Submitted:
11 July 2023
Posted:
12 July 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
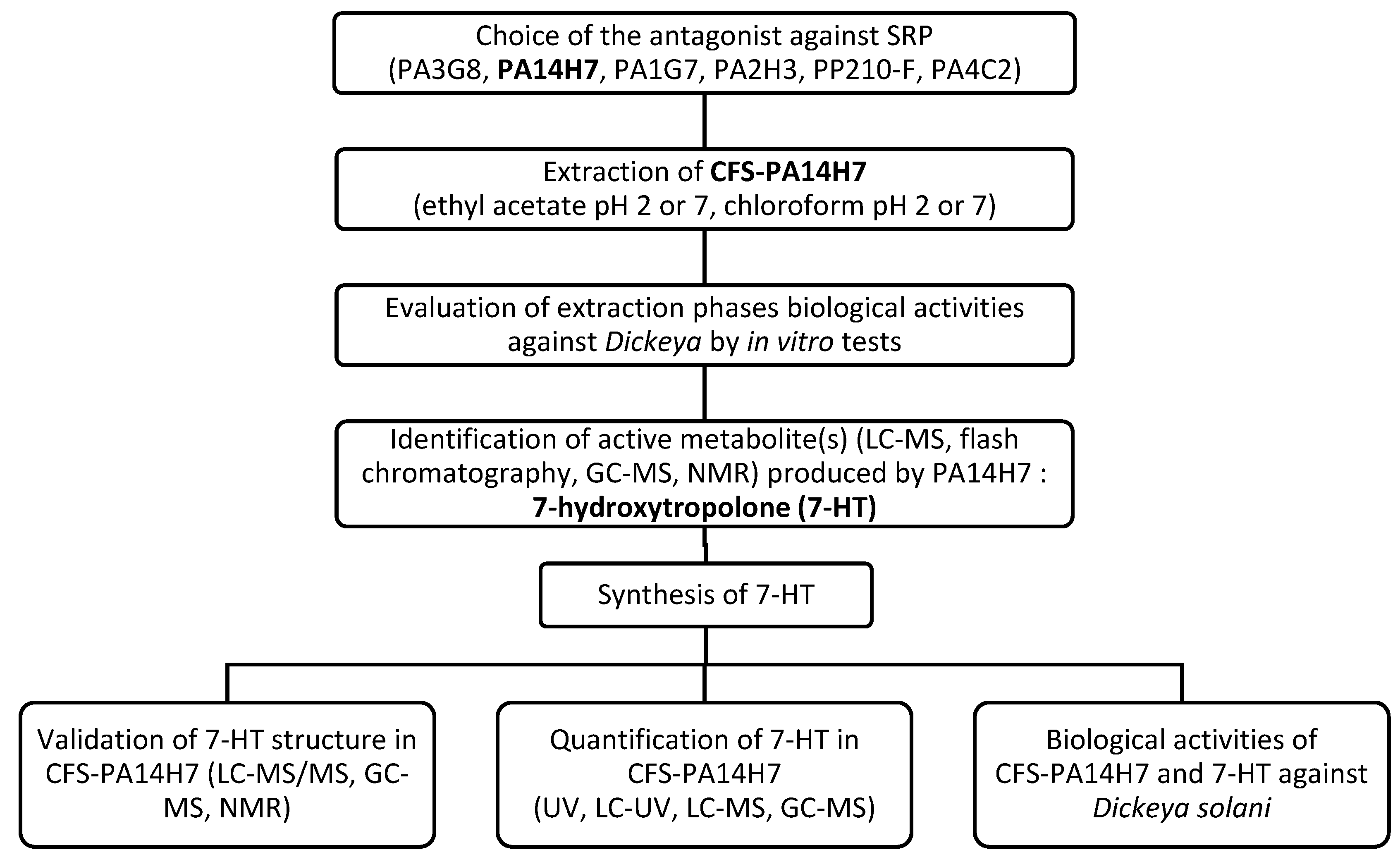
2. Results & Discussion
2.1. Choice of the main active antagonist
2.2. Evaluation of CFS-PA14H7 extraction methods
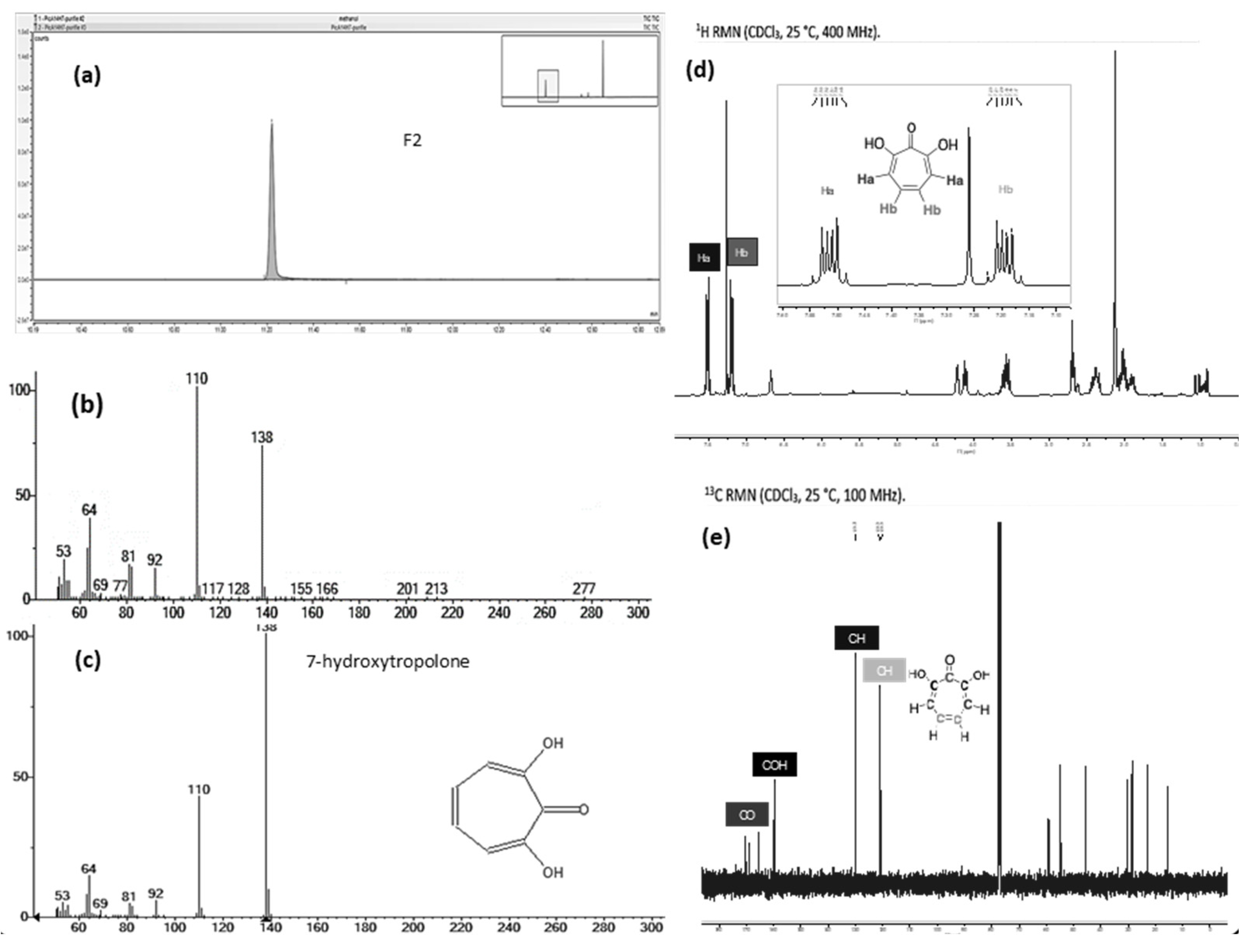
2.3. Identification of the active metabolite(s)
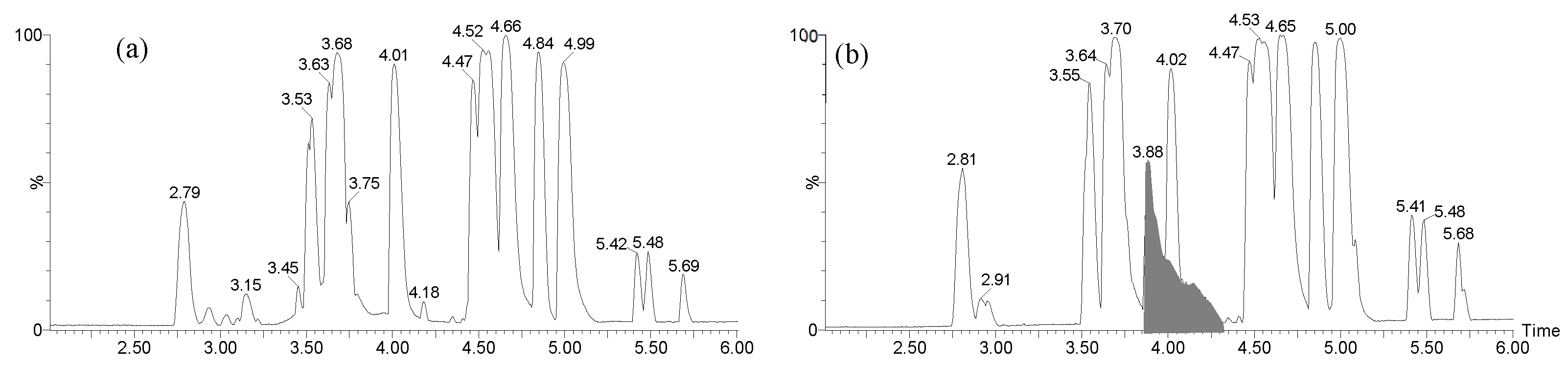
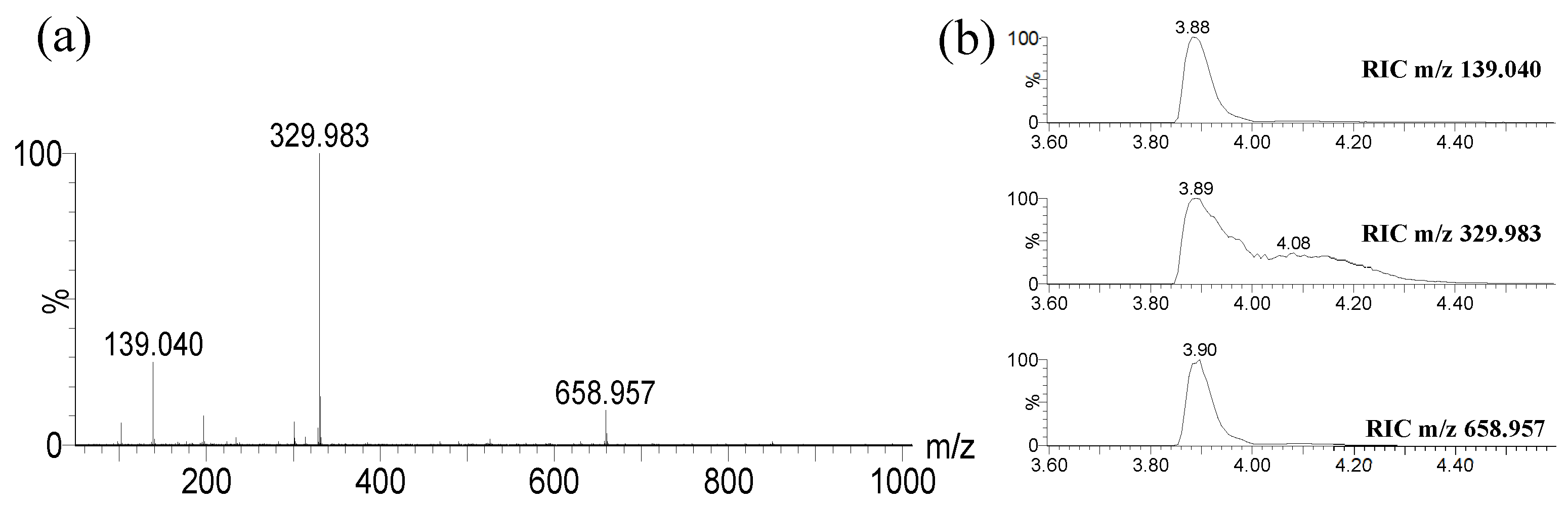
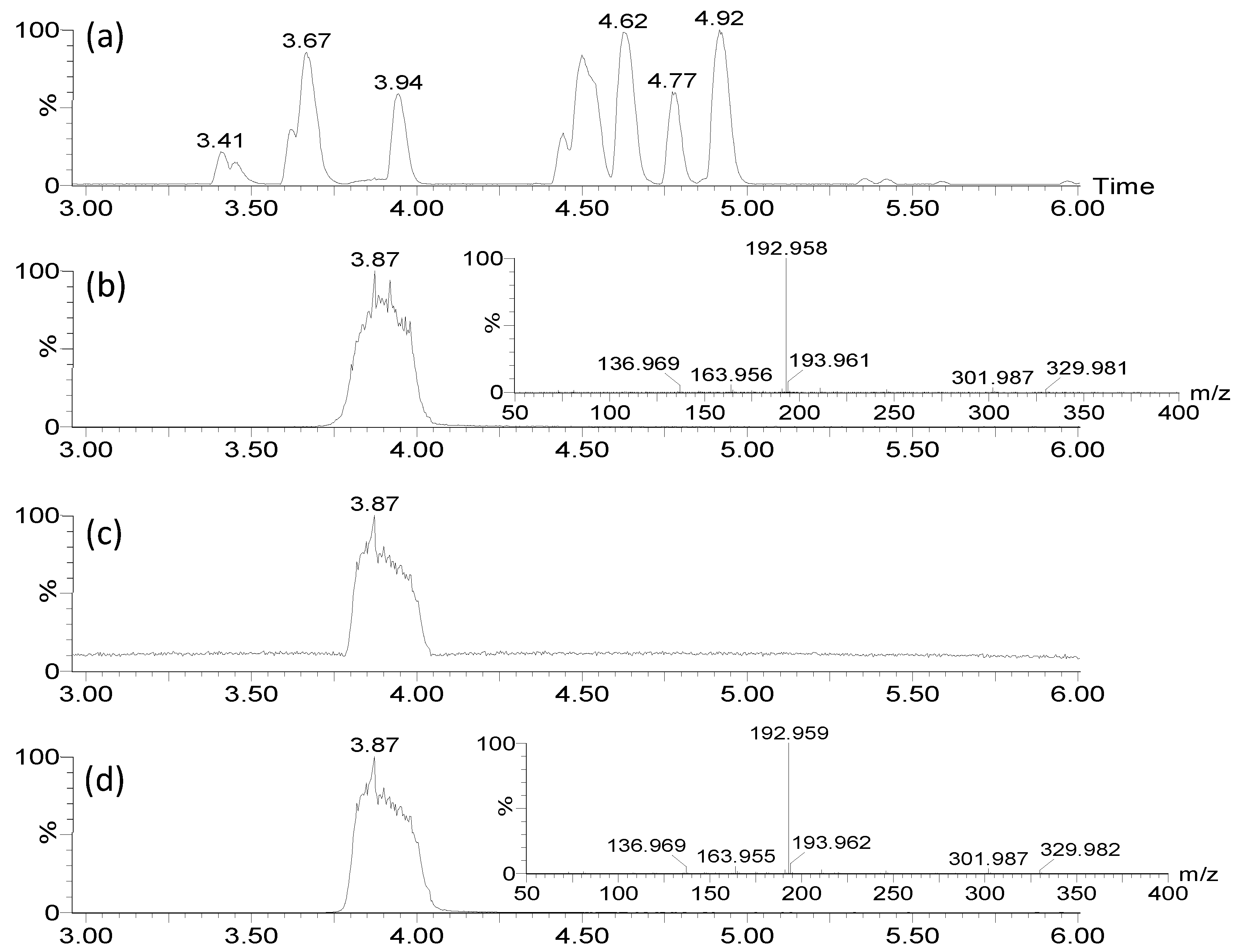
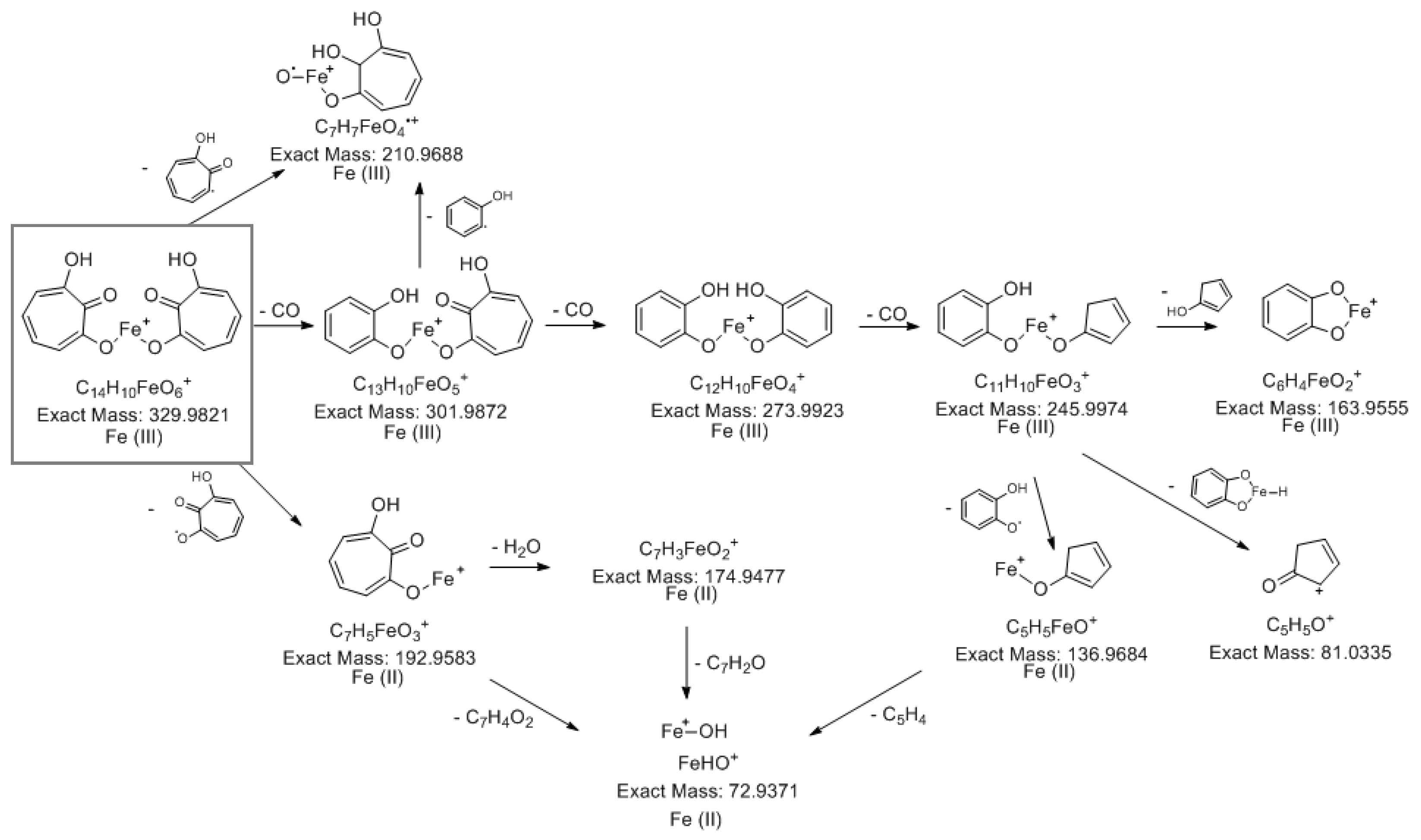
2.3.1. LC-MS analysis
