Submitted:
11 July 2023
Posted:
12 July 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemical Structures of OPs
2.2. Acquisition of OPs Targets
2.3. Prediction of NDT Targets
2.4. Venn Analysis and Construction of Protein-Protein Interaction (PPI) Network
2.5. Topological Analysis of PPI Networks
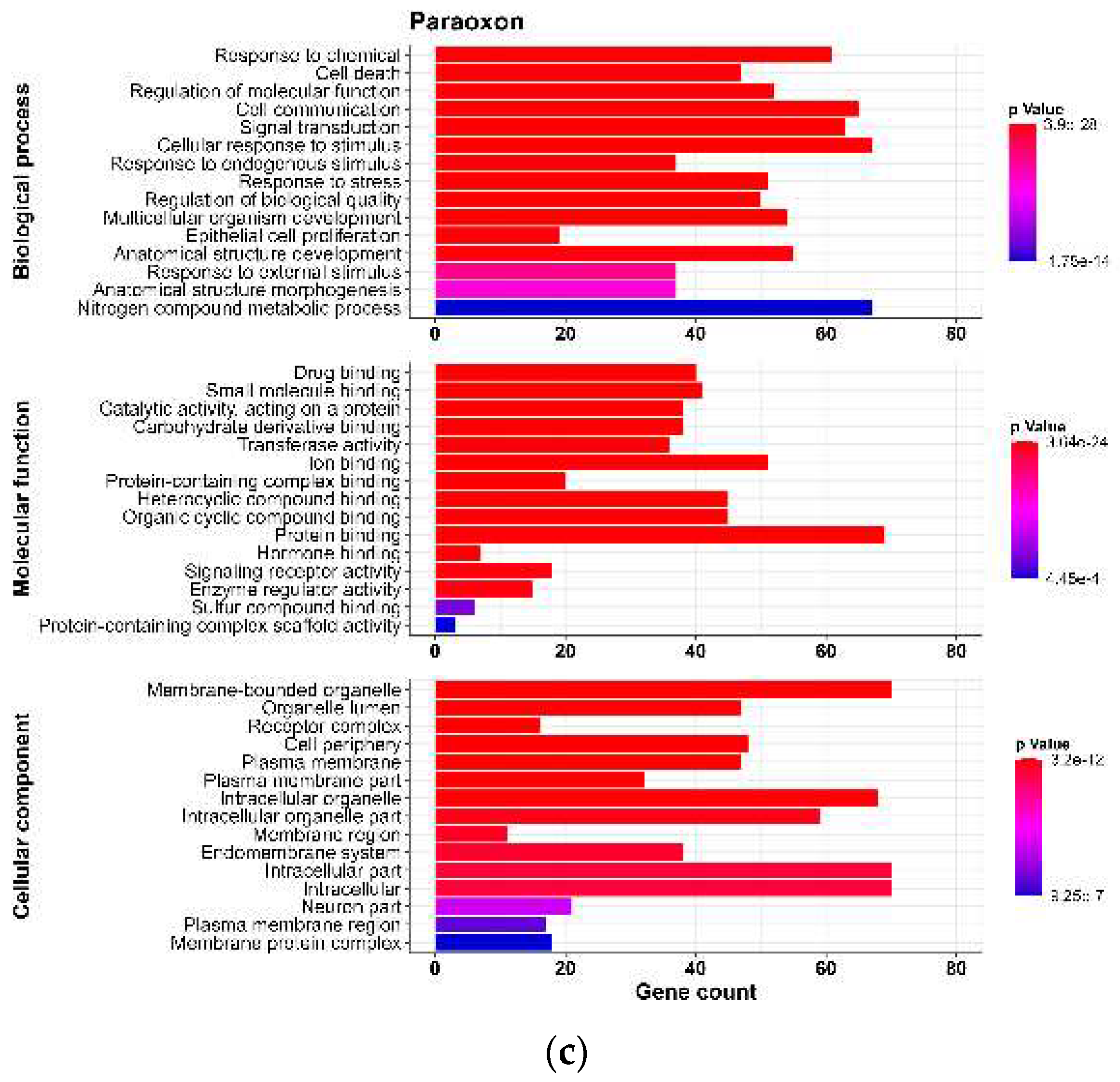
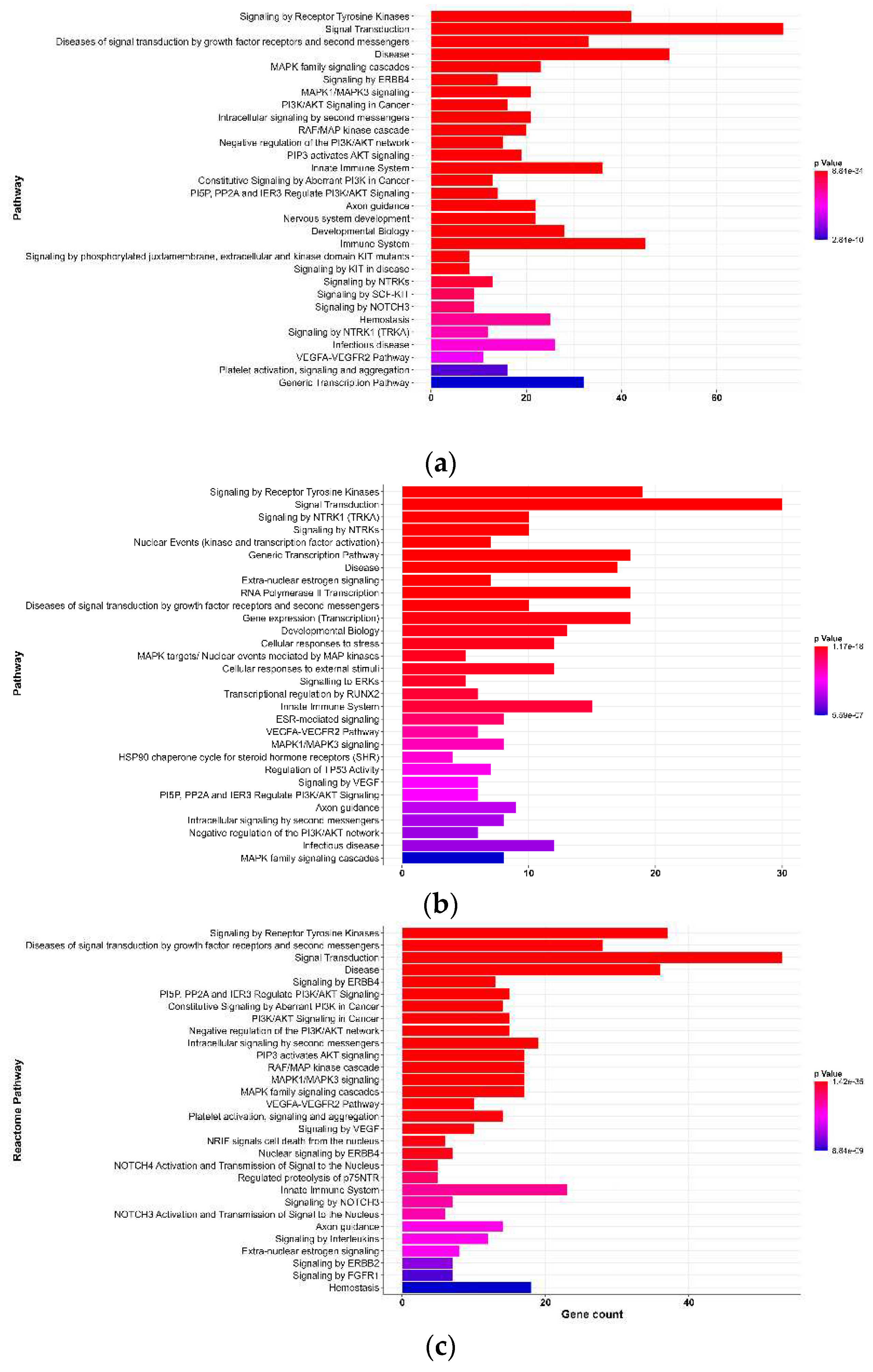
2.6. Enrichment Analysis
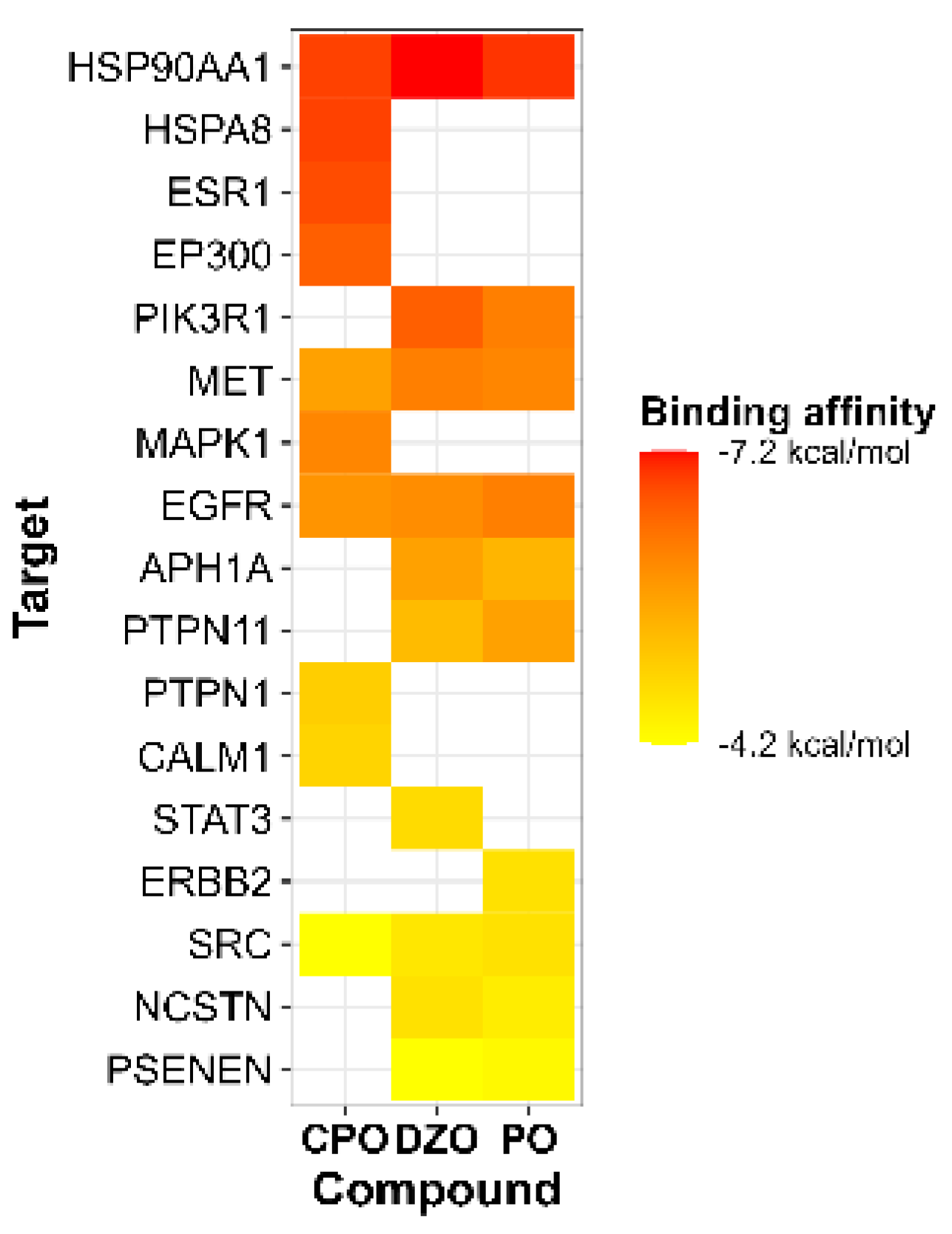
2.7. Molecular Docking:
3. Results
3.1. Candidate Targets for OP-Induced NDT
3.2. GO and Reactome Pathway analysis
3.2.1. GO analysis
3.2.2. Reactome Pathway Analysis
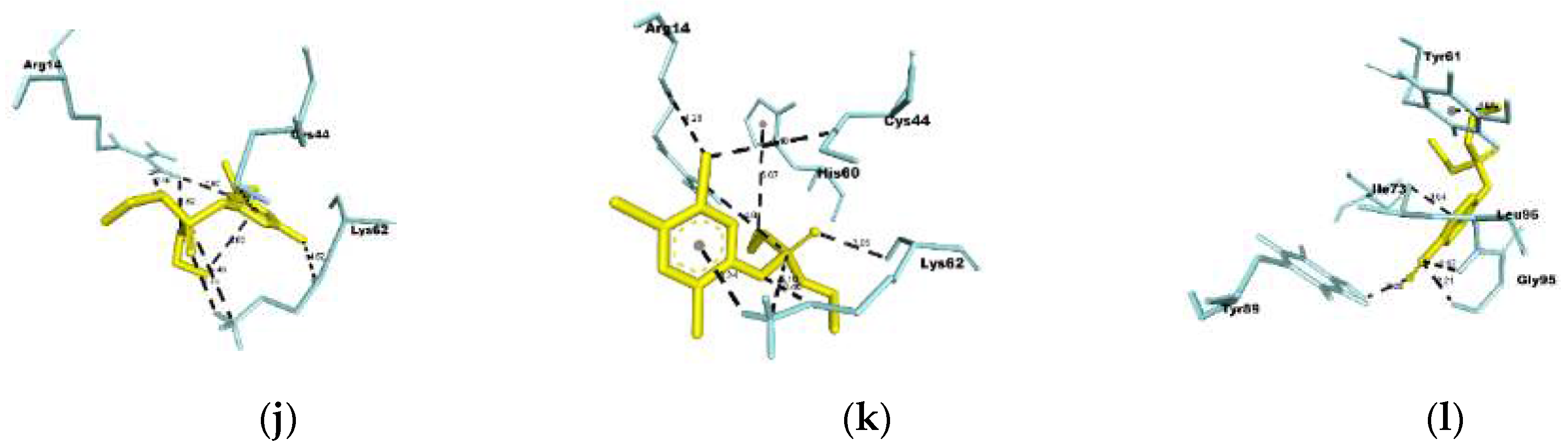
3.3. Molecular Docking
4. Discussion
4.1. Signal Transduction
4.2. Axon Guidance
4.3. Cellular Response to Stress
4.5. Activation of NMDAR
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Montuori, P.; De Rosa, E.; Di Duca, F.; De Simone, B.; Scippa, S.; Russo, I.; Sorrentino, M.; Sarnacchiaro, P.; Triassi, M. Occurrence, Distribution, and Risk Assessment of Organophosphorus Pesticides in the Aquatic Environment of the Sele River Estuary, Southern Italy. Toxics, 2022, 10(7), 377. [Google Scholar] [CrossRef]
- Sumon, K. A.; Rashid, H.; Peeters, E. T. H. M.; Bosma, R. H.; Van den Brink, P. J. Environmental Monitoring and Risk Assessment of Organophosphate Pesticides in Aquatic Ecosystems of North-West Bangladesh. Chemosphere, 2018, 206, 92–100. [Google Scholar] [CrossRef]
- Akter, R.; Pervin, M. A.; Jahan, H.; Rakhi, S. F.; Reza, A. H. M. M.; Hossain, Z. Toxic Effects of an Organophosphate Pesticide, Envoy 50 SC on the Histopathological, Hematological, and Brain Acetylcholinesterase Activities in Stinging Catfish (Heteropneustes Fossilis). The Journal of Basic and Applied Zoology, 2020, 81(1), 1–14. [Google Scholar]
- Tang, J.; Wang, W.; Jiang, Y.; Chu, W. Diazinon Exposure Produces Histological Damage, Oxidative Stress, Immune Disorders and Gut Microbiota Dysbiosis in Crucian Carp (Carassius auratus gibelio). Environmental Pollution, 2021, 269, 116129. [Google Scholar]
