Submitted:
15 August 2023
Posted:
17 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Dataset
2.2. Segmentation and Feature Extraction
2.3. Feature Selection
2.4. Modeling and Statistical Analysis
3. Results
3.1. Dataset
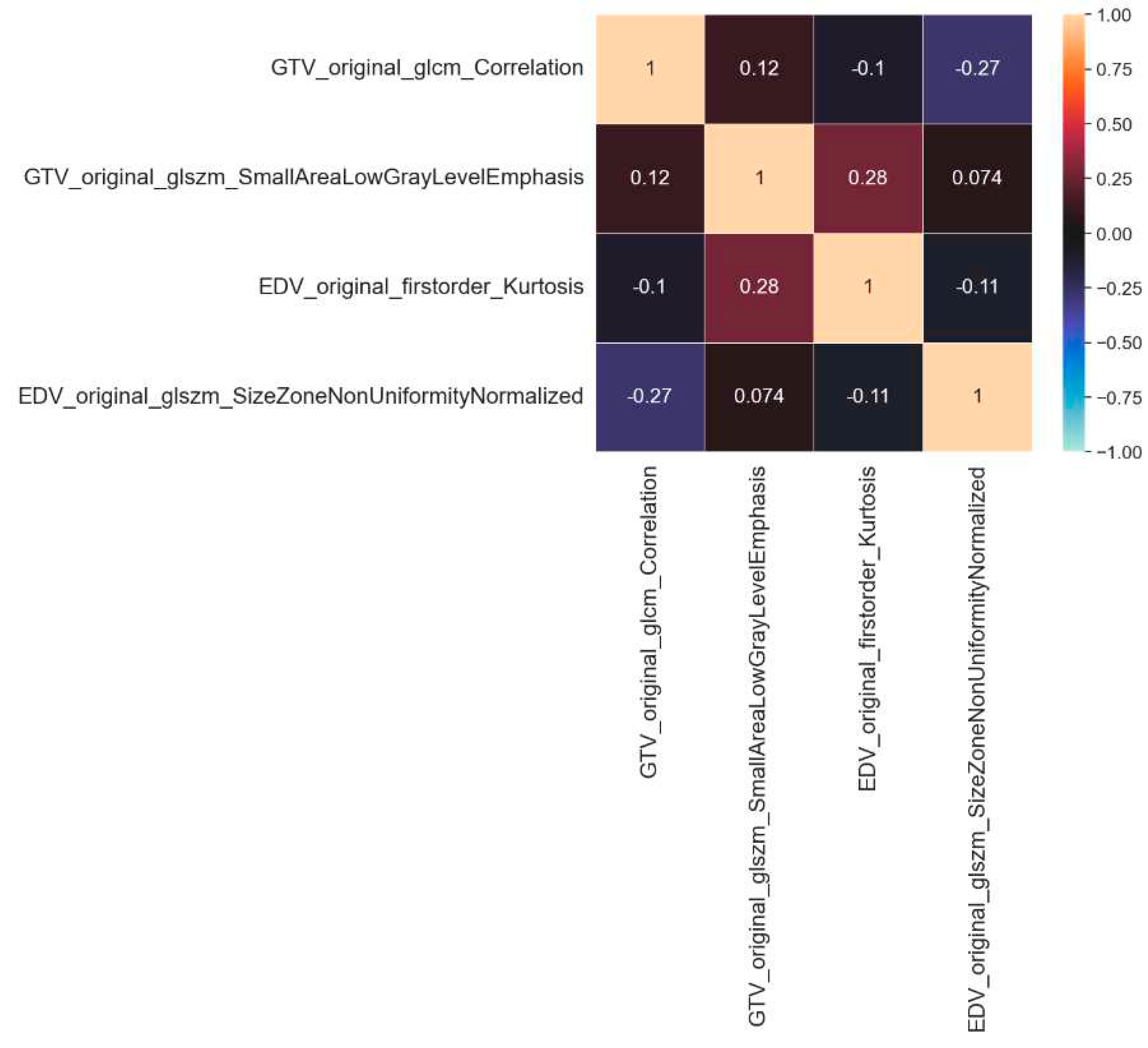
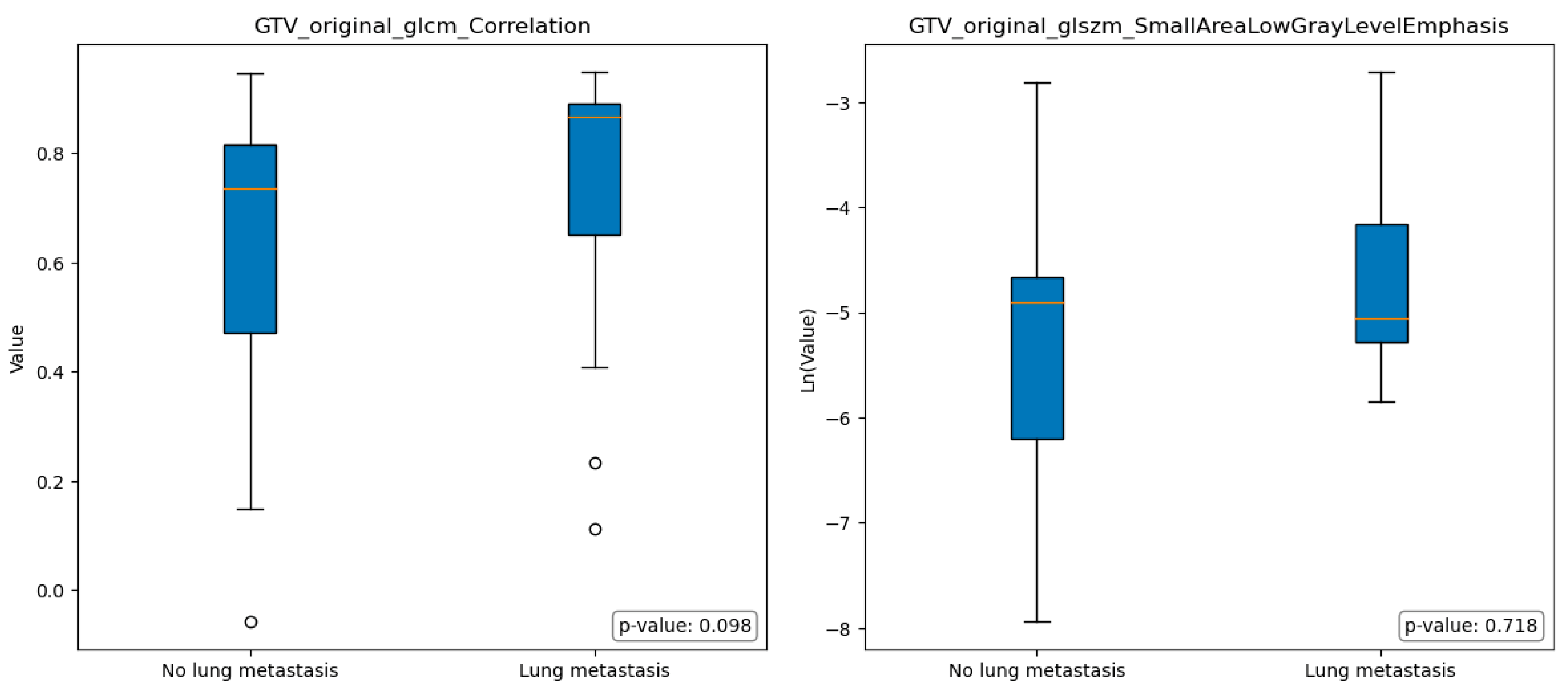
3.2. Features Extraction and Selection
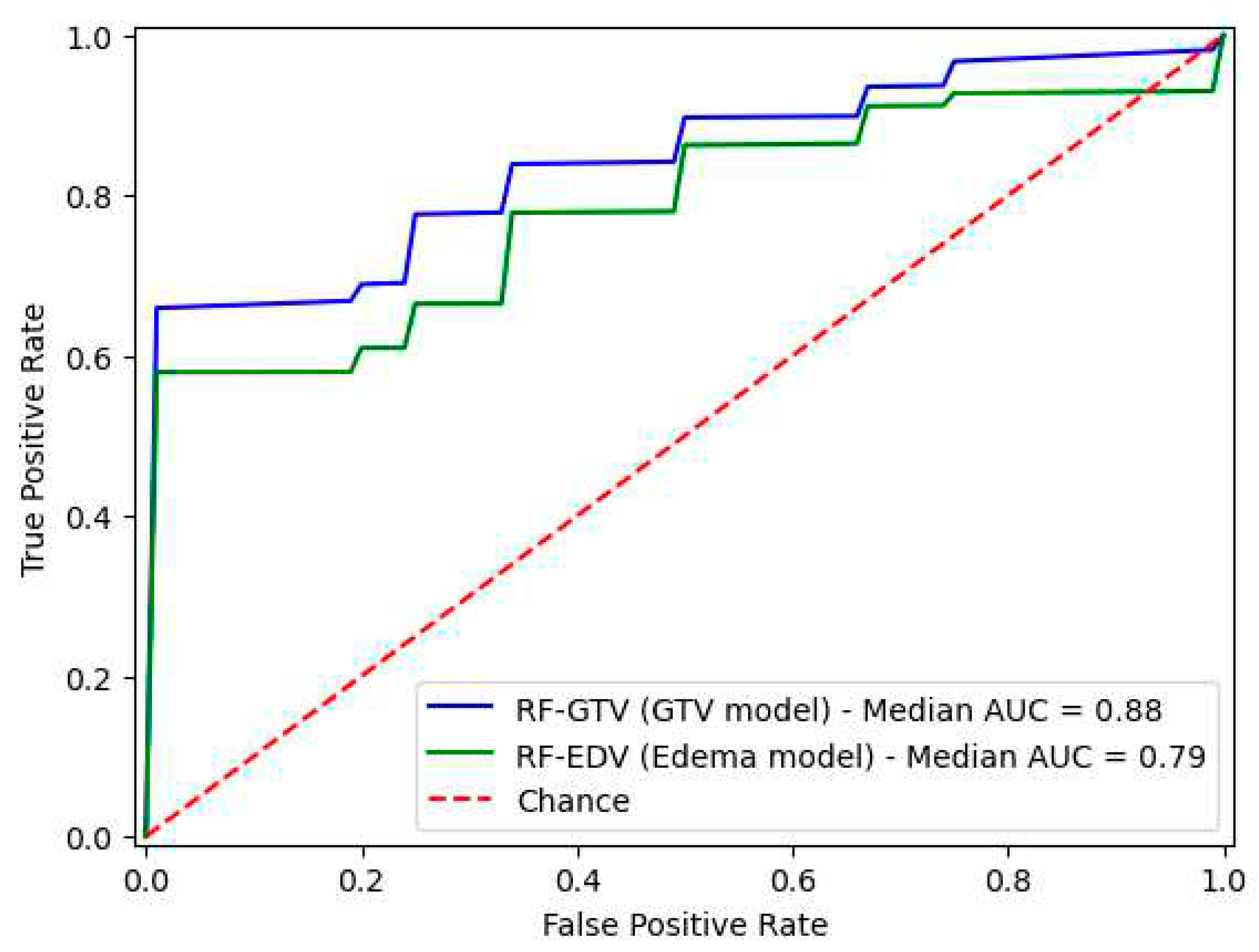
3.3. Classification Performance
4. Discussion
5. Conclusions
Data Availability Statement
Abbreviations
References
- Kransdorf, MJ. Malignant soft-tissue tumors in a large referral population: distribution of diagnoses by age, sex, and location. AJR Am J Roentgenol. 1995, 164, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Billingsley KG, Lewis JJ, Leung DH, Casper ES, Woodruff JM, Brennan MF. Multifactorial analysis of the survival of patients with distant metastasis arising from primary extremity sarcoma. Cancer. 1999, 85, 389–395. [Google Scholar] [CrossRef]