2.3.2. Biologically driven purification using flash chromatography
2.3.3. Determination of the metabolite structure
2.4. Validation of 7-HT structure and activity
2.4.1. Organic synthesis of 7-HT
2.4.2. Quantification of 7-HT produced by PA14H7
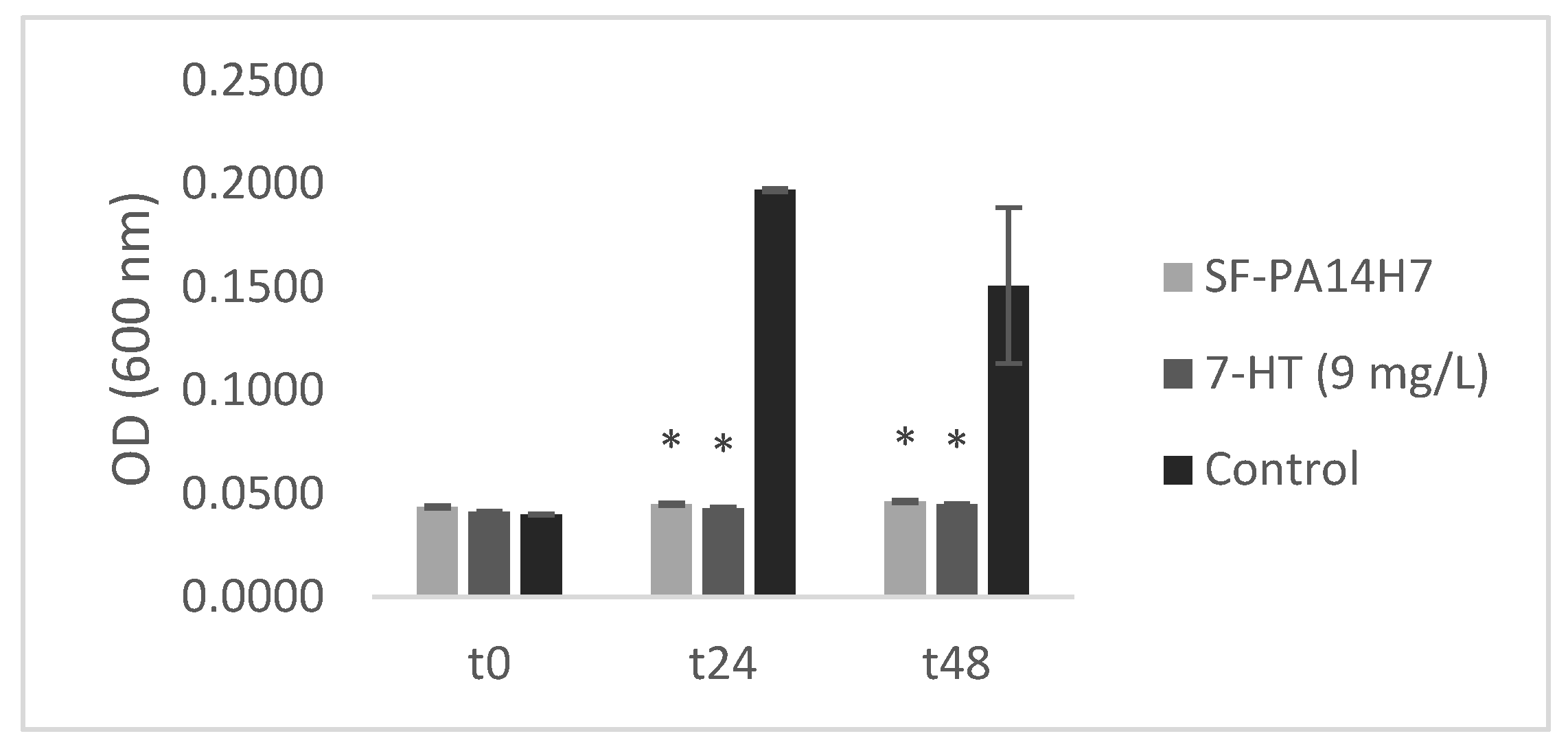
2.4.3. In vitro test of CFS-PA14H7 vs. synthetic 7-HT
3. Materials and Methods
3.1. Materials
3.2. Biological material
3.3. Biological test
3.4. Kinetics growth
3.5. Extraction method of CFS-PA14H7
3.6. LC-UV-MS
3.7. Purification by Flash chromatography
3.8. GC-MS
3.9. NMR
