Submitted:
25 June 2023
Posted:
26 June 2023
You are already at the latest version
Abstract

Keywords:
1. Introduction
2. Pathophysiology of Intracranial Meningiomas
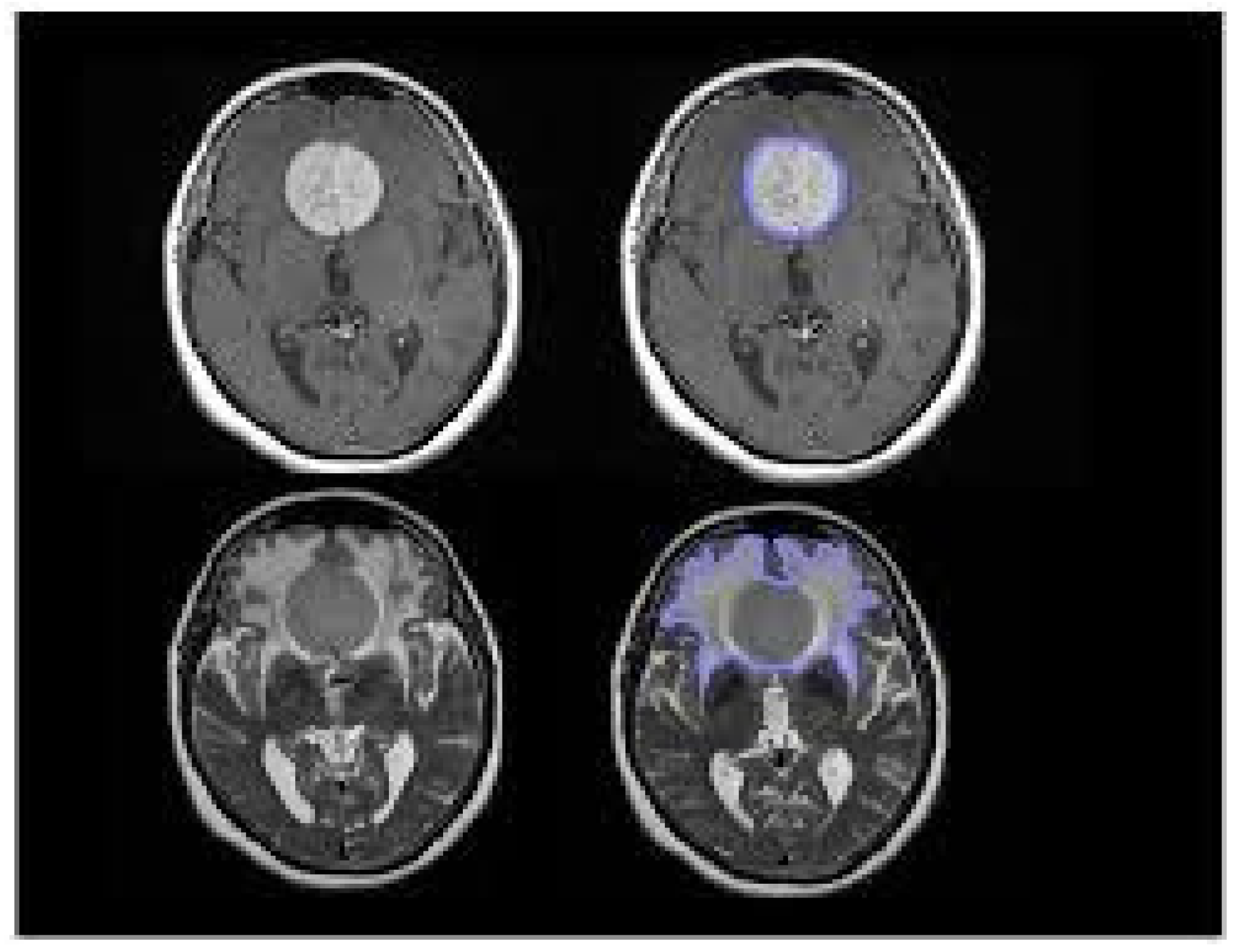
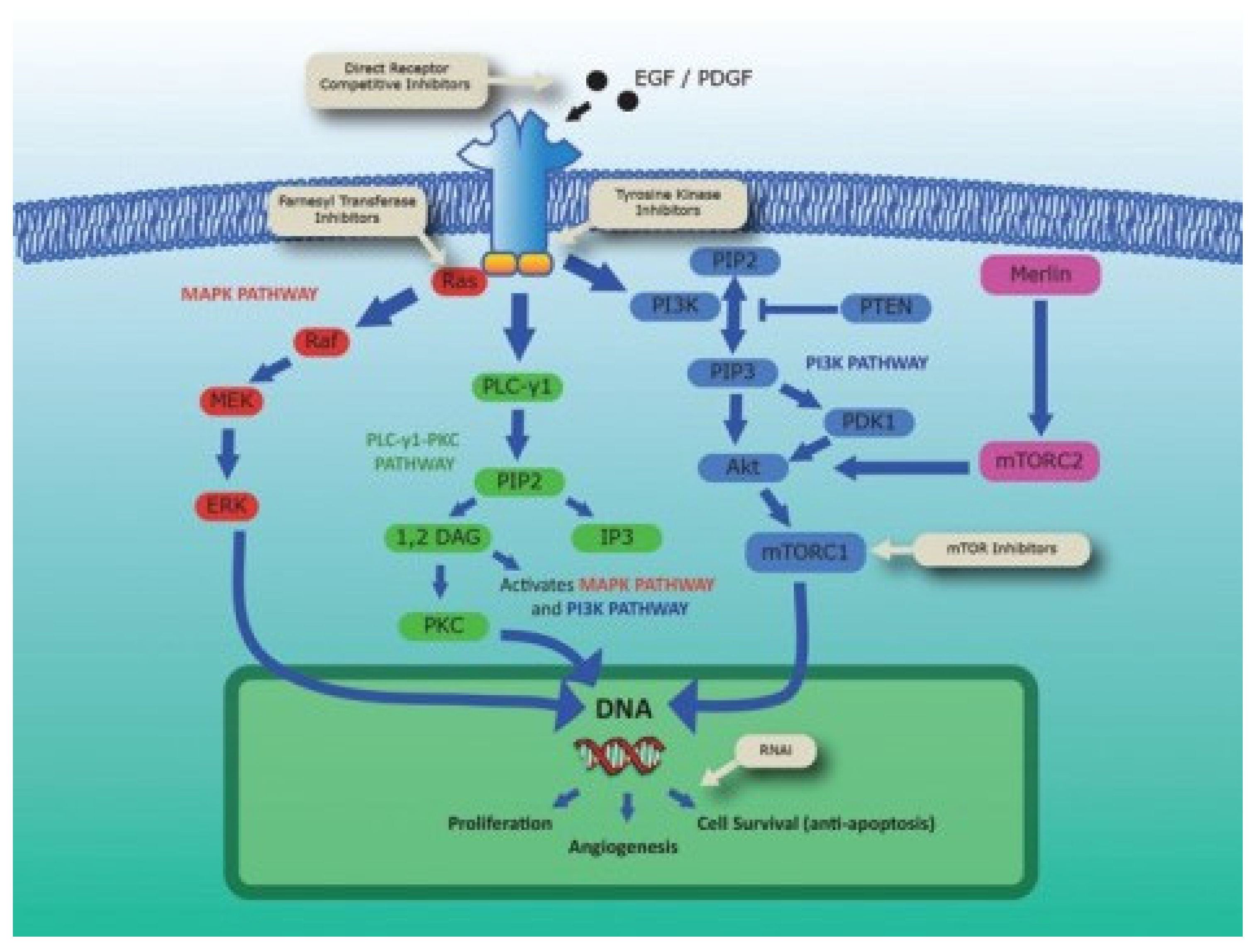
3. Potential Therapeutic Targets
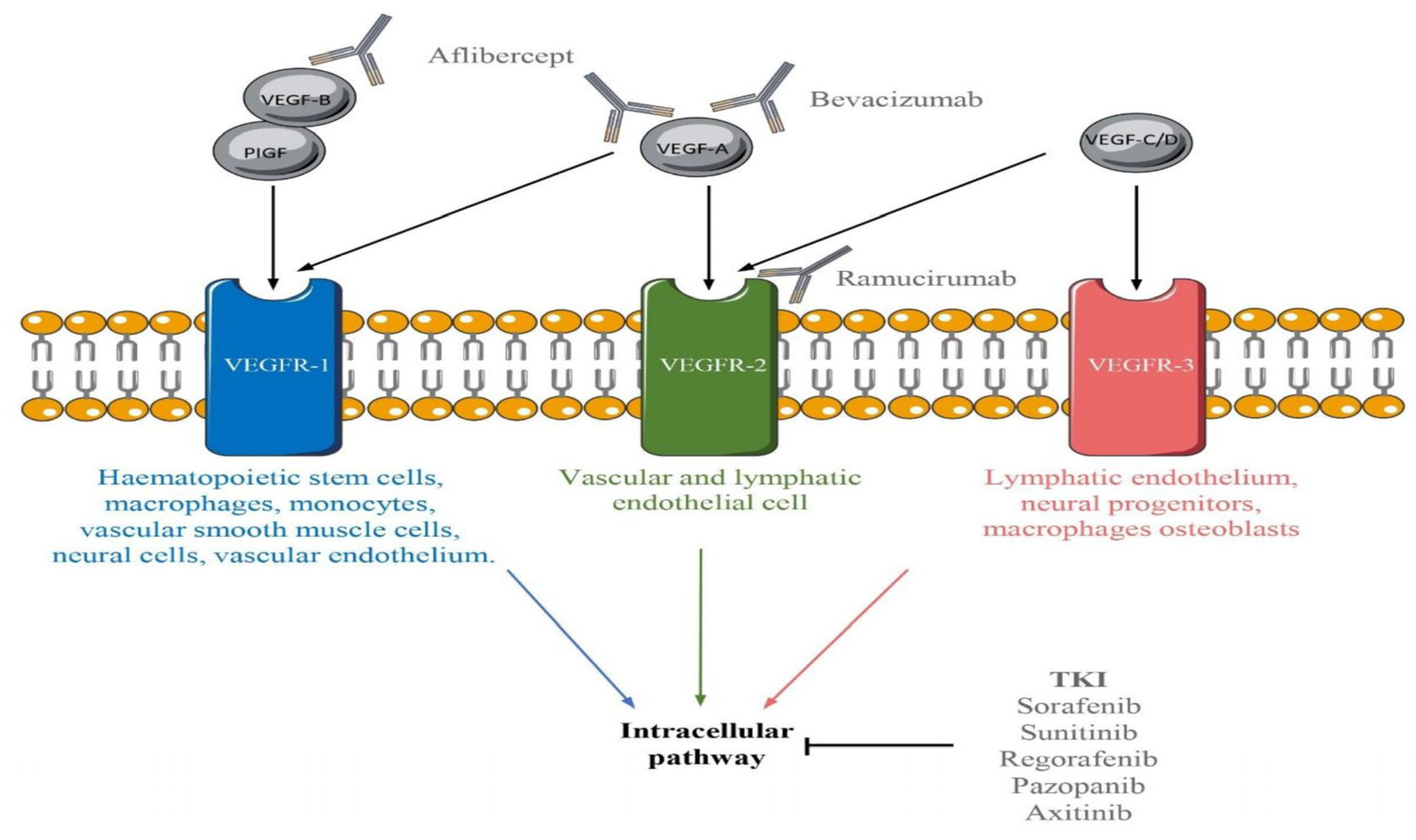
4. Current Treatment Approaches and Limitations
4.1. Surgical Resection
4.2. Radiation Therapy
4.3. Chemotherapy and Targeted Therapies
5. Cabozantinib: Mechanism of Action and Pharmacokinetics
5.1. Inhibition of VEGFR2 Signaling
5.2. Inhibition of MET Signaling
5.3. Inhibition of MET and VEGFR2 Signaling by Cabozantinib
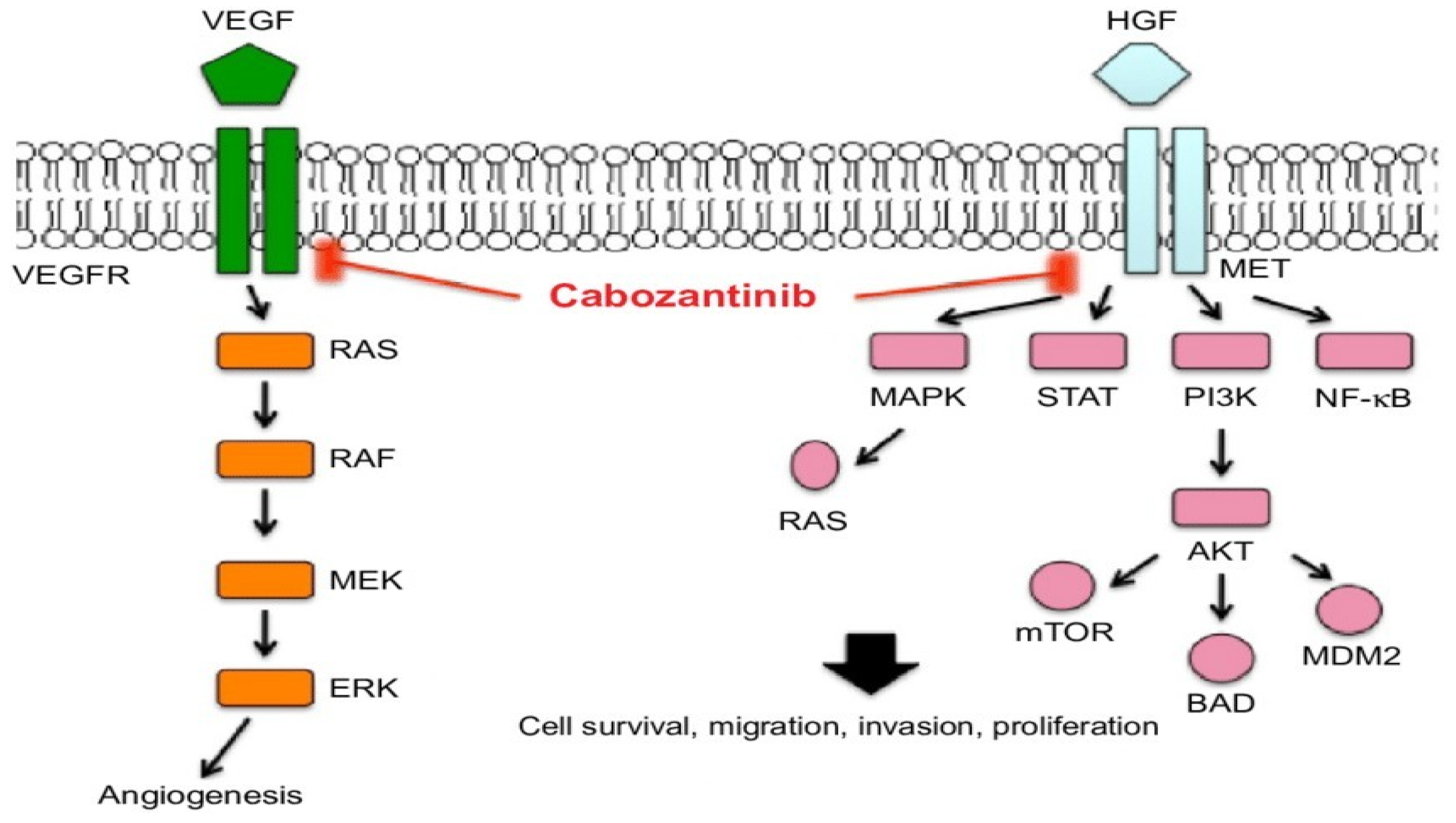
6. Preclinical Studies Highlighting Cabozantinib's Effects on Meningiomas
7. Molecular Targets and Pathways Impacted by Cabozantinib
7.1. MET Signaling Pathway and its Role in Meningioma Growth
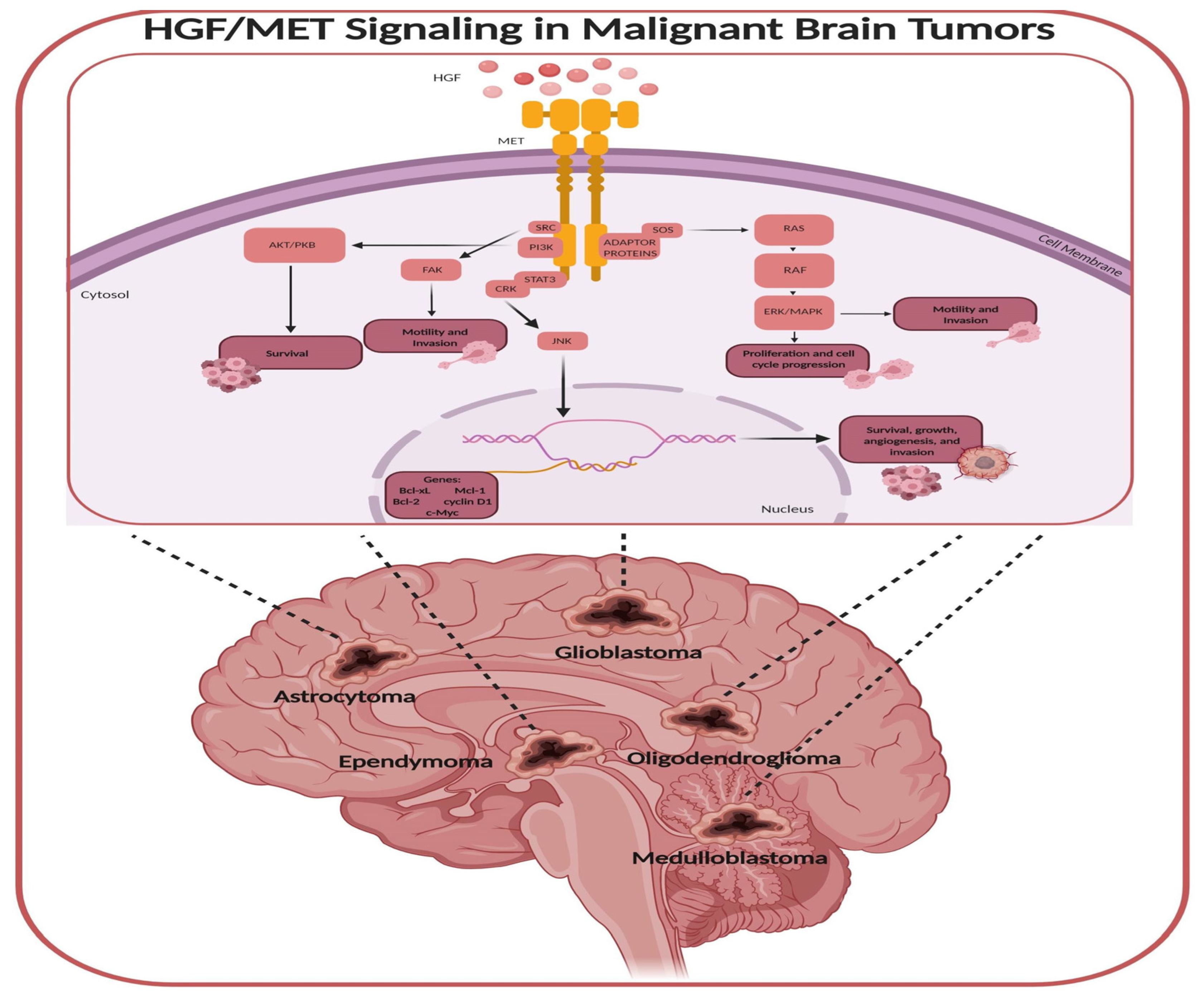
7.2. VEGFR2 Inhibition and Anti-Angiogenic Effects
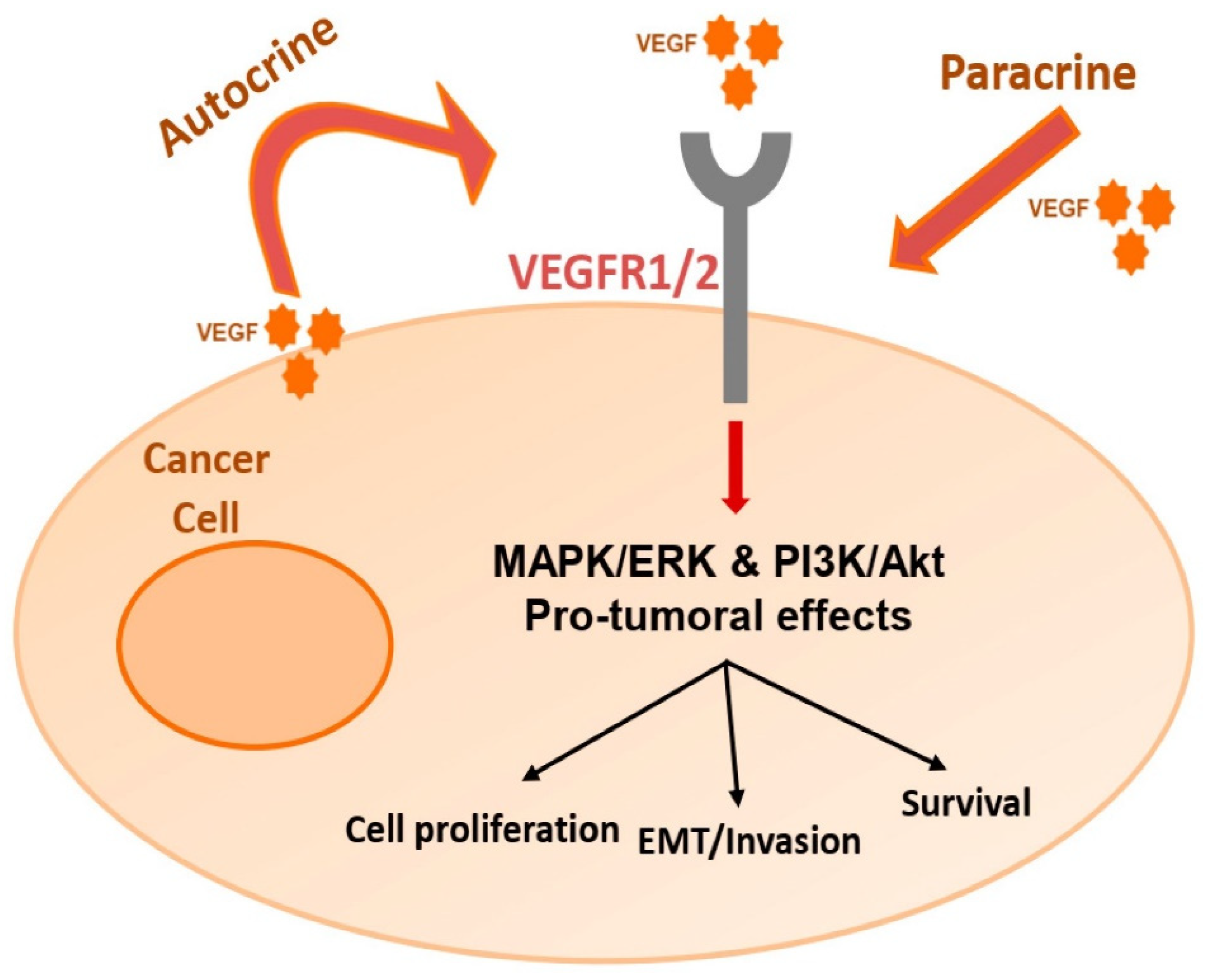
7.3. Other Potential Targets and Combinatorial Approaches
8. Mechanisms of Cabozantinib Resistance
8.1. Acquired Resistance in Meningiomas
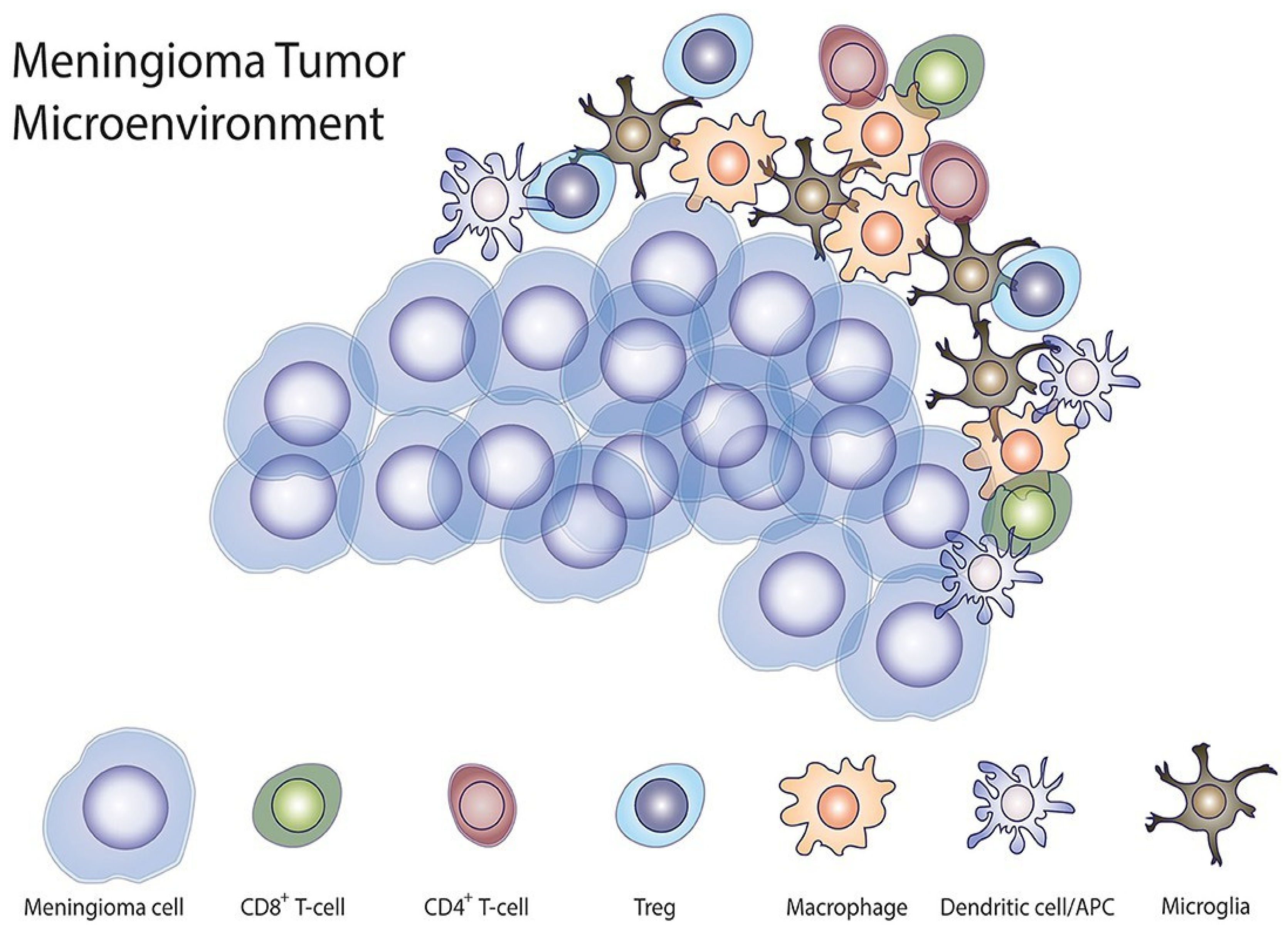
8.2. Potential Strategies to Overcome Resistance
9. Safety and Tolerability of Cabozantinib in Meningiomas
9.1. Common Adverse Events and Management Strategies
10. Future Perspectives on Safety Monitoring
11. Conclusions
Funding
Data Availability Statement
Conflicts of Interest
References
- C. L. Arteaga, “Overview of epidermal growth factor receptor biology and its role as a therapeutic target in human neoplasia,” Seminars in Oncology, vol. 29, no. 5, pp. 3–9, Jan. 2002. [CrossRef]
- S. Huang and P. M. Harari, “Epidermal Growth Factor Receptor Inhibition in Cancer Therapy: Biology, Rationale and Preliminary Clinical Results,” Investigational New Drugs, vol. 17, no. 3, pp. 259–269, Aug. 1999. [CrossRef]
- J. Woodburn, “The Epidermal Growth Factor Receptor and Its Inhibition in Cancer Therapy,” Pharmacology & Therapeutics, vol. 82, no. 2–3, pp. 241–250, May 1999. [CrossRef]
- L. Rogers, M. R. Gilbert, and M. A. Vogelbaum, “Intracranial meningiomas of atypical (WHO grade II) histology,” Journal of Neuro-oncology, vol. 99, no. 3, pp. 393–405, Aug. 2010. [CrossRef]
- E. B. Claus, M. L. Bondy, J. M. Schildkraut, J. L. Wiemels, M. Wrensch, and P. McL. Black, “Epidemiology of Intracranial Meningioma,” Neurosurgery, vol. 57, no. 6, pp. 1088–1095, Dec. 2005. [CrossRef]