- Brennan, MF. Soft tissue sarcoma: advances in understanding and management. Surgeon. 2005, 3, 216–223. [Google Scholar] [CrossRef]
- Stojadinovic A, Leung DHY, Hoos A, Jaques DP, Lewis JJ, Brennan MF. Analysis of the prognostic significance of microscopic margins in 2,084 localized primary adult soft tissue sarcomas. Ann Surg. 2002, 235, 424–434. [Google Scholar] [CrossRef] [PubMed]
- Lewis JJ, Brennan MF. Soft tissue sarcomas. Curr Probl Surg. 1996, 33, 817–872. [Google Scholar]
- Komdeur R, Hoekstra HJ, van den Berg E, Molenaar WM, Pras E, de Vries EGE, et al. Metastasis in soft tissue sarcomas: prognostic criteria and treatment perspectives. Cancer Metastasis Rev. 2002, 21, 167–183. [Google Scholar] [CrossRef]
- Vallières M, Freeman CR, Skamene SR, El Naqa I. A radiomics model from joint FDG-PET and MRI texture features for the prediction of lung metastases in soft-tissue sarcomas of the extremities. Phys Med Biol. 2015, 60, 5471–5496. [Google Scholar] [CrossRef]
- White LM, Wunder JS, Bell RS, O’Sullivan B, Catton C, Ferguson P, et al. Histologic assessment of peritumoral edema in soft tissue sarcoma. Int J Radiat Oncol Biol Phys. 2005, 61, 1439–1445. [Google Scholar] [CrossRef]
- Clark K, Vendt B, Smith K, Freymann J, Kirby J, Koppel P, et al. The Cancer Imaging Archive (TCIA): maintaining and operating a public information repository. J Digit Imaging. 2013, 26, 1045–1057. [Google Scholar] [CrossRef]
- Zhao W, Huang X, Wang G, Guo J. PET/MR fusion texture analysis for the clinical outcome prediction in soft-tissue sarcoma. Cancer Imaging. 2022, 22, 7. [Google Scholar] [CrossRef]
- Fedorov A, Beichel R, Kalpathy-Cramer J, Finet J, Fillion-Robin J-C, Pujol S, et al. 3D Slicer as an image computing platform for the Quantitative Imaging Network. Magn Reson Imaging. 2012, 30, 1323–1341. [Google Scholar] [CrossRef] [PubMed]
- van Griethuysen JJM, Fedorov A, Parmar C, Hosny A, Aucoin N, Narayan V, et al. Computational Radiomics System to Decode the Radiographic Phenotype. Cancer Res. 2017, 77, e104–e107. [Google Scholar] [CrossRef] [PubMed]
- Zwanenburg A, Vallières M, Abdalah MA, Aerts HJWL, Andrearczyk V, Apte A, et al. The Image Biomarker Standardization Initiative: Standardized Quantitative Radiomics for High-Throughput Image-based Phenotyping. Radiology. 2020, 295, 328–338. [Google Scholar] [CrossRef]
- Raschka, S. MLxtend: Providing machine learning and data science utilities and extensions to Python’s scientific computing stack. J Open Source Softw. 2018, 3, 638. [Google Scholar] [CrossRef]
- Nadeau C, Bengio Y. Inference for the Generalization Error. Mach Learn. 2003, 52, 239–281. [Google Scholar] [CrossRef]
- Bouckaert RR, Frank E. Evaluating the replicability of significance tests for comparing learning algorithms. Advances in Knowledge Discovery and Data Mining. Berlin, Heidelberg: Springer Berlin Heidelberg, 2004; pp. 3–12.
