Submitted:
31 July 2023
Posted:
02 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
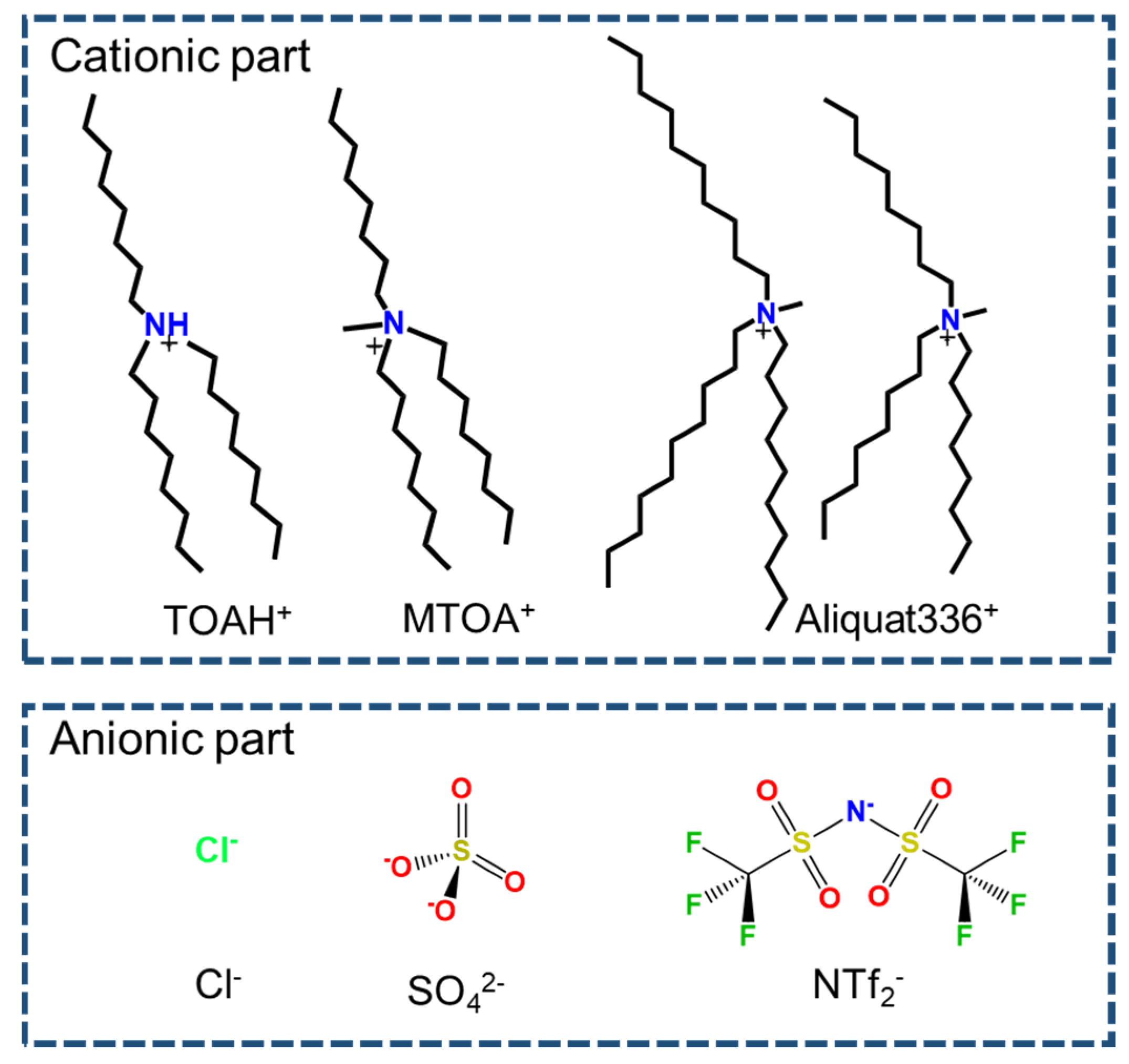
2.1. Chemicals and reagents
2.2. Instrumentation and analysis
2.3. Extraction experiments
3. Results and discussion
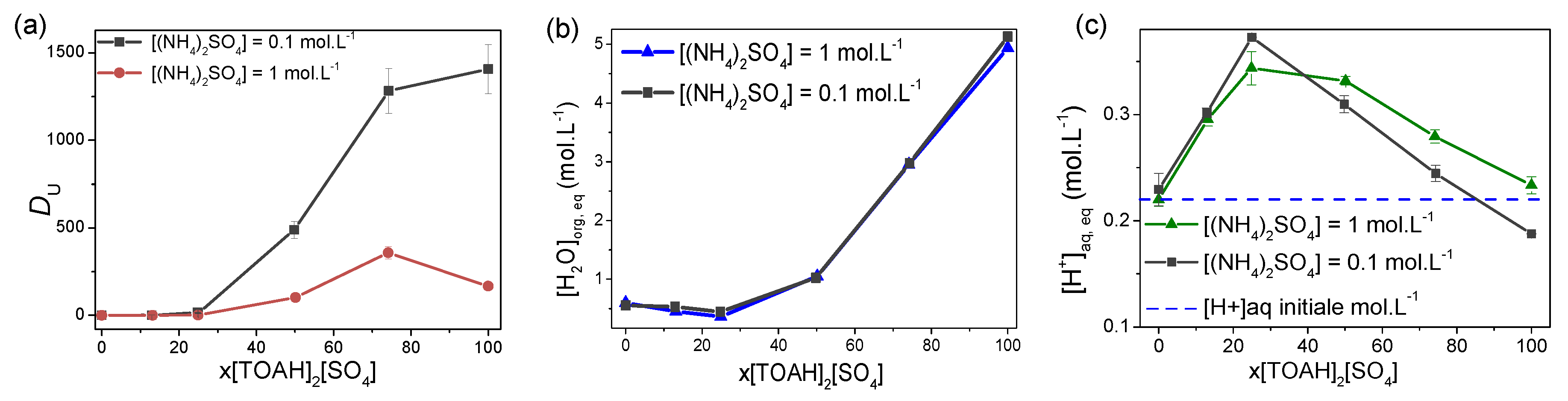
3.1. Effect of the amount of sulphates in the aqueous phase
3.2. Characterizing the extracted species
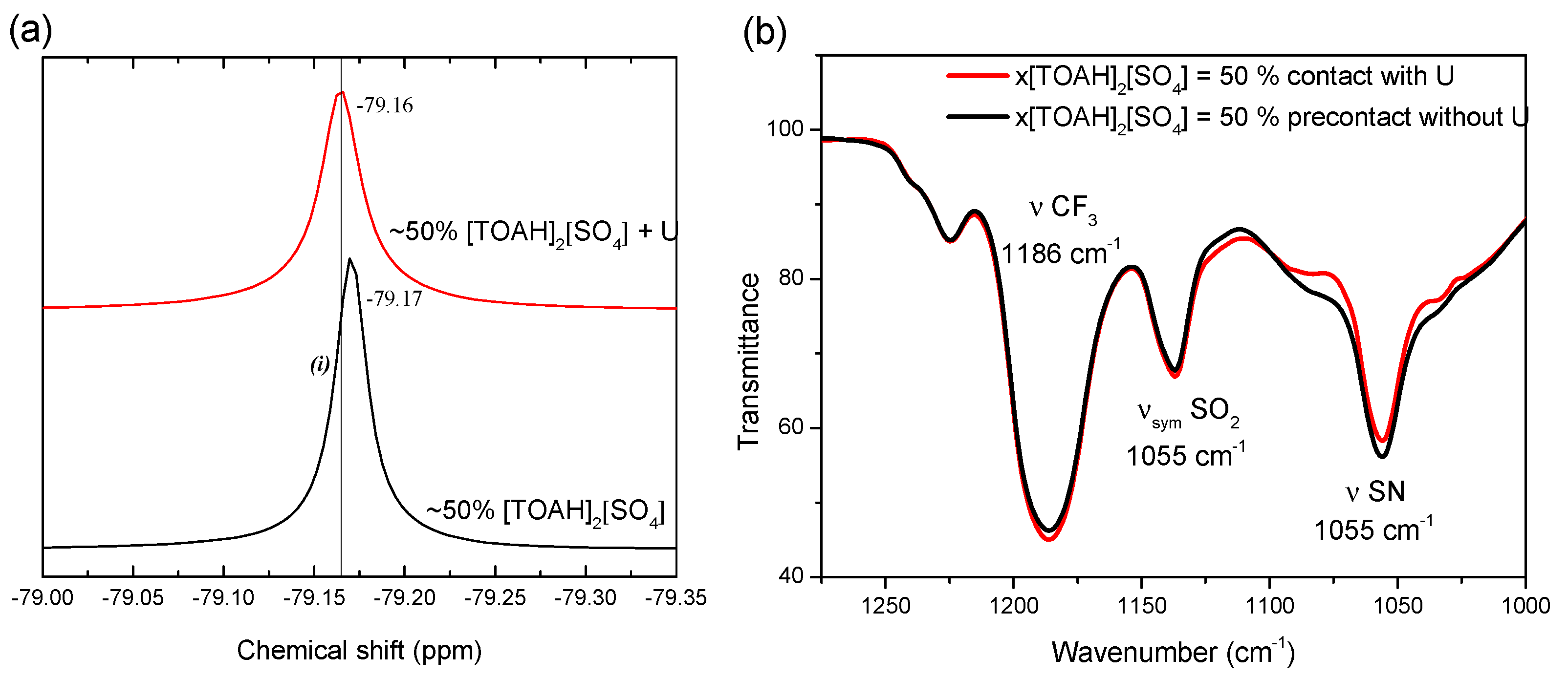
3.1.1. NTf2 participation in the uranium complex
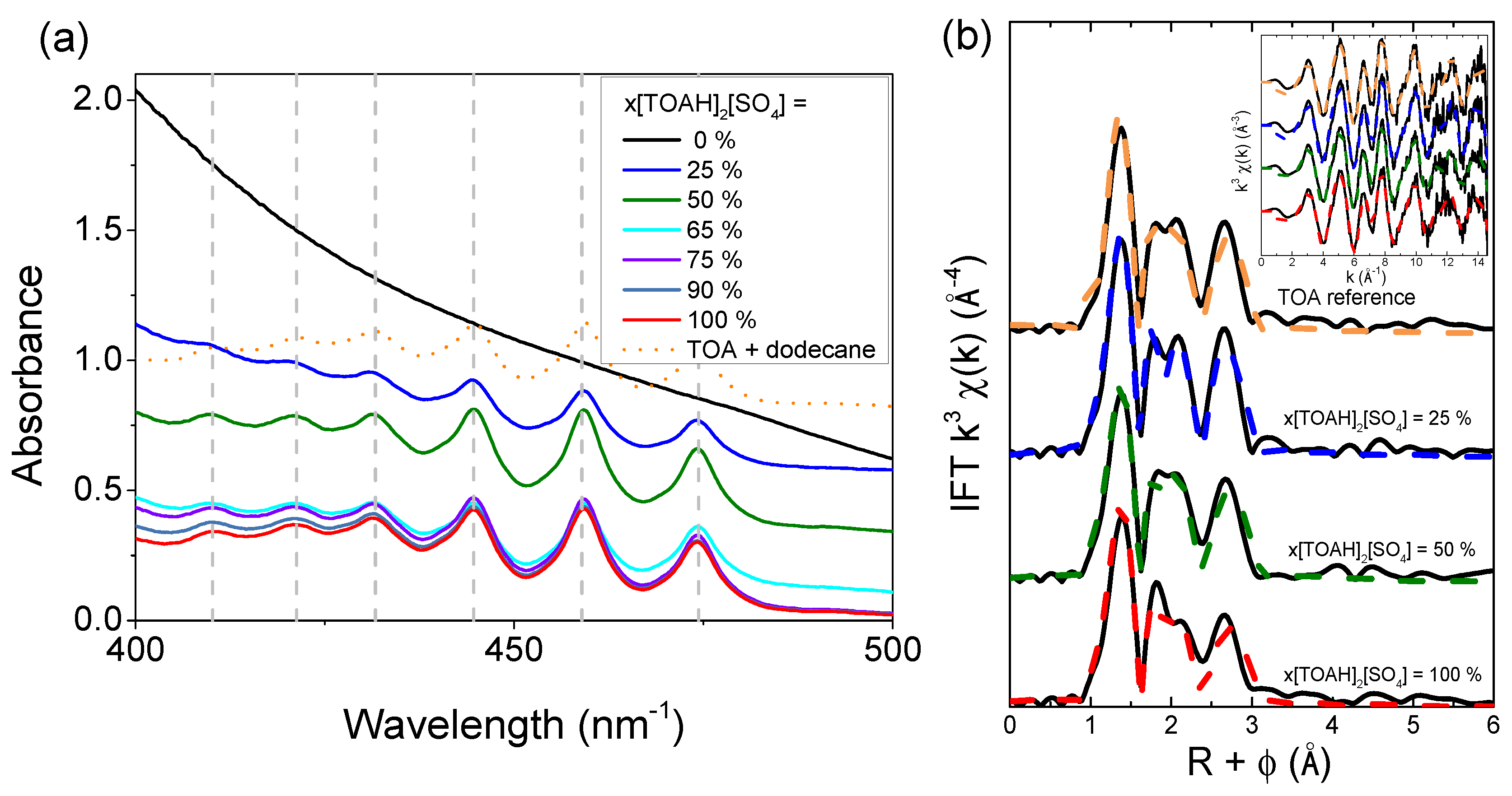
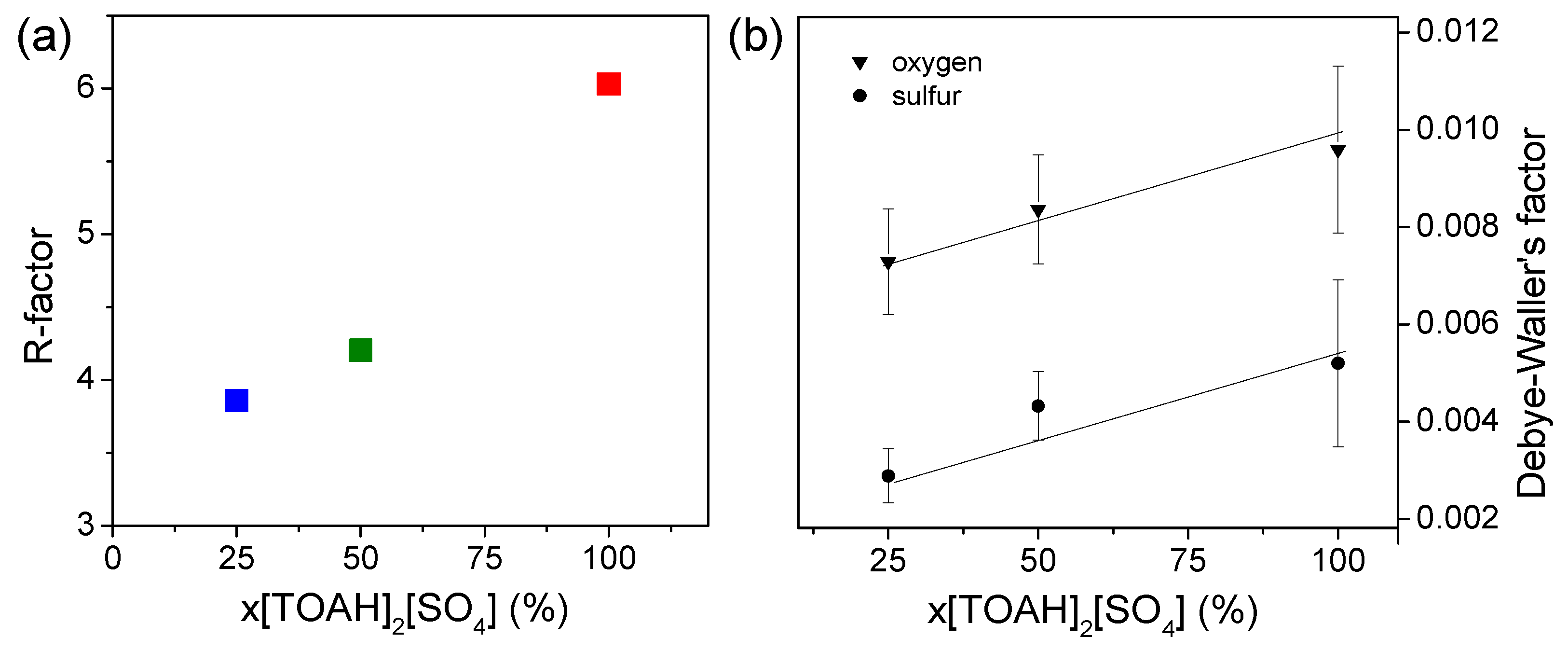
3.1.2. UV-vis spectroscopy and EXAFS, study of first neighbors
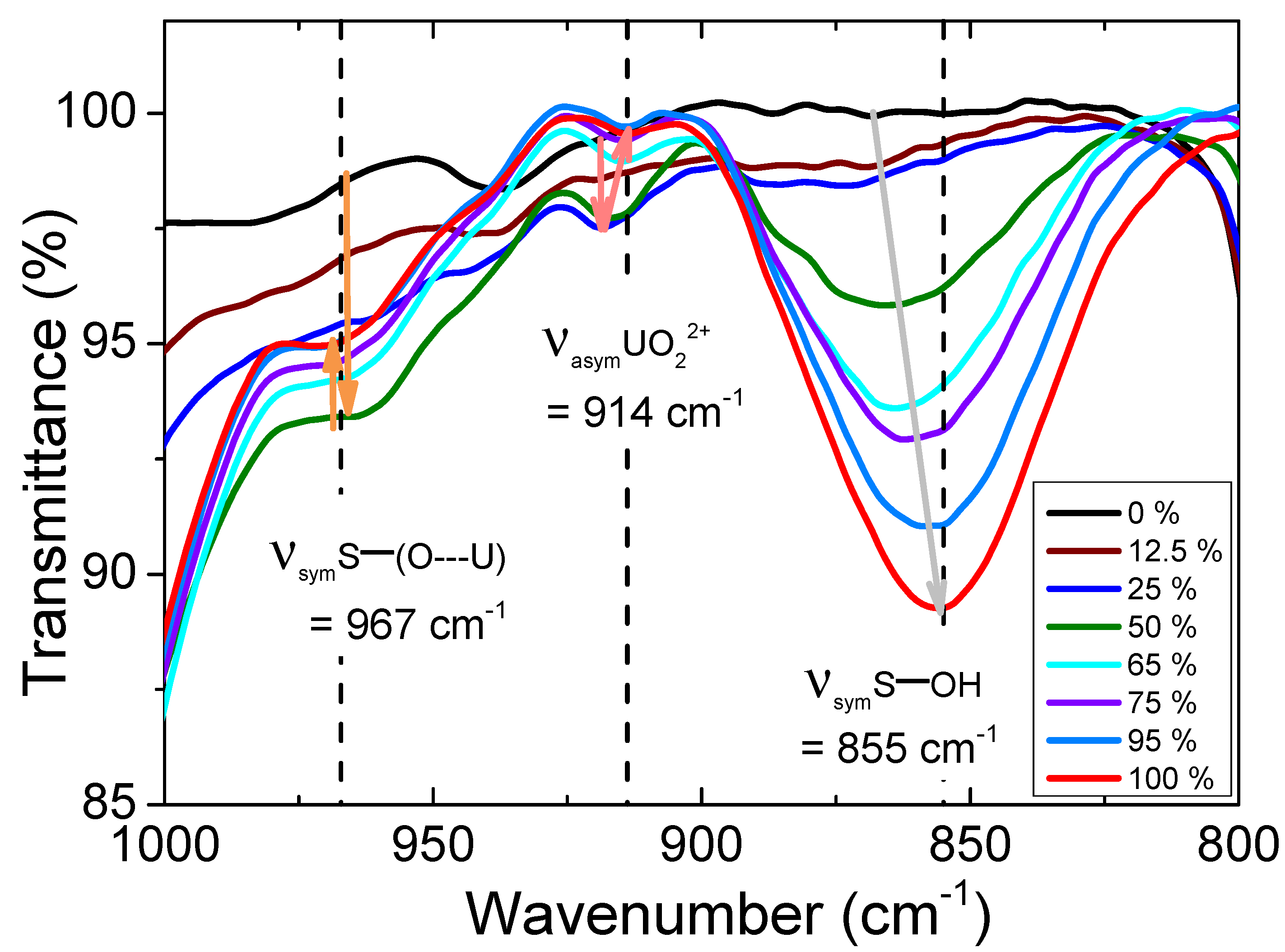
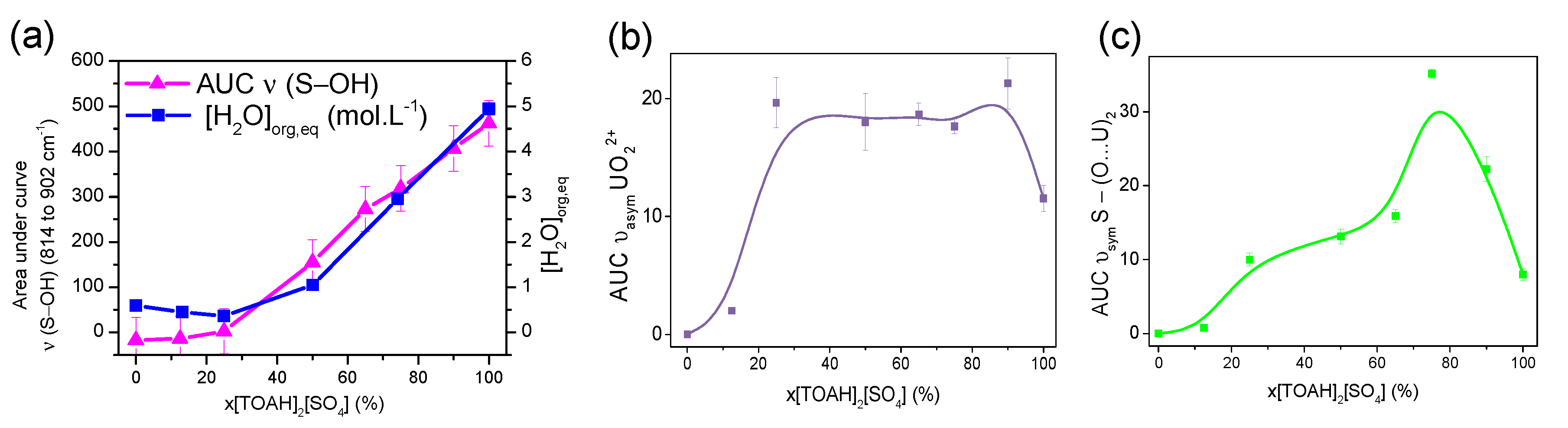
3.1.3. FT-IR spectroscopy, looking at the second coordination sphere and beyond
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Crouse, D.J.; Brown, K.B. The Amex Process for Extracting Thorium Ores with Alkyl Amines. Ind. Eng. Chem. 1959, 51, 1461–1464. [Google Scholar] [CrossRef]
- Sato, T.; Watanabe, H.; Suzuki, H. Liquid-Liquid Extraction of Molybdenum(VI) from Aqueous Acid Solutions by High-Molecular Weight Amines. Solvent Extraction and Ion Exchange 1986, 4, 987–998. [Google Scholar] [CrossRef]