- Sandoval-Herrera, N.; Mena, F.; Espinoza, M.; Romero, A. Neurotoxicity of Organophosphate Pesticides Could Reduce the Ability of Fish to Escape Predation under Low Doses of Exposure. Scientific Reports 2019, 9(1), 1–11. [Google Scholar]
- Ehrenstein, O. S. von; Ling, C.; Cui, X.; Cockburn, M.; Park, A. S.; Yu, F.; Wu, J.; Ritz, B. Prenatal and Infant Exposure to Ambient Pesticides and Autism Spectrum Disorder in Children: Population Based Case-Control Study. Br Med J, 2019, 364, 962. [Google Scholar]
- Gunier, R. B.; Bradman, A.; Harley, K. G.; Kogut, K.; Eskenazi, B. Prenatal Residential Proximity to Agricultural Pesticide Use and IQ in 7-Year-Old Children. Environ Health Perspect, 2017, 125(5), 057002. [Google Scholar] [CrossRef]
- Burke, R. D.; Todd, S. W.; Lumsden, E.; Mullins, R. J.; Mamczarz, J.; Fawcett, W. P.; Gullapalli, R. P.; Randall, W. R.; Pereira, E. F. R.; Albuquerque, E. X. Developmental Neurotoxicity of the Organophosphorus Insecticide Chlorpyrifos: From Clinical Findings to Preclinical Models and Potential Mechanisms. J Neurochem, 2017, 142 (Suppl 2), 162. [Google Scholar]
- Eddleston, M.; Buckley, N. A.; Eyer, P.; Dawson, A. H. Management of Acute Organophosphorus Pesticide Poisoning. Lancet, 2008, 371(9612), 597. [Google Scholar]
- Perez-Fernandez, C.; Morales-Navas, M.; Guardia-Escote, L.; Garrido-Cárdenas, J. A.; Colomina, M. T.; Giménez, E.; Sánchez-Santed, F. Long-Term Effects of Low Doses of Chlorpyrifos Exposure at the Preweaning Developmental Stage: A Locomotor, Pharmacological, Brain Gene Expression and Gut Microbiome Analysis. Food and Chemical Toxicology, 2020, 135, 110865. [Google Scholar]
- Rush, T.; Liu, X. Q.; Hjelmhaug, J.; Lobner, D. Mechanisms of Chlorpyrifos and Diazinon Induced Neurotoxicity in Cortical Culture. Neuroscience 2010, 166(3), 899–906. [Google Scholar] [CrossRef]
- van Melis, L. V. J.; Heusinkveld, H. J.; Langendoen, C.; Peters, A.; Westerink, R. H. S. Organophosphate Insecticides Disturb Neuronal Network Development and Function via Non-AChE Mediated Mechanisms. Neurotoxicology, 2023, 94, 35–45. [Google Scholar]
