Submitted:
19 July 2023
Posted:
19 July 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
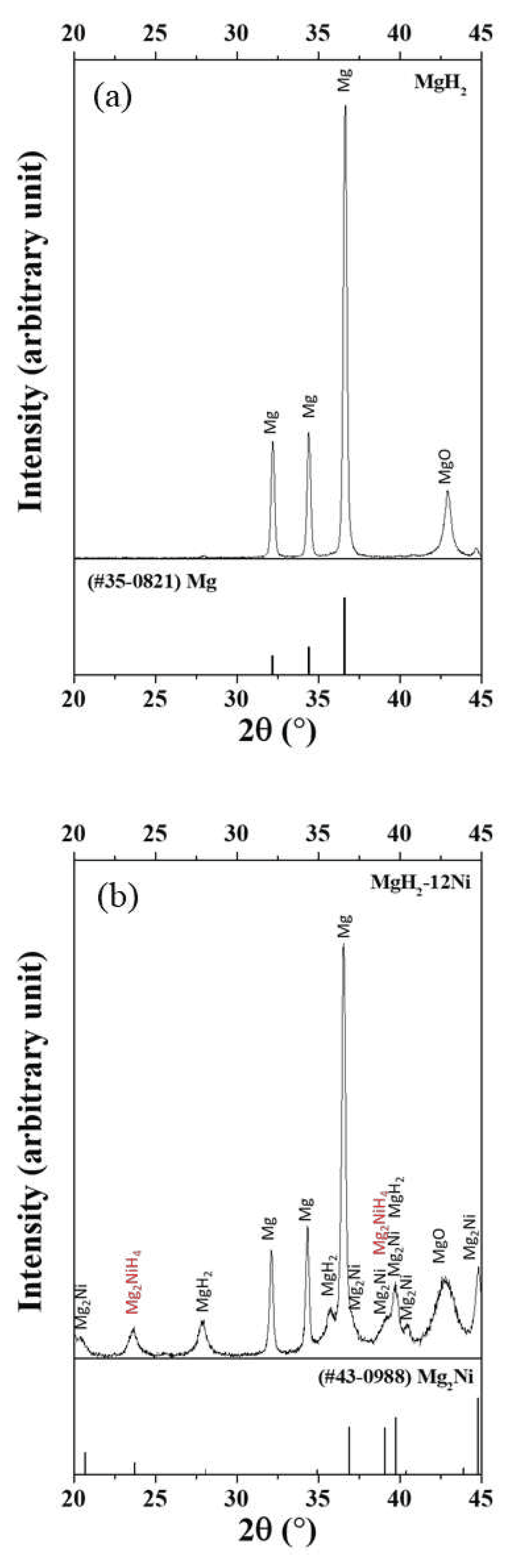
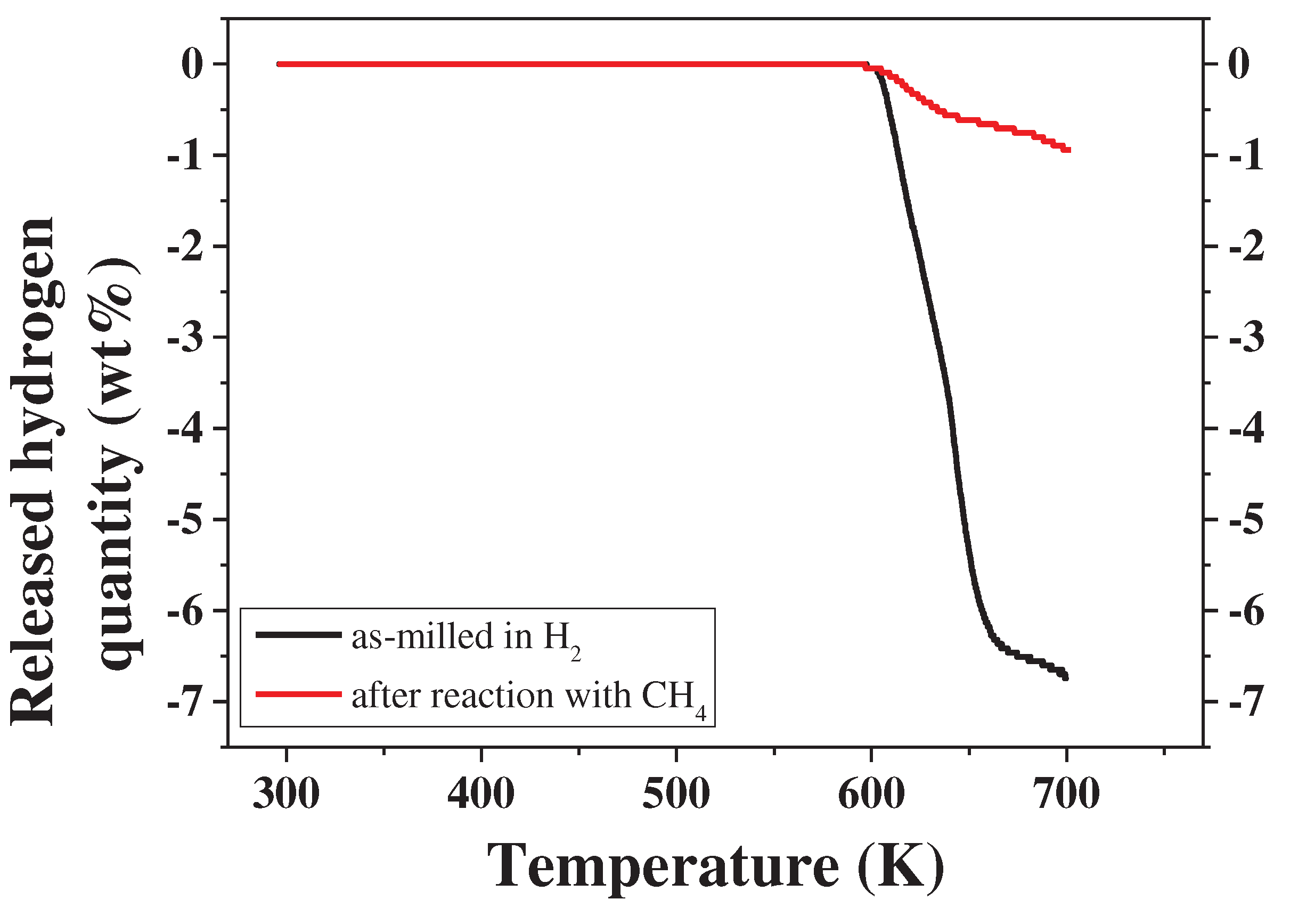
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Han, J.S.; Park, K. D. Thermal analysis of Mg hydride by Sieverts’ type automatic apparatus. Korean J. Met. Mater. 2010, 48, 1123–1129. [Google Scholar] [CrossRef]
- Hong, S.H.; Kwon, S.N.; Song, M.Y. Hydrogen storage characteristics of melt spun Mg-23.5Ni-xCu alloys and Mg-23.5Ni-2.5Cu alloy mixed with Nb2O5 and NbF5. In Korean J. Met. Mater.; 2011; Volume 49, pp. 298–303, UCI : G704-000085.2011.49.4.004. [Google Scholar]
- Kim, K.I.; Hong, T.W. Hydrogenation properties of Mg-5wt.% TiCr10NbX (x=1,3,5) composites by mechanical alloying process. Korean J. Met. Mater. 2011, 49, 264–269. [Google Scholar] [CrossRef]
- Reilly, J.J.; Wiswall, R.H. Reaction of hydrogen with alloys of magnesium and copper. Inorg. Chem. 1967, 6, 2220–2223. [Google Scholar] [CrossRef]
- Reilly, J.J.; Wiswall, R.H. Reaction of hydrogen with alloys of magnesium and nickel and the formation of Mg2NiH4. Inorg. Chem. 1968, 7, 2254–2256. [Google Scholar] [CrossRef]
- Akiba, E.; Nomura, K.; Ono, S.; Suda, S. Kinetics of the reaction between Mg-Ni alloys and H2. Int. J. Hydrogen Energy 1982, 7, 787–791. [Google Scholar] [CrossRef]
- Akiba, E.; Nomura, K.; Ono, S.; Suda, S. Kinetics of the reaction between Mg-Ni alloys and H2. Int. J. Hydrogen Energy 1982, 7, 787–791. [Google Scholar] [CrossRef]
- Zhong, H.C.; Wang, H.; Ouyang, L.Z.; Zhu., M. Microstructure and hydrogen storage properties of Mg–Sn nanocomposite by mechanical milling. J. Alloy Compd. 2011, 509(11), 4268–4272. [Google Scholar] [CrossRef]