- Sato, T.; Watanabe, H. The Extraction of Zirconium(IV) from Sulfuric Acid Solutions by Long-Chain Alkyl Quaternary Ammonium Compound. Separation Science and Technology 1982, 17, 625–634. [Google Scholar] [CrossRef]
- Chagnes, A.; Fosse, C.; Courtaud, B.; Thiry, J.; Cote, G. Chemical degradation of trioctylamine and 1-tridecanol phase modifier in acidic sulfate media in the presence of vanadium (V). Hydrometallurgy 2011, 105, 328–333. [Google Scholar] [CrossRef]
- Solgy, M.; Taghizadeh, M.; Ghoddocynejad, D. Adsorption of uranium(VI) from sulphate solutions using Amberlite IRA-402 resin: Equilibrium, kinetics and thermodynamics study. Annals of Nuclear Energy 2015, 75, 132–138. [Google Scholar] [CrossRef]
- Kumar, P.; Pal, A.; Saxena, M.K.; Ramakumar, K.L. Supercritical fluid extraction of uranium and thorium from solid matrices. Desalination 2008, 232, 71–79. [Google Scholar] [CrossRef]
- Smolinski, T.; Wawszczak, D.; Deptula, A.; Lada, W.; Olczak, T.; Rogowski, M.; Pyszynska, M.; Chmielewski, A.G. Solvent extraction of Cu, Mo, V, and U from leach solutions of copper ore and flotation tailings. J. Radioanal. Nucl. Chem. 2017, 314, 69–75. [Google Scholar] [CrossRef]
- Quijada-Maldonado, E.; Olea, F.; Sepúlveda, R.; Castillo, J.; Cabezas, R.; Merlet, G.; Romero, J. Possibilities and challenges for ionic liquids in hydrometallurgy. Separation and Purification Technology 2020, 251, 117289. [Google Scholar] [CrossRef]
