1. Introduction
Sol-gel technology has been widely used since 1980 in the history of materials science [
1,
2] in the process of synthesizing superconducting materials [
3], functional ceramic materials [
4], nonlinear optical materials [
5], catalysts and enzyme carriers for biomedical applications [
6,
7], porous glass and other materials [
8,
9]. This method for the developing of functional surfaces over multiple substrates is gaining much more attention since many applications are finding a reduction in cost and complexity compared with other methodologies. Among the substrates where these coatings can currently be applied there is a large repertoire, from fabrics for many textile applications [
10] to polymers for conferring flame retardant features [
11] or glass for the study of antifouling coatings for marine applications [
12].
Sol gel SiO
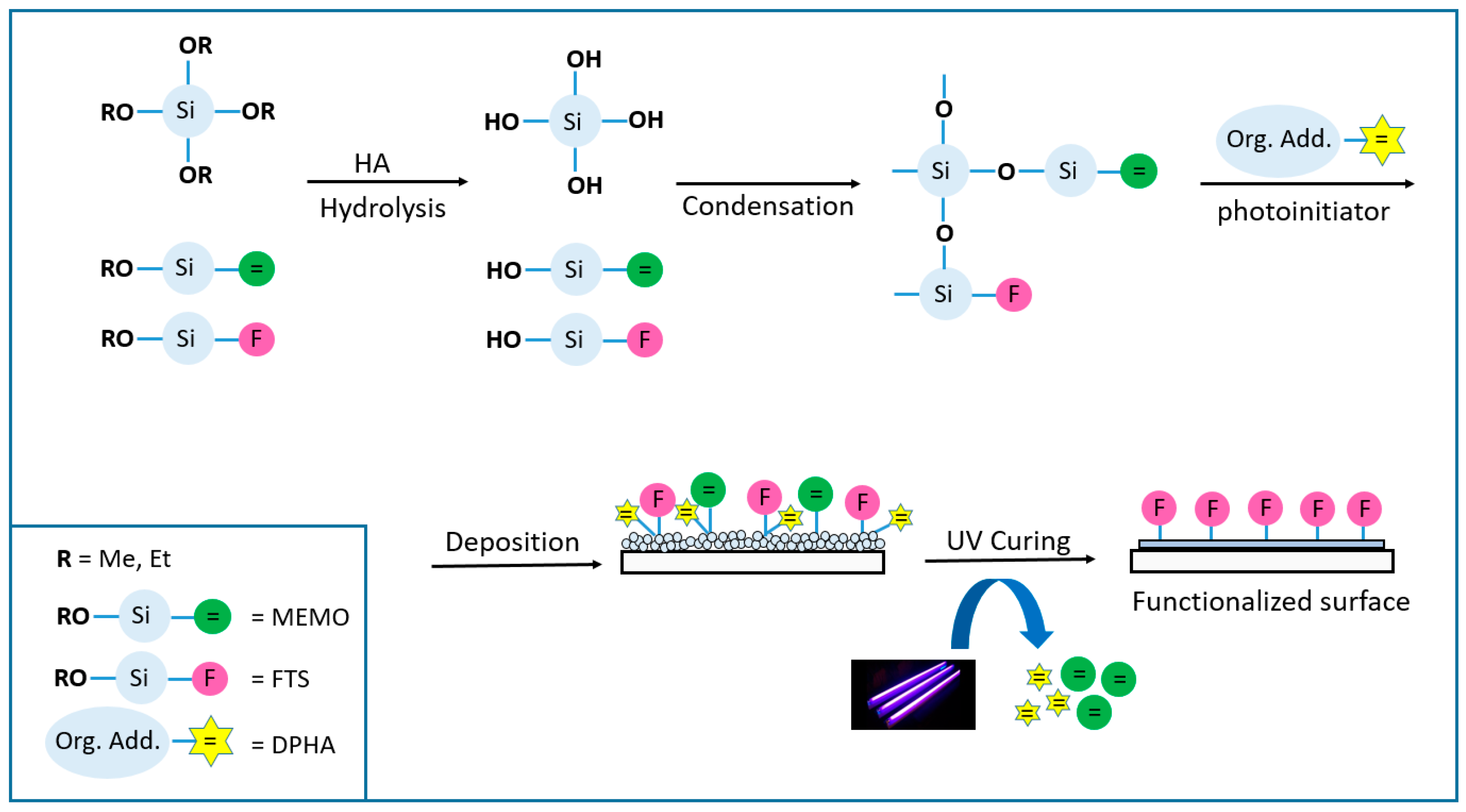
2 coatings are obtained mainly by dissolving an alkoxide precursor in alcohol together with the appropriate additives, which are then hydrolyzed in water in the presence of an acid or basic catalyst source. Once the hydrosilylation reaction takes place, the alcohol molecules are condensed forming a cross-linked polysiloxane network, and at this point, the gelation process starts. After the deposition process of the solution prepared by spinning, dipping or spraying (among others) over the substrate, it is necessary to carry out a densification of the layer to complete the gelification process by heating at high temperatures or by polymerization under UV light (technology considered in this work), leading to a compact layer free of cracks or striations, chemically and physically resistant (
Figure 1).
