Submitted:
12 August 2023
Posted:
14 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
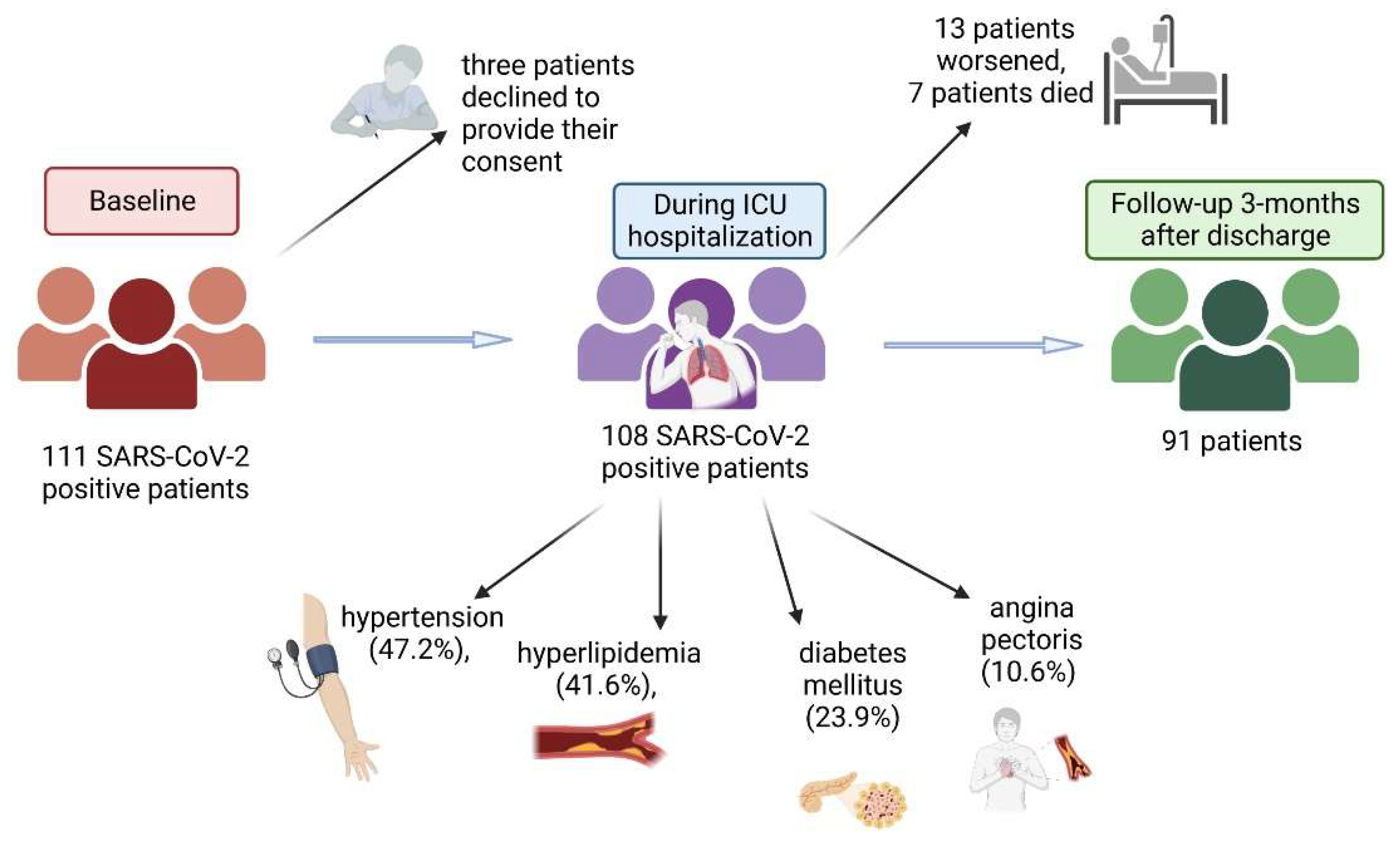
2.1. Patients and Data Extraction
2.2. Study Endpoints and Follow-Up
2.3. Blood Sampling and Antiphospholipid Antibody Measurment
2.4. Statistical Analyses
3. Results
3.1. Patient Baseline Characteristics
3.2. Laboratory Values at 4-Time Points
3.3. Specific Subgroups of Patients
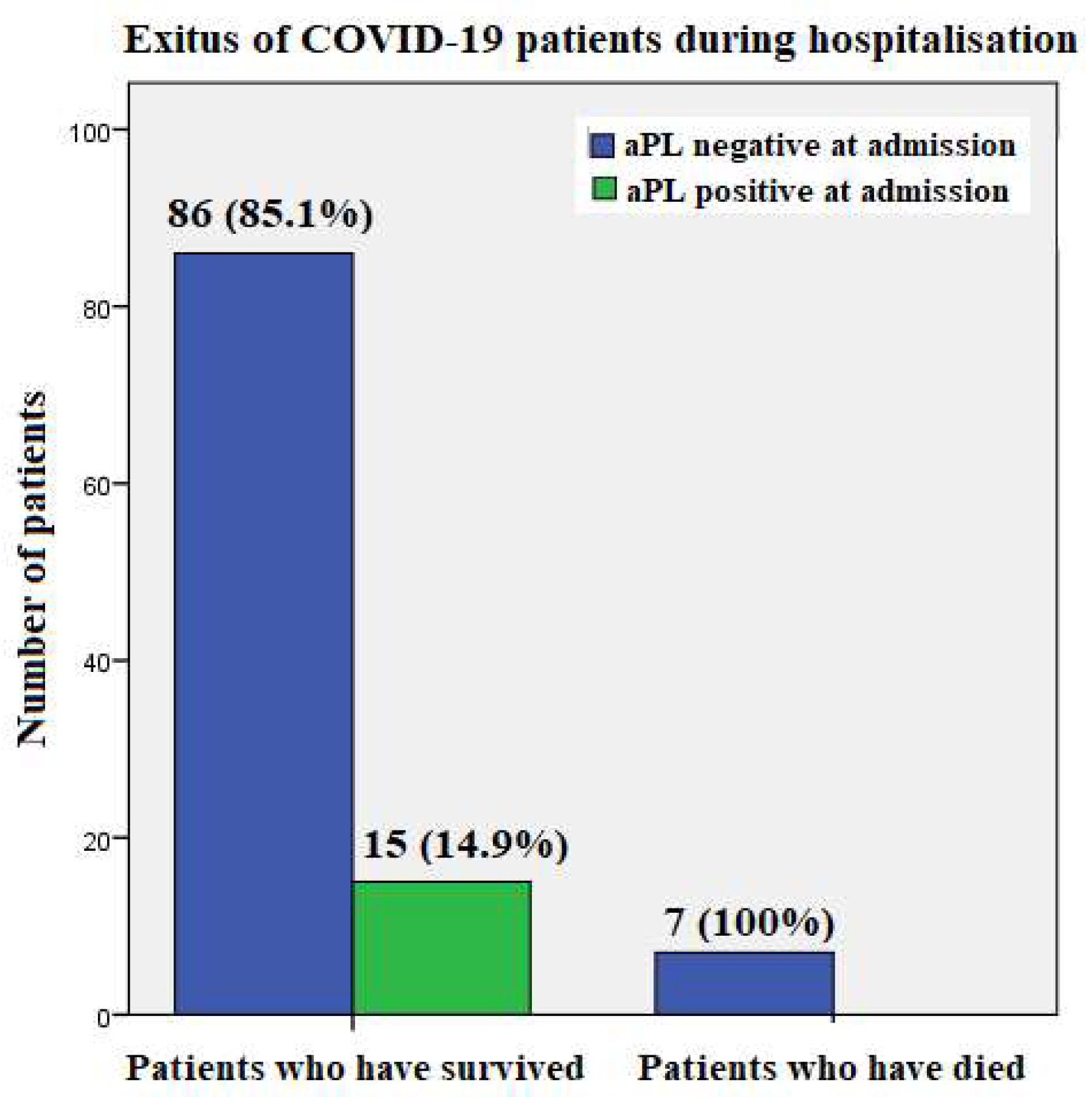
3.3.1. Patients Who Died during Hospitalization
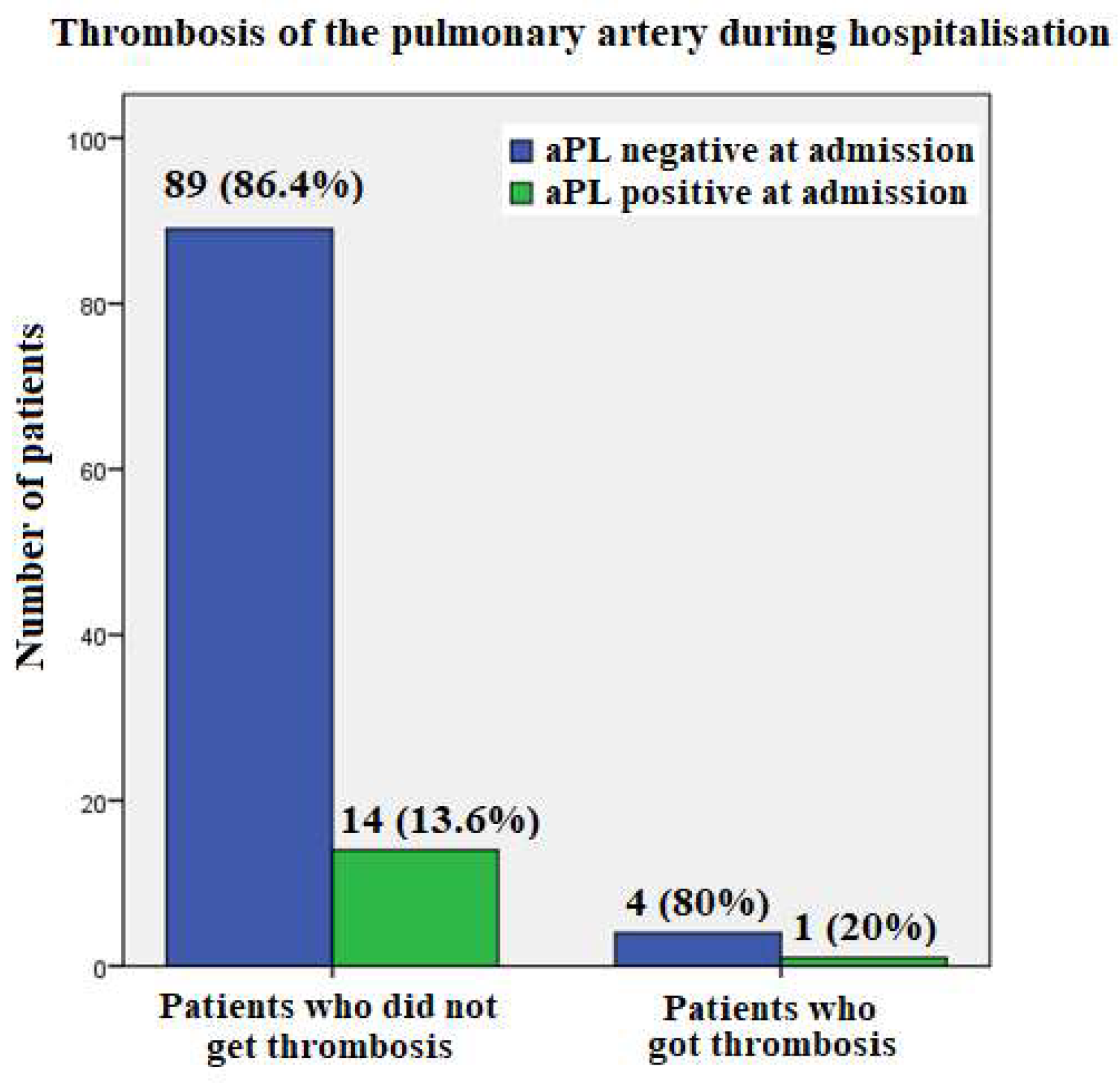
3.3.2. Patients Who Have Experienced Thrombosis during Hospitalization
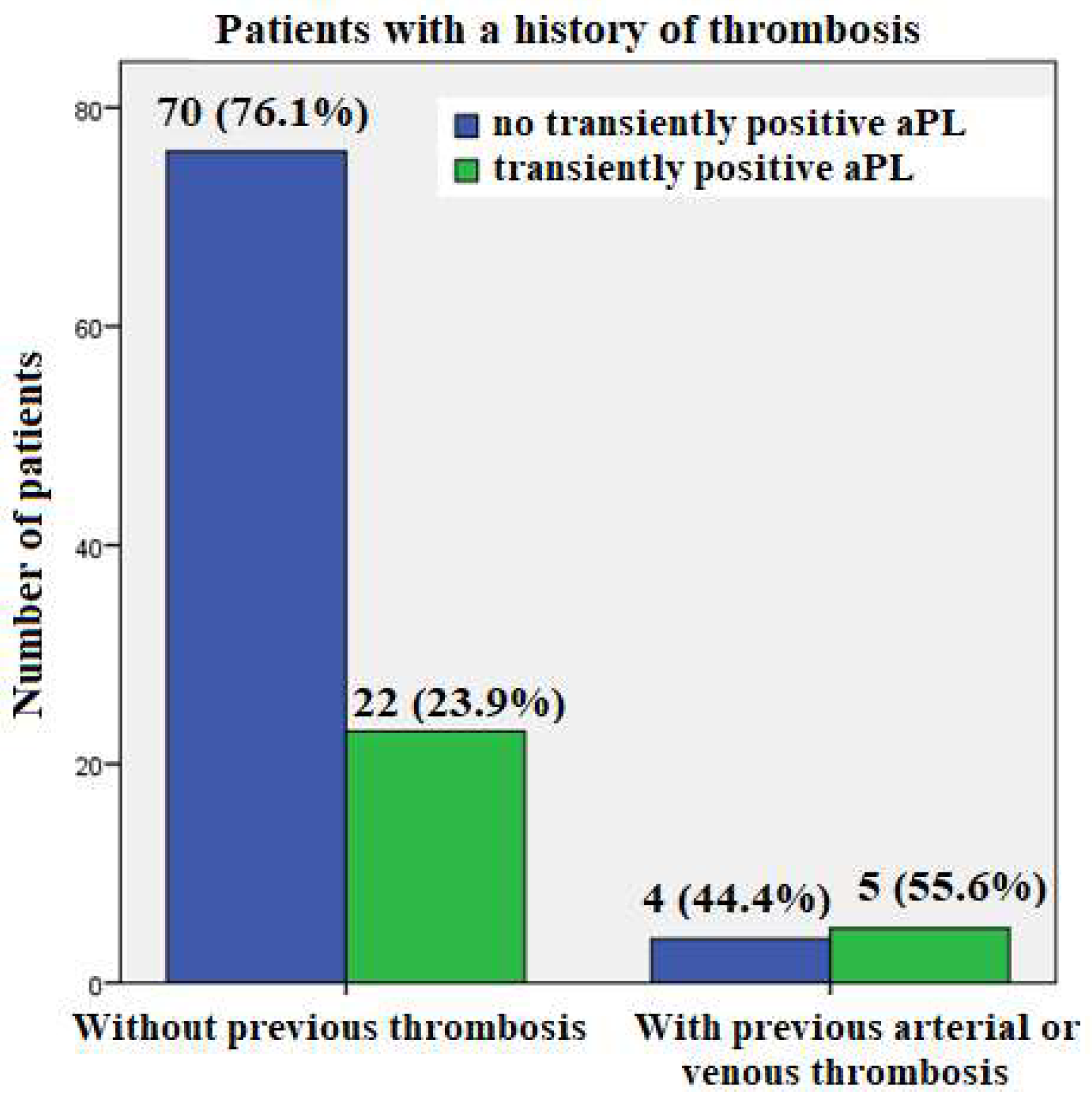
3.3.3. Patients with a History of Thrombosis
3.3.4. Patients with a History of Pregnancy Morbidity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Miyakis, S; Lockshin, M.D.; Atsumi, T.; Branch, D.W.; Brey, R.L. et al. International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS). J.Thromb.Haemost. 2006; 4:295-306. [CrossRef]
- Žigon, P.; Podovšovnik, A.; Ambrožič, A.; Tomšič, M.; Hočevar, A.; Gašperšič, N.; Rotar, Ž.; Praprotnik, S.; Šemrl, S.S.; Čučnik, S. Added value of non-criteria antiphospholipid antibodies for antiphospholipid syndrome: lessons learned from year-long routine measurements. Clinical rheumatology 2019; 38: 371-378.
- Zohoury, N.; Bertolaccini, M.L.; Rodriguez-Garcia, J.L.; Shums, Z.; Ateka-Barrutia, O.; Sorice, M.; Norman, G.L.; Khamashta, M. Closing the Serological Gap in the Antiphospholipid Syndrome: The Value of "Non-criteria" Antiphospholipid Antibodies. The Journal of rheumatology 2017; 44: 1597-1602. [CrossRef]
- Zuo, Y.; Estes, S.K.; Ali, R.A.; Gandhi, A.A.; Yalavarthi, S.; Shi, H.; Sule, G; Gockman, K; Madison, J.A.; Zuo, M; Yadav, V; Wang, J; Woodard, W; Lezak, SP; Lugogo, NL; Smith, SA; Morrissey, JH; Kanthi, Y; Knight, JS. Prothrombotic autoantibodies in serum from patients hospitalized with COVID-19. Sci Transl Med 2020;12(570):eabd3876. [CrossRef]
- Cervera, R.; Khamashta, M.A.; Shoenfeld, Y.; Camps, M.T.; Jacobsen, S. et al. Euro-Phospholipid Project Group (European Forum on Antiphospholipid Antibodies). Morbidity and mortality in the antiphospholipid syndrome during a 10-year period: a multicentre prospective study of 1000 patients. Ann Rheum Dis 2009; 68: 1428-32. [CrossRef]
- Duarte-Garcia, A.; Pham, M.M.; Crowson, C.S.; Amin, S.; Moder, K.G.; Pruthi, R.K.; Warrington, K.J.; Matteson, E.L. The Epidemiology of Antiphospholipid Syndrome: A Population-Based Study. Arthritis Rheumatol. 2019;71(9):1545-1552. [CrossRef]
- Shoenfeld, Y.; Blank, M.; Cervera, R.; Font, J.; Raschi, E ; Meron, P.L. Infectious origin of the antiphospholipid syndrome. Ann Rheum Dis 2006; 65:2–6. [CrossRef]
- Stelzer, M.; Henes, J; Saur, S. The Role of Antiphospholipid Antibodies in COVID-19. Curr Rheumatol Rep. 2021;23(9):72-4. [CrossRef]
- Taha, M.; Samavati, L. Antiphospholipid antibodies in COVID-19: a meta-analysis and systematic review. RMD Open 2021;7:e001580. [CrossRef]
- Gkrouzman, E.; Barbhaiya, M.; Erkan, D.; Lockshin, M.D. Reality Check on Antiphospholipid Antibodies in COVID-19-Associated Coagulopathy. Arthritis & rheumatology (Hoboken, N.J.) 2021; 73: 173-174. [CrossRef]
- Trahtemberg, U.; Rottapel, R.; Dos Santos, C.C.; Slutsky, A.S.; Baker, A.; Fritzler, M.J. Anticardiolipin and other antiphospholipid antibodies in critically ill COVID-19 positive and negative patients. Ann Rheum Dis. 2021;80:1236-1240. [CrossRef]
- Hasan Ali, O.; Bomze, D.; Risch, L.; Brugger, S.D.; Paprotny, M.; Weber, M.; Thiel, S.; Kern, L.; Albrich, W.C.; Kohler, P.; Kahlert, C.R.; Vernazza, P.; Bühler, P.K.;, Schüpbach, R.A.; Gómez-Mejia, A.; Popa, A.M.; Bergthaler, A.; Penninger, J.M.; Flatz, L. Severe Coronavirus Disease 2019 (COVID-19) is Associated With Elevated Serum Immunoglobulin (Ig) A and Antiphospholipid IgA Antibodies. Clin Infect Dis. 2021;73(9):e2869-e2874. [CrossRef]
- Bitterman, L.; Solhjoo, M.; Shah, V.; Kwon, S.M.; Torralba, K.; Kazbour, H. Catastrophic Antiphospholipid Syndrome as a Complication of COVID-19 Infection. J Investig Med High Impact Case Rep. 2023;11:23247096231165736. [CrossRef]