3.10. Quantification of 7-HT
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pérombelon, M.C.M. Potato Blackleg: Epidemiology, Host-Pathogen Interaction and Control. Netherlands Journal of Plant Pathology 1992, 98, 135–146. [Google Scholar] [CrossRef]
- Toth, I.K.; Barny, M.; Czajkowski, R.; Elphinstone, J.G.; Li, X.(Sean); Pédron, J.; Pirhonen, M.; Van Gijsegem, F. Pectobacterium and Dickeya: Taxonomy and Evolution. In Plant Diseases Caused by Dickeya and Pectobacterium Species; Van Gijsegem, F., van der Wolf, J.M., Toth, I.K., Eds.; Springer International Publishing: Cham, 2021; pp. 13–37. ISBN 978-3-030-61459-1. [Google Scholar]
- Dupuis, B.; Nkuriyingoma, P.; Van Gijsegem, F. Economic Impact of Pectobacterium and Dickeya Species on Potato Crops: A Review and Case Study. In Plant Diseases Caused by Dickeya and Pectobacterium Species; Van Gijsegem, F., van der Wolf, J.M., Toth, I.K., Eds.; Springer International Publishing: Cham, 2021; pp. 263–282. ISBN 978-3-030-61459-1. [Google Scholar]
- Van Der Wolf, J.M.; Acuña, I.; De Boer, S.H.; Brurberg, M.B.; Cahill, G.; Charkowski, A.O.; Coutinho, T.; Davey, T.; Dees, M.W.; Degefu, Y.; et al. Diseases Caused by Pectobacterium and Dickeya Species Around the World. In Plant Diseases Caused by Dickeya and Pectobacterium Species; Van Gijsegem, F., Van Der Wolf, J.M., Toth, I.K., Eds.; Springer International Publishing: Cham, 2021; pp. 215–261. ISBN 978-3-030-61458-4. [Google Scholar]
- Cirou, A. Développement de Stratégies d’anti-Virulence Ciblant La Régulation Quorum-Sensing Chez Pectobacterium atrosepticum, Bactérie Pathogène de La Pomme de Terre. These de doctorat, Paris 11, 2010.