- R. A. Buerki, C. Horbinski, T. J. Kruser, P. M. Horowitz, C. D. James, and R. V. Lukas, “An overview of meningiomas,” Future Oncology, vol. 14, no. 21, pp. 2161–2177, Sep. 2018. [CrossRef]
- K. J. Drummond, J.-J. Zhu, and P. McL. Black, “Meningiomas: Updating Basic Science, Management, and Outcome,” The Neurologist, vol. 10, no. 3, pp. 113–130, 04. 20 May. [CrossRef]
- L. Rogers, M. R. Gilbert, and M. A. Vogelbaum, “Intracranial meningiomas of atypical (WHO grade II) histology,” Journal of Neuro-oncology, vol. 99, no. 3, pp. 393–405, Aug. 2010. [CrossRef]
- J. Woodburn, “The Epidermal Growth Factor Receptor and Its Inhibition in Cancer Therapy,” Pharmacology & Therapeutics, vol. 82, no. 2–3, pp. 241–250, May 1999. [CrossRef]
- S. Huang and P. M. Harari, “Epidermal Growth Factor Receptor Inhibition in Cancer Therapy: Biology, Rationale and Preliminary Clinical Results,” Investigational New Drugs, vol. 17, no. 3, pp. 259–269, Aug. 1999. [CrossRef]
- D. Norden, J. Drappatz, and P. Y. Wen, “Advances in meningioma therapy,” Current Neurology and Neuroscience Reports, vol. 9, no. 3, pp. 231–240, Apr. 2009. [CrossRef]
- L. Kim, “A narrative review of targeted therapies in meningioma,” Chinese Clinical Oncology, vol. 9, no. 6, p. 76, Dec. 2020. [CrossRef]
- K. J. Dedes, D. Wetterskog, A. Ashworth, S. B. Kaye, and J. S. Reis-Filho, “Emerging therapeutic targets in endometrial cancer,” Nature Reviews Clinical Oncology, vol. 8, no. 5, pp. 261–271, Jan. 2011. [CrossRef]
- B. P. Schneider and G. W. Sledge, “Drug Insight: VEGF as a therapeutic target for breast cancer,” Nature Clinical Practice Oncology, vol. 4, no. 3, pp. 181–189, Mar. 2007. [CrossRef]
- N. Ferrara, K. J. Hillan, H.-P. Gerber, and W. Novotny, “Discovery and development of bevacizumab, an anti-VEGF antibody for treating cancer,” Nature Reviews Drug Discovery, vol. 3, no. 5, pp. 391–400, 04. 20 May. [CrossRef]
- G. W. Sledge, “VEGF-Targeting Therapy for Breast Cancer,” Journal of Mammary Gland Biology and Neoplasia, vol. 10, no. 4, pp. 319–323, Oct. 2005. [CrossRef]
- Galanis, “Targeting angiogenesis: Progress with anti-VEGF treatment with large molecules,” Nature Reviews Clinical Oncology, vol. 6, no. 9, pp. 507–518, Jul. 2009. [CrossRef]
- P. Y. Wen, E. C. Quant, J. Drappatz, R. Beroukhim, and A. D. Norden, “Medical therapies for meningiomas,” Journal of Neuro-oncology, vol. 99, no. 3, pp. 365–378, Sep. 2010. [CrossRef]
