Submitted:
04 July 2023
Posted:
10 July 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Statistical Analyses
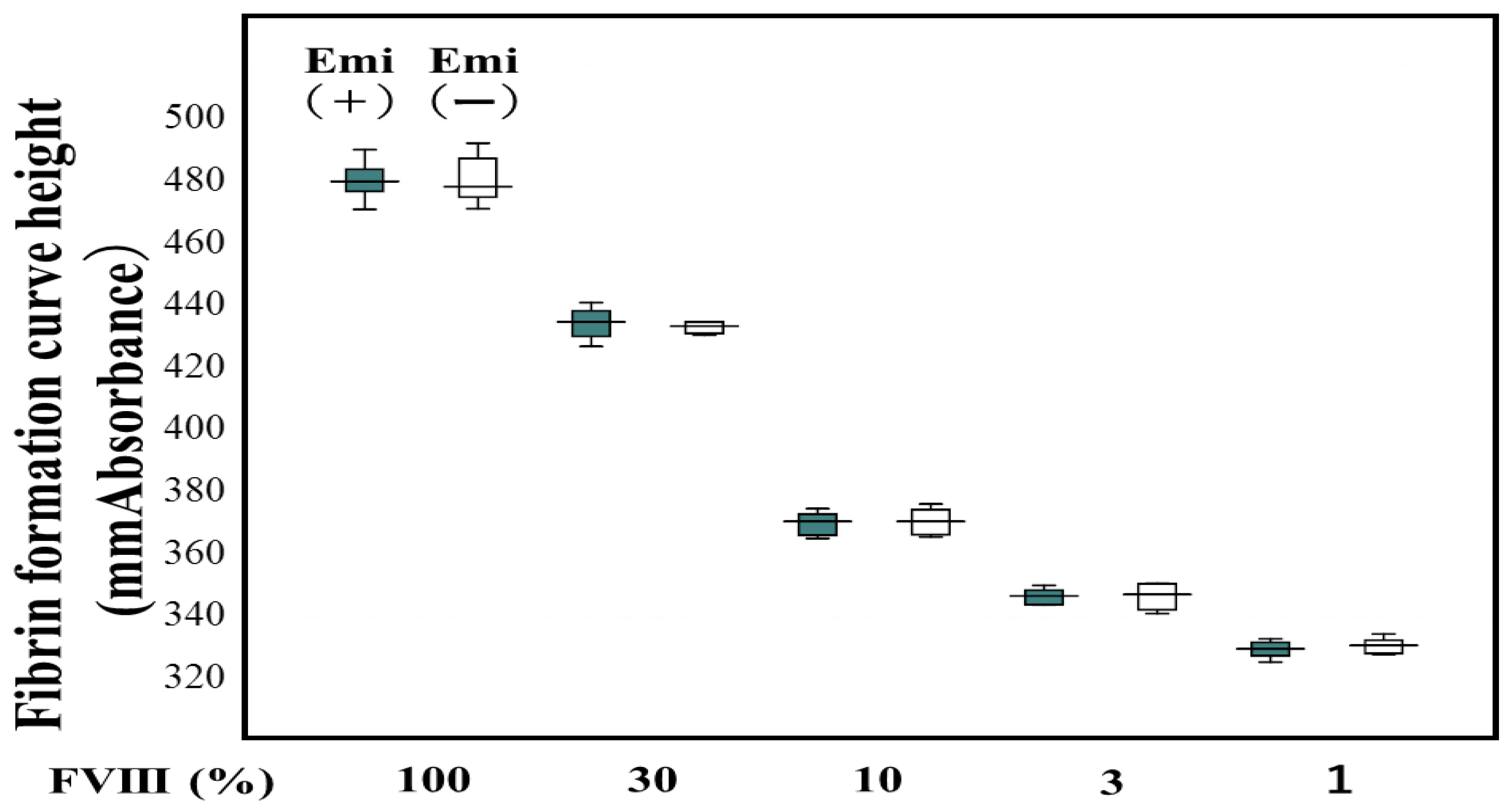
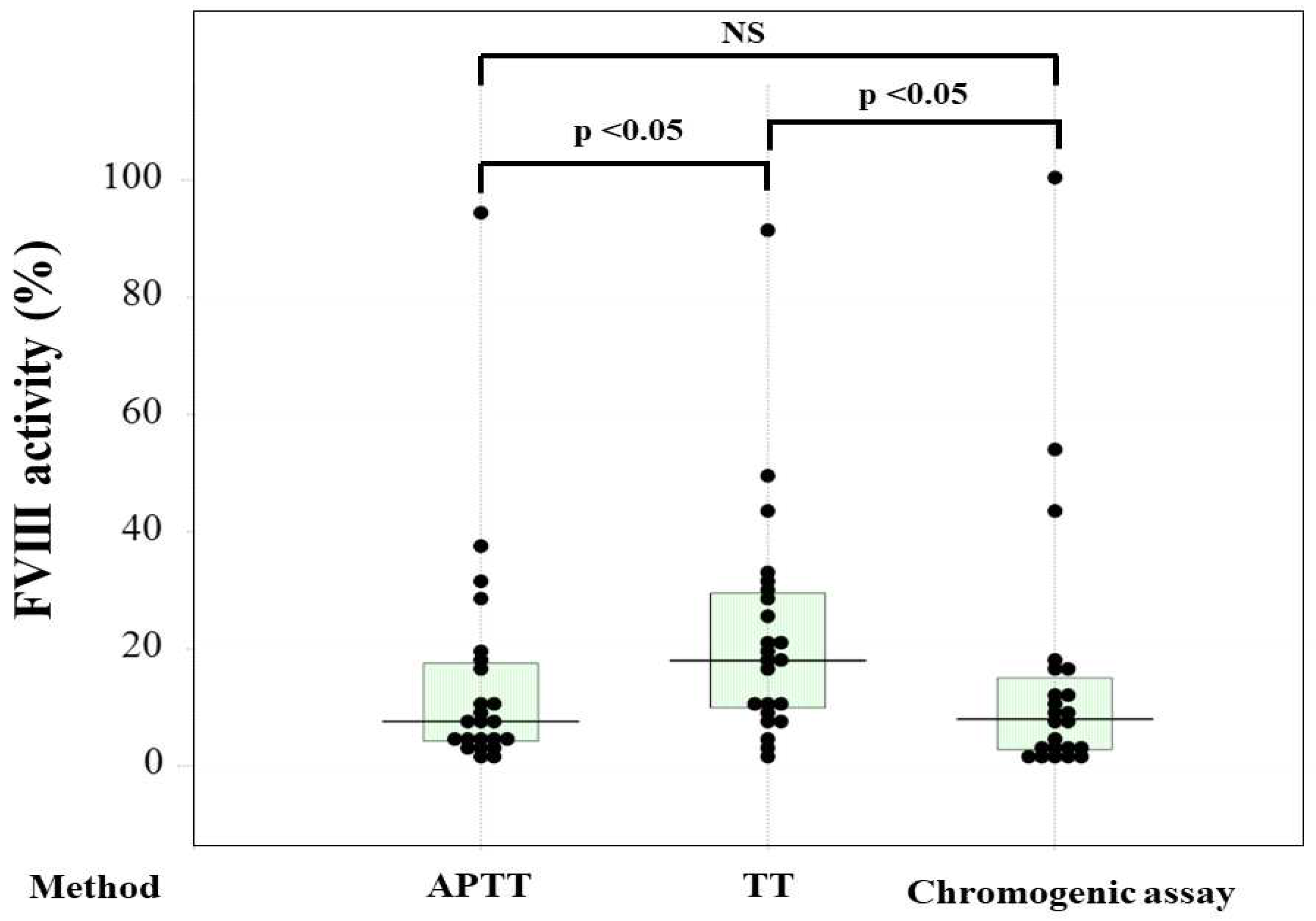
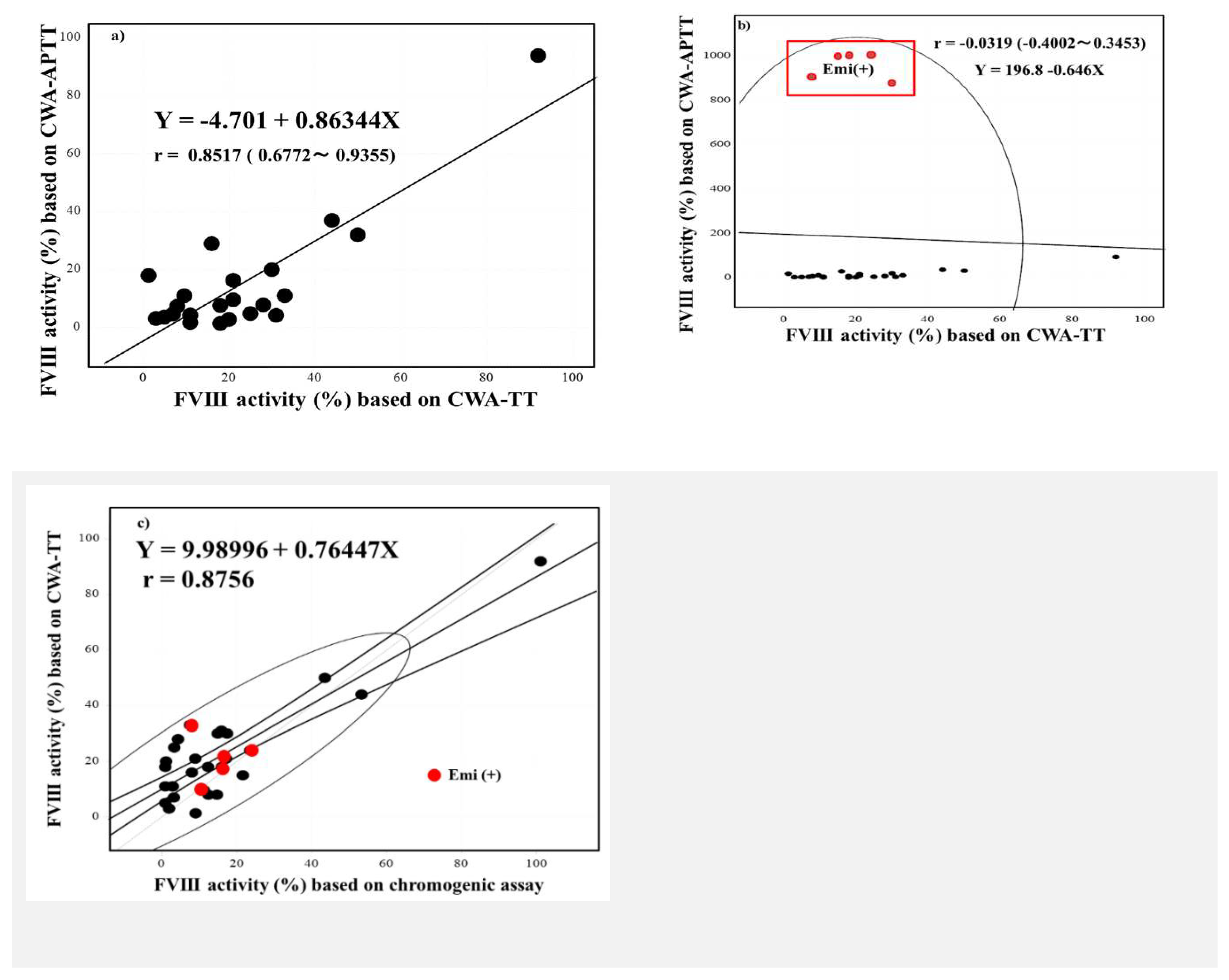
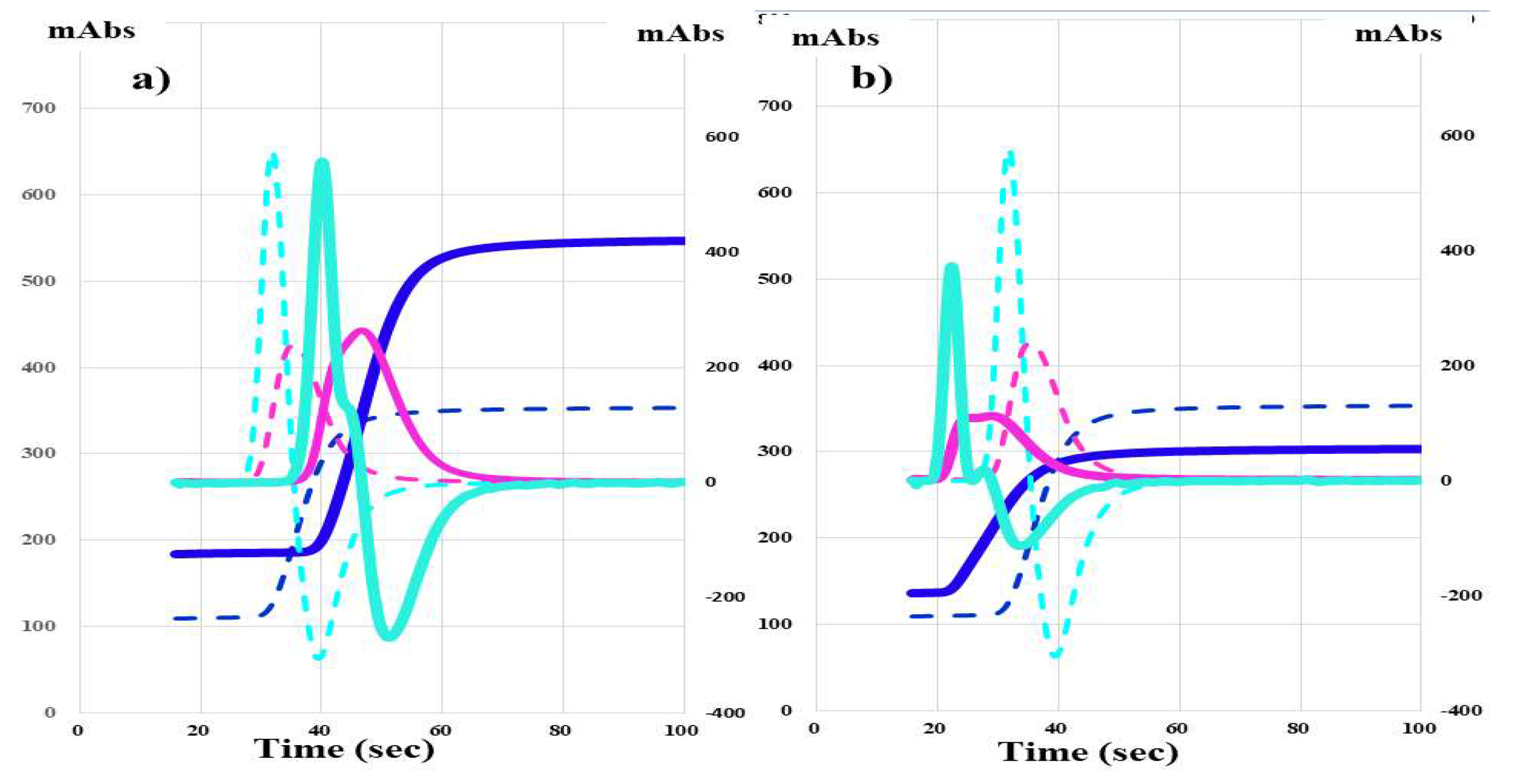
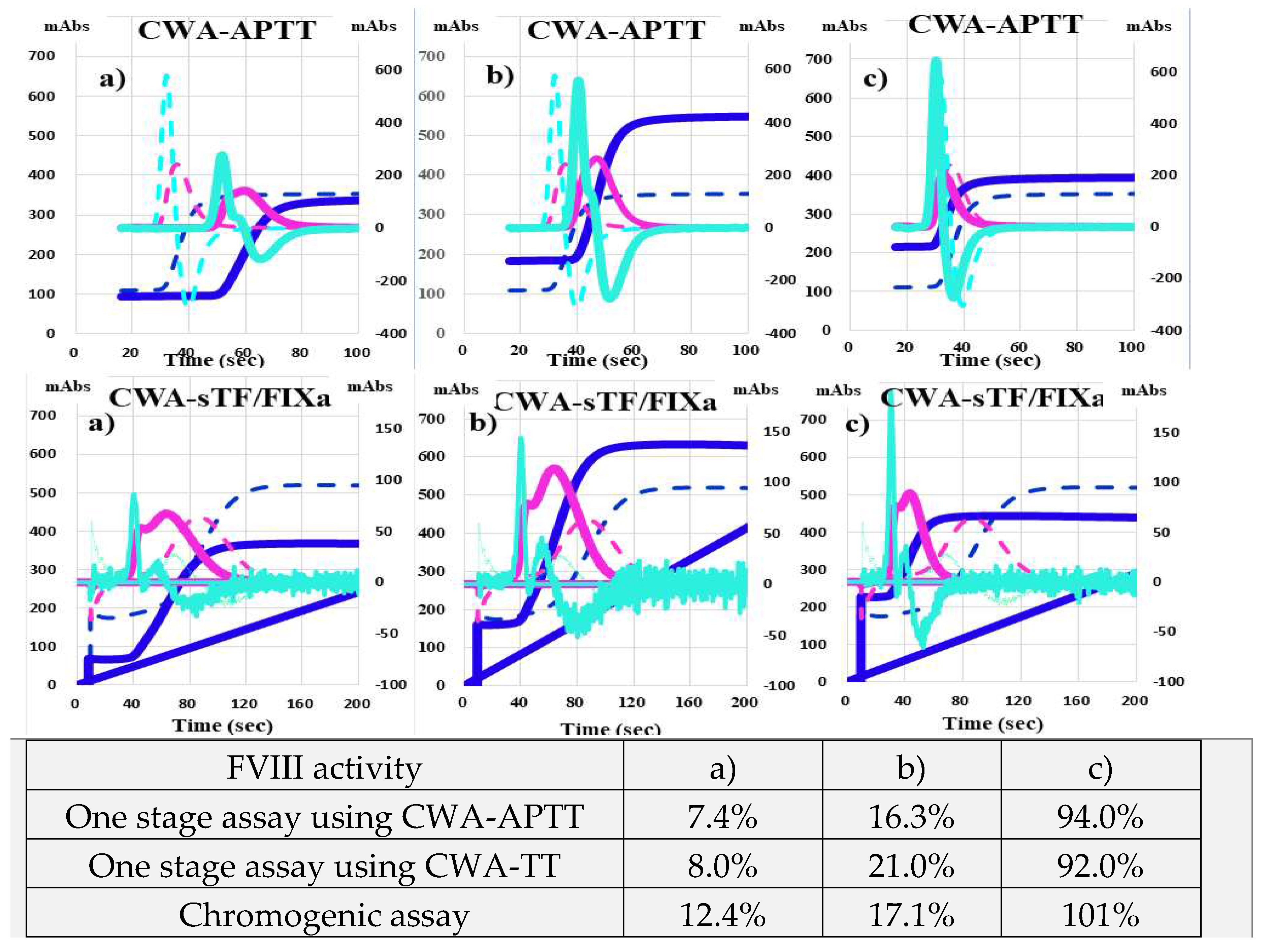
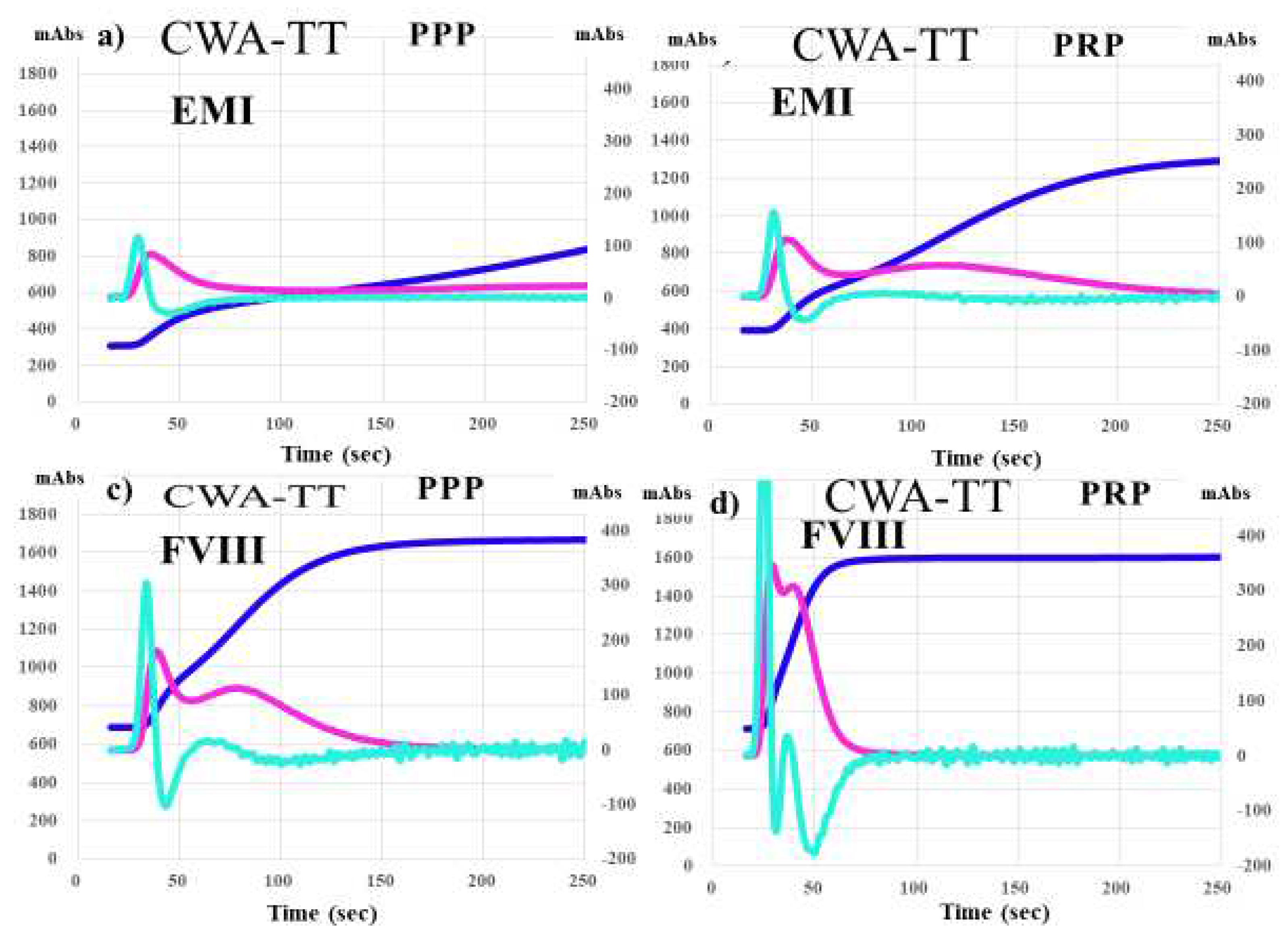
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Páramo, J.A. Treatment of haemophilia: From replacement to gene therapy. Med Clin (Barc). 2021, 157, 583–587. [Google Scholar] [CrossRef] [PubMed]
- Carcao, M.; Srivastava, A. Factor VIII/factor IX prophylaxis for severe hemophilia. Semin Hematol. 2016, 53, 3–9. [Google Scholar] [CrossRef]
- National Hemophilia Foundation. MASAC document 267- MASAC recommendation concerning prophylaxis for hemophilia A and B with and without inhibitors. Available online: http://www.hemophilia.org/NHFWeb/MainPgs/MainNHF.aspx?menuid=57&contentid=1007 (accessed on 22 June 2023).