- Yan, Q.; Cai, Y.; Wang, Z.; Dong, X.; Yuan, L.; Feng, W.; Chen, J.; Xu, C. Separation of americium from lanthanide by a Task-Specific ionic liquid decorated with 2,6-Bis-Triazolyl-Pyridine moiety. Separation and Purification Technology 2022, 299, 121752. [Google Scholar] [CrossRef]
- Brown, K.B.; Coleman, C.F.; Crouse, D.J.; Denis, J.O.; Moore, J.G. THE USE OF AMINES AS EXTRACTANTS FOR URANIUM FROM ACIDIC SULFATE LIQUORS. A Preliminary Report. 1954. [Google Scholar] [CrossRef]
- Lu, Z.; Dourdain, S.; Pellet-Rostaing, S.; Arrachart, G.; Giusti, F. Mélanges de Sels d’ammonium Quaternaire Pour l’extraction de l’uranium(VI) de Solutions Aqueuses d’acide Sulfurique. 3 June 2022. [Google Scholar]
- Ghandi, K. A Review of Ionic Liquids, Their Limits and Applications. GSC 2014, 04, 44–53. [Google Scholar] [CrossRef]
- Greaves, T.L.; Drummond, C.J. Protic Ionic Liquids: Properties and Applications. Chem. Rev. 2008, 108, 206–237. [Google Scholar] [CrossRef]
- Keshapolla, D.; Srinivasarao, K.; Gardas, R.L. Influence of temperature and alkyl chain length on physicochemical properties of trihexyl- and trioctylammonium based protic ionic liquids. The Journal of Chemical Thermodynamics 2019, 133, 170–180. [Google Scholar] [CrossRef]