- L. Kim, “A narrative review of targeted therapies in meningioma,” Chinese Clinical Oncology, vol. 9, no. 6, p. 76, Dec. 2020. [CrossRef]
- T.-Y. Kim, Y.-J. Bang, and K. D. Robertson, “Histone Deacetylase Inhibitors for Cancer Therapy,” Epigenetics, vol. 1, no. 1, pp. 15–24, Jan. 2006. [CrossRef]
- Wagle, and G. Zada, “Recent developments in chemotherapy for meningiomas: A review,” Neurosurgical Focus, vol. 35, no. 6, p. E18, Dec. 2013. [CrossRef]
- L. Kim, “A narrative review of targeted therapies in meningioma,” Chinese Clinical Oncology, vol. 9, no. 6, p. 76, Dec. 2020. [CrossRef]
- S. J. Han et al., “Expression and prognostic impact of immune modulatory molecule PD-L1 in meningioma,” Journal of Neuro-oncology, vol. 130, no. 3, pp. 543–552, Sep. 2016. [CrossRef]
- M. Preusser, P. K. Brastianos, and C. Mawrin, “Advances in meningioma genetics: Novel therapeutic opportunities,” Nature Reviews Neurology, vol. 14, no. 2, pp. 106–115, Jan. 2018. [CrossRef]
- K. Chen et al., “Antibody–Drug Conjugate to Treat Meningiomas,” Pharmaceuticals, 21. 20 May. [CrossRef]
- P. M. Pardalos, H. R. Van Loveren, and J. M. Tew, “The Surgical Resectability of Meningiomas of the Cavernous Sinus,” Neurosurgery, vol. 40, no. 2, pp. 238–247, Feb. 1997. [CrossRef]
- D. Kondziolka, J. C. Flickinger, and B. Perez, “Judicious Resection and/or Radiosurgery for Parasagittal Meningiomas: Outcomes from a Multicenter Review,” Neurosurgery, vol. 43, no. 3, pp. 405–413, Sep. 1998. [CrossRef]
- M. G. O. Sullivan, H. R. Van Loveren, and J. G. Tew, “The Surgical Resectability of Meningiomas of the Cavernous Sinus,” Journal of Neuro-ophthalmology, vol. 17, no. 4, p. 288, Dec. 1997. [CrossRef]
- L. N. Sekhar, G. Juric-Sekhar, H. B. Da Silva, and J. S. Pridgeon, “Skull Base Meningiomas,” Neurosurgery, vol. 62, no. Supplement 1, pp. 30–49, Aug. 2015. [CrossRef]
- T. L. Winther and S. H. Torp, “Significance of the Extent of Resection in Modern Neurosurgical Practice of World Health Organization Grade I Meningiomas,” World Neurosurgery, vol. 99, pp. 104–110, Mar. 2017. [CrossRef]
- T. Ichinose, T. Goto, K. Ishibashi, T. Takami, and K. Ohata, “The role of radical microsurgical resection in multimodal treatment for skull base meningioma,” Journal of Neurosurgery, vol. 113, no. 5, pp. 1072–1078, Nov. 2010. [CrossRef]
- N. Sanai, M. E. Sughrue, G. Shangari, K. K. K. Chung, M. S. Berger, and M. W. McDermott, “Risk profile associated with convexity meningioma resection in the modern neurosurgical era,” Journal of Neurosurgery, vol. 112, no. 5, pp. 913–919, 10. 20 May. [CrossRef]