- Stelzer, M.; Henes, J.; Saur, S. The Role of Antiphospholipid Antibodies in COVID-19. Curr Rheumatol Rep. 2021;23(9):72. [CrossRef]
- Božič, B.; Kveder, T.; Stegnar, M.; Morosini-Berus, E.; Kos-Golja, M.; Peternel, P.; Rozman, B. Influence of degraded phosphatidylserine on binding of antiphospholipid antibodies. Int. Arch. Allergy Immunol. 1997; 112: 19–26. [CrossRef]
- Žigon, P.; Ambrožic, A.; Cucnik, S.; Kveder, T.; Rozman, B.; Božic, B. Modified phosphatidylserine-dependent antiprothrombin ELISA enables identification of patients negative for other antiphospholipid antibodies and also detects low avidity antibodies. Clin. Chem. Lab. Med. 2011;49:1573. [CrossRef]
- Mendoza-Pinto, C.; Escárcega, R.O.; García-Carrasco, M.; Bailey, D.J.O.; Gálvez-Romero, J.L.; Cervera, R. Viral infections and their relationship with catastrophic antiphospholipid syndrome: a possible pathogenic mechanism of severe COVID-19 thrombotic complications. J Intern Med. 2020;288:737-739. [CrossRef]
- Zhang, Y.; Xiao, M.; Zhang, S.; Xia, P.; Cao, W.; Jiang, W.; Chen, H.; Ding, X.; Zhao, H.; Zhang, H.; Wang, C.; Zhao, J.; Sun, X.; Tian, R.; Wu, W.; Wu, D.; Ma. J.; Chen, Y.; Zhang, D.; Xie, J.; Yan, X.; Zhou, X.; Liu, Z.; Wang, J.; Du, B.; Qin, Y.; Gao, P.; Qin, X.; Xu, Y.; Zhang, W.; Li, T.; Zhang, F.; Zhao, Y.; Li, Y.; Zhang, S. Coagulopathy and antiphospholipid antibodies in patients with covid-19. N Engl J Med. 2020;382(17):e38. [CrossRef]
- Ahmad, F.; Kannan, M.; Ansari, A.W.; Role of SARS-CoV-2 -induced cytokines and growth factors in coagulopathy and thromboembolism. Cytokine Growth Factor Rev. 2022;63:58-68.
- Butt, A.; Erkan, D.; Lee, A.I.; COVID-19 and antiphospholipid antibodies. Best Pract Res Clin Haematol. 2022;35(3):101402. [CrossRef]
- Abdel-Wahab, N.; Lopez-Olivo, M. A.; Pinto-Patarroyo, G. P.; Suarez-Almazor, M. E. Systematic review of case reports of antiphospholipid syndrome following infection. Lupus 2016; 25: 1520–1531. [CrossRef]
- Zhang, Y.; Cao, W.; Jiang, W. Xiao, M.; Li, Y.; Tang, N.; Liu, Z.; Yan, X.; Zhao, Y.; Li, T.; Zhu, T. Profile of natural anticoagulant, coagulant factor and anti-phospholipid antibody in critically ill COVID-19 patients. J Thromb Thrombolysis. 2020;50(3):580-586.
| Admission (n = 107) |
Worsening (n = 10) |
Release (n = 101) |
Follow-up (n = 91) |
Admission/ Release |
Admission/ Follow-up |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean (SD) | p value | |||||||||||
| HAEMATOLOGY | Erytrocyte sedimentation (mm/h) | 50.2 (22.4) | 61.5 (8.5) | 28 (11.2) | 12.2 (11.9) | 0.000** | 0.000** | |||||
| C- reactive protein (mg/L) | 102.8 (72.3) | 159.9 (106.2) | 6.3 (7.9) | 5.1 (9) | 0.000** | 0.000** | ||||||
| Erytrocytes (x1012/L) | 4.51 (0.5) | 4.3 (0.4) | 4.6 (1.3) | 4.8 (0.5) | 0.242 | 0.000** | ||||||
| Haemoglobin (g/L) | 132.3 (15.7) | 129.1 (15.6) | 132.2 (13.9) | 139 (14.6) | 0.983 | 0.000** | ||||||