- Billard, I.; Ouadi, A.; Gaillard, C. Is a universal model to describe liquid–liquid extraction of cations by use of ionic liquids in reach? Dalton Transactions 2013, 42, 6203–6212. [Google Scholar] [CrossRef] [PubMed]
- Dukov, I.L.; Atanassova, M. Effect of the diluents on the synergistic solvent extraction of some lanthanides with thenoyltrifluoroacetone and quaternary ammonium salt. Hydrometallurgy 2003, 68, 89–96. [Google Scholar] [CrossRef]
- Dietz, M.L.; Dzielawa, J.A.; Laszak, I.; Young, B.A.; Jensen, M.P. Influence of solvent structural variations on the mechanism of facilitated ion transfer into room-temperature ionic liquids. Green Chemistry 2003, 5, 682–685. [Google Scholar] [CrossRef]
- Billard, I.; Ouadi, A.; Jobin, E.; Champion, J.; Gaillard, C.; Georg, S. Understanding the Extraction Mechanism in Ionic Liquids: UO22+/HNO3/TBP/C4-mimTf2N as a Case Study. Solvent Extraction and Ion Exchange 2011, 29, 577–601. [Google Scholar] [CrossRef]
- Wionczyk, B.; Apostoluk, W. Solvent extraction of chromium(III) from alkaline media with quaternary ammonium compounds. Part I. Hydrometallurgy 2004, 72, 185–193. [Google Scholar] [CrossRef]
- Rout, A.; Venkatesan, K.A.; Srinivasan, T.G.; Vasudeva Rao, P.R. Ionic liquid extractants in molecular diluents: Extraction behavior of europium (III) in quarternary ammonium-based ionic liquids. Separation and Purification Technology 2012, 95, 26–31. [Google Scholar] [CrossRef]