- R. O. Mirimanoff, D. E. Dosoretz, R. M. Linggood, R. G. Ojemann, and R. L. Martuza, “Meningioma: Analysis of recurrence and progression following neurosurgical resection,” Journal of Neurosurgery, vol. 62, no. 1, pp. 18–24, Jan. 1985. [CrossRef]
- C. Jackson, K. S. Tadokoro, E. W. Wang, G. A. Zenonos, C. H. Snyderman, and P. A. Gardner, “Approach selection for resection of petroclival meningioma,” Neurosurgical Focus: Video, vol. 6, no. 2, p. V9, Apr. 2022. [CrossRef]
- P. M. Pardalos, H. R. Van Loveren, and J. M. Tew, “The Surgical Resectability of Meningiomas of the Cavernous Sinus,” Neurosurgery, vol. 40, no. 2, pp. 238–247, Feb. 1997. [CrossRef]
- S. Y. Wayhs, G. Lepski, L. Frighetto, and N. N. Rabelo, “Petroclival meningiomas: Remaining controversies in light of minimally invasive approaches,” Clinical Neurology and Neurosurgery, vol. 152, pp. 68–75, Jan. 2017. [CrossRef]
- L. D. Lunsford, “Contemporary management of meningiomas: Radiation therapy as an adjuvant and radiosurgery as an alternative to surgical removal?,” Journal of Neurosurgery, vol. 80, no. 2, pp. 187–190, Feb. 1994. [CrossRef]
- N. M. Barbaro, P. H. Gutin, C. B. Wilson, G. E. Sheline, E. B. Boldrey, and W. M. Wara, “Radiation Therapy in the Treatment of Partially Resected Meningiomas,” Neurosurgery, vol. 20, no. 4, pp. 525–528, Apr. 1987. [CrossRef]
- M. Petty, L. E. Kun, and G. A. Meyer, “Radiation therapy for incompletely resected meningiomas,” Journal of Neurosurgery, vol. 62, no. 4, pp. 502–507, Apr. 1985. [CrossRef]
- R. Baskar, K. H. Lee, R. Yeo, and K. W. Yeoh, “Cancer and Radiation Therapy: Current Advances and Future Directions,” International Journal of Medical Sciences, vol. 9, no. 3, pp. 193– 199, Jan. 2012. [CrossRef]
- D. Kondziolka and L. D. Lunsford, “Radiosurgery of Meningiomas,” Neurosurgery Clinics of North America, vol. 3, no. 1, pp. 219–230, Jan. 1992. [CrossRef]
- H. Rodolfo et al., “Results of Linear Accelerator-based Radiosurgery for Intracranial Meningiomas,” Neurosurgery, vol. 42, no. 3, pp. 446–454, Mar. 1998. [CrossRef]
- P. Metellus et al., “Evaluation of Fractionated Radiotherapy and Gamma Knife Radiosurgery in Cavernous Sinus Meningiomas: Treatment Strategy,” Neurosurgery, vol. 57, no. 5, pp. 873–886, Nov. 2005. [CrossRef]
- T. Tanaka, T. Kobayashi, and Y. Kida, “Growth Control of Cranial Base Meningiomas by Stereotactic Radiosurgery with a Gamma Knife Unit,” Neurologia Medico-chirurgica, vol. 36, no. 1, pp. 7–10, Jan. 1996. [CrossRef]