- Perez-Fernandez, C.; Morales-Navas, M.; Guardia-Escote, L.; Colomina, M. T.; Giménez, E.; Sánchez-Santed, F. Postnatal Exposure to Low Doses of Chlorpyrifos Induces Long-Term Effects on 5C-SRTT Learning and Performance, Cholinergic and GABAergic Systems and BDNF Expression. Exp Neurol, 2020, 330, 113356. [Google Scholar]
- Richendrfer, H.; Creton, R. Chlorpyrifos and Malathion Have Opposite Effects on Behaviors and Brain Size That Are Not Correlated to Changes in AChE Activity. Neurotoxicology, 2015, 49, 50–58. [Google Scholar]
- Ireland, D.; Zhang, S.; Bochenek, V.; Hsieh, J.-H.; Rabeler, C.; Meyer, Z.; Collins, E.-M. S. Differences in Neurotoxic Outcomes of Organophosphorus Pesticides Revealed via Multi-Dimensional Screening in Adult and Regenerating Planarians. Frontiers in Toxicology, 2022, 4, 948455. [Google Scholar]
- Chen, Q.; Zhang, K.; Jiao, M.; Jiao, J.; Chen, D.; Yin, Y.; Zhang, J.; Li, F. Study on the Mechanism of Mesaconitine-Induced Hepatotoxicity in Rats Based on Metabonomics and Toxicology Network. Toxins (Basel), 2022, 14(7), 486. [Google Scholar] [CrossRef]
- Liu, C.; Zhang, C.; Wang, W.; Yuan, F.; He, T.; Chen, Y.; Wang, Q.; Huang, J. Integrated Metabolomics and Network Toxicology to Reveal Molecular Mechanism of Celastrol Induced Cardiotoxicity. Toxicol Appl Pharmacol, 2019, 383, 114785. [Google Scholar] [CrossRef]
- Flaskos, J. The Developmental Neurotoxicity of Organophosphorus Insecticides: A Direct Role for the Oxon Metabolites. Toxicol Lett, 2012, 209(1), 86–93. [Google Scholar]
- Androutsopoulos, V. P.; Hernandez, A. F.; Liesivuori, J.; Tsatsakis, A. M. A Mechanistic Overview of Health Associated Effects of Low Levels of Organochlorine and Organophosphorous Pesticides. Toxicology, 2013, 307, 89–94. [Google Scholar]
- Barabási, A. L.; Oltvai, Z. N. Network Biology: Understanding the Cell’s Functional Organization. Nature Reviews Genetics, 2004, 5(2), 101–113. [Google Scholar]
- Trott, O.; Olson, A. J. AutoDock Vina: Improving the Speed and Accuracy of Docking with a New Scoring Function, Efficient Optimization, and Multithreading. J Comput Chem, 2010, 31(2), 455–461. [Google Scholar] [CrossRef]