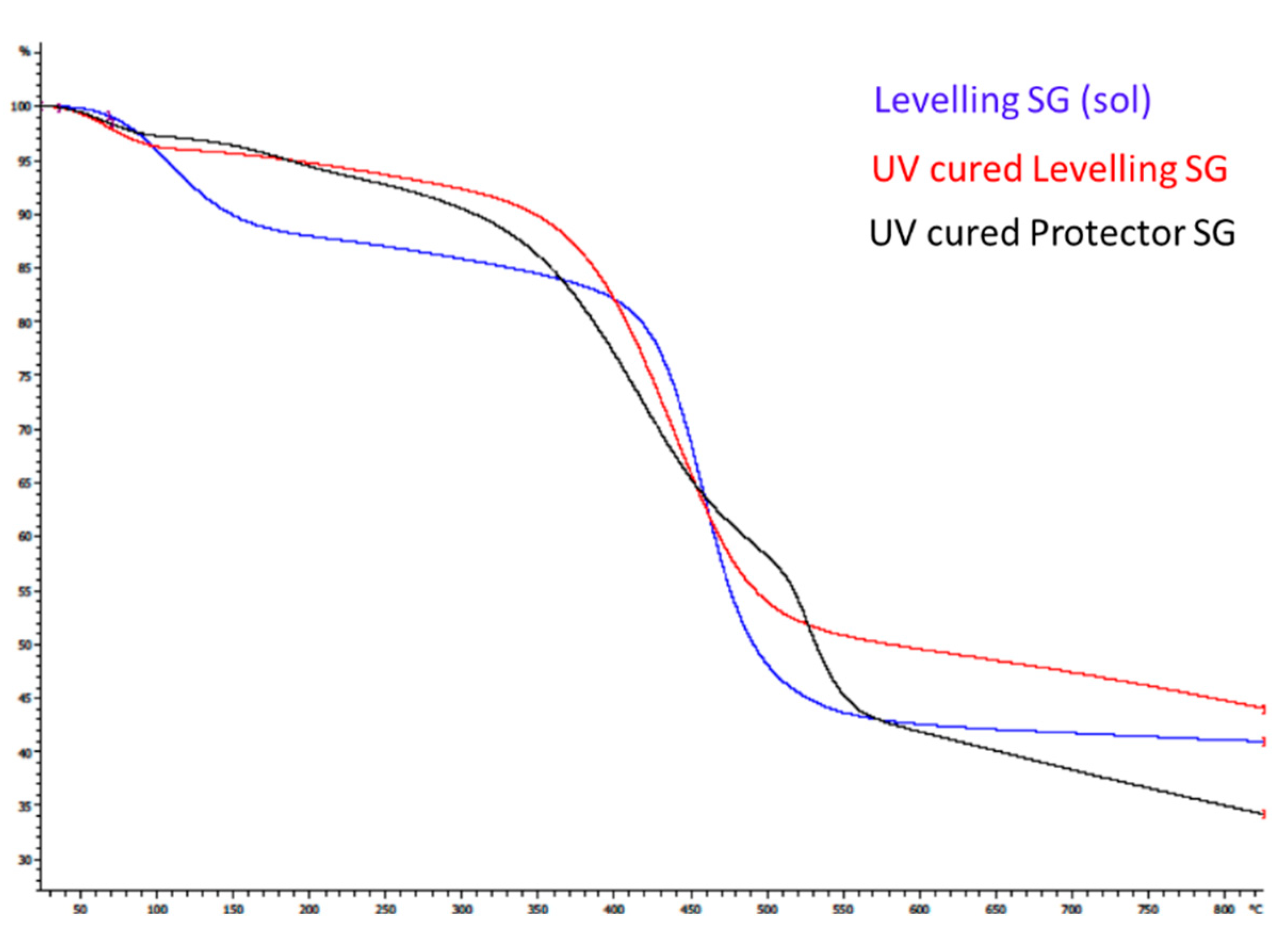
Research of sol gel coatings cured under UV light without requiring high-temperature hardening processes on plastic surfaces have recently been the center of attention. In addition to the low temperature curing process, the UV-cured films have various advantages such as high chemical and thermal stability, less harm for the environment, lower processing costs, and higher rates of hardening if the appropriate medium-pressure lamps are employed.
A photopolymerisable coating formulation essentially consists of adding a polymerisable molecule (oligomer, in the present work dipentaerythritol penta-/hexa-acrylate_ DPHA) and a light-sensitive compound or photoinitiator that is able to convert the absorbed light energy into a more useful form capable of causing the binder to polymerise into a hard solid film. The photoinitiator produces initiator radicals directly by the fragmentation of the photo-excited state, and for this kind of coatings, aromatic α-hydroxiketones like 1-hydroxycyclohexyl phenyl ketone (Irgacure 184) or 2-Hydroxy-2-methylpropiophenone (Darocure 1173) were suitable candidates [
13]. In these systems, photoinitiators mainly affect cure speed, yellowing, and cost. In addition, the light absorption properties of the selected photoinitiator need to match the emission wavelength of the light source [
14].
Figure 1 represents the general process for this technology, where it is noticeable how the double bonds of the acrylate molecules are eliminated upon radical reactions and co-polymerization of the different chemicals involved in the process (3-(Trimethoxysilyl) propyl methacrylate_MEMO and the acrylic monomer DPHA) causing the total gelification of the coating.
Organic – inorganic hybrid sol gel coatings (OIH) were studied for their application over stainless steel [
15] and other metal substrates, in order to evaluate corrosion protection [
16]. Intermolecular interactions between the macromolecular existing structures and the metallic surface are reviewed in the literature to achieve low porosity, rigidity and adhesion to the metallic substrate, creating a good physical barrier against oxidation, corrosion and erosion among other processes.
A model of protective coating produced in five steps using mainly sol–gel methods was studied by Hughes et al. in the form of a multilayer coating system with a total thickness of about 16 µm [
17]. They propose a protection with dual behavior: a protective barrier taking advantage of the OIH properties with a self-healing response. In this multilayer system, the formulation of the different layers is based on organic-inorganic hybrid sol gel matrix composition because of the advantage of being produced at room temperature conditions on an industrial scale, by using well-established and low-cost methods.