- Cirou, A.; Mondy, S.; An, S.; Charrier, A.; Sarrazin, A.; Thoison, O.; DuBow, M.; Faure, D. Efficient Biostimulation of Native and Introduced Quorum-Quenching Rhodococcus Erythropolis Populations Is Revealed by a Combination of Analytical Chemistry, Microbiology, and Pyrosequencing. Appl Environ Microbiol 2012, 78, 481–492. [Google Scholar] [CrossRef]
- Blin, P.; Robic, K.; Khayi, S.; Cigna, J.; Munier, E.; Dewaegeneire, P.; Laurent, A.; Jaszczyszyn, Y.; Hong, K.; Chan, K.; et al. Pattern and Causes of the Establishment of the Invasive Bacterial Potato Pathogen Dickeya Solani and of the Maintenance of the Resident Pathogen D. Dianthicola. Mol Ecol 2021, 30, 608–624. [Google Scholar] [CrossRef]
- Zomorodian, K.; Ghadiri, P.; Saharkhiz, M.J.; Moein, M.R.; Mehriar, P.; Bahrani, F.; Golzar, T.; Pakshir, K.; Fani, M.M. Antimicrobial Activity of Seven Essential Oils From Iranian Aromatic Plants Against Common Causes of Oral Infections. Jundishapur J Microbiol 2015, 8, e17766. [Google Scholar] [CrossRef]
- Hajian-Maleki, H.; Baghaee-Ravari, S.; Moghaddam, M. Efficiency of Essential Oils against Pectobacterium Carotovorum Subsp. Carotovorum Causing Potato Soft Rot and Their Possible Application as Coatings in Storage. Postharvest Biology and Technology 2019, 156, 110928. [Google Scholar] [CrossRef]
- Azaiez, S.; Ben Slimene, I.; Karkouch, I.; Essid, R.; Jallouli, S.; Djebali, N.; Elkahoui, S.; Limam, F.; Tabbene, O. Biological Control of the Soft Rot Bacterium Pectobacterium Carotovorum by Bacillus Amyloliquefaciens Strain Ar10 Producing Glycolipid-like Compounds. Microbiological Research 2018, 217, 23–33. [Google Scholar] [CrossRef]
- Toth, I.K.; van der Wolf, J.M.; Saddler, G.; Lojkowska, E.; Hélias, V.; Pirhonen, M.; Tsror (Lahkim), L.; Elphinstone, J.G. Dickeya Species: An Emerging Problem for Potato Production in Europe. Plant Pathology 2011, 60, 385–399. [Google Scholar] [CrossRef]
- de Weert, S.; Bloemberg, G.V. Rhizosphere Competence and the Role of Root Colonization in Biocontrol. In Plant-Associated Bacteria; Gnanamanickam, S.S., Ed.; Springer Netherlands: Dordrecht, 2006; pp. 317–333. ISBN 978-1-4020-4538-7. [Google Scholar]
- Essarts, yannick R.D. Pathogénie de Dickeya dianthicola et Dickeya solani chez Solanum tuberosum, développement et évaluation de stratégies de lutte biologique. phdthesis, Université Paris Sud - Paris XI, 2015.