- Zhao F, Ahlawat S, Farahani SJ, Weber KL, Montgomery EA, Carrino JA, et al. Can MR imaging be used to predict tumor grade in soft-tissue sarcoma? Radiology. 2014, 272, 192–201. [CrossRef]
- Lefkowitz RA, Landa J, Hwang S, Zabor EC, Moskowitz CS, Agaram NP, et al. Myxofibrosarcoma: prevalence and diagnostic value of the “tail sign” on magnetic resonance imaging. Skeletal Radiol. 2013, 42, 809–818. [Google Scholar] [CrossRef]
- Yoo HJ, Hong SH, Kang Y, Choi J-Y, Moon KC, Kim H-S, et al. MR imaging of myxofibrosarcoma and undifferentiated sarcoma with emphasis on tail sign; diagnostic and prognostic value. Eur Radiol. 2014, 24, 1749–1757. [Google Scholar] [CrossRef]
- Trojani M, Contesso G, Coindre JM, Rouesse J, Bui NB, de Mascarel A, et al. Soft-tissue sarcomas of adults; study of pathological prognostic variables and definition of a histopathological grading system. Int J Cancer. 1984, 33, 37–42. [Google Scholar] [CrossRef]
- Fernebro J, Wiklund M, Jonsson K, Bendahl P-O, Rydholm A, Nilbert M, et al. Focus on the tumour periphery in MRI evaluation of soft tissue sarcoma: infiltrative growth signifies poor prognosis. Sarcoma. 2006, 2006, 21251. [Google Scholar]
- Crombé A, Le Loarer F, Stoeckle E, Cousin S, Michot A, Italiano A, et al. MRI assessment of surrounding tissues in soft-tissue sarcoma during neoadjuvant chemotherapy can help predicting response and prognosis. Eur J Radiol. 2018, 109, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Nakamura T, Matsumine A, Matsubara T, Asanuma K, Yada Y, Hagi T, et al. Infiltrative tumor growth patterns on magnetic resonance imaging associated with systemic inflammation and oncological outcome in patients with high-grade soft-tissue sarcoma. PLoS One. 2017, 12, e0181787. [Google Scholar]