- Manco-Johnson, M.J.; Abshire, T.C.; Shapiro, A.D.; et al. Prophylaxis versus episodic treatment to prevent joint disease in boys with severe hemophilia. N Engl J Med 2007, 357, 535–544. [Google Scholar] [CrossRef] [PubMed]
- Di Minno, G.; Cerbone, A.M.; Coppola, A.; et al. Longer-acting factor VIII to overcome limitations in haemophilia management: the PEGylated liposomes formulation issue. Haemophilia 2010, 16 (Suppl. 1), 2–6. [Google Scholar] [CrossRef] [PubMed]
- von Drygalski, A.; Chowdary, P.; Kulkarni, R.; Susen, S.; Konkle, B.A.; Oldenburg, J.; Matino, D.; Klamroth, R.; Weyand, A.C.; Jimenez-Yuste, V.; Nogami, K.; Poloskey, S.; Winding, B.; Willemze, A.; Knobe, K. XTEND-1 Trial Group. : Efanesoctocog Alfa Prophylaxis for Patients with Severe Hemophilia A. N Engl J Med. 2023, 388, 310–318. [Google Scholar] [PubMed]
- Keam, S.J. Efanesoctocog Alfa: First Approval. Drugs. 2023, 83, 633–638. [Google Scholar] [CrossRef]
- Oldenburg, J.; Mahlangu, J.N.; Kim, B.; Schmitt, C.; Callaghan, M.U.; Young, G.; Santagostino, E.; Kruse-Jarres, R.; Negrier, C.; Kessler, C.; Valente, N.; Asikanius, E.; Levy, G.G.; Windyga, J.; Shima, M. Emicizumab Prophylaxis in Hemophilia A with Inhibito. N Engl J Med. 2017, 377, 809–818. [Google Scholar] [CrossRef]