- Pei, P.; Song, X.; Liu, J.; Song, A.; Zhang, P.; Chen, G. Study on the hydrogen desorption mechanism of a Mg–V composite prepared by SPS. Int. J. Hydrogen Energy 2012, 37(1), 984–989. [Google Scholar] [CrossRef]
- Li, Z.; Liu, X.; Jiang, L.; Wang, S. Characterization of Mg–20 wt% Ni–Y hydrogen storage composite prepared by reactive mechanical alloying. Int. J. Hydrogen Energy 2007, 32(12), 1869–1874. [Google Scholar] [CrossRef]
- Song, M.Y. Improvement in hydrogen storage characteristics of magnesium by mechanical alloying with nickel. J. Mater. Sci. 1995, 30, 1343–1351. [Google Scholar] [CrossRef]
- Song, M.Y.; Ivanov, E.I.; Darriet, B.; Pezat, M.; Hagenmuller, P. Hydriding properties of a mechanically alloyed mixture with a composition Mg2Ni. Int. J. Hydrogen Energy 1985, 10, 169–178. [Google Scholar] [CrossRef]
- Song, M.Y.; Ivanov, E.I.; Darriet, B.; Pezat, M.; Hagenmuller, P. Hydriding and dehydriding characteristics of mechanically alloyed mixtures Mg-xwt.%Ni (x = 5, 10, 25 and 55). J. Less-Common Met. 1987, 131, 71–79. [Google Scholar] [CrossRef]
- Song, M.Y. Effects of mechanical alloying on the hydrogen storage characteristics of Mg-xwt% Ni (x = 0, 5, 10, 25 and 55) mixtures. Int. J. Hydrogen Energy 1995, 20, 221–227. [Google Scholar] [CrossRef]
- Bobet, J.-L.; Akiba, E.; Nakamura, Y.; Darriet, B. Study of Mg-M (M=Co, Ni and Fe) mixture elaborated by reactive mechanical alloying — hydrogen sorption properties. Int. J. Hydrogen Energy 2000, 25, 987–996. [Google Scholar] [CrossRef]
- https://en.wikipedia.org/wiki/Methane_reformer (visited on June 18, 2023).