- Fadli D, Kind M, Michot A, Le Loarer F, Crombé A. Natural Changes in Radiological and Radiomics Features on MRIs of Soft-Tissue Sarcomas Naïve of Treatment: Correlations With Histology and Patients’ Outcomes. J Magn Reson Imaging. 2022, 56, 77–96. [Google Scholar] [CrossRef]
- Sedaghat S, Schmitz F, Meschede J, Sedaghat M. Systematic analysis of post-treatment soft-tissue edema and seroma on MRI in 177 sarcoma patients. Surg Oncol. 2020, 35, 218–223. [Google Scholar] [CrossRef]
- Crombé A, Périer C, Kind M, De Senneville BD, Le Loarer F, Italiano A, et al. T -based MRI Delta-radiomics improve response prediction in soft-tissue sarcomas treated by neoadjuvant chemotherapy. J Magn Reson Imaging. 2019, 50, 497–510. [Google Scholar] [CrossRef] [PubMed]
- Tsagozis P, Brosjö O, Skorpil M. Preoperative radiotherapy of soft-tissue sarcomas: surgical and radiologic parameters associated with local control and survival. Clin Sarcoma Res. 2018, 8, 19. [Google Scholar] [CrossRef] [PubMed]
- Wang L, Pretell-Mazzini J, Kerr DA, Chelala L, Yang X, Jose J, et al. MRI findings associated with microscopic residual tumor following unplanned excision of soft tissue sarcomas in the extremities. Skeletal Radiol. 2018, 47, 181–190. [Google Scholar] [CrossRef]
- Bahig H, Roberge D, Bosch W, Levin W, Petersen I, Haddock M, et al. Agreement among RTOG sarcoma radiation oncologists in contouring suspicious peritumoral edema for preoperative radiation therapy of soft tissue sarcoma of the extremity. Int J Radiat Oncol Biol Phys. 2013, 86, 298–303. [Google Scholar] [CrossRef]
