Submitted:
18 August 2023
Posted:
18 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
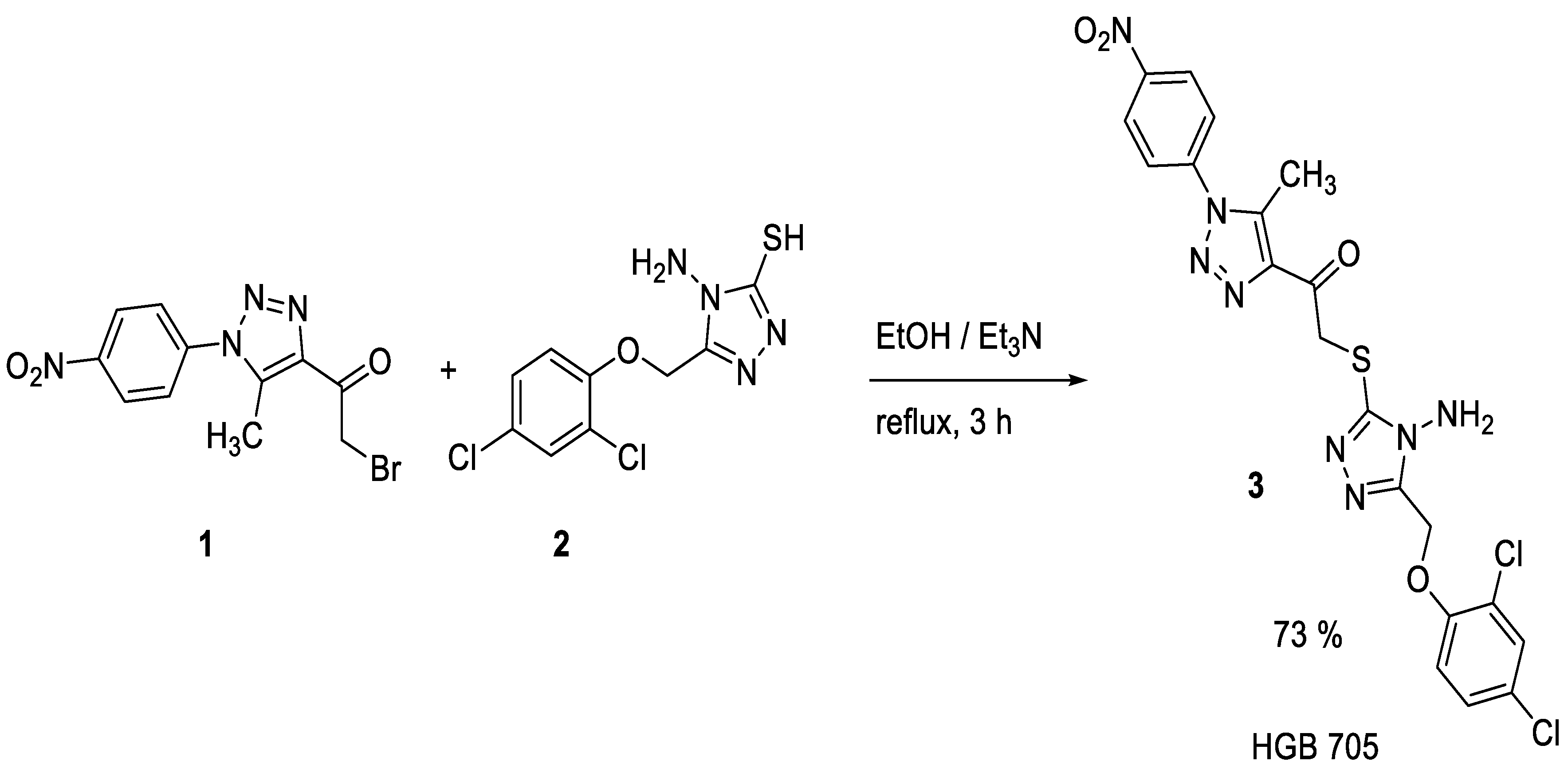
2.1. Synthesis
2.2. NMR Spectroscopy
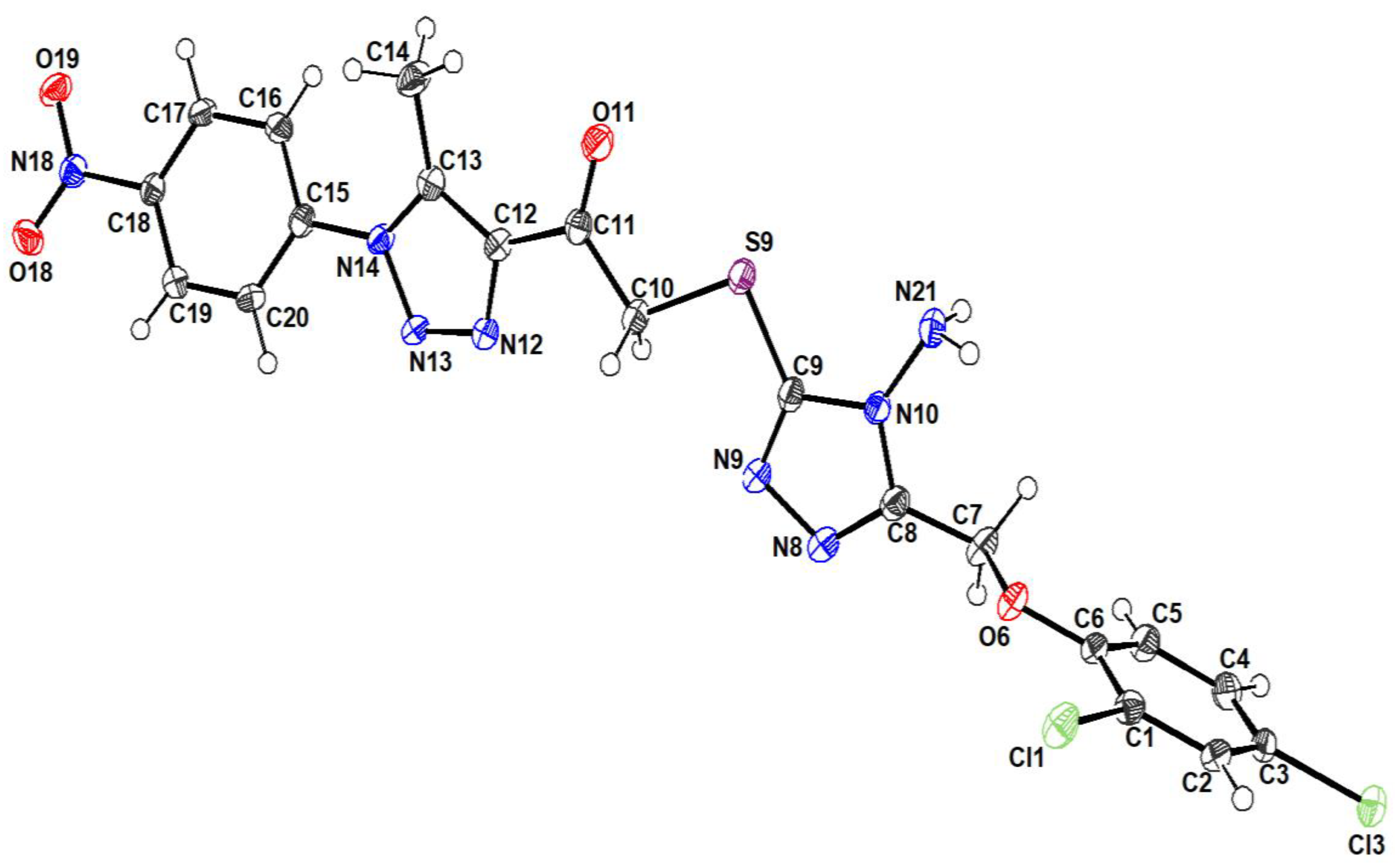
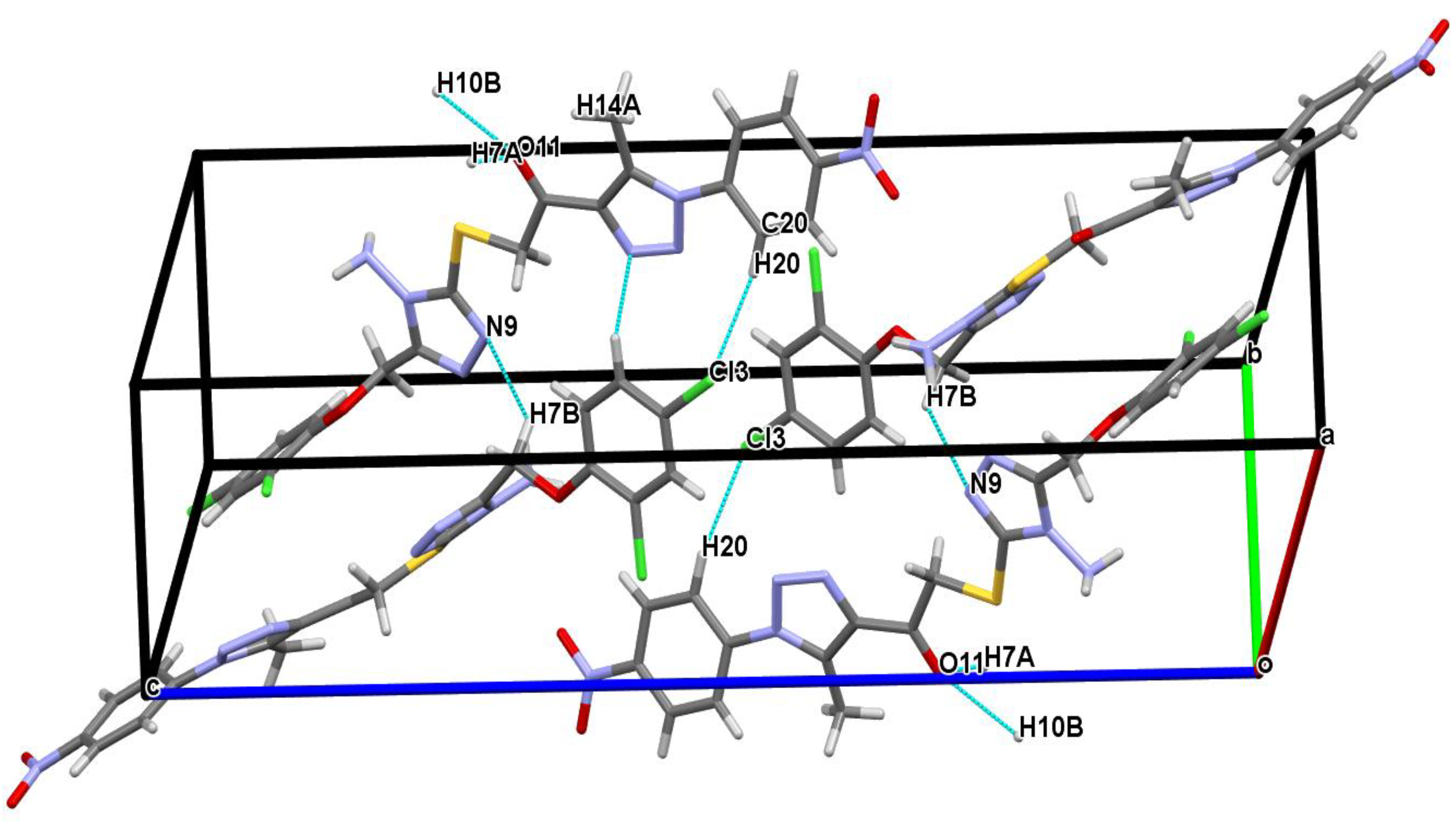
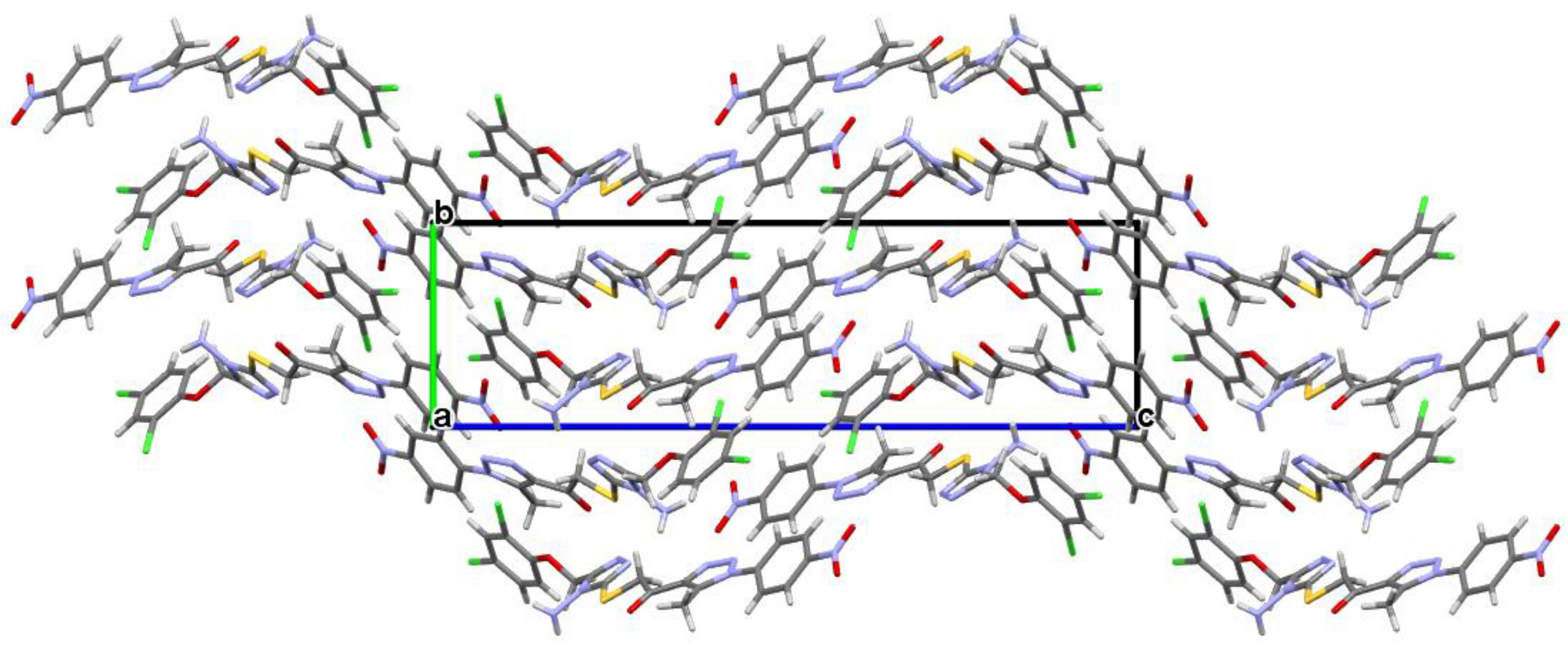
2.3. Crystal Structure Analysis
3. Materials and Methods
3.1. General
3.2. Synthesis of 3
3.3. X-ray Crystallography
4. Conclusions
Supplementary Materials
Author Contributions
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Liang, T.; Sun, X.; Li, W.; Hou, G.; Gao, F. 1,2,3-Triazole-Containing Compounds as Anti–Lung Cancer Agents: Current Developments, Mechanisms of Action, and Structure–Activity Relationship. Front. Pharmacol. 2021, 12, 661173. [CrossRef]
- Bozorov, K.; Zhao, J.; Aisa, H.A. 1,2,3-Triazole-containing hybrids as leads in medicinal chemistry: A recent overview. Bioorg Med Chem. 2019, 27, 3511-3531. [CrossRef]
- Khan, S.A.; Akhtar, M.J.; Gogoi, U.; Meenakshi, D.U.; Das, A. An Overview of 1,2,3-triazole-Containing Hybrids and Their Potential Anticholinesterase Activities. Pharmaceuticals 2023, 16, 179. [CrossRef]
- Kumar, S.; Khokra, S.L.; Yadav, A. Triazole analogues as potential pharmacological agents: a brief review. Futur J. Pharm. Sci. 2021, 7, 106. [CrossRef]
- H. A. Mohamed, M. S. Bekheit, E. F. Ewies, H. M. Awad, R. Betz, E. C. Hosten, B. F. Abdel-Wahab,Design of new hybrids indole/phthalimide/oxadiazole-1,2,3 triazole agents and their anticancer properties,Journal of Molecular Structure, Volume 1274, 2023, 134415. [CrossRef]
- Abdelli, S. Azzouni, R. Plais, A. Gaucher, M. L. Efrit, D. Prim, Recent advances in the chemistry of 1,2,4-triazoles: Synthesis, reactivity and biological activities, Tetrahedron Letters, Volume 86, 2021, 153518. [CrossRef]
- Yi-Nan Cheng, Zhen-Hua Jiang, Lian-Sheng Sun, Zi-Yang Su, Meng-Meng Zhang, Hong-Lian Li, Synthesis of 1,2,4-triazole benzoyl arylamine derivatives and their high antifungal activities, European Journal of Medicinal Chemistry, Volume 200, 2020, 112463. [CrossRef]
- Dai, J.; Tian, S.; Yang, X.; Liu, Z. Synthesis methods of 1,2,3-/1,2,4-triazoles: A review. Front. Chem., 2022, 10. [CrossRef]
- Kumari, M.; Tahlan, S.; Narasimhan, B. et al. Synthesis and biological evaluation of heterocyclic 1,2,4-triazole scaffolds as promising pharmacological agents. BMC Chemistry 2022, 15, 5. [CrossRef]
- Abdel-Wahab, B. F.; Mohamed, H. A.; Farahat, A. A.; Kariuki, B. M.; El-Hiti, G. A. Reactivity of 4-bromoacetyl-1,2,3-triazoles towards amines and phenols: synthesis and antimicrobial activity of novel heterocycles. Heterocycles 2022, 104, 1601–1613. [CrossRef]
- Abdel-Wahab, B.F.; Farahat, A.A.; Kariuki, B.M.; El-Hiti, G.A. (E)-1-(5-Methyl-1-(4-nitrophenyl)-1H-1,2,3-triazol-4-yl)ethan-1-one Oxime. Molbank 2023, 2023, M1593. [CrossRef]
- Kariuki, B.M.; Abdel-Wahab, B.F.; Farahat, A.A.; El-Hiti, G.A. Synthesis and structure determination of 1-(4-methoxyphenyl)-5-methyl-N’-(2-oxoindolin-3-ylidene)-1H-1,2,3-triazole-4-carbohydrazide. Molbank 2022, 2022, M1374. [CrossRef]
- Abdel-Wahab, B.F.; Mabied, A.F.; Fettinger, J.C.; Hassan, A.H.E.; Farahat, A.A. 2-((5-(5-Methyl-2-phenyl-1H-imidazol-4-yl)-1,3,4-oxadiazol-2-yl)thio)-1-phenylethan-1-one. Molbank 2023, 2023, M1666. [CrossRef]
- Orpen, A.G.; Brammer, L.; Allen, F.H.; Kennard, O.; Watson, D.G.; Taylor, R. Supplement. Tables of bond lengths determined by X-ray and neutron diffraction. Part 2. Organometallic compounds and co-ordination complexes of the d- and f-block metals. J. Chem. Soc. Dalton Trans. 1989, 12, S1–S83. [CrossRef]
- Bunev, A. S.; Trushkova, Yu. O.; Ostapenko, G. I.; Statsyuk, V. E.; Peregudov, A. S. Synthesis of 4-(1H-1,2,3-triazol-4-yl)-1,3-thiazole-2-amine derivatives. Chem. Heterocycl. Cpds. 2014, 50, 1027-1031. [CrossRef]
- Bano, Q.; Tiwari, N.; Giri, S.; Nizamuddin; Synthesis and fungicidal activities of 3-(aryloxymethyl)-6-substistuted 1,2,4- triazolo[3,4-b]-1,3,4- thiadiazoles. Indian J. Chem. 1992, 31B, 714-18.
- Bruker. APEX III; Bruker AXS Inc.: Madison, WI, USA, 2019.
- Sheldrick, G.M. A short history of SHELX. Acta Cryst. 2008, A64, 112–122. [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [CrossRef]
- Spek, A.L. Structure Validation in Chemical Crystallography. Acta Cryst. 2009, D65, 148–155. [CrossRef]
- Macrae, C.F.; Sovago, I.; Cottrell, S.J.; Galek, P.T.A.; McCabe, P.; Pidcock, E.; Platings, M.; Shields, G.P.; Stevens, J.S.; Towler, M.; et al. Mercury 4.0: From visualization to analysis, design and prediction. J. Appl. Crystallogr. 2020, 53, 226–235.
- Farrugia, L.J.WinGX and ORTEP for Windows: An update. J. Appl. Cryst. 2012, 45, 849–854.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