- H. H. Engelhard, “Current status of radiation therapy and radiosurgery in the treatment of intracranial meningiomas,” Neurosurgical Focus, vol. 2, no. 4, p. E8, Apr. 1997. [CrossRef]
- D. Kondziolka, L. D. Lunsford, R. J. Coffey, and J. C. Flickinger, “Stereotactic radiosurgery of meningiomas,” Journal of Neurosurgery, vol. 74, no. 4, pp. 552–559, Apr. 1991. [CrossRef]
- C. Cordova and S. Kurz, “Advances in Molecular Classification and Therapeutic Opportunities in Meningiomas,” Current Oncology Reports, vol. 22, no. 8, Jul. 2020. [CrossRef]
- F. Nigim, H. Wakimoto, E. M. Kasper, L. Ackermans, and Y. Temel, “Emerging Medical Treatments for Meningioma in the Molecular Era,” Biomedicines, vol. 6, no. 3, p. 86, Aug. 2018. [CrossRef]
- J. Jagannathan, R. J. Oskouian, H. K. Yeoh, D. Saulle, and A. S. Dumont, “Molecular Biology of Unreresectable Meningiomas: Implications for New Treatments and Review of the Literature,” Skull Base Surgery, vol. 18, no. 3, pp. 173–187, May 2008. [CrossRef]
- L. Rogers et al., “Meningiomas: Knowledge base, treatment outcomes, and uncertainties. A RANO review,” Journal of Neurosurgery, vol. 122, no. 1, pp. 4–23, Jan. 2015. [CrossRef]
- L. Rogers et al., “Meningiomas: Knowledge base, treatment outcomes, and uncertainties. A RANO review,” Journal of Neurosurgery, vol. 122, no. 1, pp. 4–23, Jan. 2015. [CrossRef]
- Wagle, and G. Zada, “Recent developments in chemotherapy for meningiomas: A review,” Neurosurgical Focus, vol. 35, no. 6, p. E18, Dec. 2013. [CrossRef]
- M. Preusser, P. K. Brastianos, and C. Mawrin, “Advances in meningioma genetics: Novel therapeutic opportunities,” Nature Reviews Neurology, vol. 14, no. 2, pp. 106–115, Jan. 2018. [CrossRef]
- P. D. Tatman et al., “High-Throughput Mechanistic Screening of Epigenetic Compounds for the Potential Treatment of Meningiomas,” Journal of Clinical Medicine, vol. 10, no. 14, p. 3150, Jul. 2021. [CrossRef]
- M. D. Johnson and S. A. Toms, “Mitogenic Signal Transduction Pathways in Meningiomas: Novel Targets for Meningioma Chemotherapy?,” Journal of Neuropathology and Experimental Neurology, vol. 64, no. 12, pp. 1029–1036, Dec. 2005. [CrossRef]
- J. De Stricker Borch, J. Haslund-Vinding, F. Vilhardt, A. B. Maier, and T. Mathiesen, “Meningioma–Brain Crosstalk: A Scoping Review,” Cancers, vol. 13, no. 17, p. 4267, Aug. 2021. [CrossRef]