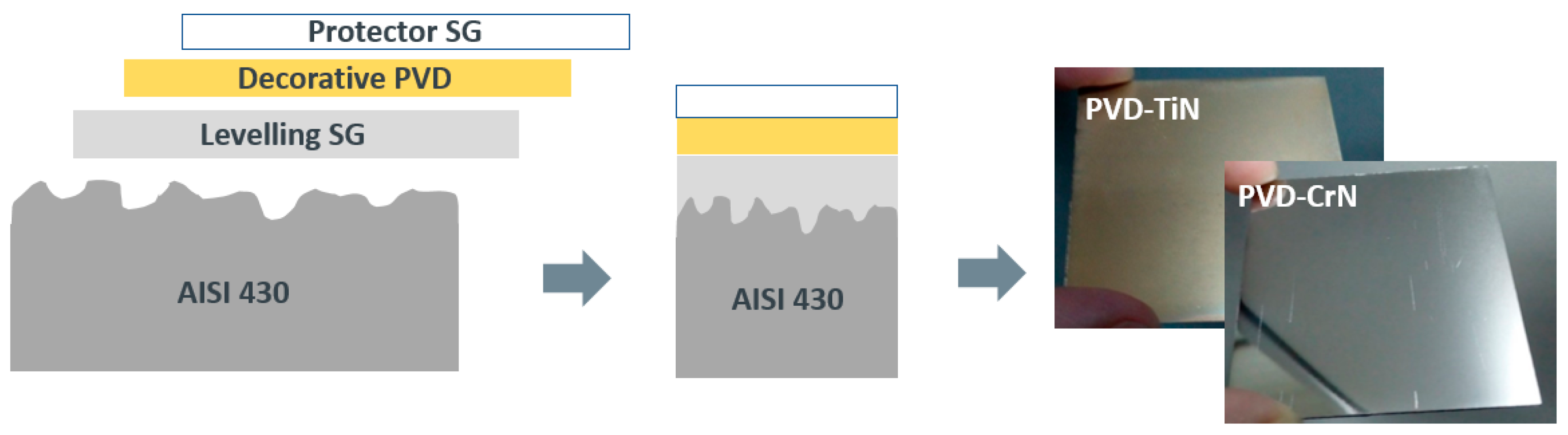
The first aim of this work is to analyze the levelling capability of a sol gel coating onto low-cost metallic substrates to prepare the material for an additional decorative PVD layer, which requires a smooth surface to be deposited. Although there are many studies in the literature based on levelling thermally cured sol gel coatings, to our knowledge, the UV curing version for this application has not been studied.
For example, Guillen et al. studied the levelling capability of sol gel SiO
2 layers onto titanium and Kovar (Fe54 Ni29 Co17) foil substrates [
18]. In this work, the coatings were prepared by immersion techniques and the roughness of the coating was determined for different withdrawal rates, and therefore thickness of the film, after the dip-coating deposition method. The sintering step of the sol-gel layers was obtained by heat treating in air at 400 °C for 1 h, and the Ra levelling parameter was improved (decreased) by adding successive sol gel SiO
2 layers with their corresponding heating treatment.
Similarly, it was recently described a method for the functionalization of low-cost steel with application on solar cells, with the properties of surface levelling, electrical insulation and anti-diffusion barrier [
