color from blue to yellow? Stud. Univ. Babes-Bolyai Chem. 2011, 56, 231–238. [Google Scholar]
- Mot, A.C.; Pârvu, M.; Damian, G.; Irimie, F.D.D.; Darula, Z.; Medzihradszky, K.F.K.F.; Brem, B.; Silaghi-Dumitrescu, R.; Parvu, M.; Damian, G.; et al. A “yellow” laccase with “blue” spectroscopic features, from Sclerotinia sclerotiorum. Process Biochem. 2012, 47, 968–975. [Google Scholar] [CrossRef]
- Mot, A.C.; Coman, C.; Hadade, N.; Damian, G.; Silaghi-Dumitrescu, R.; Heering, H. “Yellow” laccase from Sclerotinia sclerotiorum is a blue laccase that enhances its substrate affinity by forming a reversible tyrosyl-product adduct. PLoS One 2020, 15, e0225530. [Google Scholar] [CrossRef]
- Pozdnyakova, N.N.; Rodakiewicz-Nowak, J.; Turkovskaya, O. V.; Haber, J. Oxidative degradation of polyaromatic hydrocarbons and their derivatives catalyzed directly by the yellow laccase from Pleurotus ostreatus D1. J. Mol. Catal. B Enzym. 2006, 41. [Google Scholar] [CrossRef]
- Pozdnyakova, N. .; Rodakiewicz-Nowak, J.; Turkovskaya, O.. Catalytic properties of yellow laccase from Pleurotus ostreatus D1. J. Mol. Catal. B Enzym. 2004, 30, 19–24. [Google Scholar] [CrossRef]
- Leontievsky, A.; Myasoedova, N.; Pozdnyakova, N.; Golovleva, L. `Yellow’ laccase of Panus tigrinus oxidizes non-phenolic substrates without electron-transfer mediators. FEBS Lett. 1997, 413, 446–448. [Google Scholar] [CrossRef]
- Enguita, F.J.; Martins, L.O.; Henriques, A.O.; Carrondo, M.A. Crystal Structure of a Bacterial Endospore Coat Component. J. Biol. Chem. 2003, 278, 19416–19425. [Google Scholar] [CrossRef]
- Mendonca, R.; Ferraz, A.; Kordsachia, O.; Koch, G. Cellular UV-microspectrophotometric investigations on pine wood (Pinus taeda and Pinus elliottii) delignification during biopulping with Ceriporiopsis subvermispora (Pil�t) Gilbn. & Ryv. and alkaline sulfite/anthraquinone treatment. Wood Sci. Technol. 2004, 38, 567–575. [Google Scholar] [CrossRef]
- Sundberg, A.; Holmbom, B.; Willför, S.; Pranovich, A. Weakening of paper strength by wood resin. Nord. Pulp Pap. Res. J. 2000, 15, 46–53. [Google Scholar] [CrossRef]
- Zhang, X.; Eigendorf, G.; Stebbing, D. .; Mansfield, S..; Saddler, J.. Degradation of trilinolein by laccase enzymes. Arch. Biochem. Biophys. 2002, 405, 44–54. [Google Scholar] [CrossRef]
- Fackler, K.; Kuncinger, T.; Ters, T.; Srebotnik, E. Laccase-catalyzed functionalization with 4-hydroxy-3-methoxybenzylurea significantly improves internal bond of particle boards. Holzforschung 2008, 62, 223–229. [Google Scholar] [CrossRef]
- Tsuchiya, R.; Petersen, B.R.; Christensen, S. Reduction of maladour 2000, 15.
- Zerva, A.; Simić, S.; Topakas, E.; Nikodinovic-Runic, J. Applications of Microbial Laccases: Patent Review of the Past Decade (2009–2019). Catalysts 2019, 9, 1023. [Google Scholar] [CrossRef]
- Kunamneni, A.; Plou, F.; Ballesteros, A.; Alcalde, M. Laccases and Their Applications: A Patent Review. Recent Pat. Biotechnol. 2008, 2, 10–24. [Google Scholar] [CrossRef]
- Raghukumar, C. Fungi from marine habitats: an application in bioremediation. Mycol. Res. 2000, 104, 1222–1226. [Google Scholar] [CrossRef]
- Soares, G.M.B.; Costa-Ferreira, M.; Pessoa de Amorim, M. . Decolorization of an anthraquinone-type dye using a laccase formulation. Bioresour. Technol. 2001, 79, 171–177. [Google Scholar] [CrossRef]
- Osiadacz, J.; Al-Adhami, A.J.H.; Bajraszewska, D.; Fischer, P.; Peczyñska-Czoch, W. On the use of Trametes versicolor laccase for the conversion of 4-methyl-3-hydroxyanthranilic acid to actinocin chromophore. J. Biotechnol. 1999, 72, 141–149. [Google Scholar] [CrossRef]
- Niebisch, C.H.; Malinowski, A.K.; Schadeck, R.; Mitchell, D.A.; Kava-Cordeiro, V.; Paba, J. Decolorization and biodegradation of reactive blue 220 textile dye by Lentinus crinitus extracellular extract. J. Hazard. Mater. 2010, 180, 316–322. [Google Scholar] [CrossRef]
- Larsson, S.; Cassland, P.; Jönsson, L.J. Development of a Saccharomyces cerevisiae Strain with Enhanced Resistance to Phenolic Fermentation Inhibitors in Lignocellulose Hydrolysates by Heterologous Expression of Laccase. Appl. Environ. Microbiol. 2001, 67, 1163–1170. [Google Scholar] [CrossRef]
- Flander, L.; Rouau, X.; Morel, M.-H.; Autio, K.; Seppänen-Laakso, T.; Kruus, K.; Buchert, J. Effects of Laccase and Xylanase on the Chemical and Rheological Properties of Oat and Wheat Doughs. J. Agric. Food Chem. 2008, 56, 5732–5742. [Google Scholar] [CrossRef]
- Selinheimo, E.; Kruus, K.; Buchert, J.; Hopia, A.; Autio, K. Effects of laccase, xylanase and their combination on the rheological properties of wheat doughs. J. Cereal Sci. 2006, 43, 152–159. [Google Scholar] [CrossRef]
- Chavez, M.D.; Schrimsher, J.L.; Morar, A.S. PEGylation of Proteins: A Structural Approach. BioPharm Int. 2006, 19, 04-01–2006. [Google Scholar]
- Portoro, I.; Kocsis, L.; Herman, P.; Caccia, D.; Perrella, M.; Ronda, L.; Bruno, S.; Bettati, S.; Micalella, C.; Mozzarelli, A.; et al. Towards a novel haemoglobin-based oxygen carrier: Euro-PEG-Hb, physico-chemical properties, vasoactivity and renal filtration. Biochim Biophys Acta 2008, 1784, 1402–1409. [Google Scholar] [CrossRef] [PubMed]
- Mot, A.C.; Roman, A.; Lupan, I.; Kurtz Jr., D. M.; Silaghi-Dumitrescu, R. Towards the development of hemerythrin-based blood substitute. Protein J. 2010, 29, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Nojima, Y.; Iguchi, K.; Suzuki, Y.; Sato, A. The pH-Dependent Formation of PEGylated Bovine Lactoferrin by Branched Polyethylene Glycol (PEG)-N-Hydroxysuccinimide (NHS) Active Esters. Biol. Pharm. Bull. 2009, 32, 523–526. [Google Scholar] [CrossRef] [PubMed]
- Nojima, Y.; Suzuki, Y.; Iguchi, K.; Shiga, T.; Iwata, A.; Fujimoto, T.; Yoshida, K.; Shimizu, H.; Takeuchi, T.; Sato, A. Development of Poly(ethylene glycol) Conjugated Lactoferrin for Oral Administration. Bioconjug. Chem. 2008, 19, 2253–2259. [Google Scholar] [CrossRef]
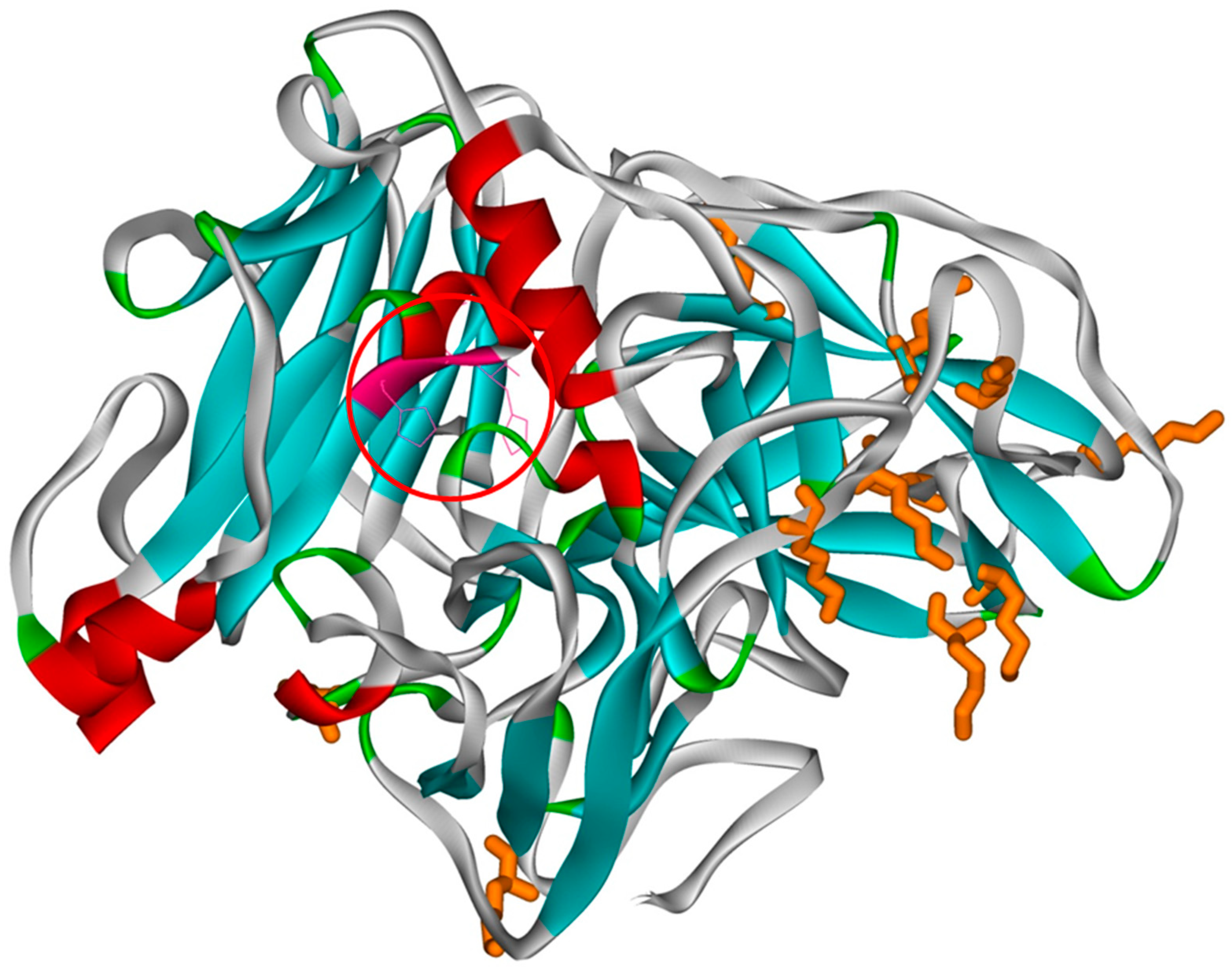
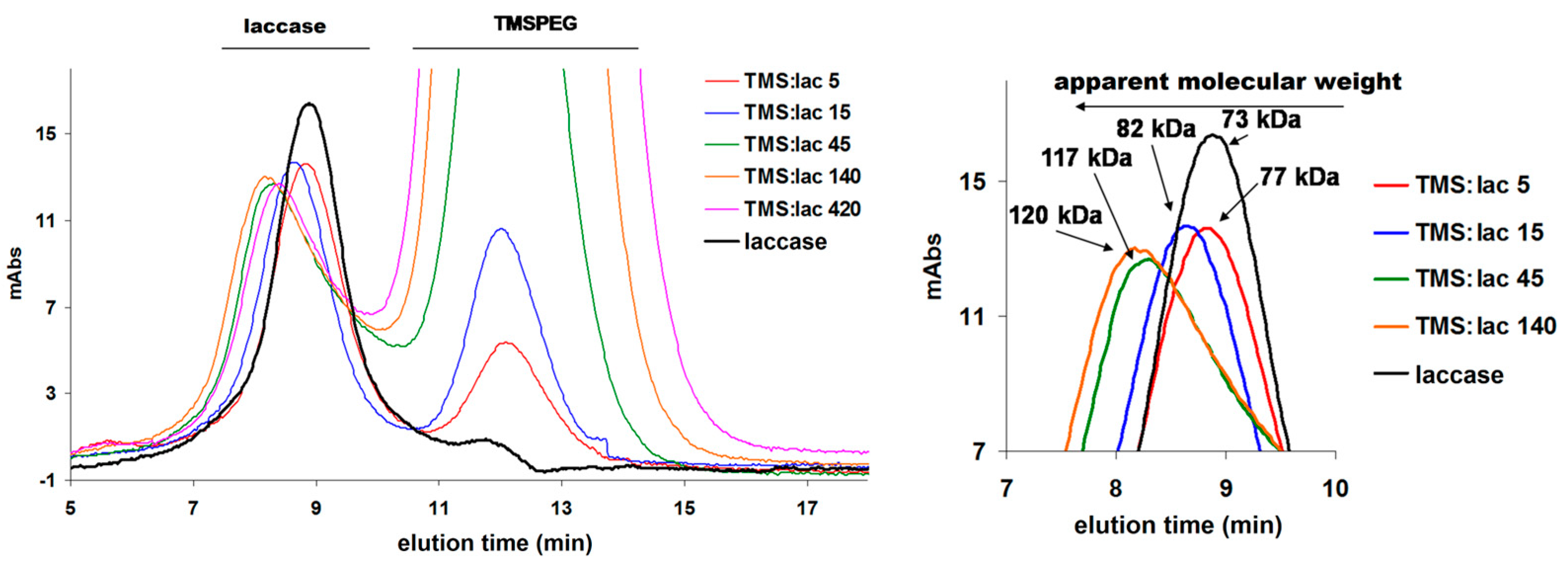
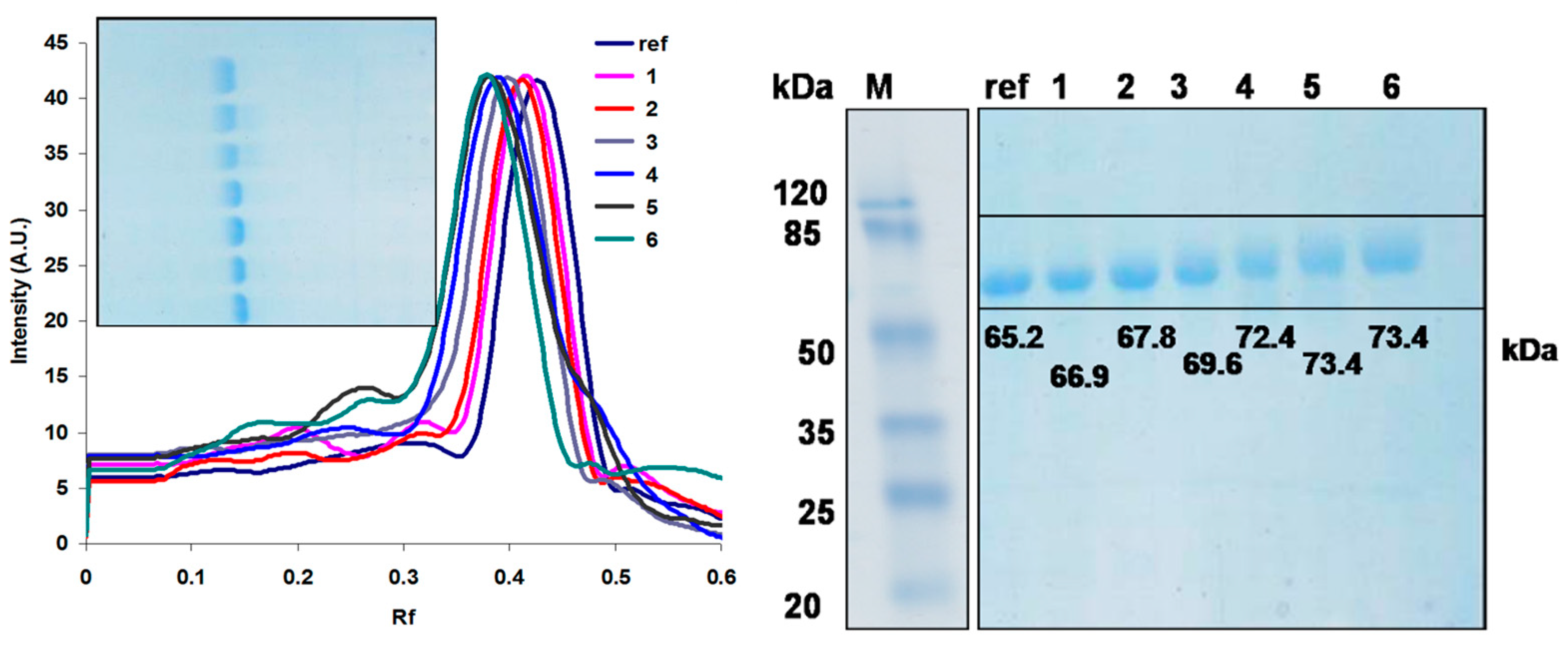
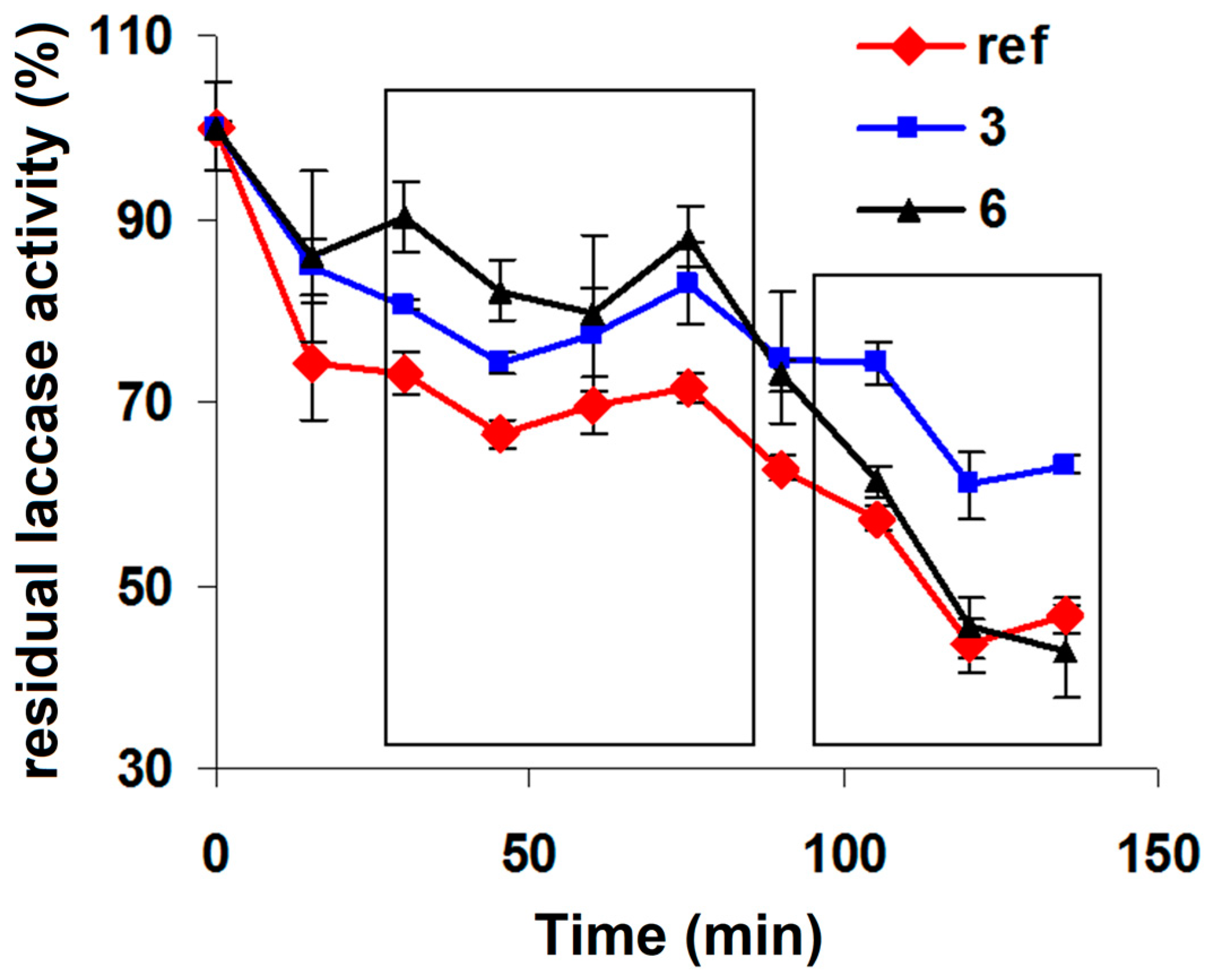
| 1 | 2 | 3 | 4 | 5 | 6 | |
| laccase (µM) | 10 | 10 | 10 | 10 | 10 | 10 |
| [TMSPEG] (µM) | 10 | 50 | 150 | 450 | 1400 | 4200 |
| ratio | 1 | 5 | 15 | 45 | 140 | 420 |
| Rf | Sample | log(MW) | M (kDa) | Excess weight (kDa) | Number of PEG units |
| 0.29 | laccase | 1.814 | 65.21 | ||
| 0.28 | 1 | 1.826 | 66.94 | 1.73 | 0.72 |
| 0.27 | 2 | 1.831 | 67.82 | 2.62 | 1.09 |
| 0.26 | 3 | 1.843 | 69.63 | 4.42 | 1.84 |
| 0.25 | 4 | 1.860 | 72.42 | 7.21 | 3.00 |
| 0.24 | 5 | 1.866 | 73.37 | 8.16 | 3.40 |
| 0.24 | 6 | 1.866 | 73.37 | 8.16 | 3.40 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