- S. E. Tollefsen, O. Solheim, P. Mjønes, and S. H. Torp, “Meningiomas and Somatostatin Analogs: A Systematic Scoping Review on Current Insights and Future Perspectives,” International Journal of Molecular Sciences, vol. 24, no. 5, p. 4793, Mar. 2023. [CrossRef]
- F. M. Yakes et al., “Cabozantinib (XL184), a Novel MET and VEGFR2 Inhibitor, Simultaneously Suppresses Metastasis, Angiogenesis, and Tumor Growth,” Molecular Cancer Therapeutics, vol. 10, no. 12, pp. 2298–2308, Dec. 2011. [CrossRef]
- C. Grüllich, “Cabozantinib: A MET, RET, and VEGFR2 Tyrosine Kinase Inhibitor,” in Springer eBooks, 2014, pp. 207–214. [CrossRef]
- F. M. Yakes et al., “Cabozantinib (XL184), a Novel MET and VEGFR2 Inhibitor, Simultaneously Suppresses Metastasis, Angiogenesis, and Tumor Growth,” Molecular Cancer Therapeutics, vol. 10, no. 12, pp. 2298–2308, Dec. 2011. [CrossRef]
- N. Rathi, B. L. Maughan, N. Agarwal, and U. Swami, “Mini-Review: Cabozantinib in the Treatment of Advanced Renal Cell Carcinoma and Hepatocellular Carcinoma,” Cancer Management and Research, vol. Volume 12, pp. 3741–3749, 20. 20 May. [CrossRef]
- C. Grüllich, “Cabozantinib: A MET, RET, and VEGFR2 Tyrosine Kinase Inhibitor,” Springer eBooks, pp. 207–214, Jan. 2014. [CrossRef]
- X. Zhang, Y. Shao, and K. Wang, “Incidence and risk of hypertension associated with cabozantinib in cancer patients: A systematic review and meta-analysis,” Expert Review of Clinical Pharmacology, vol. 9, no. 8, pp. 1109–1115, Jun. 2016. [CrossRef]
- S. S. Yu, D. I. Quinn, and T. B. Dorff, “Clinical use of cabozantinib in the treatment of advanced kidney cancer: Efficacy, safety, and patient selection,” OncoTargets and Therapy, vol. Volume 9, pp. 5825–5837, Sep. 2016. [CrossRef]
- D. Smith et al., “Cabozantinib in Patients With Advanced Prostate Cancer: Results of a Phase II Randomized Discontinuation Trial,” Journal of Clinical Oncology, vol. 31, no. 4, pp. 412–419, Feb. 2013. [CrossRef]
- L. A. Nguyen, N. Benrimoh, Y. Xie, E. Offman, and S. Lacy, “Pharmacokinetics of cabozantinib tablet and capsule formulations in healthy adults,” Anti-Cancer Drugs, vol. 27, no. 7, pp. 669–678, Aug. 2016. [CrossRef]
- V. R. Belum, C. Serna-Tamayo, S. L. Wu, and M. E. Lacouture, “Incidence and risk of hand- foot skin reaction with cabozantinib, a novel multikinase inhibitor: A meta-analysis,” Clinical and Experimental Dermatology, vol. 41, no. 1, pp. 8–15, 15. 20 May. [CrossRef]
- R. Elisei et al., “Cabozantinib in Progressive Medullary Thyroid Cancer,” Journal of Clinical Oncology, vol. 31, no. 29, pp. 3639–3646, Oct. 2013. [CrossRef]