- Mahlangu, J.; Oldenburg, J.; Paz-Priel, I.; Negrier, C.; Niggli, M.; Mancuso, M.E.; Schmitt, C.; Jiménez-Yuste, V.; Kempton, C.; Dhalluin, C.; Callaghan, M.U.; Bujan, W.; Shima, M.; Adamkewicz, J.I.; Asikanius, E.; Levy, G.G.; Kruse-Jarres, R. Emicizumab Prophylaxis in Patients Who Have Hemophilia A without Inhibitors. N Engl J Med. 2018, 379, 811–822. [Google Scholar] [CrossRef]
- Nogami, K.; Soeda, T.; Matsumoto, T.; Kawabe, Y.; Kitazawa, T.; Shima, M. Routine measurements of factor VIII activity and inhibitor titer in the presence of emicizumab utilizing anti-idiotype monoclonal antibodies. J Thromb Haemost. 2018, 16, 1383–1390. [Google Scholar] [CrossRef]
- Peyvandi, F.; Kenet, G.; Pekrul, I.; Pruthi, R.K.; Ramge, P.; Spannagl, M.J. Laboratory testing in hemophilia: Impact of factor and non-factor replacement therapy on coagulation assays. Thromb Haemost. 2020, 18, 1242–1255. [Google Scholar] [CrossRef]
- Gray, E.; Kitchen, S.; Bowyer, A.; Chowdary, P.; Jenkins, P.V.; Murphy, P.; Platton, S.; Riddell, A.; Lester, W. Laboratory measurement of factor replacement therapies in the treatment of congenital haemophilia: A United Kingdom Haemophilia Centre Doctors' Organisation guideline. Haemophilia. 2020, 26, 6–16. [Google Scholar] [CrossRef] [PubMed]
- Jeanpierre, E.; Pouplard, C.; Lasne, D.; Le Cam Duchez, V.; Eschwege, V.; Flaujac, C.; Galinat, H.; Harzallah, I.; Proulle, V.; Smahi, M.; Sobas, F.; Stepina, N.; Toulon, P.; Voisin, S.; Ternisien, C.; Nougier, C. French Study Group on the Biology of Hemorrhagic Diseases (the BIMHO group). Factor VIII and IX assays for post-infusion monitoring in hemophilia patients: Guidelines from the French BIMHO group (GFHT). Eur J Haematol. 2020, 105, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Wada, H.; Matsumoto, T.; Ohishi, K.; Shiraki, K.; Shimaoka, M. Update on the Clot Waveform Analysis. Clin Appl Thromb Hemost; 26:1076029620912027.
- Wada, H.; Ichikawa, Y.; Ezaki, E.; Matsumoto, T.; Yamashita, Y.; Shiraki, K.; Shimaoka, M.; Shimpo, H. The reevaluation of thrombin time using a clot waveform analysis, J. Clin. Med. 2021, 10, 4840. [Google Scholar] [CrossRef] [PubMed]
- Wada, H.; Shiraki, K.; Matsumoto, T.; Suzuki, K.; Yamashita, Y.; Tawara, I.; Shimpo, H.; Shimaoka, M. A Clot Waveform Analysis of Thrombin Time Using a Small Amount of Thrombin Is Useful for Evaluating the Clotting Activity of Plasma Independent of the Presence of Emicizumab. J Clin Med. 2022, 11, 6142. [Google Scholar] [CrossRef]
- Wada, H.; Shiraki, K.; Matsumoto, T.; Ohishi, K.; Shimpo, H.; Shimaoka, M. Effects of platelet and phospholipids on clot formation activated by a small amount of tissue factor. Thromb Res 2020, 193, 146–153. [Google Scholar] [CrossRef]
- Matsumoto, T.; Wada, H.; Toyoda, H.; Hirayama, M.; Yamashita, Y.; Katayama, N. Modified clot waveform analysis to measure plasma coagulation potential in the presence of the anti-factor IXa/factor X bispecific antibody emicizumab: comment. J Thromb Haemost. 2018, 16, 1665–1666. [Google Scholar] [CrossRef]
- Iorio, A.; Königs, C.; Reding, M.T.; Rotellini, D.; Skinner, M.W.; Mancuso, M.E.; Berntorp, E. Prophylaxis use of clotting factor replacement products in people with non-severe haemophilia: A review of the literature. Haemophilia. 2023, 29, 33–44. [Google Scholar] [CrossRef]
- Schmitt, C.; Mancuso, M.E.; Chang, T.; Podolak-Dawidziak, M.; Petry, C.; Sidonio Jr, R.; Yoneyama, K.; Key, N.S.; Niggli, M.; Lehle, M.; Peyvandi, F.; Oldenburg, J. Emicizumab dose up-titration in case of suboptimal bleeding control in people with haemophilia, A. Haemophilia. 2023, 29, 90–99. [Google Scholar] [CrossRef]
- Nogami, K.; Shima, M. Current and future therapies for haemophilia-Beyond factor replacement therapies. Br J Haematol. 2023, 200, 23–34. [Google Scholar] [CrossRef]
- López-Jaime, F.J.; Benítez, O.; Díaz Jordán, B.L.; Montaño, A.; Coll, J.; Quintana París, L.; Gómez-Del Castillo Solano, M.D.C. Expert opinion paper on the treatment of hemophilia a with emicizumab. Hematology. 2023, 28, 2166334. [Google Scholar] [CrossRef]
- Nardi, M.A. Hemophilia A: Emicizumab monitoring and impact on coagulation testing. Adv Clin Chem. 2023, 113, 273–315. [Google Scholar] [PubMed]
- Dev, P.; Ekhlak, M.; Dash, D.; Pathak, A. Platelet function suggests cardioembolic aetiology in cryptogenic stroke. Sci Rep. 2023, 13, 7615. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K.; Wada, H.; Shinkai, T.; Tanemura, A.; Matsumoto, T.; Mizuno, S. Evaluation of hemostatic abnormalities in patients who underwent major hepatobiliary pancreatic surgery using activated partial thromboplastin time-clot waveform analysis. Thromb Res. 2021, 201, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Nogami, K.; Shima, M. A combined approach using global coagulation assays quickly differentiates coagulation disorders with prolonged aPTT and low levels of FVIII activity. Int J Hematol. 2017, 105, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Nogami, K. Semin Thromb Hemost.: Clot Waveform Analysis for Monitoring Hemostasis. 2023 (in press).