- Jaree, A.; Khunphakdee, N. Separation of concentrated platinum(IV) and rhodium(III) in acidic chloride solution via liquid–liquid extraction using tri-octylamine. Journal of Industrial and Engineering Chemistry 2011, 17, 243–247. [Google Scholar] [CrossRef]
- Biswas, S.; Basu, S. Extraction of zirconium(IV) from hydrochloric acid solutions by tri-octylamine and neutral donors. Journal of Radioanalytical and Nuclear Chemistry 1999, 242, 253–258. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, X.; Yang, A.; Lv, P.; Zhang, L.; Li, Y.; Yang, Y. Extraction and separation on Au(III) and Pt(IV) from HCl media using novel piperazine-based ionic liquid as an ionic exchanger. Journal of Molecular Liquids 2022, 353, 118846. [Google Scholar] [CrossRef]
- Xue, W.; Liu, R.; Liu, X.; Wang, Y.; Lv, P.; Yang, Y. Selective extraction of Nd(III) by novel carboxylic acid based ionic liquids without diluent from waste NdFeB magnets. Journal of Molecular Liquids 2022, 364, 119919. [Google Scholar] [CrossRef]
- Micheau, C.; Lejeune, M.; Arrachart, G.; Draye, M.; Turgis, R.; Michel, S.; Legeai, S.; Pellet-Rostaing, S. Recovery of tantalum from synthetic sulfuric leach solutions by solvent extraction with phosphonate functionalized ionic liquids. Hydrometallurgy 2019, 189, 105107. [Google Scholar] [CrossRef]