- T. K. Choueiri et al., “Cabozantinib versus Everolimus in Advanced Renal-Cell Carcinoma,” The New England Journal of Medicine, vol. 373, no. 19, pp. 1814–1823, Nov. 2015. [CrossRef]
- F. M. Yakes et al., “Cabozantinib (XL184), a Novel MET and VEGFR2 Inhibitor, Simultaneously Suppresses Metastasis, Angiogenesis, and Tumor Growth,” Molecular Cancer Therapeutics, vol. 10, no. 12, pp. 2298–2308, Dec. 2011. [CrossRef]
- Q. Xiang et al., “Cabozantinib Suppresses Tumor Growth and Metastasis in Hepatocellular Carcinoma by a Dual Blockade of VEGFR2 and MET,” Clinical Cancer Research, vol. 20, no. 11, pp. 2959–2970, May 2014. [CrossRef]
- N. Rathi, B. L. Maughan, N. Agarwal, and U. Swami, “Mini-Review: Cabozantinib in the Treatment of Advanced Renal Cell Carcinoma and Hepatocellular Carcinoma,” Cancer Management and Research, vol. Volume 12, pp. 3741–3749, May 2020. [CrossRef]
- M. Santoni, R. Iacovelli, V. Colonna, S. G. Klinz, G. Mauri, and M. Nuti, “Antitumor effects of the multi-target tyrosine kinase inhibitor cabozantinib: A comprehensive review of the preclinical evidence,” Expert Review of Anticancer Therapy, vol. 21, no. 9, pp. 1029–1054, Aug. 2021. [CrossRef]
- X. Zhang et al., “Cabozantinib, a Multityrosine Kinase Inhibitor of MET and VEGF Receptors Which Suppresses Mouse Laser-Induced Choroidal Neovascularization,” Journal of Ophthalmology, vol. 2020, pp. 1–16, Jun. 2020. [CrossRef]
- C. Grüllich, “Cabozantinib: Multi-kinase Inhibitor of MET, AXL, RET, and VEGFR2,” in Recent results in cancer research, Springer Science+Business Media, 2018, pp. 67–75. [CrossRef]
- Prete, A. Cammarota, N. Personeni, and L. Rimassa, “The Role of Cabozantinib as a Therapeutic Option for Hepatocellular Carcinoma: Current Landscape and Future Challenges,” Journal of Hepatocellular Carcinoma, vol. Volume 8, pp. 177–191, Mar. 2021. [CrossRef]
- J. N. Markowitz and K. S. Fancher, “Cabozantinib: A Multitargeted Oral Tyrosine Kinase Inhibitor,” Pharmacotherapy, vol. 38, no. 3, pp. 357–369, Feb. 2018. [CrossRef]
- Small, “Treatment of advanced renal cell carcinoma patients with cabozantinib, an oral multityrosine kinase inhibitor of MET, AXL and VEGF receptors,” Future Oncology, vol. 15, no. 20, pp. 2337–2348, Jul. 2019. [CrossRef]
- D. Vesque, M. Decraecker, and J.-F. Blanc, “Profile of Cabozantinib for the Treatment of Hepatocellular Carcinoma: Patient Selection and Special Considerations,” Journal of Hepatocellular Carcinoma, vol. Volume 7, pp. 91–99, Jun. 2020. [CrossRef]