- Gillespie, M.; Jassal, B.; Stephan, R.; Milacic, M.; Rothfels, K.; Senff-Ribeiro, A.; Griss, J.; Sevilla, C.; Matthews, L.; Gong, C.; et al. The Reactome Pathway Knowledgebase 2022. Nucleic Acids Res, 2022, 50(D1), D687. [Google Scholar] [CrossRef]
- van der Geer, P. Signal Transduction. In Brenner’s Encyclopedia of Genetics, 2nd ed, 2013, Stanley Maloy, A., Kelly Hughes, B., Elsevier, 436–439.
- Katz, M.; Amit, I.; Yarden, Y. Regulation of MAPKs by Growth Factors and Receptor Tyrosine Kinases. Biochimica et Biophysica Acta (BBA) - Molecular Cell Research 2007, 1773(8), 1161–1176. [Google Scholar]
- Lee, J. E.; Park, J. H.; Shin, I. C.; Koh, H. C. Reactive Oxygen Species Regulated Mitochondria-Mediated Apoptosis in PC12 Cells Exposed to Chlorpyrifos. Toxicol Appl Pharmacol, 2012, 263(2), 148–162. [Google Scholar]
- Ki, Y. W.; Park, J. H.; Lee, J. E.; Shin, I. C.; Koh, H. C. JNK and P38 MAPK Regulate Oxidative Stress and the Inflammatory Response in Chlorpyrifos-Induced Apoptosis. Toxicol Lett, 2013, 218(3), 235–245. [Google Scholar]
- Jahan, S.; Kumar, D.; Singh, S.; Kumar, V.; Srivastava, A.; Pandey, A.; Rajpurohit, C. S.; Khanna, V. K.; Pant, A. B. Resveratrol Prevents the Cellular Damages Induced by Monocrotophos via PI3K Signaling Pathway in Human Cord Blood Mesenchymal Stem Cells. Mol Neurobiol, 2018, 55(11), 8278–8292. [Google Scholar] [CrossRef]
- Lasagna, M.; Ventura, C.; Hielpos, M. S.; Mardirosian, M. N.; Martín, G.; Miret, N.; Randi, A.; Núñez, M.; Cocca, C. Endocrine Disruptor Chlorpyrifos Promotes Migration, Invasion, and Stemness Phenotype in 3D Cultures of Breast Cancer Cells and Induces a Wide Range of Pathways Involved in Cancer Progression. Environ Res, 2022, 204, 111989. [Google Scholar]
- Suriyo, T.; Tachachartvanich, P.; Visitnonthachai, D.; Watcharasit, P.; Satayavivad, J. Chlorpyrifos Promotes Colorectal Adenocarcinoma H508 Cell Growth through the Activation of EGFR/ERK1/2 Signaling Pathway but Not Cholinergic Pathway. Toxicology, 2015, 338, 117–129. [Google Scholar]