- Duy Hung N, Tam N-T, Khanh Huyen D, Thi N-V, Minh Duc N. Diagnostic performance of magnetic resonance imaging in discriminating benign and malignant soft tissue tumors. Int J Gen Med. 2023, 16, 1383–1391. [Google Scholar] [CrossRef]
- Crombé A, Bertolo F, Fadli D, Kind M, Le Loarer F, Perret R, et al. Distinct patterns of the natural evolution of soft tissue sarcomas on pre-treatment MRIs captured with delta-radiomics correlate with gene expression profiles. Eur Radiol. 2023, 33, 1205–1218. [Google Scholar]
- Chen W, Samuelson FW, Gallas BD, Kang L, Sahiner B, Petrick N. On the assessment of the added value of new predictive biomarkers. BMC Med Res Methodol. 2013, 13, 98. [Google Scholar]
- Kang L, Chen W, Petrick NA, Gallas BD. Comparing two correlated C indices with right-censored survival outcome: a one-shot nonparametric approach. Stat Med. 2015, 34, 685–703. [Google Scholar] [CrossRef]
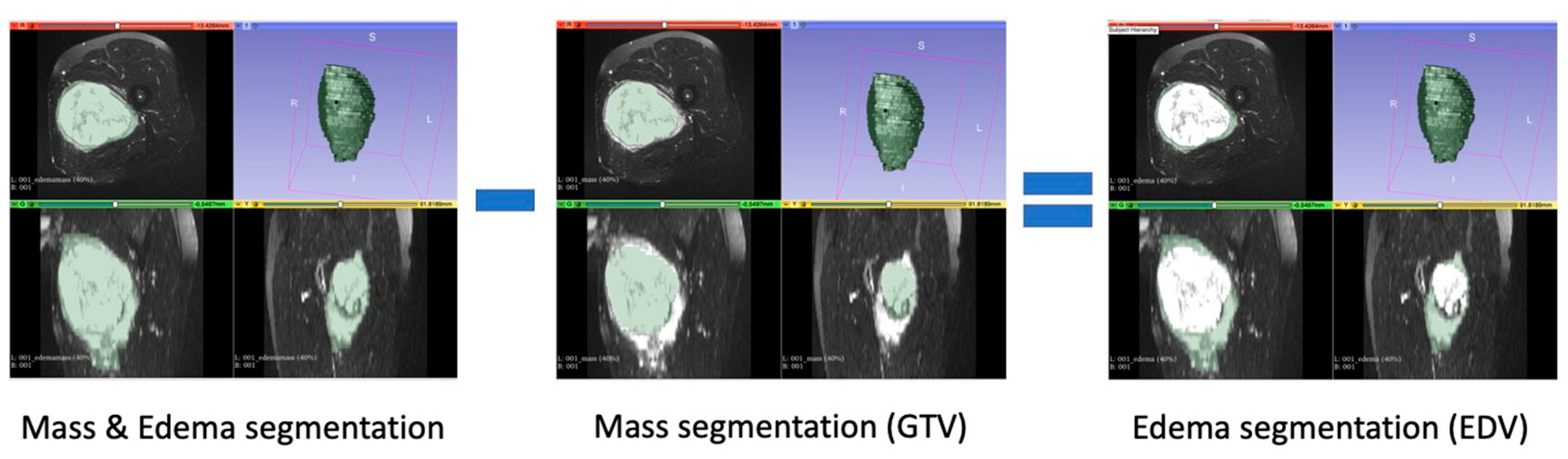
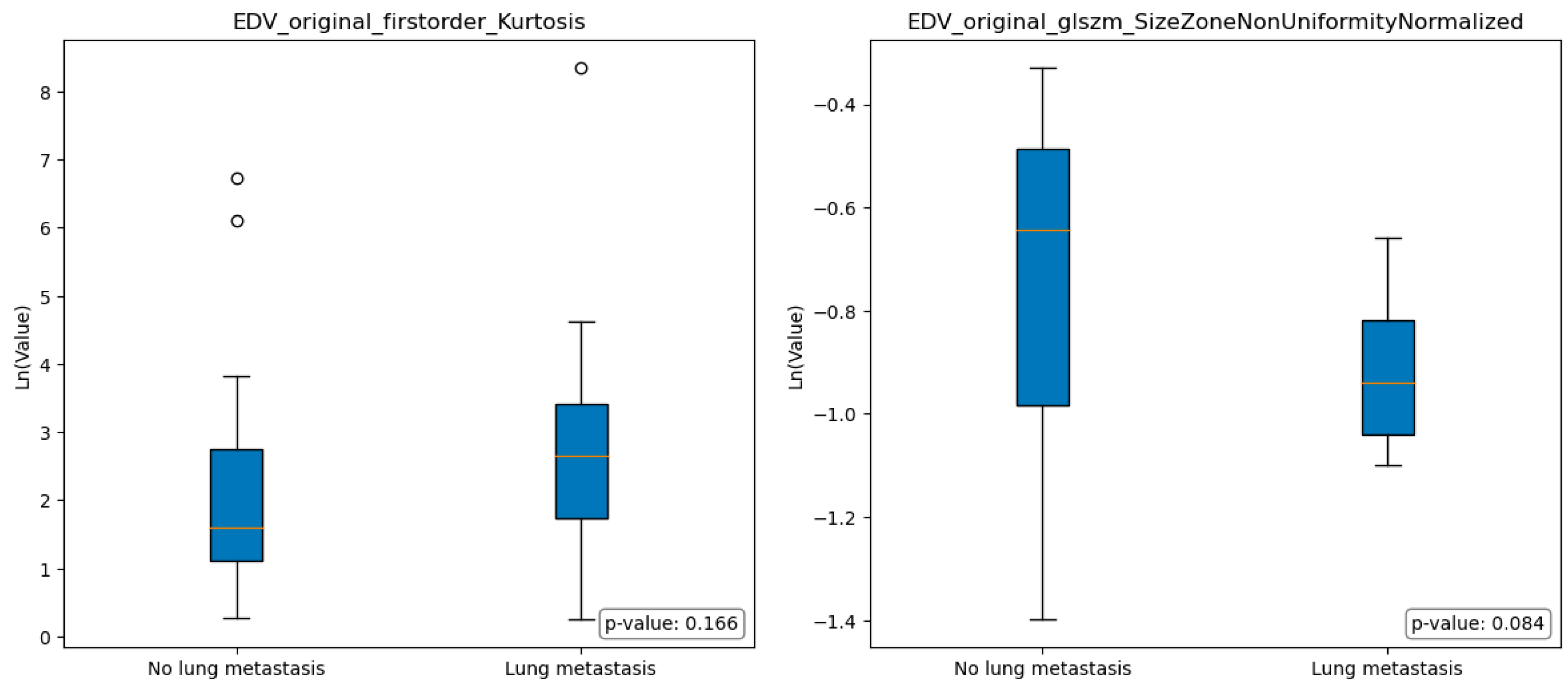
| Group A (No lung metastasis) | Group B (Lung metastasis) | |
|---|---|---|
| Number of patients | 18 | 14 |
| Gender ratio (M/F) | 5/13 | 9/5 |
| Age, y, median (range) | 53.5 (16-83) | 62.5 (44-74) |
| Grade ratio (Low/Intermediate/High) | 1/9/8 | 0/4/10 |
| Selected features | |
|---|---|
| Gross Tumor Volume (GTV) | Edema Tumor Volume (EDV) |
| original_glcm_Correlation | original_firstorder_Kurtosis |
| original_glszm_SmallAreaLowGrayLevelEmphasis | original_glszm_SizeZoneNonUniformityNormalized |
| RF-GTV median [interquartile range] | RF-EDV median [interquartile range] | |
|---|---|---|
| Accuracy | 0.83 [0.17] | 0.75 [0.17] |
| Sensitivity | 0.67 [0.50] | 0.67 [0.50] |
| Specificity | 1.00 [0.33] | 0.80 [0.33] |
| AUC | 0.88 [0.23] | 0.79 [0.38] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