- Raoul des Essarts, Y.; Cigna, J.; Quêtu-Laurent, A.; Caron, A.; Munier, E.; Beury-Cirou, A.; Hélias, V.; Faure, D. Biocontrol of the Potato Blackleg and Soft Rot Diseases Caused by Dickeya Dianthicola. Applied and Environmental Microbiology 2016, 82, 268–278. [Google Scholar] [CrossRef] [PubMed]
- Cigna, J.; Robic, K.; Dewaegeneire, P.; Hélias, V.; Beury, A.; Faure, D. Efficacy of Soft-Rot Disease Biocontrol Agents in the Inhibition of Production Field Pathogen Isolates. Microorganisms 2023, 11, 372. [Google Scholar] [CrossRef]
- Syed Ab Rahman, S.F.; Singh, E.; Pieterse, C.M.J.; Schenk, P.M. Emerging Microbial Biocontrol Strategies for Plant Pathogens. Plant Science 2018, 267, 102–111. [Google Scholar] [CrossRef]
- Mishra, J.; Arora, N.K. Secondary Metabolites of Fluorescent Pseudomonads in Biocontrol of Phytopathogens for Sustainable Agriculture. Applied Soil Ecology 2018, 125, 35–45. [Google Scholar] [CrossRef]
- Dimkić, I.; Janakiev, T.; Petrović, M.; Degrassi, G.; Fira, D. Plant-Associated Bacillus and Pseudomonas Antimicrobial Activities in Plant Disease Suppression via Biological Control Mechanisms - A Review. Physiological and Molecular Plant Pathology 2022, 117, 101754. [Google Scholar] [CrossRef]
- Dowling, D.; O’Gara, F. Metabolites of Pseudomonas Involved in the Biocontrol of Plant Disease. Trends in Biotechnology 1994, 12, 133–141. [Google Scholar] [CrossRef]
- Muzio, F.M.; Agaras, B.C.; Masi, M.; Tuzi, A.; Evidente, A.; Valverde, C. 7-hydroxytropolone Is the Main Metabolite Responsible for the Fungal Antagonism of Pseudomonas Donghuensis Strain SVBP6. Environ Microbiol 2020, 22, 2550–2563. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, M.; Pagnani, G.; Bernardi, M.; Mattedi, A.; Spera, D.M.; Gallo, M.D. Cell-Free Supernatants of Plant Growth-Promoting Bacteria: A Review of Their Use as Biostimulant and Microbial Biocontrol Agents in Sustainable Agriculture. Sustainability 2020, 12, 9917. [Google Scholar] [CrossRef]
- Jain, R.; Pandey, A. A Phenazine-1-Carboxylic Acid Producing Polyextremophilic Pseudomonas Chlororaphis (MCC2693) Strain, Isolated from Mountain Ecosystem, Possesses Biocontrol and Plant Growth Promotion Abilities. Microbiological Research 2016, 190, 63–71. [Google Scholar] [CrossRef]
- Winter, N.; Trauner, D. Thiocarbonyl Ylide Chemistry Enables a Concise Synthesis of (±)-Hippolachnin A. J. Am. Chem. Soc. 2017, 139, 11706–11709. [Google Scholar] [CrossRef] [PubMed]
- Takeshita, H.; Mori, A. An Improved Synthesis of 2,7-Dihydroxytropolone (3-Hydroxytropolone). Synthesis 1986, 578–579. [Google Scholar] [CrossRef]
- Hamor, T.A.; Watkin, D.J. The Crystal Structure of Tris(Tropolonato)Iron( III )(“Ferric Tropolone”). J. Chem. Soc. D 1969, 0, 440–441. [Google Scholar] [CrossRef]
- Jiang, Z.; Chen, M.; Yu, X.; Xie, Z. 7-Hydroxytropolone Produced and Utilized as an Iron-Scavenger by Pseudomonas Donghuensis. Biometals 2016, 29, 817–826. [Google Scholar] [CrossRef]