- Kobayashi, M.; Wada, H.; Fukui, S.; Mizutani, H.; Ichikawa, Y.; Shiraki, K.; Moritani, I.; Inoue, H.; Shimaoka, M.; Shimpo, H. A Clot Waveform Analysis Showing a Hypercoagulable State in Patients with Malignant Neoplasms. J Clin Med. 2021, 10, 5352. [Google Scholar] [CrossRef]
- Kamon, T.; Horie, S.; Inaba, T.; Ito, N.; Shiraki, K.; Ichikawa, Y.; Ezaki, M.; Shimpo, H.; Shimaoka, M.; Nishigaki, A.; Shindo, A.; Wada, H. The Detection of Hypercoagulability in Patients with Acute Cerebral Infarction Using a Clot Waveform Analysis. Clin Appl Thromb Hemost. 2023, 29, 10760296231161591. [Google Scholar] [CrossRef]
| No | Disease | Severity | FVIII activity | Inhibitor | RRT | Drug |
|---|---|---|---|---|---|---|
| HA-1 | HA | Severe | ≤1.0% | Negative | Yes | Rurioctocog alfa pegol |
| HA-2 | HA | Moderate | 1.8% | Negative | Yes | Efraloctocog alfa |
| HA-3 | HA | Severe | ≤1.0% | Negative | Yes | Rurioctocog alfa pegol |
| HA-4 | HA | Severe | ≤1.0% | Negative | Yes | Rurioctocog alfa pegol |
| HA-5 | HA | Mild | 23.3% | Negative | No | Octocog beta |
| HA-6 | HA | Severe | ≤1.0% | Positive | Yes | Emicizumab |
| HA-7 | HA | Severe | ≤1.0% | Negative | Yes | Octocog beta |
| HA-8 | HA | Moderate | ≤1.0% | Negative | No | Rurioctocog alfa pegol |
| HA-9 | HA | Severe | ≤1.0% | Negative | Yes | Emicizumab |
| HA-10 | HA | Severe | ≤1.0% | Negative | Yes | Lonoctocog alfa |
| HA-11 | HA | Severe | 1.0% | Negative | Yes | Octocog beta |
| HA-12 | HA | Mild | 14.1% | Negative | No | FDCHB-FVIII |
| HA-13 | HA | Severe | ≤1.0% | Negative | Yes | Rurioctocog alfa pegol |
| HA-14 | HA | Moderate | 1.6% | Negative | No | ― |
| HA-15 | HA | Moderate | 3.9% | Negative | No | Rurioctocog alfa pegol |
| HA-16 | HA | Mild | 6.7% | Negative | No | Rurioctocog alfa |
| HA-17 | HA | Mild | 5.4% | Negative | No | Rurioctocog alfa |
| HA-18 | HA | Moderate | 2.2% | Negative | No | ― |
| HA-19 | HA | Severe | ≤1.0% | Negative | Yes | Rurioctocog alfa pegol |
| HA-20 | HA | Severe | ≤1.0% | Negative | Yes | Emicizumab |
| HA-21 | HA | Moderate | 1.0% | Negative | Yes | Emicizumab |
| HA-22 | HA | Severe | ≤1.0% | Negative | Yes | Emicizumab |
| HA-23 | HA | Severe | ≤1.0% | Negative | Yes | Efraloctocog alfa |
| 24 | HA* | Mild | 20.4% | Negative | No | ― |
| 25-1 | AHA | Severe | ≤1.0% | Positive | No | APCC |
| 25-2 | AHA | Severe | ≤1.0% | Positive | No | APCC |
| 25-3 | AHA | Severe | ≤1.0% | Positive | No | APCC |
| 25-4 | AHA | Severe | ≤1.0% | Positive | No | ― |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