- Subramaniam, S.; Unsicker, K. ERK and Cell Death: ERK1/2 in Neuronal Death. FEBS J, 2010, 277(1), 22–29. [Google Scholar]
- Botella, J. A.; Kretzschmar, D.; Kiermayer, C.; Feldmann, P.; Hughes, D. A.; Schneuwly, S. Deregulation of the Egfr/Ras Signaling Pathway Induces Age-Related Brain Degeneration in the Drosophila Mutant Vap. Mol Biol Cell, 2003, 14(1), 241. [Google Scholar] [CrossRef]
- Stoeckli, E. T. Understanding Axon Guidance: Are We Nearly There Yet? Development, 2018, 145(10), dev151415. [Google Scholar]
- Romano, R.; Bucci, C. Role of EGFR in the Nervous System. Cells, 2020, 9(8), 1887. [Google Scholar]
- Chen, Z.; Oh, D.; Dubey, A. K.; Yao, M.; Yang, B.; Groves, J. T.; Sheetz, M. EGFR Family and Src Family Kinase Interactions: Mechanics Matters? Curr Opin Cell Biol, 2018, 51, 97–102. [Google Scholar] [CrossRef]
- Knöll, B.; Drescher, U. Src Family Kinases Are Involved in EphA Receptor-Mediated Retinal Axon Guidance. The Journal of Neuroscience, 2004, 24(28), 6248. [Google Scholar] [CrossRef]
- Yam, P. T.; Langlois, S. D.; Morin, S.; Charron, F. Sonic Hedgehog Guides Axons through a Noncanonical, Src-Family-Kinase-Dependent Signaling Pathway. Neuron, 2009, 62(3), 349–362. [Google Scholar] [CrossRef]
- Poliak, S.; Morales, D.; Croteau, L. P.; Krawchuk, D.; Palmesino, E.; Morton, S.; Cloutier, J. F.; Charron, F.; Dalva, M. B.; Ackerman, S. L.; et al. Synergistic Integration of Netrin and Ephrin Axon Guidance Signals by Spinal Motor Neurons. Elife, 2015, 4, e10841. [Google Scholar]
- Desole, C.; Gallo, S.; Vitacolonna, A.; Montarolo, F.; Bertolotto, A.; Vivien, D.; Comoglio, P.; Crepaldi, T. HGF and MET: From Brain Development to Neurological Disorders. Front Cell Dev Biol, 2021, 9, 683609. [Google Scholar]
- Howard, A. S.; Bucelli, R.; Jett, D. A.; Bruun, D.; Yang, D.; Lein, P. J. Chlorpyrifos Exerts Opposing Effects on Axonal and Dendritic Growth in Primary Neuronal Cultures. Toxicol Appl Pharmacol, 2005, 207(2), 112–124. [Google Scholar] [CrossRef]