- Carrette, L.; Friedrich, K.A.; Stimming, U. Fuel cells- Fundamentals and applications. Fuel Cells 2001, 1(1), 5–35. [Google Scholar] [CrossRef]
- Larminie, J.; Dicks, A. Fuel Cell Systems Explained, 2nd ed.; Wiley, 2003; Available online: https://sv.20file.org/up1/482_0.pdf.
- Ngyuen, N.Q. Ceramic fuel cells. J. Amer. Chem. Soc. 1993, 76, 563–588. [Google Scholar] [CrossRef]
- Song, M.Y.; Ahn, D.S.; Kwon, I.H.; Ahn, H.J. Development of hydrogen-storage alloys by mechanical alloying Mg with Fe and Co. Met. Mater. Int. 1999, 5, 485–490. [Google Scholar] [CrossRef]
- Song, M.Y.; Ivanov, E.I.; Darriet, B.; Pezat, M.; Hagenmuller, P. Hydriding properties of a mechanically alloyed mixture with a composition Mg2Ni. Int. J. Hydrogen Energy 1985, 10, 169–178. [Google Scholar] [CrossRef]
- https://www.sigmaaldrich.com/KR/ko/technical-documents/technical-article/analytical-chemistry/photometry-and-reflectometry/ir-spectrum-table. (visited on June 29, 2023).
- https://chem.libretexts.org/Ancillary_Materials/Reference/Reference_Tables/Spectroscopic_Reference_Tables/Infrared_Spectroscopy_Absorption_Table. (visited on June 29, 2023).
- Stampfer, J.F. Jr.; Holley, C.E. Jr.; Suttle, J.F. The magnesium-hydrogen system. J. Amer. Chem. Soc. 1960, 82, 3504–3508. [Google Scholar] [CrossRef]
- Song, M.Y.; Park, H.R. Pressure–composition isotherms in the Mg2Ni–H2 system. J. Alloys. Compd. 1998, 270, 164–167. [Google Scholar] [CrossRef]
- Song, M.Y.; Pezat, M.; Darriet, B.; Hagenmuller, P. Hydriding mechanism of Mg2Ni in the presence of oxygen impurity in hydrogen. J. Mater. Sci. 1985, 20, 2958–2964. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