- Cox, R.J.; Al-Fahad, A. Chemical Mechanisms Involved during the Biosynthesis of Tropolones. Current Opinion in Chemical Biology 2013, 17, 532–536. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Roman, D.; Beemelmanns, C. Tropolone Natural Products. Nat. Prod. Rep. 2019, 36, 1137–1155. [Google Scholar] [CrossRef]
- Cigna, J.; Raoul des Essarts, Y.; Mondy, S.; Hélias, V.; Beury-Cirou, A.; Faure, D. Draft Genome Sequences of Pseudomonas Fluorescens Strains PA4C2 and PA3G8 and Pseudomonas Putida PA14H7, Three Biocontrol Bacteria against Dickeya Phytopathogens. Genome Announc 2015, 3, e01503–14. [Google Scholar] [CrossRef] [PubMed]
- Pédron, J.; Mondy, S.; Raoul des Essarts, Y.; Van Gijsegem, F.; Faure, D. Genomic and Metabolic Comparison with Dickeya Dadantii 3937 Reveals the Emerging Dickeya Solani Potato Pathogen to Display Distinctive Metabolic Activities and T5SS/T6SS-Related Toxin Repertoire. BMC Genomics 2014, 15, 283. [Google Scholar] [CrossRef]
- Haluk, J.-P.; Roussel-Bousta, C. Biosynthèse de tropolones dans les cals et les suspensions cellulaires à partir d’ébauches foliaires de plantules de Thuja plicata Donn. Ann. For. Sci. 2003, 60, 271–276. [Google Scholar] [CrossRef]
- Czajkowski, R.; Pérombelon, M.C.M.; van Veen, J.A.; van der Wolf, J.M. Control of Blackleg and Tuber Soft Rot of Potato Caused by Pectobacterium and Dickeya Species: A Review. Plant Pathology 2011, 60, 999–1013. [Google Scholar] [CrossRef]
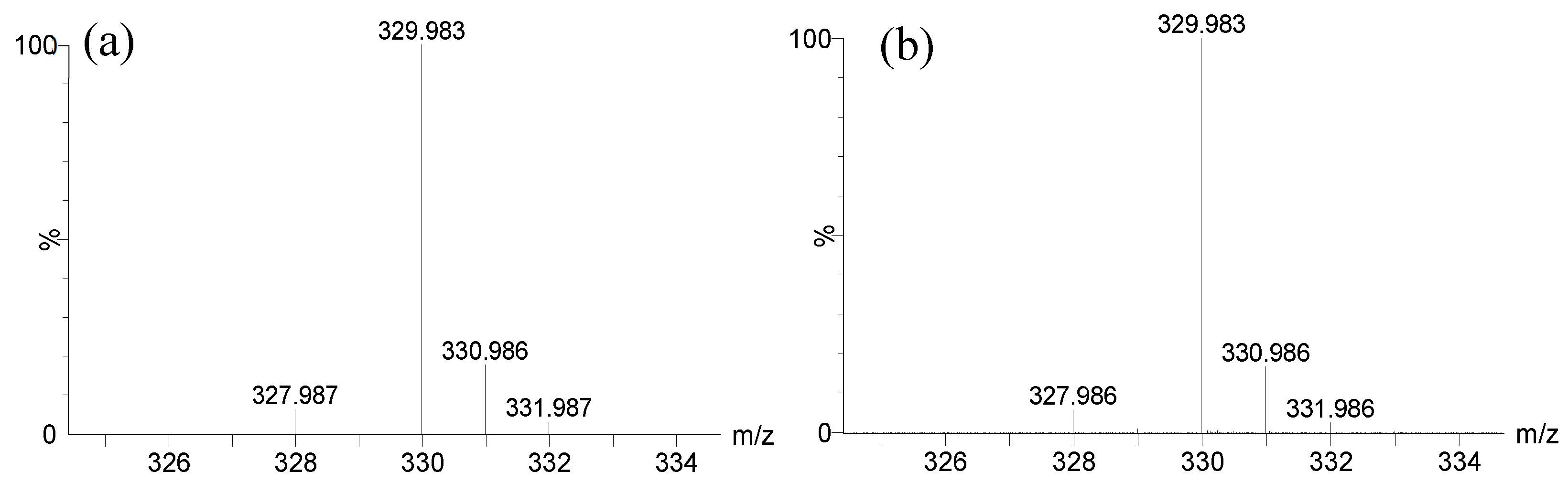
| Antagonist bacteria strains | ||||||
|---|---|---|---|---|---|---|
| SRP strains | PA3G8 | PA1G7 | PA4C2 | PA14H7 | PA2H3 | PP1-210F |
| P. atrosepticum P8-Me25a | - | ++ | - | ++ | - | ++ |
| P. parmentieri P13-CH22 | - | - | - | + | - | - |
| P. brasiliense P1-15C1 | - | - | - | + | - | - |
| P. polaris P1-10C1 | - | - | - | + | - | - |
| D. dianthicola P15-29 | - | - | - | + | - | - |
| D. solani P5-Sp1a | - | - | - | + | - | - |
| D. solani 3337 | - | - | - | + | - | - |
| Aqueous phase (dilution factor) |
Organic phase (dilution factor) |
||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Extraction conditions | 1 | 0.5 | 0.25 | 0.1 | 0.05 | 0.01 | 1 | 0.5 | 0.25 | 0.1 | 0.05 | 0.01 | |
| Ethyl acetate | pH 2 | - | - | - | - | - | - | ++ | ++ | ++ | ++ | ++ | - |
| pH 7 | - | - | - | - | - | - | ++ | ++ | ++ | ++ | + | - | |
| Chloroform | pH 2 | - | - | - | - | - | - | ++ | ++ | ++ | ++ | ++ | - |
| pH 7 | - | - | - | - | - | - | ++ | ++ | + | + | + | - | |
| Dilution factor | ||||||||
|---|---|---|---|---|---|---|---|---|
| Fraction | 1 | 0.5 | 0.25 | 0.1 | 0.05 | 0.01 | 0.005 | 0.001 |
| F1 | - | - | - | - | - | - | - | - |
| F2 | ++ | ++ | ++ | ++ | ++ | + | + | - |
| F3 | ++ | ++ | + | - | - | - | - | - |
| Analytical methods | |||||
|---|---|---|---|---|---|
| Extraction conditions | LC-UV | LC-MS | GC-MS | ||
|
Extraction of CFS PA14H71 |
Ethyl acetate | pH 2 | 8.9 | 8.5 | 12.4 |
| pH 7 | 5.7 | 6.0 | 7.3 | ||
| Chloroform | pH 2 | 7.9 | 8.2 | 10.0 | |
| pH 7 | 10.3 | 8.5 | 8.9 | ||
| Synthetic 7-HT in water (9.6 mg/L) | Ethyl acetate | pH 2 | n.d. | 8.9 | 9.7 |
| Chloroform | pH 7 | n.d. | 11.3 | 9.2 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




