Submitted:
27 July 2023
Posted:
28 July 2023
You are already at the latest version
Abstract
Keywords:
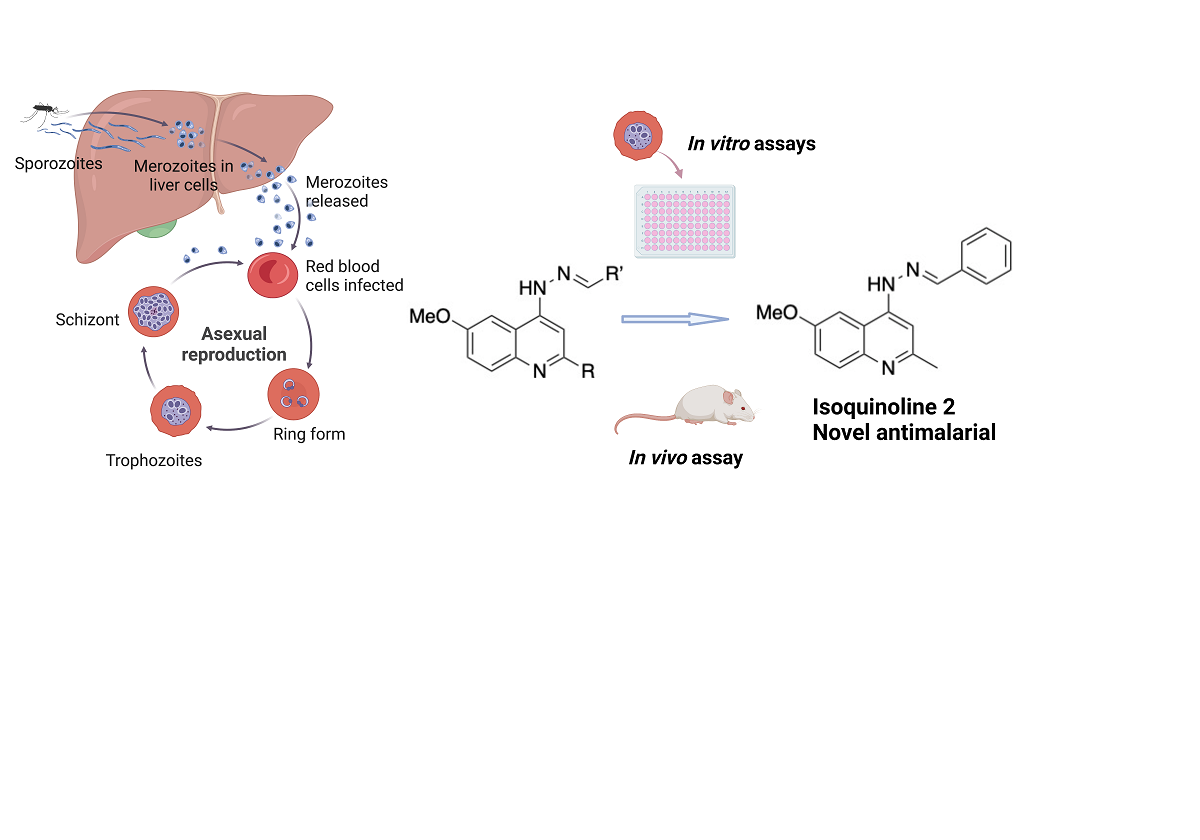
1. Introduction
2. Results
2.1. Determination of IC50 values for the activity of 4-aminoquinoline hydrazones against K1 strain of P. falciparum
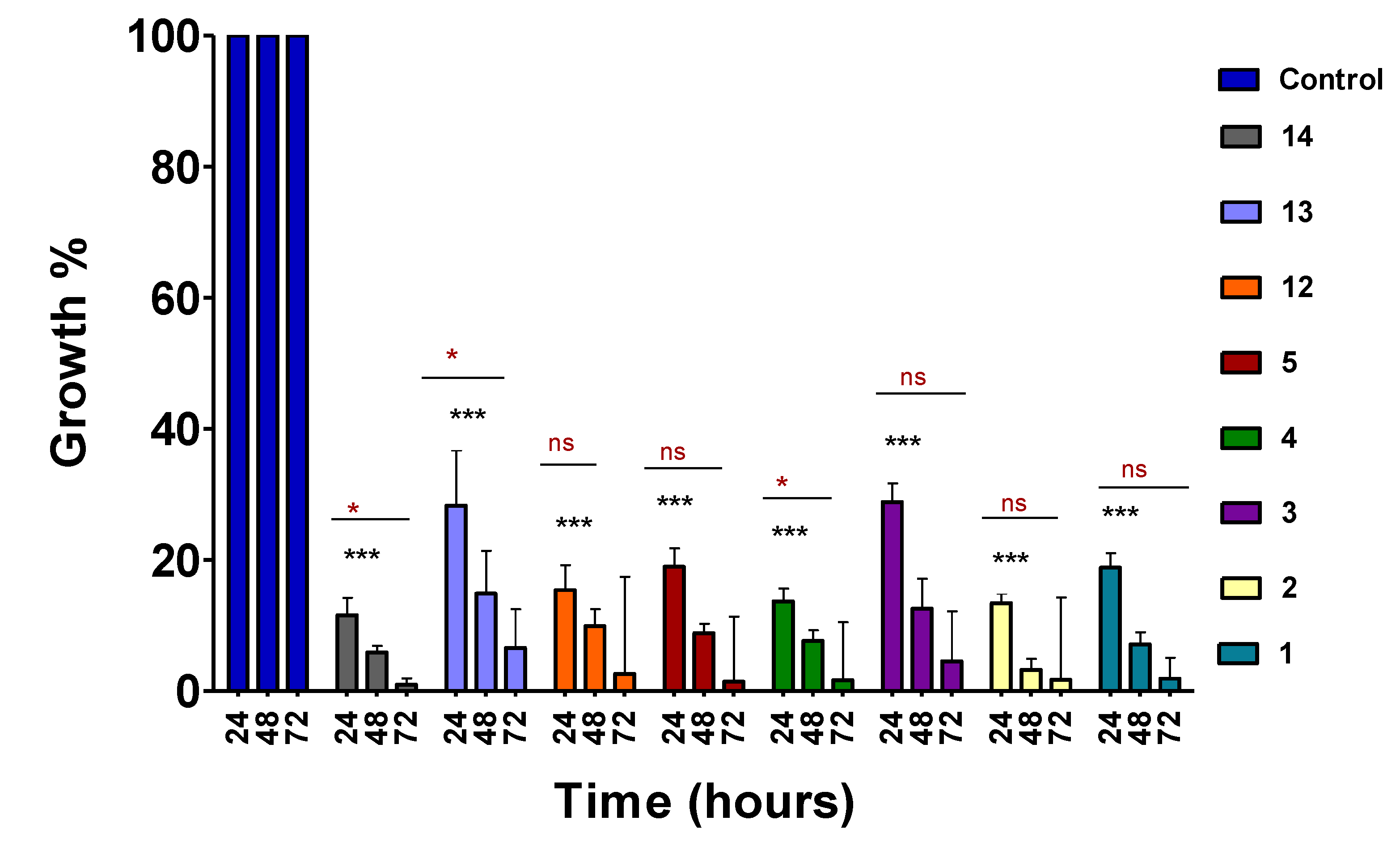
2.2. Time-course assay
2.3. Dose-response of lead compounds after 72 hours incubation period
2.4. MTT cytotoxicity assay
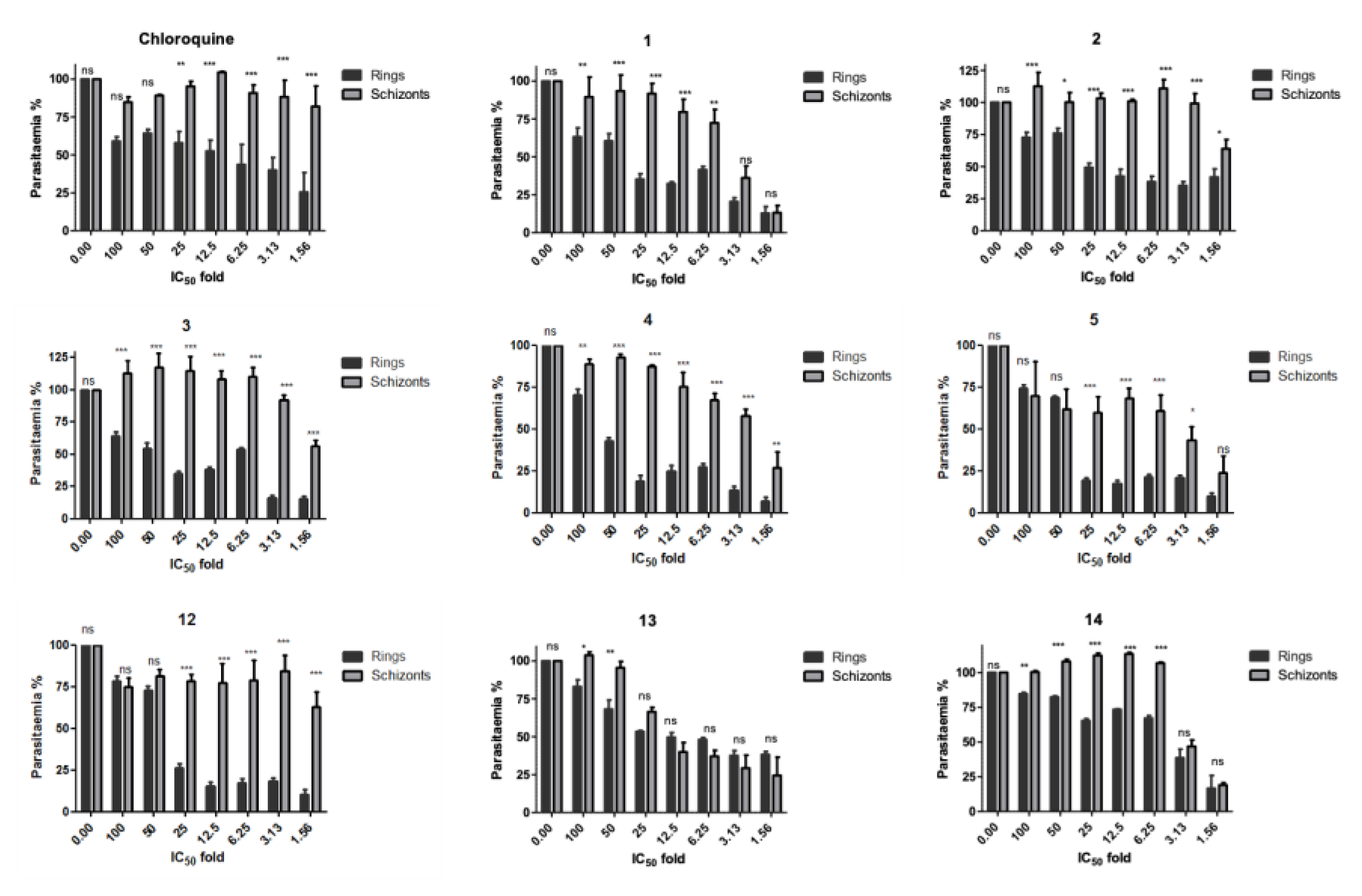
2.5. Stage–specificity analysis
2.6. In vitro IC50 values against 3D7 and Dd2 P. falciparum strains
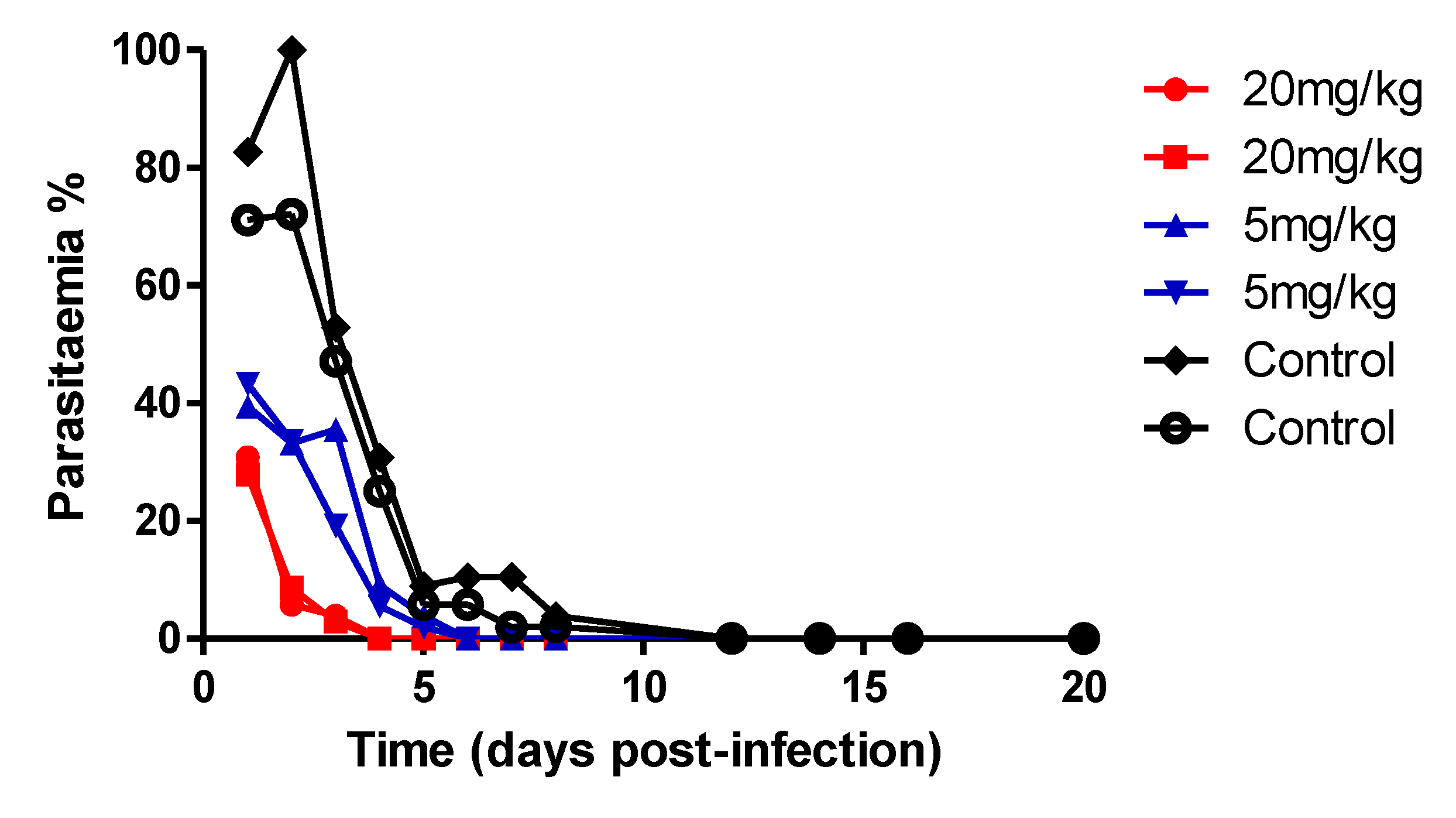
2.7. In vivo assay on P. yoelii mouse model
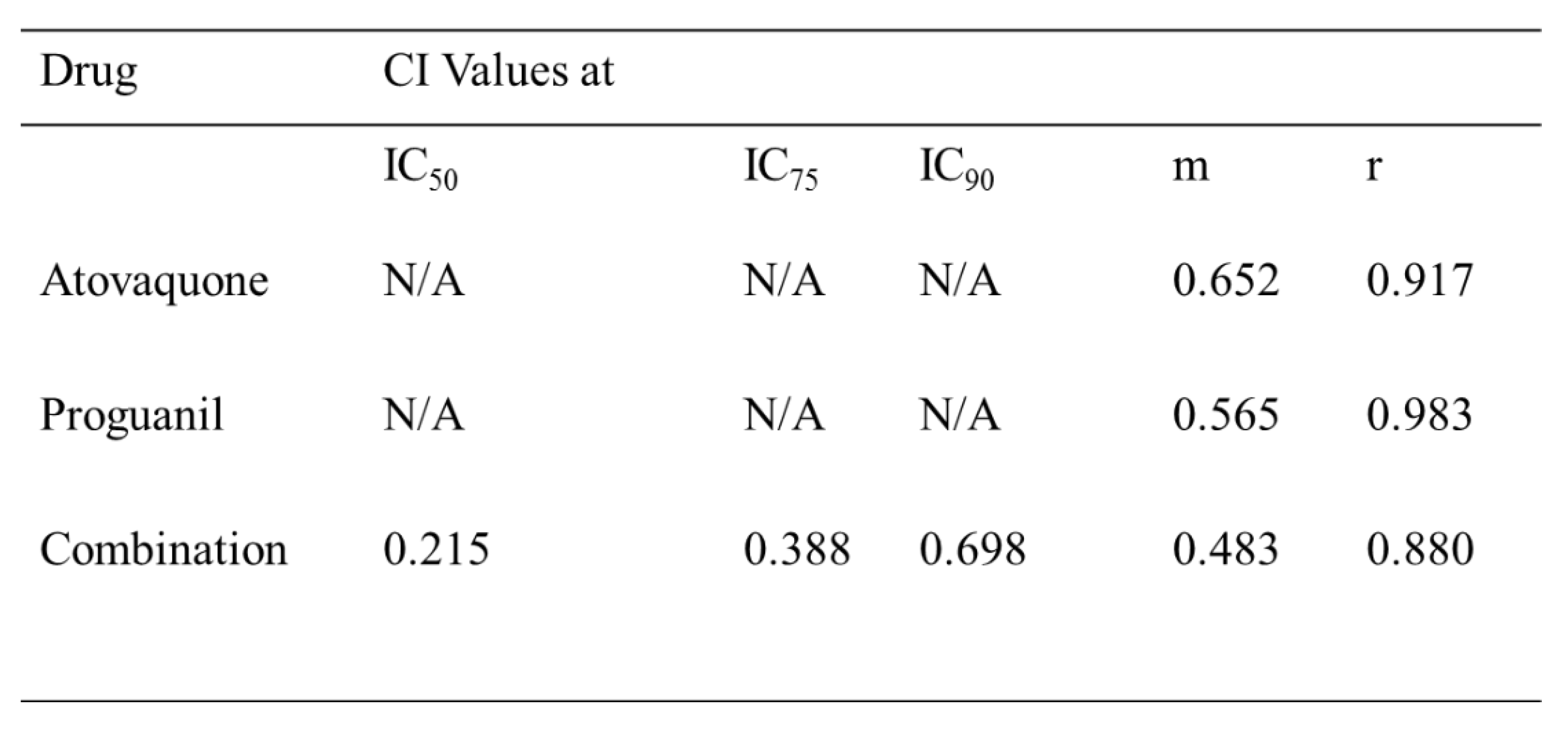
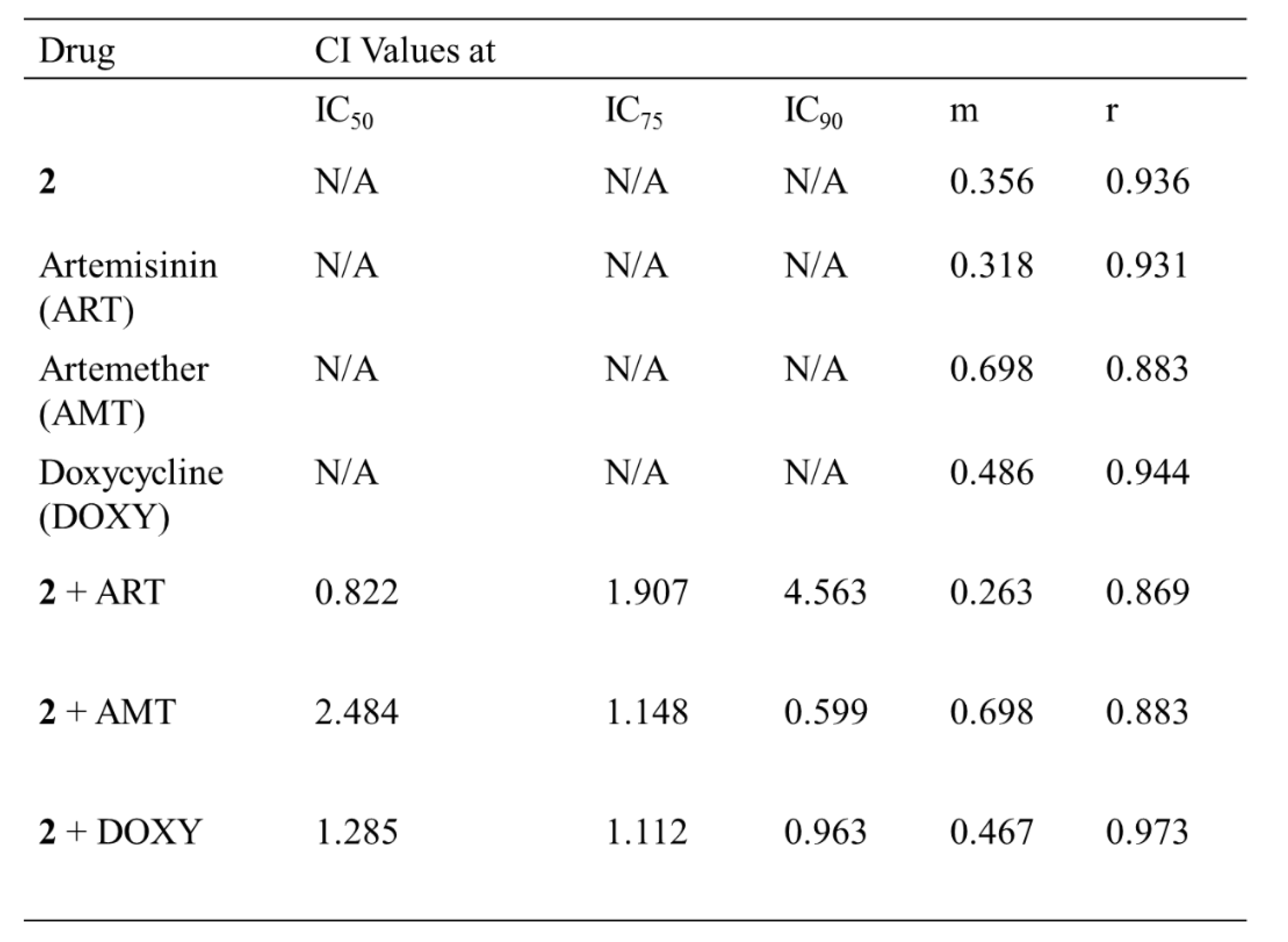
2.8. CalcuSyn assay for combination therapy assay
3. Discussion
4. Materials and Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization, “World Malaria Report 2022.”.
- C. J. Sutherland, N. Tanomsing, D. Nolder, M. Oguike, C. Jennison, S. Pukrittayakamee, C. Dolecek, T. T. Hien, V. E. do Rosário, A. P. Arez, J. Pinto, P. Michon, A. A. Escalante, F. Nosten, M. Burke, R. Lee, M. Blaze, T. D. Otto, J. W. Barnwell, A. Pain, J. Williams, N. J. White, N. P. J. Day, G. Snounou, P. J. Lockhart, P. L. Chiodini, M. Imwong, and S. D. Polley, “ Two Nonrecombining Sympatric Forms of the Human Malaria Parasite Plasmodium ovale Occur Globally ,” J. Infect. Dis., vol. 201, no. 10, pp. 1544–1550, 2010. [CrossRef]
- K. Karunamoorthi, “Vector control: A cornerstone in the malaria elimination campaign,” Clin. Microbiol. Infect., vol. 17, no. 11, pp. 1608–1616, 2011. [CrossRef]
- M. Kaiser, P. Mäser, L. P. Tadoori, J. R. Ioset, R. Brun, and D. J. Sullivan, “Antiprotozoal activity profiling of approved drugs: A starting point toward drug repositioning,” PLoS One, vol. 10, no. 8, pp. 1–10, 2015.
- World Health Organization, World Malaria Report 2019. Geneva. 2019. [CrossRef]
- E. B. Magbity, N. T. Marbiah, G. Maude, C. F. Curtis, D. J. Bradley, B. M. Greenwood, E. Petersen, and J. D. Lines, “Effects of community-wide use of lambdacyhalothrin-impregnated bednets on malaria vectors in rural Sierra Leone,” Med. Vet. Entomol., vol. 11, no. 1, pp. 79–86, 1997. [CrossRef]
- A. Ouattara and M. B. Laurens, “Vaccines against malaria,” Clin. Infect. Dis., vol. 60, no. 6, pp. 930–936, 2015.
- J. Okombo and K. Chibale, “Recent updates in the discovery and development of novel antimalarial drug candidates,” Medchemcomm, vol. 9, no. 3, pp. 437–453, 2018.
- S. R. Meshnick, T. E. Taylor, and S. Kamchonwongpaisan, “Artemisinin and the antimalarial endoperoxides: from herbal remedy to targeted chemotherapy.,” Microbiol. Rev., vol. 60, no. 2, pp. 301–315, 1996.
- H. Noedl, Y. Se, K. Schaecher, B. L. Smith, D. Socheat, and M. M. Fukuda, “Evidence of artemisinin-resistant malaria in Western Cambodia,” N. Engl. J. Med., vol. 359, no. 24, pp. 2619–2620, 2008. [CrossRef]
- A. M. Dondorp, F. Nosten, P. Yi, D. Das, A. P. Phyo, J. Tarning, D. Ph, K. M. Lwin, F. Ariey, W. Hanpithakpong, S. J. Lee, P. Ringwald, K. Silamut, T. Herdman, S. S. An, S. Yeung, D. Socheat, and N. J. White, “Artemisinin Resistance in,” Drug Ther. (NY)., vol. 361, no. 5, pp. 455–467, 2009.
- A. B. Panosian, “Economic Access to Effective Drugs for Falciparum Malaria,” Clin. Infect. Dis., vol. 40, no. 5, pp. 713–717, 2005. [CrossRef]
- R. Musiol, T. Magdziarz, and A. Kurczyk, “Quinoline scaffold as a privileged substructure in antimicrobial drugs,” Sci. against Microb. Pathog. Commun. Curr. Res. Technol. Adv., no. January, pp. 72–83, 2011.
- F. Loeb, W. M. Clark, G. R. Coatney, L. T. Coggeshall, F. R. Dieuaide, A. R. Dochez, E. G. Hakansson, E. K. Marshall Jr., C. S. Marvel, O. R. McCoy, J. J. Sapero, W. H. Sebrell, J. A. Shannon, and G. A. Carden Jr., “Activity of a new antimalarial agent, chloroquine (SN 7618): Statement approved by the board for coordination of malarial studies,” J. Am. Med. Assoc., vol. 130, no. 16, pp. 1069–1070, 1946.
- P. A. Winstanley, S. A. Ward, and R. W. Snow, “Clinical status and implications of antimalarial drug resistance,” Microbes Infect., vol. 4, no. 2, pp. 157–164, 2002. [CrossRef]
- K. Kaur, M. Jain, R. P. Reddy, and R. Jain, “Quinolines and structurally related heterocycles as antimalarials,” Eur. J. Med. Chem., vol. 45, no. 8, pp. 3245–3264, 2010. [CrossRef]
- B. Hussein, B. Ikhmais, M. Kadirvel, R. N. Magwaza, G. Halbert, R. A. Bryce, I. J. Stratford, and S. Freeman, “Discovery of potent 4-aminoquinoline hydrazone inhibitors of NRH:quinoneoxidoreductase-2 (NQO2),” Eur. J. Med. Chem., vol. 182, p. 111649, 2019. [CrossRef]
- N. Fisher, P. G. Bray, S. A. Ward, and G. A. Biagini, “The malaria parasite type II NADH : quinone oxidoreductase : an alternative enzyme for an alternative lifestyle,” TRENDS Parasitol., vol. 23, no. 7, 2007. [CrossRef]
- S. Kondaparla, P. Agarwal, K. Srivastava, S. K. Puri, and S. B. Katti, “Design, synthesis and in vitro antiplasmodial activity of some bisquinolines against chloroquine-resistant strain,” Chem. Biol. Drug Des., vol. 89, no. 6, pp. 901–906, 2017. [CrossRef]
- H. Matthews, J. Deakin, M. Rajab, M. Idris-Usman, and N. J. Nirmalan, “Investigating antimalarial drug interactions of emetine dihydrochloride hydrate using CalcuSyn-based interactivity calculations,” PLoS One, vol. 12, no. 3, pp. 1–19, 2017.
- P. Panwar, K. K. Burusco, M. Abubaker, H. Matthews, A. Gutnov, E. Fernández-Álvaro, R. A. Bryce, J. Wilkinson, and N. Nirmalan, “Lead optimization of dehydroemetine for repositioned use in malaria,” Antimicrob. Agents Chemother., vol. 64, no. 4, pp. 1–22, 2020.
- T. C. Chou, “Drug combination studies and their synergy quantification using the chou-talalay method,” Cancer Res., vol. 70, no. 2, pp. 440–446, 2010. [CrossRef]
- M. A. Biamonte, J. Wanner, and K. G. Le Roch, “Recent advances in malaria drug discovery,” Bioorganic Med. Chem. Lett., vol. 23, no. 10, pp. 2829–2843, 2013. [CrossRef]
- J. N. Burrows, R. Hooft Van Huijsduijnen, J. J. Möhrle, C. Oeuvray, and T. N. Wells, “Designing the next generation of medicines for malaria control and eradication,” Malar. J., vol. 12, no. 1, pp. 1–20, 2013.
- D. A. Fidock, P. J. Rosenthal, S. L. Croft, R. Brun, and S. Nwaka, “Antimalarial drug discovery: Efficacy models for compound screening,” Nat. Rev. Drug Discov., vol. 3, no. 6, pp. 509–520, 2004. [CrossRef]
- R. T. Eastman and D. A. Fidock, “Artemisinin-based combination therapies: A vital tool in efforts to eliminate malaria,” Nat. Rev. Microbiol., vol. 7, no. 12, pp. 864–874, 2009. [CrossRef]
- H. A. Poindexter, “Human Malaria Parasites in Continuous Culture,” J. Natl. Med. Assoc., vol. 68, no. 6, pp. 530–533, 1976.
- C. Le Manach, C. Scheurer, S. Sax, S. Schleiferböck, D. G. Cabrera, Y. Younis, T. Paquet, L. Street, P. Smith, X. C. Ding, D. Waterson, M. J. Witty, D. Leroy, K. Chibale, and S. Wittlin, “Fast in vitro methods to determine the speed of action and the stage-specificity of anti-malarials in Plasmodium falciparum,” Malar. J., vol. 12, no. 1, pp. 1–7, 2013. [CrossRef]
- H. Matthews, M. Usman-idris, F. Khan, M. Read, and N. Nirmalan, “Drug repositioning as a route to anti-malarial drug discovery : preliminary investigation of the in vitro anti-malarial efficacy of emetine dihydrochloride hydrate,” Malar. J., vol. 12, no. 395, pp. 1–11, 2013. [CrossRef]
- T. C. Chou, “Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies,” Pharmacol. Rev., vol. 58, no. 3, pp. 621–681, 2006. [CrossRef]
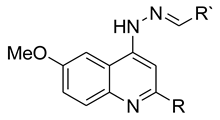 | |||||
| Plasmodium IC50(µM) ± SE | Cell toxicity IC50(µM) ± SE | Cell toxicity IC50(µM) ± SE | |||
| Compound | R | R’ | HepG2 | MDBK | |
| 1 | -Me | 4-Fluorophenyl | 0.612 ± 0.35 | 0.872 ± 0.79 | 3.77 ± 1.5 |
| 2 | -Me | Phenyl | 2.26 ± 2.2 | 1.90 ± 0.11 | 3.44 ± 2.7 |
| 3 | -Me | 3-Pyridinyl | 3.29 ± 1.6 | 3.00 ± 0.27 | 7.34 ± 7.6 |
| 4 | -Me | 2-Nitrofuranyl | 0.600 ± 0.84 | 1.59 ± 0.18 | 1.66 ± 0.21 |
| 5 | -Me | 2-Hydroxy-3-methoxyphenyl | 3.82 (wide) | 6.73 ± 0.73 | >25 |
| 6 | -Me | 4-Imidazoyl | 48.8 (wide) | 7.26 ± 1.00 | 11.7 ± 2.5 |
| 7 | -Me | 4-Nitrophenyl | 20.1 (wide) | 1.69 (wide) | 6.68 ± 1.7 |
| 8 | -Me | 4-Hydroxyphenyl | 22.3 (wide) | 1.97 ± 0.42 | 3.73 ± 1.2 |
| 9 | -Me | 4-Benzoic acid | >200 | >25 | >25 |
| 10 | -Me | 3-Hydroxyphenyl | 18.9 (wide) | 2.64 ± 0.20 | 9.71 ± 3.5 |
| 11 | -Me | 3,5-Dihydroxylphenyl | 35.9 ± 43 | 11.1 ± 2.9 | >25 |
| 12 | -Ph | 2-Hydroxy-3-methoxyphenyl | 5.67 ± 13 | 1.80 ± 0.78 | 6.60 |
| 13 | -Ph | Benzyl | 3.33 ± 2.4 | >25 | >25 |
| 14 | -Ph | 4-Nitrophenyl | 2.20 ± 1.5 | 0.89 ± 0.13 | 4.24 ± 1.0 |
| 15 | -Ph | 4-Imidazoyl | 28.3 ± 29 | 2.21 ± 0.15 | 9.31 ± 3.5 |
| 16 | -Ph | 4-N,N-dimethylaniline | 8.06 ± 4.5 | 4.15 ± 2.4 | 6.24 (wide) |
| Chloroquine | 0.199 ± 0.04 | ||||
| Cisplatin | 3.92 ± 0.64 | 9.15 ± 3.5 | |||
| Compound |
Plasmodium IC50 (µM) ± SE |
Selectivity indices (MDBK) |
Selectivity indices (HepG2) |
| 1 | 0.0257 ± 0.14 | 147 | 34 |
| 2 | 0.0329 ± 0.007 | 101 | 58 |
| 3 | 0.175 ± 0.04 | 42 | 17 |
| 4 | 0.219 ± 0.02 | 7 | 7 |
| 5 | 0.174 ± 0.03 | N/Aa | 39 |
| 12 | 0.133 ± 0.06 | 50 | 14 |
| 13 | 1.61 (wide) | N/Aa | N/Aa |
| 14 | 0.176 ± 0.06 | 31 | 5 |
| Compounds |
3D7 |
Dd2 IC50 ± SE (µM) |
K1 |
| 1 | 0.183 ± 1.04 | 0.133 ± 1.18 | 0.0257 ± 0.14 |
| 2 | 0.0554 ± 1.04 | 0.0244 ± 1.13 | 0.0329 ± 0.007 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




