Submitted:
04 August 2023
Posted:
07 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. HGF-MET signaling
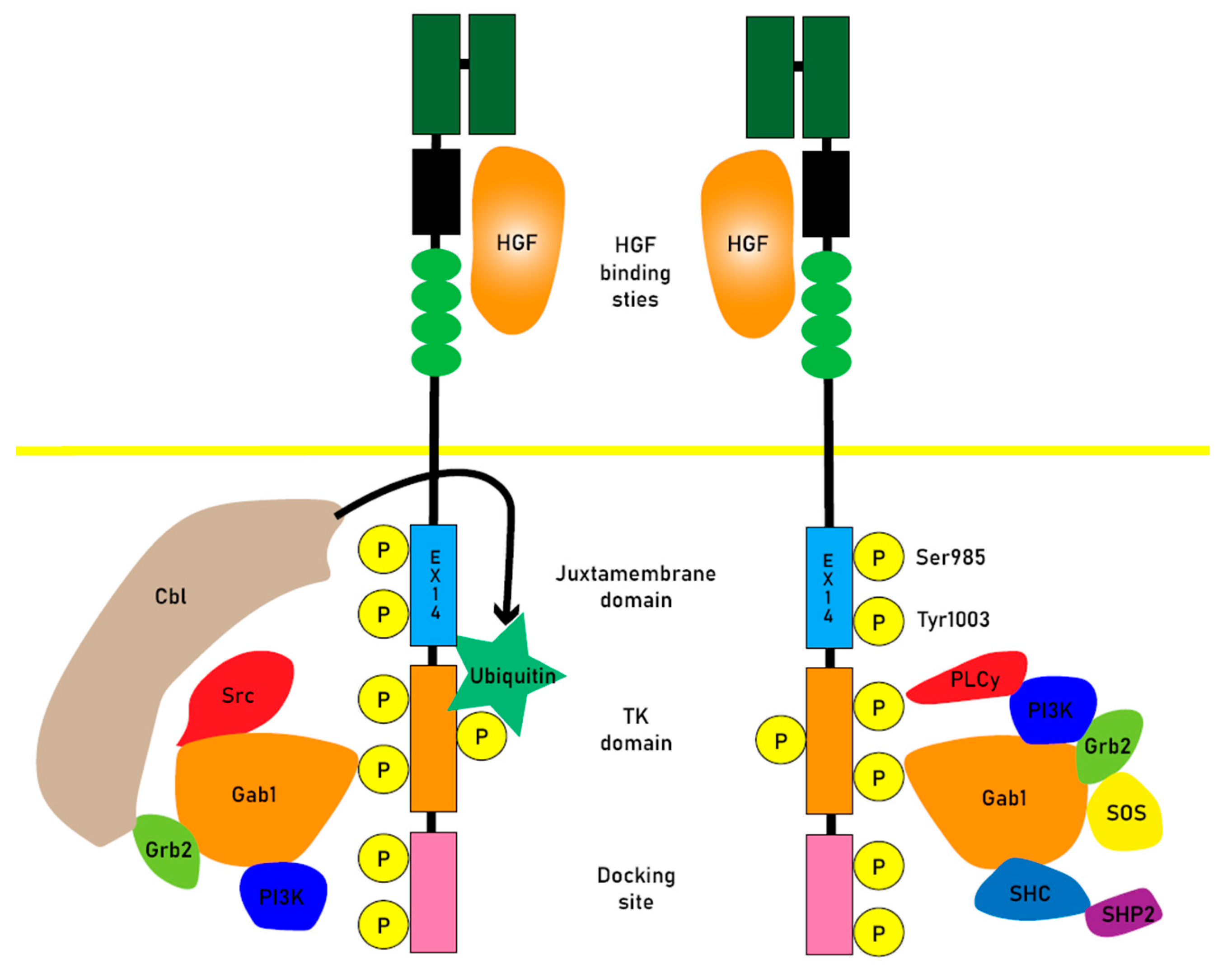
2.1. HGF-MET signaling
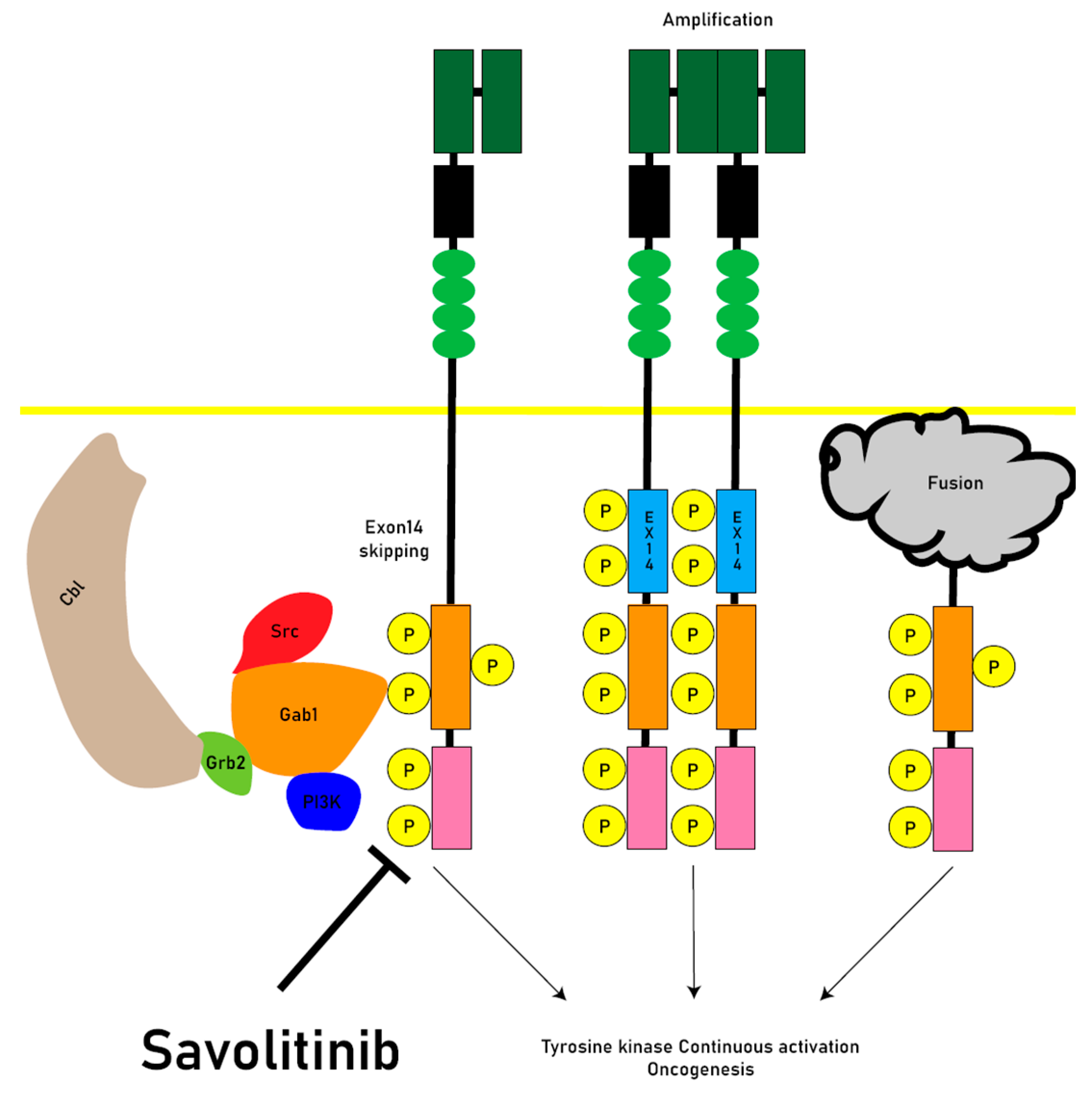
2.2. MET mutation
2.3. Oncogenesis
3. Savolitinib
3.1. Introducing savolitinib
3.2. Metabolism
3.3. Side effects and safety
3.4. In vivo and xenograft study
3.5. First in human phase I trial
3.6. Pivotal phase 2 trial
3.7. Phase 3 trials on the way
3.8. Pivotal phase 2 trial
3.8.1. Savolitinib + Osimertinib
3.8.2. Savolitinib + Gefatinib
3.8.3. Savolitinib + Docetaxel
3.8.4. Savolitinib + Durvalumab
4. Resistance
5. Biomarker
6. Non-Small Cell Lung Cancer (NSCLC)
6.1. Non-Small Cell Lung Cancer
6.2. Neoadjuvant therapy in NSCLC
6.3. Lung sarcomatoid carcinoma
7. Renal cell carcinoma
8. Gastric cancer
9. Hepatocellular carcinoma
10. Colorectal cancer
11. Pancreatic cancer
12. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hammerman, P.S. , et al., Comprehensive genomic characterization of squamous cell lung cancers. Nature, 2012. 489(7417): p. 519-525.