- Yang, D.; Lauridsen, H.; Buels, K.; Chi, L. H.; La Du, J.; Bruun, D. A.; Olson, J. R.; Tanguay, R. L.; Lein, P. J. Chlorpyrifos-Oxon Disrupts Zebrafish Axonal Growth and Motor Behavior. Toxicological Sciences, 2011, 121(1), 146. [Google Scholar] [CrossRef]
- Dowell, J.; Elser, B. A.; Schroeder, R. E.; Stevens, H. E. Cellular Stress Mechanisms of Prenatal Maternal Stress: Heat Shock Factors and Oxidative Stress. Neurosci Lett, 2019, 709, 134368. [Google Scholar]
- Miller, D. J.; Fort, P. E. Heat Shock Proteins Regulatory Role in Neurodevelopment. Front Neurosci, 2018, 12. [Google Scholar]
- Zohn, I. E. Hsp90 and Complex Birth Defects: A Plausible Mechanism for the Interaction of Genes and Environment. Neurosci Lett, 2020, 716, 134680. [Google Scholar] [PubMed]
- Xing, H.; Liu, T.; Zhang, Z.; Wang, X.; Xu, S. Acute and Subchronic Toxic Effects of Atrazine and Chlorpyrifos on Common Carp (Cyprinus Carpio L.): Immunotoxicity Assessments. Fish Shellfish Immunol, 2015, 45(2), 327–333. [Google Scholar] [PubMed]
- Eder, K. J.; Leutenegger, C. M.; Köhler, H. R.; Werner, I. Effects of Neurotoxic Insecticides on Heat-Shock Proteins and Cytokine Transcription in Chinook Salmon (Oncorhynchus tshawytscha). Ecotoxicol Environ Saf, 2009, 72(1), 182–190. [Google Scholar] [CrossRef]
- Fulda, S.; Gorman, A. M.; Hori, O.; Samali, A. Cellular Stress Responses: Cell Survival and Cell Death. Int J Cell Biol, 2010, 214074. [Google Scholar]
- Slotkin, T. A.; Seidler, F. J. Developmental Neurotoxicity of Organophosphates Targets Cell Cycle and Apoptosis, Revealed by Transcriptional Profiles in Vivo and in Vitro. Neurotoxicol Teratol, 2012, 34(2), 232–241. [Google Scholar] [CrossRef]
- Lin, J. W.; Fu, S. C.; Liu, J. M.; Liu, S. H.; Lee, K. I.; Fang, K. M.; Hsu, R. J.; Huang, C. F.; Liu, K. M.; Chang, K. C.; et al. Chlorpyrifos Induces Neuronal Cell Death via Both Oxidative Stress and Akt Activation Downstream-Regulated CHOP-Triggered Apoptotic Pathways. Toxicology in Vitro, 2023, 86, 105483. [Google Scholar]
- Ewald, R. C.; Cline, H. T. NMDA Receptors and Brain Development. In Biology of the NMDA Receptor. CRC Press/Taylor & Francis: Florida, United States, 2009.
- Burnashev, N.; Szepetowski, P. NMDA Receptor Subunit Mutations in Neurodevelopmental Disorders. Curr Opin Pharmacol, 2015, 20, 73–82. [Google Scholar]
- Chen, Z.; Wang, X.; Ashraf, U.; Zheng, B.; Ye, J.; Zhou, D.; Zhang, H.; Song, Y.; Chen, H.; Zhao, S.; et al. Activation of Neuronal N-Methyl-d-Aspartate Receptor Plays a Pivotal Role in Japanese Encephalitis Virus-Induced Neuronal Cell Damage. J Neuroinflammation, 2018, 15(1), 238. [Google Scholar] [CrossRef]
- Jiang, S.; Li, X.; Jin, W.; Duan, X.; Bo, L.; Wu, J.; Zhang, R.; Wang, Y.; Kang, R.; Huang, L. Ketamine-Induced Neurotoxicity Blocked by N-Methyl-d-Aspartate Is Mediated through Activation of PKC/ERK Pathway in Developing Hippocampal Neurons. Neurosci Lett, 2018, 673, 122–131. [Google Scholar]
- Eisenkraft, A.; Falk, A.; Finkelstein, A. The Role of Glutamate and the Immune System in Organophosphate-Induced CNS Damage. Neurotoxicity Research, 2013, 24(2), 265–279. [Google Scholar] [PubMed]
- Slotkin, T. A.; Seidler, F. J. Comparative Developmental Neurotoxicity of Organophosphates in Vivo: Transcriptional Responses of Pathways for Brain Cell Development, Cell Signaling, Cytotoxicity and Neurotransmitter Systems. Brain Res Bull, 2007, 72(4–6), 232–274. [Google Scholar] [PubMed]
- Banerjee, A.; Wang, H. Y.; Borgmann-Winter, K. E.; MacDonald, M. L.; Kaprielian, H.; Stucky, A.; Kvasic, J.; Egbujo, C.; Ray, R.; Talbot, K.; et al. Src Kinase as a Mediator of Convergent Molecular Abnormalities Leading to NMDAR Hypoactivity in Schizophrenia. Mol Psychiatry, 2015, 20(9), 1091. [Google Scholar] [PubMed]
- Tang, Y.; Ye, M.; Du, Y.; Qiu, X.; Lv, X.; Yang, W.; Luo, J. EGFR Signaling Upregulates Surface Expression of the GluN2B-Containing NMDA Receptor and Contributes to Long-Term Potentiation in the Hippocampus. Neuroscience, 2015, 304, 109–121. [Google Scholar] [PubMed]
- Gallo, S.; Vitacolonna, A.; Comoglio, P.; Crepaldi, T. MET Oncogene Controls Invasive Growth by Coupling with NMDA Receptor. Cancers (Basel), 2022, 14(18), 4408. [Google Scholar] [CrossRef] [PubMed]
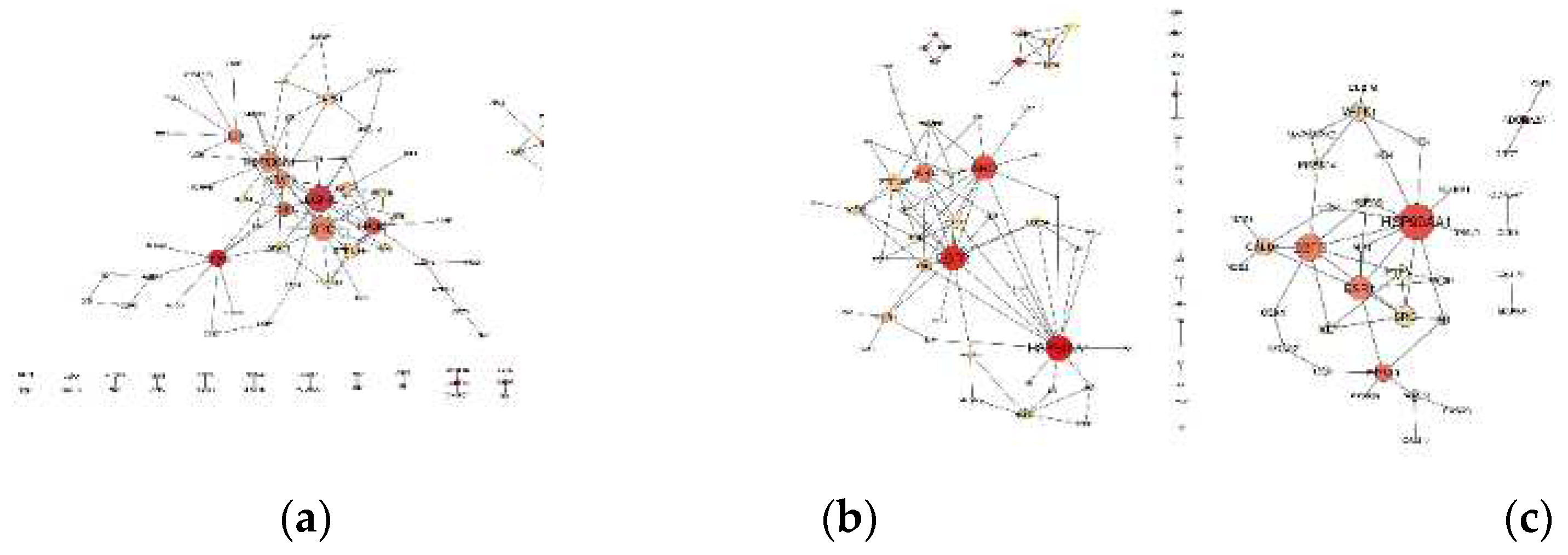


| Protein | Node | k | Clustering Coefficient | BC | ASPL | CC |
|---|---|---|---|---|---|---|
| Diazinon-oxon | ||||||
| Proto-oncogene tyrosine kinase SRC | SRC | 13 | 0.218 | 0.154 | 2.286 | 0.438 |
| Epidermal growth factor receptor | EGFR | 14 | 0.165 | 0.275 | 2.143 | 0.467 |