19]. The application method for this work was the spray coating and the samples were cured by near-infrared technique (NIR) at an irradiation power of 90%. A curing time of 2 min was applied in all cases (reaching a peak metal temperature of 198 °C), which represents a clear time reduction compared to conventional hot-plate curing (typically close to one hour).
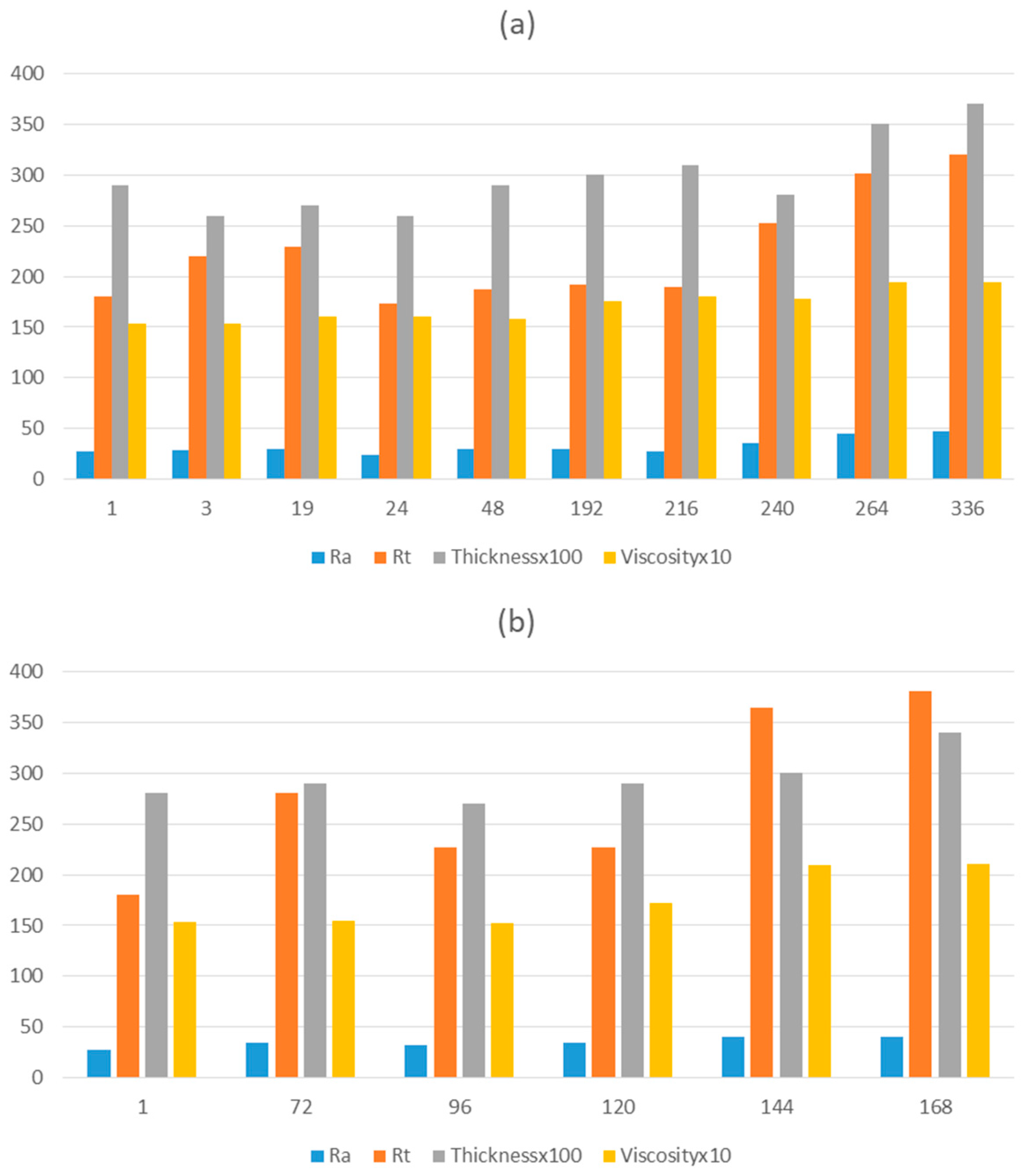
In a thermal curing process, densification of the layer is usually obtained by heating at temperatures over 400°C. By using the fotocurable method, the levelling layer could be performed under mild processing conditions (low temperature and atmospheric pressure). For this purpose, AISI 430 stainless steel foils were covered with the optimal formulation parameters at various thicknesses, first by spin coating and later by spraying techniques, in a large-scale study. The analysis of the roughness, topography and morphology of these samples allowed us to conclude that a levelling functionality of the film has been achieved, with good adhesion properties and employing ultrafast curing methods.
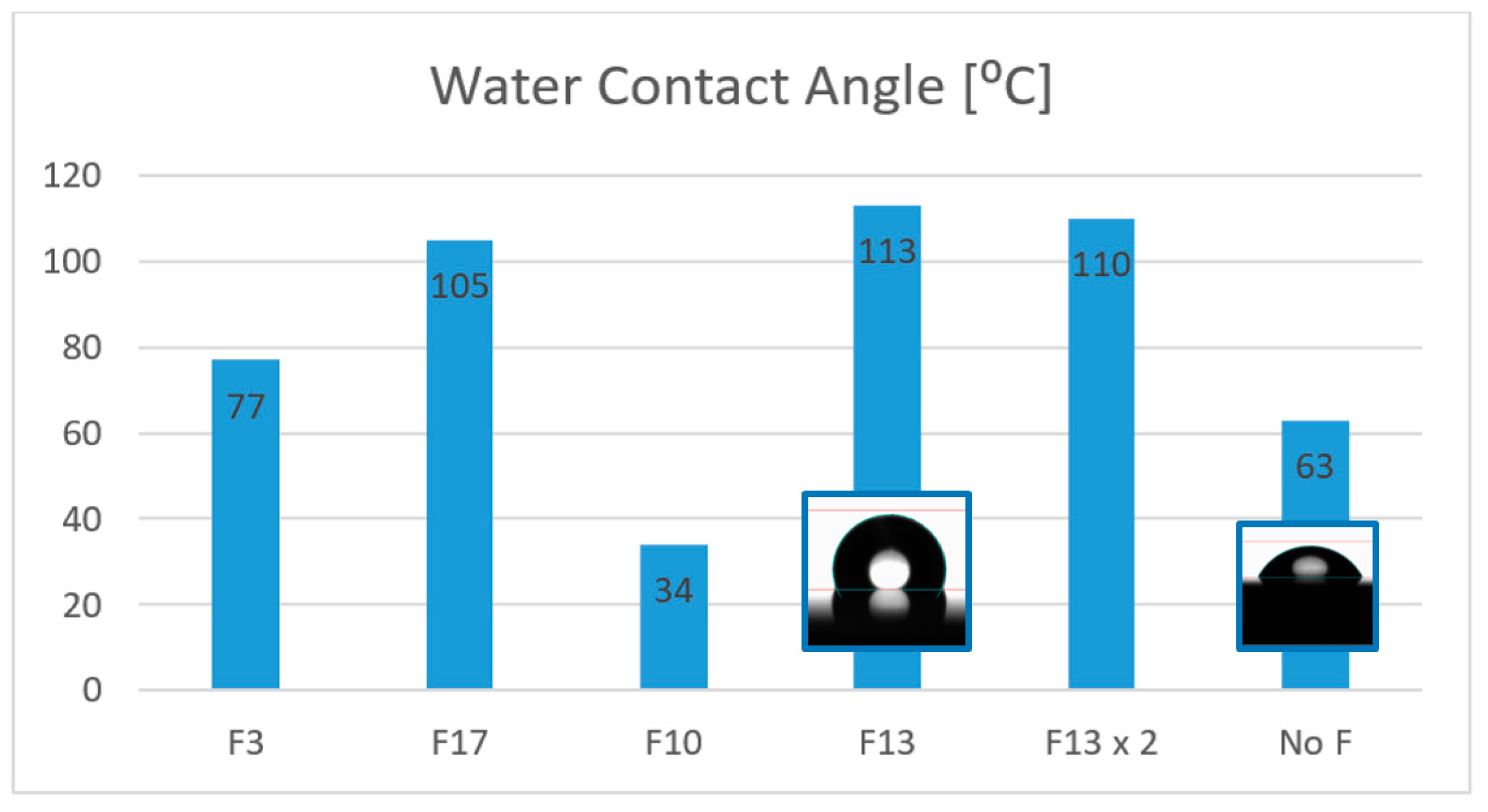
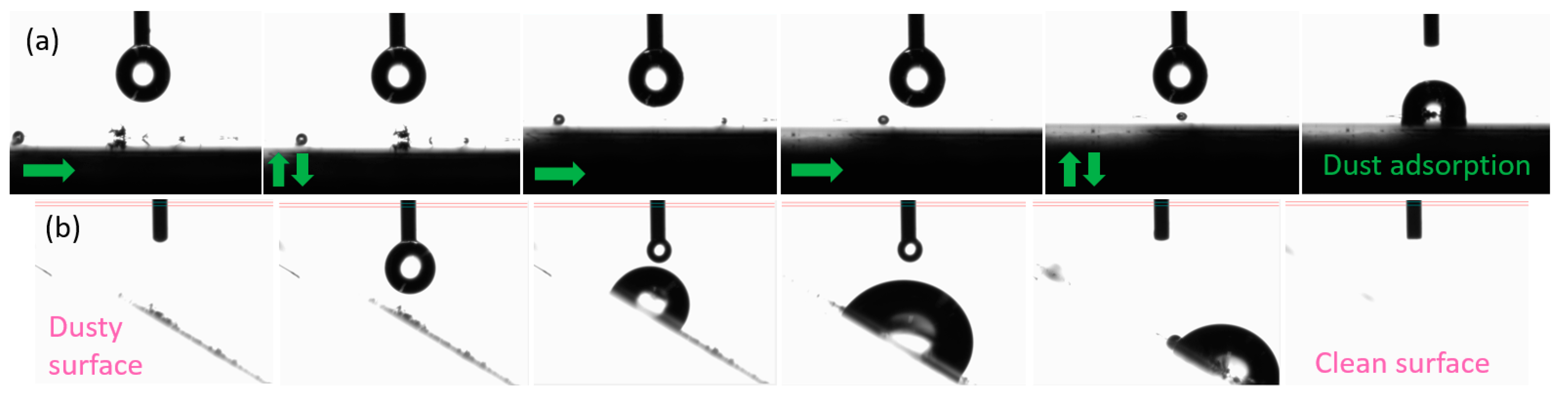
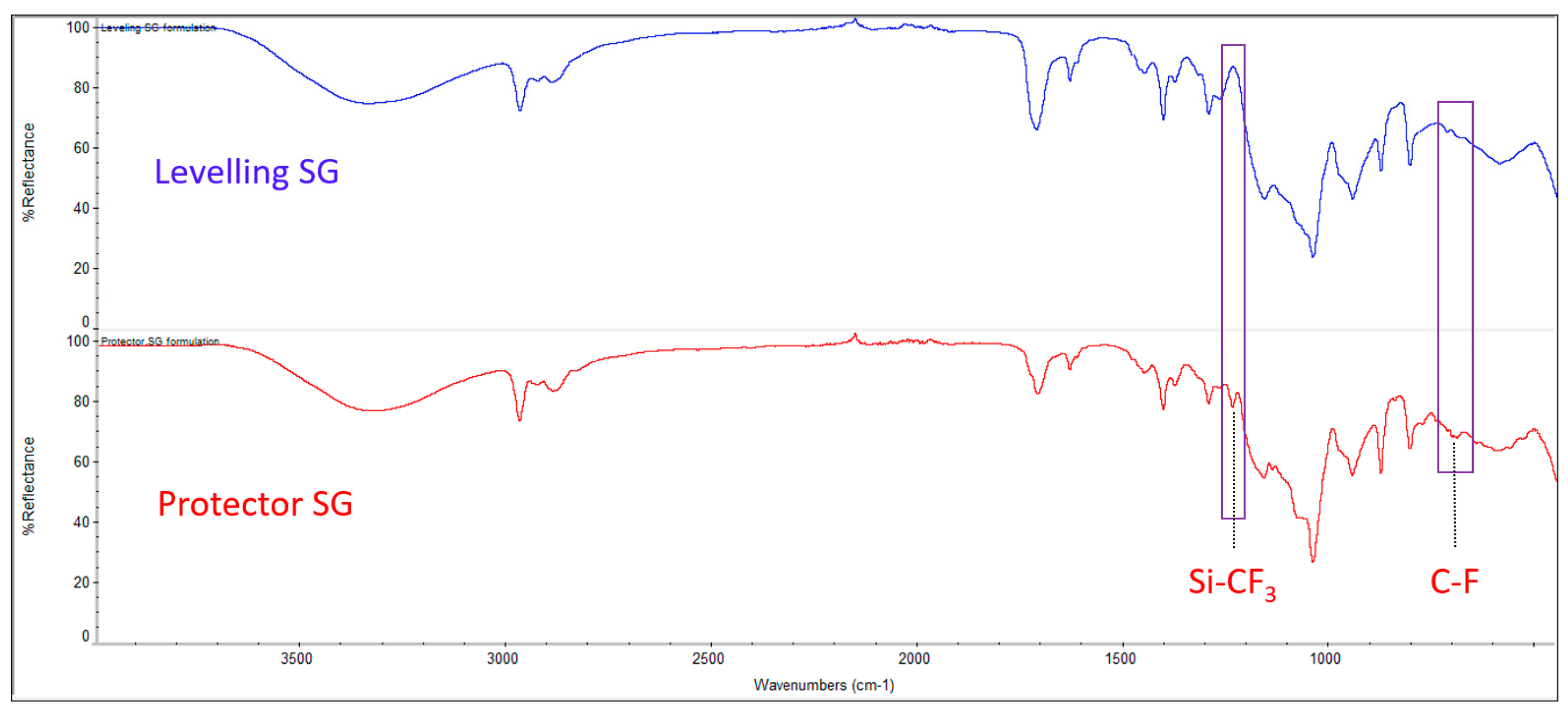
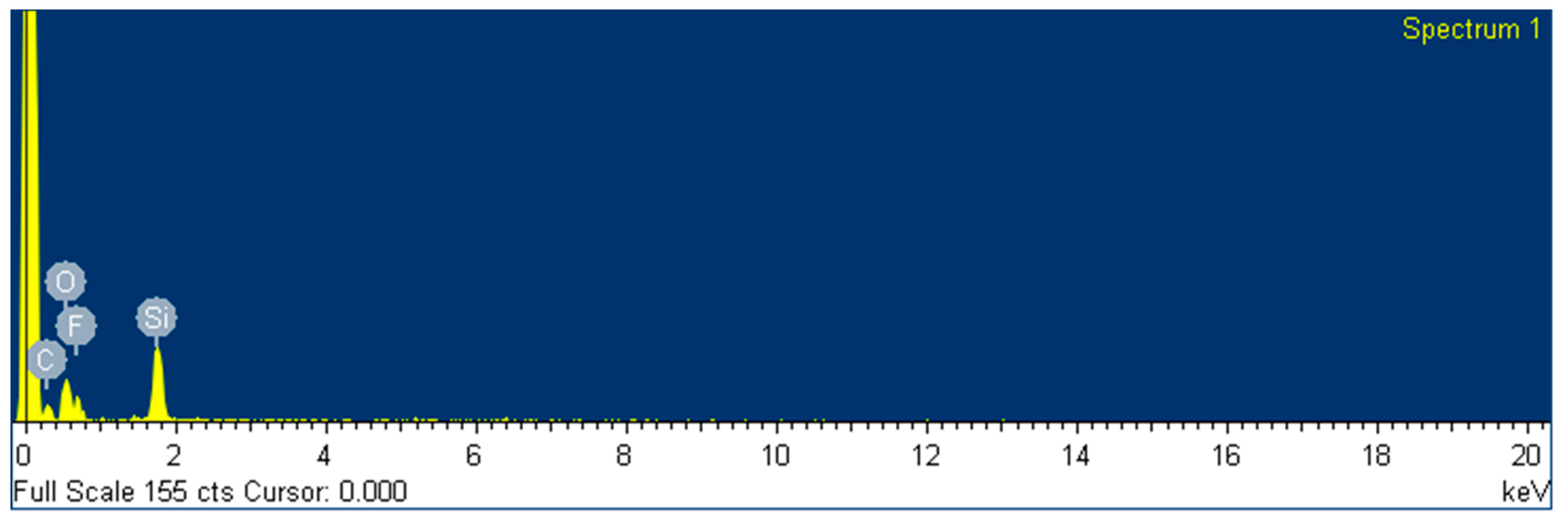
On the other hand, we studied the addition of a fluoro-compound with a low surface energy in the appropriate step of the formulation to convert the previously designed coating onto a hydrophobic surface, thereby easily achieving a new functionality for different materials just by adding an extra additive. There are few examples in the literature for this kind of UV-curable coating [
20,
21] but all of them are focused exclusively on the coating of plastics such as polymethyl methacrylate or polycarbonate, and not applicable to metallic surfaces, requiring an extra step of heating before UV curing and applied by different methods in small samples.
Here we report the optimized formula for a dual-functional coating onto stainless steel surfaces, not only for laboratory scale but also for covering large areas and different geometries by employing different deposition methods.