- Collisson, E.A. , et al., Comprehensive molecular profiling of lung adenocarcinoma. Nature, 2014. 511(7511): p. 543-550.
- Govindan, R. , et al., Genomic landscape of non-small cell lung cancer in smokers and never-smokers. Cell, 2012. 150(6): p. 1121-34.
- Rosell, R. , et al., Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol, 2012. 13(3): p. 239-46.
- Yang, J.C. , et al., Afatinib versus cisplatin-based chemotherapy for EGFR mutation-positive lung adenocarcinoma (LUX-Lung 3 and LUX-Lung 6): analysis of overall survival data from two randomised, phase 3 trials. Lancet Oncol, 2015. 16(2): p. 141-51.
- Weidner, K.M. , et al., Scatter factor: molecular characteristics and effect on the invasiveness of epithelial cells. J Cell Biol, 1990. 111(5 Pt 1): p. 2097-108.
- Montesano, R. , et al., Identification of a fibroblast-derived epithelial morphogen as hepatocyte growth factor. Cell, 1991. 67(5): p. 901-8.
- Bladt, F. , et al., Essential role for the c-met receptor in the migration of myogenic precursor cells into the limb bud. Nature, 1995. 376(6543): p. 768-71.
- Weidner, K.M. , et al., Interaction between Gab1 and the c-Met receptor tyrosine kinase is responsible for epithelial morphogenesis. Nature, 1996. 384(6605): p. 173-176.
- Birchmeier, C. , et al., Met, metastasis, motility and more. Nature Reviews Molecular Cell Biology, 2003. 4(12): p. 915-925.
- Sipeki, S. , et al., Phosphatidylinositol 3-kinase Contributes to Erk1/Erk2 MAP Kinase Activation Associated with Hepatocyte Growth Factor-induced Cell Scattering. Cellular Signalling, 1999. 11(12): p. 885-890.
- Zhang, Y.-W. , et al., Requirement of Stat3 signaling for HGF/SF-Met mediated tumorigenesis. Oncogene, 2002. 21(2): p. 217-226.
- Van Der Steen, N. , et al., cMET in NSCLC: Can We Cut off the Head of the Hydra? From the Pathway to the Resistance. Cancers, 2015. 7(2): p. 556-573.
- Zaborowska-Szmi, M. , et al., Savolitinib for non-small cell lung cancer. Drugs Today (Barc), 2023. 59(1): p. 17-36.
- Schmidt, L. , et al., Germline and somatic mutations in the tyrosine kinase domain of the MET proto-oncogene in papillary renal carcinomas. Nat Genet, 1997. 16(1): p. 68-73.
- Coleman, N. , et al., Targeting un-MET needs in advanced non-small cell lung cancer. Lung Cancer, 2022. 164: p. 56-68.
- Tong, J.H. , et al., MET Amplification and Exon 14 Splice Site Mutation Define Unique Molecular Subgroups of Non–Small Cell Lung Carcinoma with Poor Prognosis. Clinical Cancer Research, 2016. 22(12): p. 3048-3056.
- Safi, D., T. Abu Hejleh, and M. Furqan, Narrative review: mesenchymal-epithelial transition inhibitors-meeting their target. Transl Lung Cancer Res, 2021. 10(1): p. 462-474.
- Peinado, H. , et al., Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat Med, 2012. 18(6): p. 883-91.
- Matsumoto, K. , et al., Hepatocyte growth factor/MET in cancer progression and biomarker discovery. Cancer Sci, 2017. 108(3): p. 296-307.
- Markham, A. , Savolitinib: First Approval. Drugs, 2021. 81(14): p. 1665-1670.
- Gu, Y. , et al., Preclinical pharmacokinetics, disposition, and translational pharmacokinetic/pharmacodynamic modeling of savolitinib, a novel selective cMet inhibitor. Eur J Pharm Sci, 2019. 136: p. 104938.
- Ren, S. , et al., Clinical evaluation of the potential drug-drug interactions of savolitinib: Interaction with rifampicin, itraconazole, famotidine or midazolam. Br J Clin Pharmacol, 2022. 88(2): p. 655-668.
- Cortot, A. , et al., Safety of MET Tyrosine Kinase Inhibitors in Patients With MET Exon 14 Skipping Non-small Cell Lung Cancer: A Clinical Review. Clin Lung Cancer, 2022. 23(3): p. 195-207.
- Sahota, T. , et al., A Randomized, Double-Blind, Placebo- and Positive-Controlled, Three-Way Crossover Study in Healthy Participants to Investigate the Effect of Savolitinib on the QTc Interval. Clin Pharmacol Drug Dev, 2021. 10(5): p. 521-534.
- Schalkwijk, S. , et al., Parent and Metabolite Concentration-QT Modeling to Evaluate QT-Interval Prolongation at Savolitinib Therapeutic Doses. AAPS J, 2021. 23(3): p. 46.
- Xiong, Y. , et al., Case report: Savolitinib induced severe adverse reactions resembling septic shock in an HIV-1-positive patient with advanced non-small cell lung cancer. Front Pharmacol, 2023. 14: p. 1089184.
- Schuller, A.G. , et al., The MET Inhibitor AZD6094 (Savolitinib, HMPL-504) Induces Regression in Papillary Renal Cell Carcinoma Patient-Derived Xenograft Models. Clin Cancer Res, 2015. 21(12): p. 2811-9.
- Gan, H.K. , et al., First-in-Human Phase I Study of the Selective MET Inhibitor, Savolitinib, in Patients with Advanced Solid Tumors: Safety, Pharmacokinetics, and Antitumor Activity. Clin Cancer Res, 2019. 25(16): p. 4924-4932.
- Wang, Y. , et al., Phase Ia/Ib Study of the Selective MET Inhibitor, Savolitinib, in Patients with Advanced Solid Tumors: Safety, Efficacy, and Biomarkers. Oncologist, 2022. 27(5): p. 342-e383.
- Lu, S. , et al., Long-Term Efficacy, Safety, and Subgroup Analysis of Savolitinib in Chinese Patients With NSCLCs Harboring MET Exon 14 Skipping Alterations. JTO Clin Res Rep, 2022. 3(10): p. 100407.
- Lu, S. , et al., 2MO Final OS results and subgroup analysis of savolitinib in patients with MET exon 14 skipping mutations (METex14+) NSCLC. Volume : 33: p. Copyright © 2022 European Society for Medical Oncology. Published by Elsevier Ltd. All rights reserved.
- Lu, S. , et al., Once-daily savolitinib in Chinese patients with pulmonary sarcomatoid carcinomas and other non-small-cell lung cancers harbouring MET exon 14 skipping alterations: a multicentre, single-arm, open-label, phase 2 study. Lancet Respir Med, 2021. 9(10): p. 1154-1164.
- Hong, L. , et al., Current and future treatment options for MET exon 14 skipping alterations in non-small cell lung cancer. Therapeutic Advances in Medical Oncology, 2021. 13: p. 1758835921992976.
- Zhu, X., Y. Lu, and S. Lu, Landscape of Savolitinib Development for the Treatment of Non-Small Cell Lung Cancer with MET Alteration-A Narrative Review. Cancers (Basel), 2022. 14(24).
- Sequist, L.V. , et al., Osimertinib plus savolitinib in patients with EGFR mutation-positive, MET-amplified, non-small-cell lung cancer after progression on EGFR tyrosine kinase inhibitors: interim results from a multicentre, open-label, phase 1b study. Lancet Oncol, 2020. 21(3): p. 373-386.
- Oxnard, G.R. , et al., TATTON: a multi-arm, phase Ib trial of osimertinib combined with selumetinib, savolitinib, or durvalumab in EGFR-mutant lung cancer. Ann Oncol, 2020. 31(4): p. 507-516.
- Hartmaier, R.J. , et al., Osimertinib + Savolitinib to Overcome Acquired MET-Mediated Resistance in Epidermal Growth Factor Receptor-Mutated, MET-Amplified Non-Small Cell Lung Cancer: TATTON. Cancer Discov, 2023. 13(1): p. 98-113.
- Oxnard, G.R. , et al., SAVANNAH: A Phase II trial of osimertinib plus savolitinib for patients (pts) with EGFR-mutant, MET-driven (MET+), locally advanced or metastatic non-small cell lung cancer (NSCLC), following disease progression on osimertinib. Journal of Clinical Oncology, 2019. 37(15_suppl): p. TPS9119-TPS9119.
- Li, A., H. J. Chen, and J.J. Yang, Design and Rationale for a Phase II, Randomized, Open-Label, Two-Cohort Multicenter Interventional Study of Osimertinib with or Without Savolitinib in De Novo MET Aberrant, EGFR-Mutant Patients with Advanced Non-Small-Cell Lung Cancer: The FLOWERS Trial. Clin Lung Cancer, 2023. 24(1): p. 82-88.
- Yang, J.J. , et al., A phase Ib study of the highly selective MET-TKI savolitinib plus gefitinib in patients with EGFR-mutated, MET-amplified advanced non-small-cell lung cancer. Invest New Drugs, 2021. 39(2): p. 477-487.
- Kim, S.T. , et al., Combination of Docetaxel Plus Savolitinib in Refractory Cancer Patients: A Report on Phase I Trial. Transl Oncol, 2019. 12(4): p. 597-601.
- Suárez, C. , et al., Phase II Study Investigating the Safety and Efficacy of Savolitinib and Durvalumab in Metastatic Papillary Renal Cancer (CALYPSO). J Clin Oncol, 2023. 41(14): p. 2493-2502.
- Henry, R.E. , et al., Acquired savolitinib resistance in non-small cell lung cancer arises via multiple mechanisms that converge on MET-independent mTOR and MYC activation. Oncotarget, 2016. 7(36): p. 57651-57670.
- Frigault, M.M. , et al., Mechanisms of Acquired Resistance to Savolitinib, a Selective MET Inhibitor in MET-Amplified Gastric Cancer. JCO Precis Oncol, 2020. 4.
- Zhang, Y., Z. Du, and M. Zhang, Biomarker development in MET-targeted therapy. Oncotarget, 2016. 7(24): p. 37370-37389.
- Matsumoto, K. and T. Nakamura, Hepatocyte growth factor: renotropic role and potential therapeutics for renal diseases. Kidney Int, 2001. 59(6): p. 2023-38.
- Srivastava, A.K. , et al., Pharmacodynamic Response of the MET/HGF Receptor to Small-Molecule Tyrosine Kinase Inhibitors Examined with Validated, Fit-for-Clinic Immunoassays. Clinical Cancer Research, 2016. 22(14): p. 3683-3694.
- Lee, J. , et al., Tumor Genomic Profiling Guides Patients with Metastatic Gastric Cancer to Targeted Treatment: The VIKTORY Umbrella Trial. Cancer Discov, 2019. 9(10): p. 1388-1405.
- Yu, Y. , et al., Circulating tumour DNA biomarkers in savolitinib-treated patients with non-small cell lung cancer harbouring MET exon 14 skipping alterations: a post hoc analysis of a pivotal phase 2 study. Ther Adv Med Oncol, 2022. 14: p. 17588359221133546.
- Subramanian, J. and O. Tawfik, Detection of MET exon 14 skipping mutations in non-small cell lung cancer: overview and community perspective. Expert Rev Anticancer Ther, 2021. 21(8): p. 877-886.
- Drusbosky, L.M. , et al., Therapeutic strategies in METex14 skipping mutated non-small cell lung cancer. J Hematol Oncol, 2021. 14(1): p. 129.
- Gong, C. , et al., MET alterations in advanced pulmonary sarcomatoid carcinoma. Front Oncol, 2022. 12: p. 1017026.
- Xu, Z. , et al., Incidence and PD-L1 Expression of MET 14 Skipping in Chinese Population: A Non-Selective NSCLC Cohort Study Using RNA-Based Sequencing. Onco Targets Ther, 2020. 13: p. 6245-6253.
- Sabari, J.K. , et al., PD-L1 expression, tumor mutational burden, and response to immunotherapy in patients with MET exon 14 altered lung cancers. Ann Oncol, 2018. 29(10): p. 2085-2091.
- Xu, L., F. Wang, and F. Luo, MET-targeted therapies for the treatment of non-small-cell lung cancer: A systematic review and meta-analysis. Front Oncol, 2022. 12: p. 1013299.
- Gu, L. , et al., A durable response to savolitinib in a patient with lung adenocarcinoma harboring two novel MET exon 14 skipping sites. Anticancer Drugs, 2023.
- Fu, M. , et al., Neoadjuvant Savolitinib targeted therapy stage IIIA-N2 primary lung adenocarcinoma harboring MET Exon 14 skipping mutation: A case report. Front Oncol, 2022. 12: p. 954886.
- Deng, H.Y. , et al., The safety and feasibility of preoperative induction therapy of Savolitinib in non-small cell lung cancer patients with MET exon 14 skipping mutation. J Cancer Res Clin Oncol, 2022.
- Tian, J. , et al., Dramatic response to neoadjuvant savolitinib in marginally resectable lung adenocarcinoma with MET exon 14 skipping mutation: A case report and literature review. Front Oncol, 2022. 12: p. 1006634.
- Yang, F. and Q.F. Chen, A case of lung adenocarcinoma with MET∆ex14 mutation regressed after preoperative treatment with savolitinib, and successfully underwent radical resection. Anticancer Drugs, 2023. 34(2): p. 302-305.
- Zhang, L. , et al., Multimodality Treatment of Pulmonary Sarcomatoid Carcinoma: A Review of Current State of Art. J Oncol, 2022. 2022: p. 8541157.
- Choueiri, T.K. , et al., Biomarker-Based Phase II Trial of Savolitinib in Patients With Advanced Papillary Renal Cell Cancer. Journal of Clinical Oncology, 2017. 35(26): p. 2993-3001.
- Choueiri, T.K. , et al., Efficacy of Savolitinib vs Sunitinib in Patients With MET-Driven Papillary Renal Cell Carcinoma: The SAVOIR Phase 3 Randomized Clinical Trial. JAMA Oncol, 2020. 6(8): p. 1247-1255.
- He, X. and G. An, Significant role of savolitinib in a case of advanced gastric cancer with abnormal mesenchymal-epithelial transition factor (MET): A case report. Medicine (Baltimore), 2022. 101(48): p. e32072.
- Ye, W. , et al., Case Report: Prompt Response to Savolitinib in a Case of Advanced Gastric Cancer With Bone Marrow Invasion and MET Abnormalities. Front Oncol, 2022. 12: p. 868654.
- Yan, N. , et al., Advanced HCC with amplified mesenchymal epithelial transition factor receptor responds well to savolitinib: a case report. Front Med (Lausanne), 2023. 10: p. 1130012.
- Jia, J. , et al., A phase II study of savolitinib (volitinib, AZD6094, HMPL-504) in subjects with MET amplified metastatic colorectal cancer (mCRC) detected by cell-free (cf)DNA. Journal of Clinical Oncology, 2020. 38(4_suppl): p. TPS270-TPS270.
- Pothula, S.P. , et al., Targeting HGF/c-MET Axis in Pancreatic Cancer. Int J Mol Sci, 2020. 21(23).
- Hage, C. , et al., The novel c-Met inhibitor cabozantinib overcomes gemcitabine resistance and stem cell signaling in pancreatic cancer. Cell Death Dis, 2013. 4(5): p. e627.
- Avan, A. , et al., Crizotinib Inhibits Metabolic Inactivation of Gemcitabine in c-Met–driven Pancreatic Carcinoma. Cancer Research, 2013. 73(22): p. 6745-6756.
- Brandes, F. , et al., Targeting cMET with INC280 impairs tumour growth and improves efficacy of gemcitabine in a pancreatic cancer model. BMC Cancer, 2015. 15(1): p. 71.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