- Hu, Q.; Zhao, J.; Wang, F.; Huo, F.; Liu, H. Selective extraction of vanadium from chromium by pure [C8mim][PF6]: An anion exchange process. Separation and Purification Technology 2014, 131, 94–101. [Google Scholar] [CrossRef]
- Zuo, Y.; Liu, Y.; Chen, J.; Li, D.Q. The Separation of Cerium(IV) from Nitric Acid Solutions Containing Thorium(IV) and Lanthanides(III) Using Pure [C8mim]PF6 as Extracting Phase. Ind. Eng. Chem. Res. 2008, 47, 2349–2355. [Google Scholar] [CrossRef]
- Villemejeanne, B.; Legeai, S.; Meux, E.; Dourdain, S.; Mendil-Jakani, H.; Billy, E. Halide based ionic liquid mixture for a sustainable electrochemical recovery of precious metals. Journal of Environmental Chemical Engineering 2021, 10, 107063. [Google Scholar] [CrossRef]
- Ouadi, A.; Klimchuk, O.; Gaillard, C.; Billard, I. Solvent extraction of U(vi) by task specific ionic liquids bearing phosphoryl groups. Green Chem. 2007, 9, 1160–1162. [Google Scholar] [CrossRef]
- Rout, A.; Binnemans, K. Solvent Extraction of Neodymium(III) by Functionalized Ionic Liquid Trioctylmethylammonium Dioctyl Diglycolamate in Fluorine-free Ionic Liquid Diluent. Ind. Eng. Chem. Res. 2014, 53, 6500–6508. [Google Scholar] [CrossRef]
- Llorens, I.; Solari, P.L.; Sitaud, B.; Bes, R.; Cammelli, S.; Hermange, H.; Othmane, G.; Safi, S.; Moisy, P.; Wahu, S.; et al. X-ray absorption spectroscopy investigations on radioactive matter using MARS beamline at SOLEIL synchrotron. Radiochimica Acta 2014, 102, 957–972. [Google Scholar] [CrossRef]
- Sitaud, B.; Solari, P.L.; Schlutig, S.; Llorens, I.; Hermange, H. Characterization of radioactive materials using the MARS beamline at the synchrotron SOLEIL. Journal of Nuclear Materials 2012, 425, 238–243. [Google Scholar] [CrossRef]
- Sukhbaatar, T.; Duvail, M.; Dumas, T.; Dourdain, S.; Arrachart, G.; Solari, P.L.; Guilbaud, P.; Pellet-Rostaing, S. Probing the existence of uranyl trisulfate structures in the AMEX solvent extraction process. Chem. Commun. 2019, 55, 7583–7586. [Google Scholar] [CrossRef] [PubMed]
- Hanke, K.; Kaufmann, M.; Schwaab, G.; Havenith, M.; Wolke, C.T.; Gorlova, O.; Johnson, M.A.; Kar, B.P.; Sander, W.; Sanchez-Garcia, E. Understanding the ionic liquid [NC4111][NTf2] from individual building blocks: an IR-spectroscopic study. Phys. Chem. Chem. Phys. 2015, 17, 8518–8529. [Google Scholar] [CrossRef] [PubMed]
- Servaes, K.; Hennig, C.; Billard, I.; Gaillard, C.; Binnemans, K.; Görller-Walrand, C.; Van Deun, R. Speciation of Uranyl Nitrato Complexes in Acetonitrile and in the Ionic Liquid 1-Butyl-3-methylimidazolium Bis(trifluoromethylsulfonyl)imide. European Journal of Inorganic Chemistry 2007, 2007, 5120–5126. [Google Scholar] [CrossRef]
- Görller-Walrand, C.; De Jaegere, S. Étude comparative des spectres d’absorption de complexes d’uranyle en solution et a l’état solide. Complexes de symétrie Cs, D2h (6) ET D3h (6). J. Chim. Phys. 1972, 69, 726–736. [Google Scholar] [CrossRef]
- Hennig, C.; Kraus, W.; Emmerling, F.; Ikeda, A.; Scheinost, A.C. Coordination of a Uranium(IV) Sulfate Monomer in an Aqueous Solution and in the Solid State. Inorg. Chem. 2008, 47, 1634–1638. [Google Scholar] [CrossRef] [PubMed]
- Lipovskii, A.A.; Kuzina, M.G. The Infrared Absorption Spectra and Structure of Tridecylammonium Sulphate, Tridecylammonium Hydrogensulphate, and Tridecylammonium Dioxotrisulphatouranate (VI). 1965, 10, 740–745. [Google Scholar]
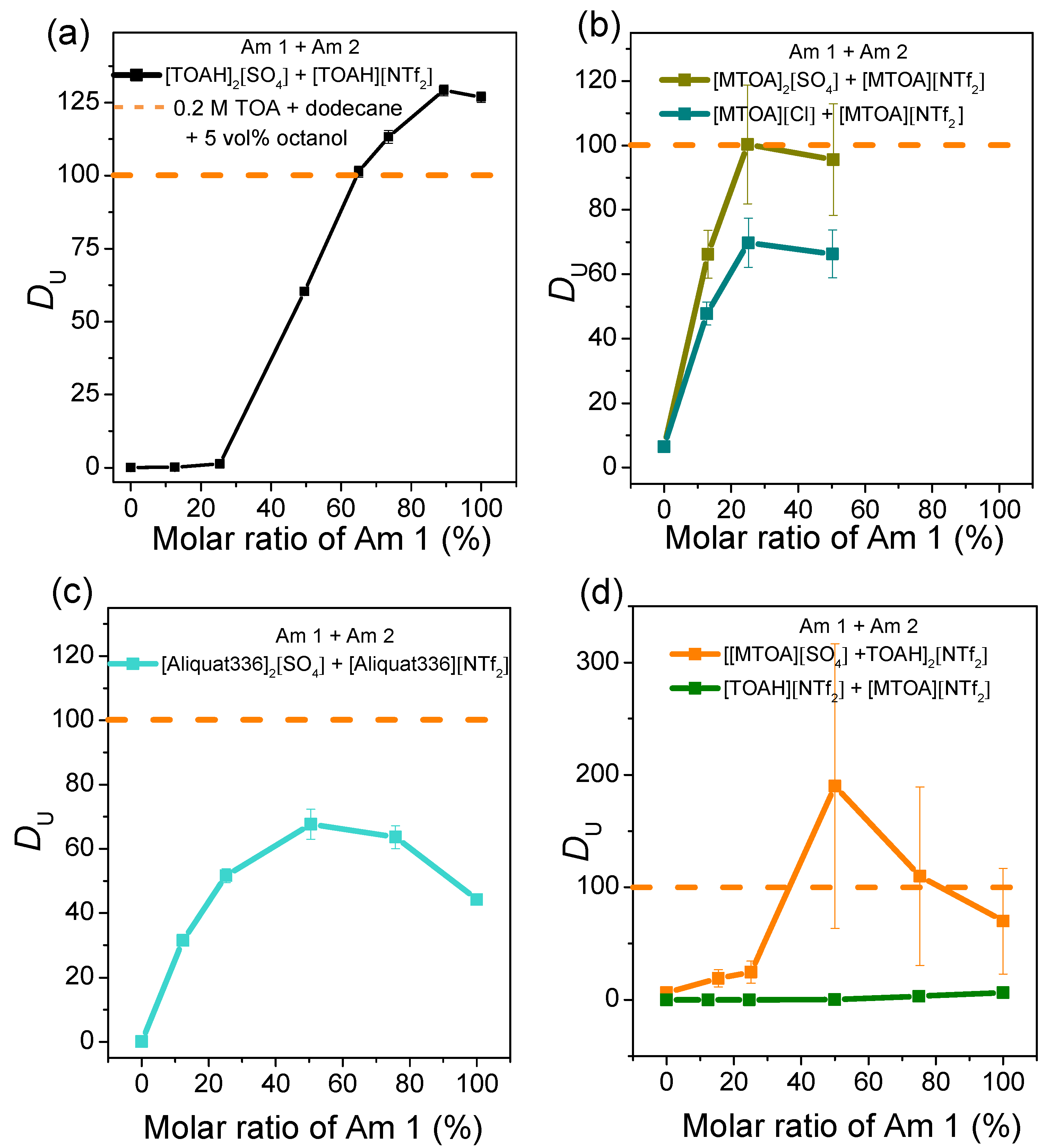
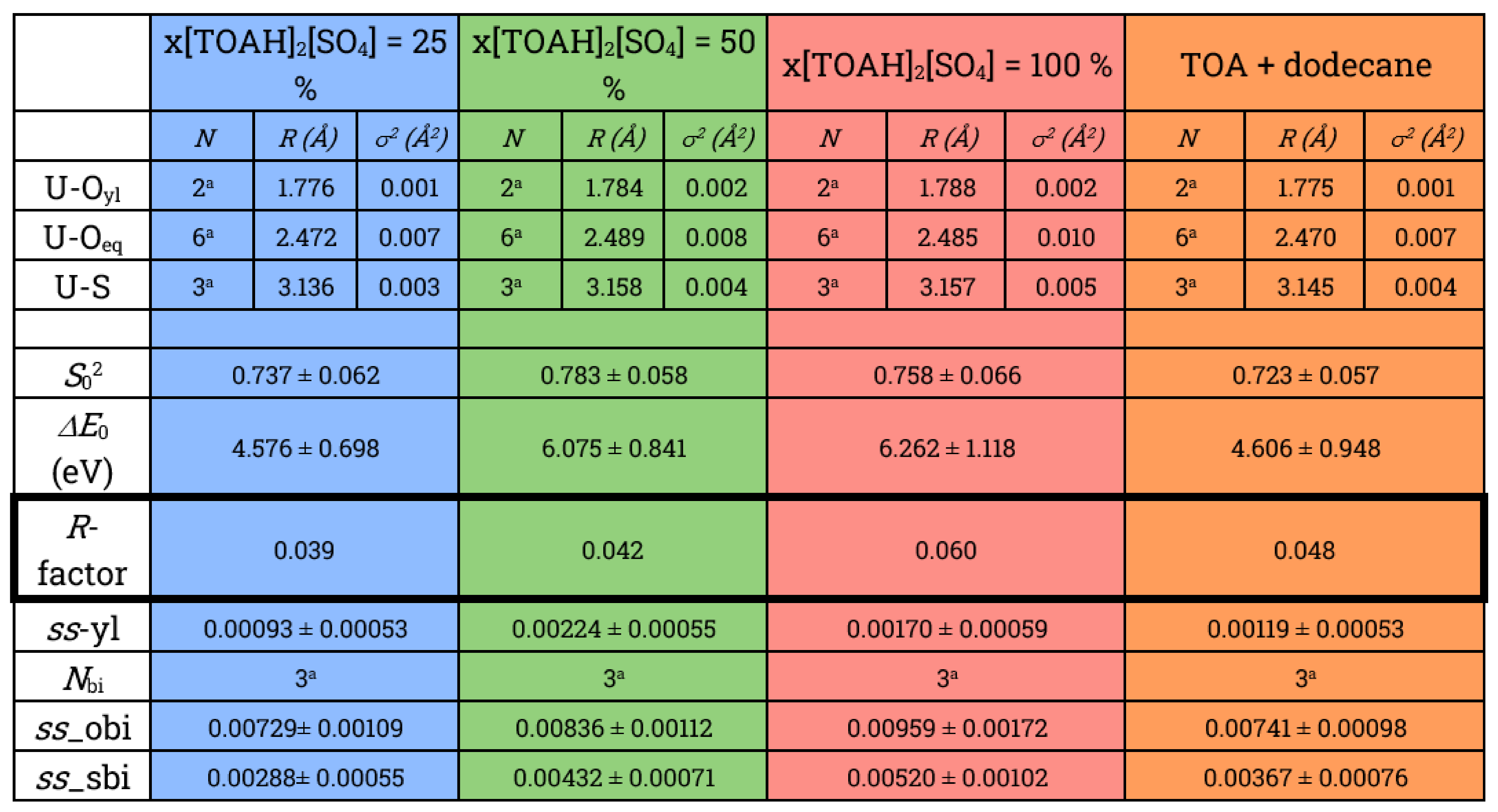
| [MTOA]2[SO4] + [MTOA][NTf2] | [MTOA][Cl] + [MTOA][NTf2] | [Aliquat 336]2[SO4] + [Aliquat 336][NTf2] | [MTOA][NTf2] + [TOAH]2[SO4] | [TOAH]2[SO4] + [TOAH][NTf2] | |
|---|---|---|---|---|---|
| Ratio (%) | 25 | 25 | 50 | 50 | 90 |
| DU | 100 | 70 | 78 | 196 | 128 |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