- D. Vesque, M. Decraecker, and J.-F. Blanc, “Profile of Cabozantinib for the Treatment of Hepatocellular Carcinoma: Patient Selection and Special Considerations,” Journal of Hepatocellular Carcinoma, vol. Volume 7, pp. 91–99, Jun. 2020. [CrossRef]
- T. A. Mannsåker et al., “Cabozantinib Is Effective in Melanoma Brain Metastasis Cell Lines and Affects Key Signaling Pathways,” International Journal of Molecular Sciences, vol. 22, no. 22, p. 12296, Nov. 2021. [CrossRef]
- J. Lolli et al., “Impressive reduction of brain metastasis radionecrosis after cabozantinib therapy in metastatic renal carcinoma: A case report and review of the literature,” Frontiers in Oncology, vol. 13, Mar. 2023. [CrossRef]
- Q. Xiang et al., “Cabozantinib Suppresses Tumor Growth and Metastasis in Hepatocellular Carcinoma by a Dual Blockade of VEGFR2 and MET,” Clinical Cancer Research, vol. 20, no. 11, pp. 2959–2970, May 2014. [CrossRef]
- F. M. Yakes et al., “Cabozantinib (XL184), a Novel MET and VEGFR2 Inhibitor, Simultaneously Suppresses Metastasis, Angiogenesis, and Tumor Growth,” Molecular Cancer Therapeutics, vol. 10, no. 12, pp. 2298–2308, Dec. 2011. [CrossRef]
- M. Sameni et al., “Cabozantinib (XL184) Inhibits Growth and Invasion of Preclinical TNBC Models,” Clinical Cancer Research, vol. 22, no. 4, pp. 923–934, Feb. 2016. [CrossRef]
- O. Saydam et al., “Downregulated MicroRNA-200a in Meningiomas Promotes Tumor Growth by Reducing E-Cadherin and Activating the Wnt/β-Catenin Signaling Pathway,” Molecular and Cellular Biology, vol. 29, no. 21, pp. 5923–5940, Nov. 2009. [CrossRef]
- E. Daudigeos-Dubus et al., “Dual inhibition using cabozantinib overcomes HGF/MET signaling mediated resistance to pan-VEGFR inhibition in orthotopic and metastatic neuroblastoma tumors,” International Journal of Oncology, vol. 50, no. 1, pp. 203–211, Dec. 2016. [CrossRef]
- R. Elisei et al., “Cabozantinib in Progressive Medullary Thyroid Cancer,” Journal of Clinical Oncology, vol. 31, no. 29, pp. 3639–3646, Oct. 2013. [CrossRef]
- D. Vesque, M. Decraecker, and J.-F. Blanc, “Profile of Cabozantinib for the Treatment of Hepatocellular Carcinoma: Patient Selection and Special Considerations,” Journal of Hepatocellular Carcinoma, vol. Volume 7, pp. 91–99, Jun. 2020. [CrossRef]
- R. Kotecha et al., “Regression of Intracranial Meningiomas Following Treatment with Cabozantinib,” Current Oncology, vol. 28, no. 2, pp. 1537–1543, Apr. 2021. [CrossRef]
- Prete, A. