Submitted:
21 July 2023
Posted:
24 July 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Local infiltration analgesia (LIA)
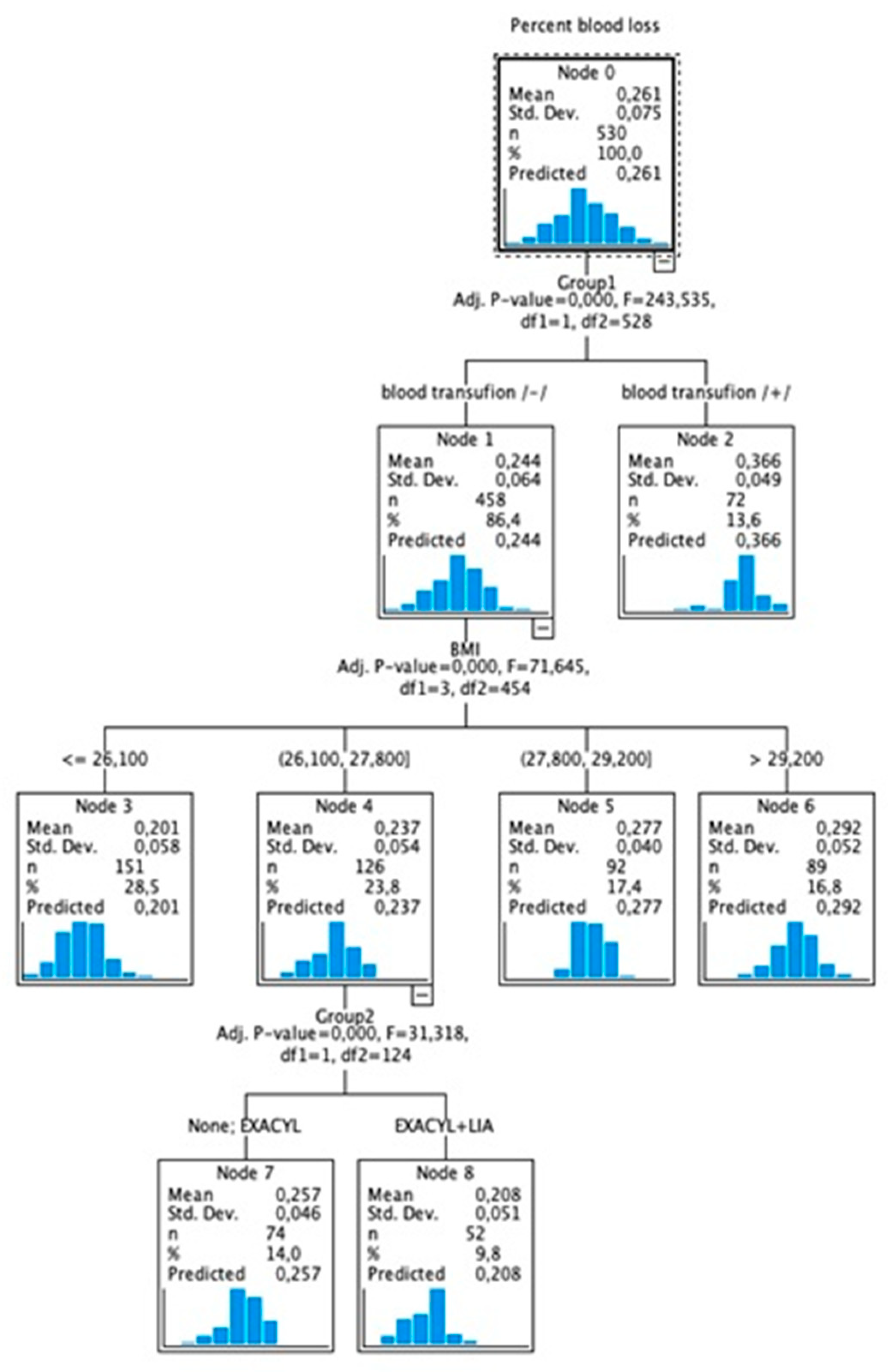
2. Materials and methods
2.1. Surgical technique
3. Results
3.1. Complications
3.2. Statistical analysis
4. Discussion
4.1. Study limitations
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Consent to Participate
Consent to Publish
Ethical Approval
Abbreviations
| TKA | Total knee arthroplasty. |
| THA | Total hip arthroplasty. |
| TXA | Tranexamic acid. |
| LIA | Local infiltration analgesia. |
| NSAIDs | Non-steroidal anti-inflammatory drugs. |
| STROBE | Strengthening the Reporting of Observational Studies in Epidemiology. |
| LMWH | Low Molecular Weight Heparin. |
| I&D | Irrigation & debridement procedure. |
| VTED | Venous thromboembolic disease. |
| DVT | Deep venous thrombosis. |
| PE | Pulmonary embolism. |
References
- Park, J.H.; Rasouli, M.R.; Mortazavi, S.M.; et al. Predictors of perioperative blood loss in total joint arthroplasty. J Bone Joint Surg AM 2013, 95, 1777–83. [Google Scholar] [CrossRef]
- Schwab, P.E.; Lavand'homme, P.; Yombi, J.C.; Thienpont, E. Lower blood loss after unicompartmental than total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc 2015, 23, 3494–500. [Google Scholar] [CrossRef] [PubMed]
- Spahn, D.R. Anemia and patient blood management in hip and knee surgery: a systematic review of the literature. Anesthesiology 2010, 113, 482–495. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Kapadia, B.H.; Issa, K.; et al. Postoperative blood loss prevention in total knee arthroplasty. J Knee Surg 2013, 26, 395–400. [Google Scholar] [CrossRef]
- Vamvakas, E.C.; Blajchman, M.A. Transfusion-related mortality: the ongoing risks of allogeneic blood transfusion and the available strategies for their prevention. Blood 2009, 113, 3406–17. [Google Scholar] [CrossRef]
- Shander, A.; Hofmann, A.; Ozawa, S.; et al. Activity-based costs of blood transfusions in surgical patients at four hospitals. Transfusion 2010, 50, 753–65. [Google Scholar] [CrossRef]
- Grant, A.L.; Letson, H.L.; Morris, J.L.; McEwen, P.; Hazratwala, K.; Wilkinson, M.; Dobson, G.P. Tranexamic acid is associated with selective increase in inflammatory markers following total knee arthroplasty (TKA): a pilot study. J Orthop Surg Res 2018, 13, 149. [Google Scholar] [CrossRef]
- Tengborn, L.; Blomback, M.; Berntorp, E. Tranexamic acid–an old drug still going strong and making a revival. Thromb Res 2015, 135, 231–242. [Google Scholar] [CrossRef]
- Eubanks, J.D. Antifibrinolytics in major orthopaedic surgery. J Am Acad orthop surg 2010, 18, 132–138. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Model List of Essential Medicines: 21st List 2019; World Health Organization: Geneva, 2019. [Google Scholar]
- Wei, W.; Dang, S.; Duan, D.; Wei, L. Comparison of intravenous and topical tranexamic acid in total knee arthroplasty. BMC Musculoskelet Disord 2018, 19, 191. [Google Scholar] [CrossRef]
- Wang, H.; Shen, B.; Zeng, Y. Comparison of topical versus intravenous tranexamic acid in primary total knee arthroplasty: a meta-analysis of randomized controlled and prospective cohort trials. Knee 2014, 21, 987–993. [Google Scholar] [CrossRef]
- Laoruengthana, A.; Rattanaprichavej, P.; Rasamimongkol, S.; Galassi, M.; Weerakul, S.; Pongpirul, K. Intra-articular tranexamic acid mitigates blood loss and morphine use after total knee arthroplasty. A randomized controlled trial. J Arthroplasty 2019, 34, 877–881. [Google Scholar]
- Whiting, D.R.; Sierra, R.J. Efficacy of combined use of intraarticular and intravenous tranexamic acid in total knee arthroplasty. Ann Transl Med 2015, 3 (Suppl 1), S39. [Google Scholar]
- Akgul, T.; Buget, M.; Salduz, A.; et al. Efficacy of preoperative admin- istration of single high dose intravenous tranexamic acid in reducing blood loss in total knee arthroplasty: a prospective clinical study. Acta Orthop Traumatol Turc 2016, 50, 429–431. [Google Scholar] [CrossRef]
- Lei, Y.; Xie, J.; Huang, Q.; Huang, W.; Pei, F. Additional benefits of multiple-dose tranexamic acid to anti-fibrinolysis and antiinflammation in total knee arthroplasty: a randomized controlled trial. Arch Orthop Trauma Surg 2020, 140, 1087–1095. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Li, B.; Wang, Q.; et al. Comparison of 3 routes of administration of tranexamic acid on primary unilateral total knee arthroplasty: a prospective, randomized, controlled study. J Arthroplasty 2017, 32, 2738–43. [Google Scholar] [CrossRef]
- Lee, Q.J.; Ching, W.Y.; Wong, Y.C. Blood sparing efficacy of oral tranexamic acid in primary total knee arthroplasty: a randomized controlled trial. Knee Surg Relat Res 2017, 29, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Kerr, D.R.; Kohan, L. Local infiltration analgesia: a technique for the control of acute postoperative pain following knee and hip surgery: a case study of 325 patients. Acta Orthop 2008, 79, 174–8. [Google Scholar] [CrossRef] [PubMed]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. ; STROBE Initiative The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. Lancet 2007, 370, 1453–1457. [Google Scholar] [CrossRef]
- Hourlier, H.; Reina, n.; Fennema, P. Single dose intravenous tranexamic acid as effective as continuous infusion in primary total knee arthroplasty: a randomised clinical trial. Arch orthop trauma surg 2015, 135, 465–471. [Google Scholar] [CrossRef]
- Lin, s.Y.; Chen, C.H.; Fu, Y.C.; et al. The efficacy of combined use of intraarticular and intravenous tranexamic acid on reducing blood loss and transfusion rate in total knee arthroplasty. J Arthroplasty 2015, 30, 776–80. [Google Scholar] [CrossRef]
- Levine, B.R.; Haughom, B.D.; Belkin, M.n.; et al. Weighted versus uniform dose of tranexamic acid in patients undergoing pri- mary, elective knee arthroplasty: a prospective randomized controlled trial. J Arthroplasty 2014, 29 (9 suppl), 186–188. [Google Scholar] [CrossRef]
- Iwai, T.; Tsuji, S.; Tomita, T.; et al. Repeat-dose intravenous tranexamic acid further decreases blood loss in total knee arthroplasty. Int orthop 2013, 37, 441–445. [Google Scholar] [CrossRef]
- Shen, P.F.; Hou, W.L.; Chen, J.B.; et al. Effectiveness and safety of tranexamic acid for total knee arthroplasty: a prospective randomized controlled trial. Med sci Monit 2015, 21, 576–581. [Google Scholar] [CrossRef]
- Karaaslan, F.; Karaoğlu, S.; Mermerkaya, M.U.; et al. Reducing blood loss in simultaneous bilateral total knee arthroplasty: combined intravenous-intra-articular tranexamic acid admin- istration. A prospective randomized controlled trial. Knee 2015, 22, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, X.; Martínez-Zapata, M.J.; Hinarejos, P.; et al. Topical and intravenous tranexamic acid reduce blood loss compared to routine hemostasis in total knee arthroplasty: a multicenter, randomized, controlled trial. Arch orthop trauma surg 2015, 135, 1017–1025. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.P.; nisthane, P.P.; Shah, N.A. Combined administration of systemic and topical tranexamic acid for total knee arthroplasty: can it be a better regimen and yet safe? A randomized controlled trial. J Arthroplasty 2016, 31, 542–547. [Google Scholar]
- Marra, F.; Rosso, F.; Bruzzone, M.; Bonasia, D.E.; Dettoni, F.; Rossi, R. Use of tranexamic acid in total knee arthroplasty. Joints 2017, 4, 202–213. [Google Scholar] [CrossRef] [PubMed]
- Shahi, A.; Chen, A.F.; Tan, T.L.; Maltenfort, M.G.; Kucukdurmaz, F.; Parvizi, J. The incidence and economic burden of in-hospital venous throm- boembolism in the United States. J Arthroplasty 2017, 32, 1063–1066. [Google Scholar] [CrossRef]
- Zhang, Z.H.; Shen, B.; Yang, J.; Zhou, Z.K.; Kang, P.D.; Pei, F.X. Risk factors for venous thromboembolism of total hip arthroplasty and total knee arthroplasty: a systematic review of evidences in ten years. BMC Musculoskelet Disord 2015, 16, 24. [Google Scholar] [CrossRef]
- Bjerke-Kroll, B.T.; Sculco, P.K.; McLawhorn, A.S.; Christ, A.B.; Gladnick, B.P.; Mayman, D.J. The increased total cost associated with post-operative drains in total hip and knee arthroplasty. J Arthroplasty 2014, 29, 895–899. [Google Scholar] [CrossRef]
- Ovadia, D.; Luger, E.; Bickels, J.; Menachem, A.; Dekel, S. Efficacy of closed wound drainage after total joint arthroplasty. A prospective randomized study. J Arthroplasty 1997, 12, 317–321. [Google Scholar] [CrossRef]
- Kumar, S.; Penematsa, S.; Parekh, S. Are drains required fol- lowing a routine primary total joint arthroplasty? Int Orthop 2007, 31, 593–596. [Google Scholar] [CrossRef]
- Omonbude, D.; El Masry, M.A.; O’Connor, P.J.; Grainger, A.J.; Allgar, V.L.; Calder, S.J. Measurement of joint effusion and haematoma formation by ultrasound in assessing the effectiveness of drains after total knee replacement: a prospective randomised study. J Bone Joint Surg (Br) 2010, 92, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, R.O.; Parkinson, R.W. Closed suction drains do not increase the blood transfusion rates in patients undergoing total knee arthroplasty. Int Orthop 2007, 31, 613–616. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.; Ramankutty, S.; Board, T.; et al. Does intraarticular steroid infiltration increase the rate of infection in subsequent total knee replacements. Knee 2009, 16, 262–264. [Google Scholar] [CrossRef]
- Fan, L.; Zhu, C.; Zan, P.; Yu, X.; Liu, J.; Sun, Q.; Li, G. The Comparison of Local Infiltration Analgesia with Peripheral Nerve Block following Total Knee Arthroplasty (TKA): A Systematic Review with Meta-Analysis. J Arthroplasty 2015, 30, 1664–71. [Google Scholar] [CrossRef] [PubMed]
- Innerhofer, P.; Klingler, A.; Klimmer, C.; Fries, D.; Nussbaumer, W. Risk for postoperative infection after transfusion of white blood cell-filtered allogeneic or autologous blood components in orthopedic patients undergoing primary arthroplasty. Transfusion 2005, 45, 103–10. [Google Scholar] [CrossRef]
- Başdelioğlu, K. Effects of body mass index on outcomes of total knee arthroplasty. Eur J Orthop Surg Traumatol 2021, 31, 595–600. [Google Scholar] [CrossRef]

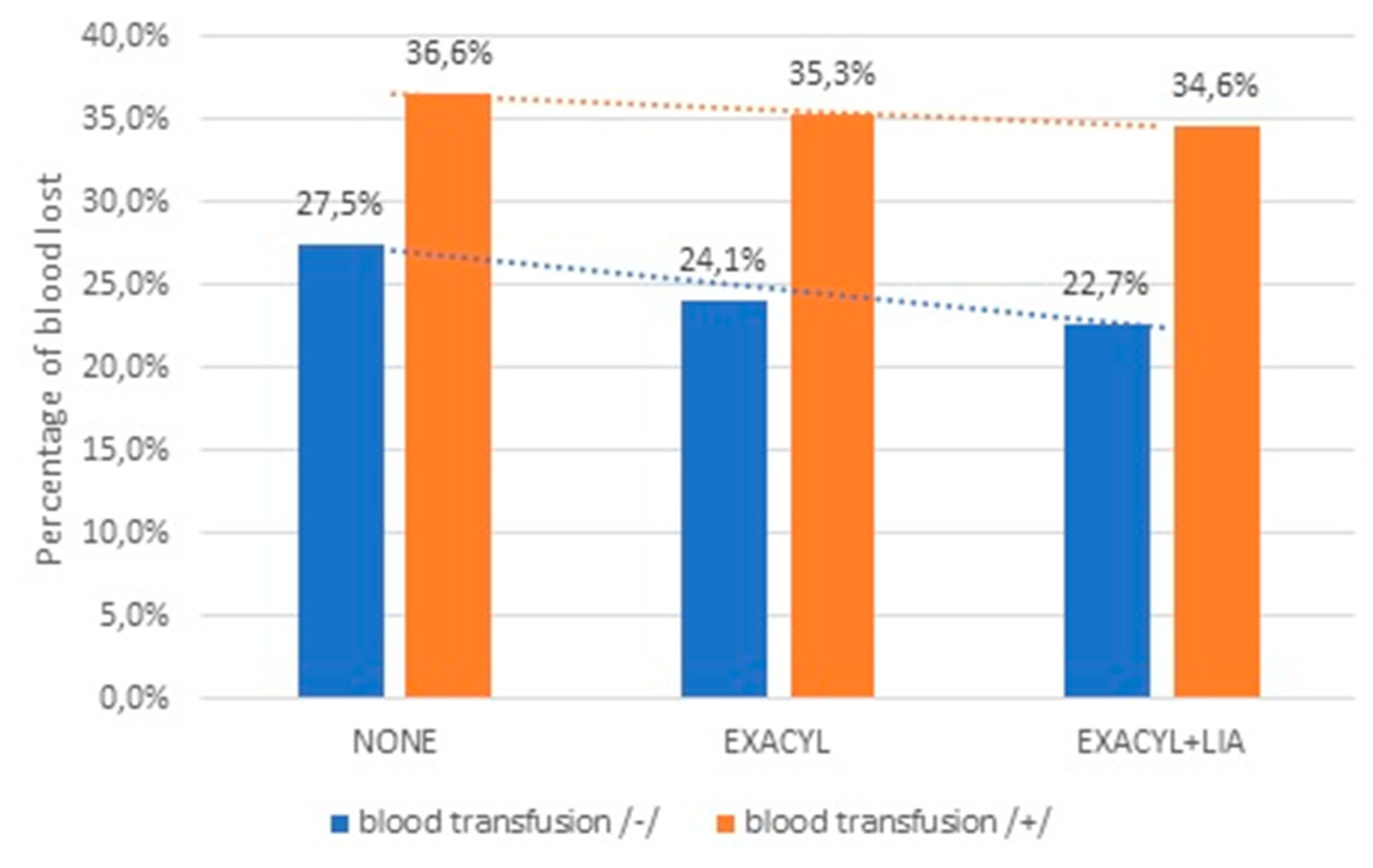
| No protocol group | TXA group | TXA+ LIA group | ||||
| blood transfusion /+/ | blood transfusion /-/ | blood transfusion /+/ | blood transfusion /-/ | blood transfusion /+/ | blood transfusion /-/ | |
| No | 40 | 124 | 14 | 127 | 18 | 207 |
| Age [mean] | 70,82 |
68,98 |
75,93 |
69,77 |
72,61 |
70,57 |
| Sex [M/F] | 8 / 32 | 34 / 90 | 2 / 12 | 40 / 87 | 2 / 16 | 53 / 154 |
| BMI | 27,625 (SD 1,93) |
27,04 (SD 1,99) | 27,80 (SD 1,98) | 26,97 (SD 2,03) | 27,96 (SD 1,59) | 27,46 (SD 2,3) |
| Hospital stay [day] | 7,32 (SD 1,55) |
6,92 (SD 1,26) |
6,71 (SD 0,82) |
6,01 (SD 1,06) |
5,88 (SD 0,9) |
5,54 (SD 0,78) |
| Preoperative Hb (g/dl) | 13,36 (SD 0,95) | 14,13 (SD 1,15) |
12,83 (SD 0,74) |
14,13 (SD 1,18) |
12,37 (SD 1,19) |
13,81 (SD 1,15) |
| Lowest Hb (g/dl) | 8,25 (SD 0,51) |
10,24 (SD 1,09) |
8,3 (SD 0,36) |
10,71 (SD 1,14) |
8,06 (SD 0,69) |
10,67 (SD 1,21) |
|
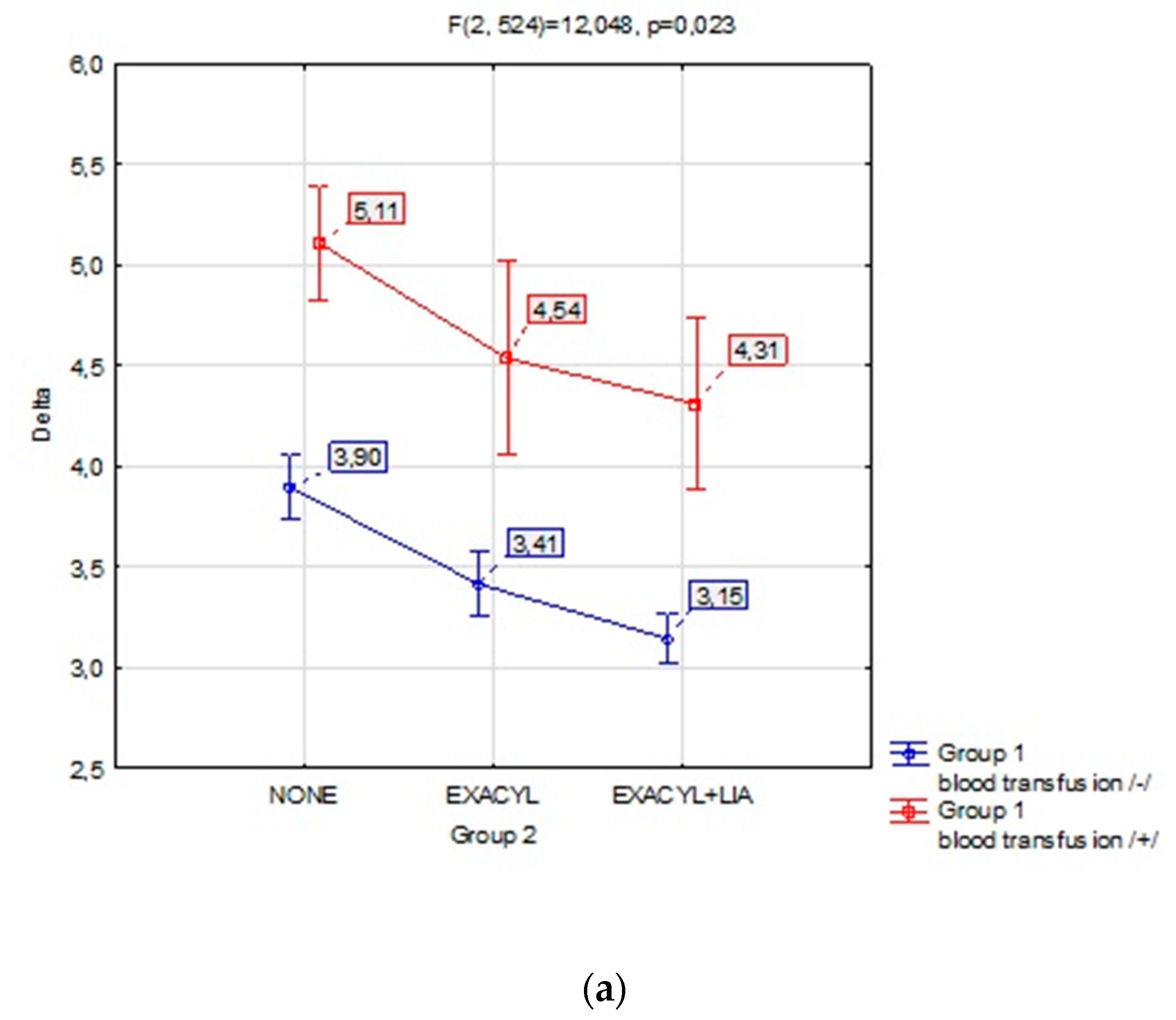
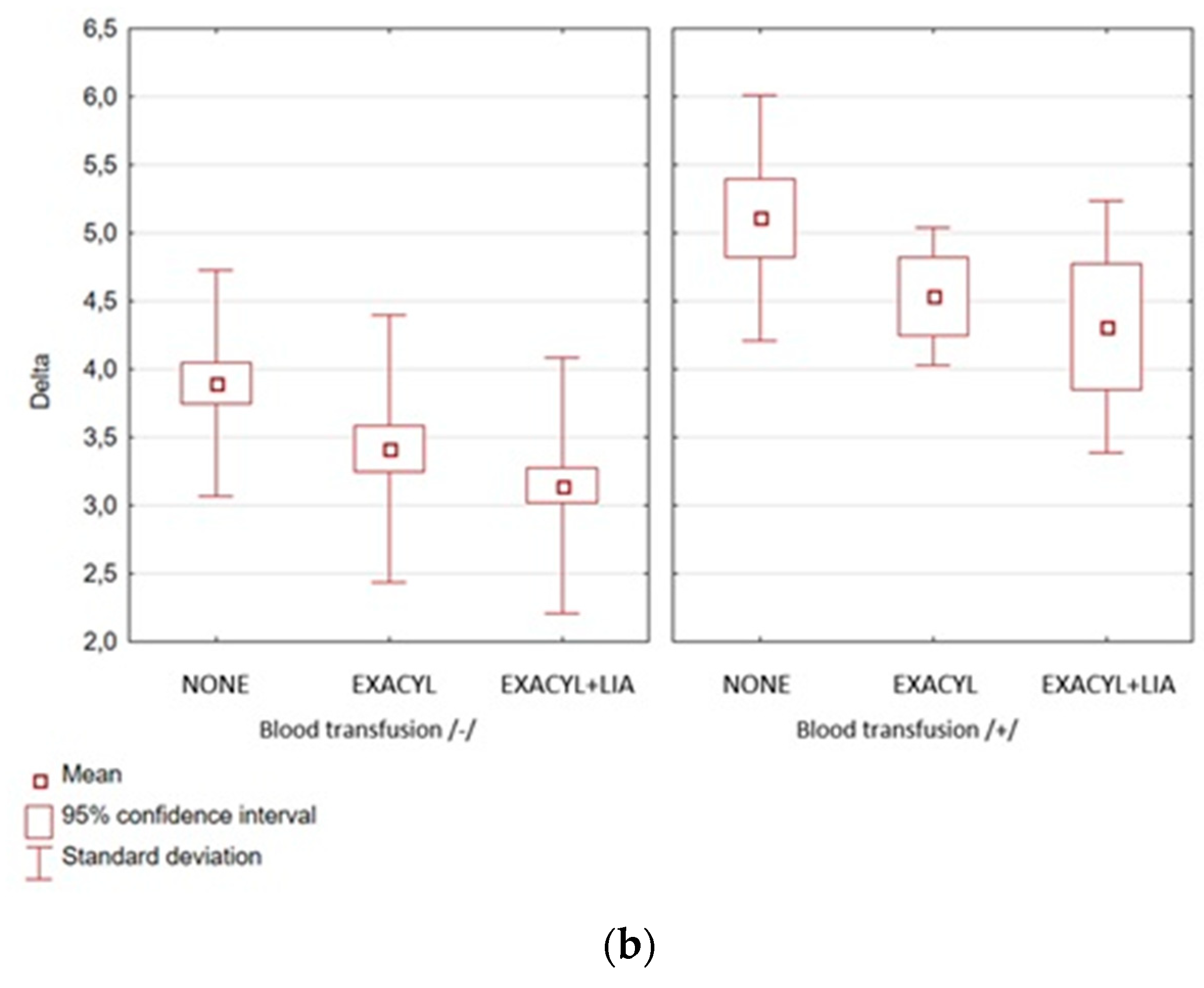
Maximum Hb loss (g/dl) |
5,11 (SD 0,90) |
3,89 (SD 0,82) |
4,53 (SD 0,50) |
3,41 (SD 0,98) |
4,31 (SD 0,92) |
3,14 (SD 0,90) |
| blood transfusion /-/ | blood transfusion /+/ | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| hemoglobin (Hb-g/dl) | decrease | % | hemoglobin (Hb-g/dl) | decrease | % | ||||
| post-op | pre-op | decrease | post-op | pre-op | decrease | ||||
| Control group | M | 10,2 | 14,1 | 3,9 | 27,5% | 8,3 | 13,4 | 5,1 | 38,0% |
| SD | 1,1 | 1,2 | 0,8 | 5,2% | 0,5 | 1 | 0,9 | 4,9% | |
| MD | 10,1 | 14 | 3,8 | 26,6% | 8,5 | 13,5 | 5,1 | 37,0% | |
| MIN | 8,3 | 11,9 | 1,9 | 13,7% | 7 | 10,3 | 2 | 19,4% | |
| MAX | 15,2 | 19 | 6,5 | 40,9% | 8,9 | 15,4 | 7,3 | 49,3% | |
| N | 124 | 40 | |||||||
| TXA | M | 10,7 | 14,1 | 3,4 | 24,1% | 8,3 | 12,8 | 4,5 | 35,3% |
| SD | 1,1 | 1,2 | 1 | 6,3% | 0,4 | 0,7 | 0,5 | 2,2% | |
| MD | 10,7 | 14 | 3,5 | 24,6% | 8,4 | 12,7 | 4,5 | 35,4% | |
| MIN | 8,8 | 10,9 | 0,4 | 3,4% | 7,5 | 11,8 | 3,8 | 32,2% | |
| MAX | 13,5 | 17 | 5,6 | 36,2% | 8,8 | 14,3 | 5,6 | 39,2% | |
| N | 127 | 14 | |||||||
| TXA + LIA | M | 10,7 | 13,8 | 3,1 | 22,7% | 8,1 | 12,4 | 4,3 | 34,6% |
| SD | 1,2 | 1,2 | 0,9 | 6,4% | 0,7 | 1,2 | 0,9 | 5,4% | |
| MD | 10,5 | 13,7 | 3,2 | 23,4% | 8,3 | 12,7 | 4,6 | 36,5% | |
| MIN | 8,2 | 10,5 | 0,9 | 7,4% | 5,9 | 9,9 | 2,5 | 23,9% | |
| MAX | 14,7 | 17,8 | 6,3 | 41,7% | 8,8 | 14,1 | 5,8 | 41,7% | |
| N | 207 | 18 | |||||||
| Overall | M | 10,6 | 14 | 3,4 | 24,4% | 8,2 | 13 | 4,8 | 36,6% |
| SD | 1,2 | 1,2 | 1 | 6,4% | 0,5 | 1,1 | 0,9 | 4,9% | |
| MD | 10,4 | 13,9 | 3,4 | 24,8% | 8,4 | 13,1 | 4,8 | 36,6% | |
| MIN | 8,2 | 10,5 | 0,4 | 3,4% | 5,9 | 9,9 | 2 | 19,4% | |
| MAX | 15,2 | 19 | 6,5 | 41,7% | 8,9 | 15,4 | 7,3 | 49,3% | |
| N | 458 | 72 | |||||||
| SS | df | MS | F | p | |
| Intercept term. | 3450,016 | 1 | 3450,016 | 4130,919 | p<0,01 |
| Group 1 (blood transfusion /-/ and blood transfusion /+/) | 70,930 | 1 | 70,930 | 84,929 | p<0,01 |
| Gruup 2 (no blood protocol; TXA; TXA + LIA) | 28,244 | 2 | 14,122 | 16,909 | p<0,01 |
| Group 1* Group 2 | 10,083 | 2 | 6,041 | 12,048 | p=0,023 |
| Error | 437,629 | 524 | 0,835 |
| {1} M=3,90 27,5%↓ |
{2} M=3,42 24,1%↓ |
{3} M=3,15 22,7%↓ |
{4} M=5,11 36,6%↓ |
{5} M=4,54 35,3%↓ |
{6} M=4,31 34,6%↓ |
|||
| p- Value | ||||||||
| 1 |
blood transfusion /-/ |
CONTROL | 0,028 | 0,001 | 0,000 | 0,005 | 0,038 | |
| 2 |
blood transfusion /-/ |
TXA | 0,028 | 0,219 | 0,000 | 0,000 | 0,000 | |
| 3 |
blood transfusion /-/ |
TXA+LIA | 0,001 | 0,219 | 0,000 | 0,000 | 0,000 | |
| 4 | blood transfusion /+/ | CONTROL | 0,000 | 0,000 | 0,000 | 0,009 | 0,000 | |
| 5 | blood transfusion /+/ | TXA | 0,005 | 0,000 | 0,000 | 0,009 | 0,041 | |
| 6 | blood transfusion /+/ | TXA+LIA | 0,038 | 0,000 | 0,000 | 0,000 | 0,041 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