| Phosphatidylinositol 3-kinase regulatory subunit alpha | PIK3R1 | 9 | 0.250 | 0.238 | 2.408 | 0.415 |
| Tyrosine-protein phosphatase non-receptor type 11 | PTPN11 | 7 | 0.4286 | 0.0164 | 2.7143 | 0.368 |
| Heat shock protein HSP 90-alpha | HSP90AA1 | 11 | 0.109 | 0.154 | 2.429 | 0.412 |
| Gamma-secretase subunit APH-1A | APH1A | 4 | 0.8333 | 0.0667 | 1.5000 | 0.667 |
| Nicastrin | NCSTN | 4 | 0.833 | 0.067 | 1.500 | 0.667 |
| Gamma-secretase subunit PEN-2 | PSENEN | 4 | 0.8333 | 0.0667 | 1.5000 | 0.667 |
| Signal transducer and activator of transcription 3 | STAT3 | 8 | 0.179 | 0.142 | 2.327 | 0.430 |
| Hepatocyte growth factor receptor | MET | 4 | 0.8333 | 0.0002 | 2.9184 | 0.343 |
| Chlorpyrifos oxon | ||||||
| Heat shock protein HSP 90-alpha | HSP90AA1 | 11 | 0.109 | 0.382 | 2.034 | 0.492 |
| Epidermal growth factor receptor | EGFR | 8 | 0.179 | 0.276 | 2.172 | 0.460 |
| Proto-oncogene tyrosine kinase SRC | SRC | |||||
| Estrogen receptor | ESR1 | 7 | 0.143 | 0.269 | 2.103 | 0.475 |
| Tyrosine-protein phosphatase non-receptor type 1 | PTPN1 | 4 | 0.500 | 0.017 | 2.759 | 0.363 |
| Hepatocyte growth factor receptor | MET | 3 | 1.000 | 0.000 | 2.793 | 0.358 |
| Mitogen-activated protein kinase1 | MAPK1 | 5 | 0.100 | 0.095 | 3.172 | 0.315 |
| Calmodulin 1 | CALM1 | 5 | 0.000 | 0.158 | 2.414 | 0.414 |
| Histone acetyltransferase p300 | EP300 | 5 | 0.000 | 0.308 | 2.552 | 0.392 |
| Heat shock cognate 71 kDa protein | HSPA8 | 3 | 0.667 | 0.008 | 2.379 | 0.420 |
| Paraoxon | ||||||
| Proto-oncogene tyrosine kinase SRC | SRC | 14 | 0.220 | 0.264 | 1.875 | 0.533 |
| Epidermal growth factor receptor | EGFR | 14 | 0.198 | 0.312 | 1.800 | 0.556 |
| Tyrosine-protein phosphatase non-receptor type 11 | PTPN11 | 9 | 0.361 | 0.059 | 2.225 | 0.449 |
| Phosphatidylinositol 3-kinase regulatory subunit alpha | PIK3R1 | 10 | 0.289 | 0.180 | 2.150 | 0.465 |
| Steroid hormone receptor ERR2 | ERRB2 | 6 | 0.667 | 0.026 | 2.100 | 0.476 |
| Heat shock protein HSP 90-alpha | HSP90AA1 | 15 | 0.095 | 0.378 | 1.925 | 0.519 |
| Gamma-secretase subunit APH-1A | APH1A | 4 | 0.833 | 0.067 | 1.200 | 0.833 |
| Nicastrin | NCSTN | 4 | 0.833 | 0.067 | 1.200 | 0.833 |
| Hepatocyte growth factor receptor | MET | 4 | 0.833 | 0.001 | 2.450 | 0.408 |
| Gamma-secretase subunit PEN-2 | PSENEN | 4 | 0.833 | 0.067 | 1.200 | 0.833 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