| Haematocrite (%) | 0.4 (0.04) | 0.4 (0.04) | 0.4 (0.04) | 0.4 (0.05) | 0.347 | 0.000** | ||||||
| Leucocytes (x109/L) | 6 (2.5) | 8.4 (5) | 10.8 (4.2) | 7.1 (2.2) | 0.000** | 0.000** | ||||||
| Neutrophiles (x109/L) | 71.3 (20.2) | 86.1 (8.6) | 75.6 (13.4) | 57.3 (8.2) | 0.040* | 0.000** | ||||||
| Lymphocytes (x109/L) | 16.9 (9.9) | 10.4 (7.2) | 16.7 (10) | 37.1 (8.4) | 0.451 | 0.000** | ||||||
| Platelets (x109/L) | 214.9 (78) | 224.3 (87.1) | 315.6 (105.7) | 246 (55.4) | 0.000** | 0.004* | ||||||
| BIOCHEMIASTRY | D-dimer (µg/L) | 1.3 (3.08) | 5.7 (11.9) | 0.7 (0.6) | 0.5 (0.7) | 0.173 | 0.022* | |||||
| Ferritin (µg/L) | 997 (664.8) | 1523.3 (420.2) | 692 (533.2) | 195.3 (185) | 0.000** | 0.000** | ||||||
| Troponin (ng/L) | 23.2 (88.2) | 142.2 (347.1) | 6.3 (10.8) | 4.6 (6) | 0.016* | 0.025* | ||||||
| Procalcitonin (ng/mL) | 0.3 (1.8) | 0.2 (0.2) | 0.03 (0.03) | 0.03 (0.03) | 0.175 | 0.196 | ||||||
| LDH (U/L) | 666.8 (282) | 1107.5 (487.8) | 394 (111.2) | 370.3(65.9) | 0.000** | 0.000** | ||||||
|
ANTIPHOSPHOLIPID ANTIBODIES |
aCL IgG (AUG) | 4.49 (6.22) | 7.00 (7.90) | 9.13 (9.54) | 5.99 (5.19) | 0.000** | 0.050* | |||||
| aCL IgM (AUM) | 3.34 (3.77) | 4.0 (1.79) | 9.92 (12.10) | 3.21 (6.53) | 0.000** | 0.506 | ||||||
| aCL IgA (AUA) | 1.71 (2.66) | 1.71 (2.66) | 1.65 (1.79) | 0.45 (0.87) | 0.727 | 0.000** | ||||||
| anti-β2GPI IgG (AUG) | 0.10 (0.31) | 0.14 (0.36) | 0.44 (1.42) | 0.40 (0.99) | 0.120 | 0.004** | ||||||
| anti-β2GPI IgM (AUM) | 0.04 (0.24) | 0.00 (0.00) | 0.10 (0.41) | 0.20 (0.88) | 0.320 | 0.139 | ||||||
| anti-β2GPI IgA (AUA) | 0.23 (0.75) | 0.21 (0.43) | 0.25 (0.83) | 0.20 (0.62) | 0.807 | 0.426 | ||||||
| aPS/PT IgG (AUG) | 1.03 (0.42) | 1.21 (0.69) | 0.93 (0.59) | 1.22 (0.51) | 0.096 | 0.001** | ||||||
| aPS/PT IgM (AUM) | 0.78 (3.60) | 3.50 (12.26) | 0.99 (4.52) | 0.98 (4.71) | 0.363 | 1.000 | ||||||
| aPS/PT IgA (AUA) | 1.54 (1.19) | 2.14 (1.23) | 1.52 (1.22) | 0.82 (1.24) | 0.940 | 0.000 | ||||||
| Admission (n= 107) |
Worsening (n= 10) |
Release (n= 101) |
Follow-up (n= 91) |
Admission/ Release |
Admission/ Follow-up |
|
|---|---|---|---|---|---|---|
| aPL positivity | Number (%) | p value | ||||
| aCL IgG | 9 (8.4) | 3 (21.4) | 28 (27.7) | 13 (14.3) | 0.000** | 0.285 |
| aCL IgM | 4 (3.7) | 0 (0.0) | 23 (22.8) | 6 (6.6) | 0.000** | 0.705 |
| aCL IgA | 3 (2.8) | 0 (0.0) | 1 (1) | 0 (0.0) | 0.317 | 0.083 |
| anti-β2GPI IgG | 0 (0.0) | 0 (0.0) | 7 (6.9) | 5 (5.5) | 0.008** | 0.025* |
| anti-β2GPI IgM | 1 (0.9) | 0 (0.0) | 2 (2) | 4 (4.4) | 1.000 | 0.157 |
| anti-β2GPI IgA | 4 (3.7) | 0 (0.0) | 4 (4) | 4 (4.4) | 1.000 | 1.000 |
| aPS/PT IgG | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1.000 | 1.000 |
| aPS/PT IgM | 5 (4.7) | 1 (7.1) | 5 (5) | 4 (4.4) | 1.000 | 0.317 |
| aPS/PT IgA | 1 (0.9) | 0 (0.0) | 2 (2) | 1 (1.1) | 0.317 | 1.000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




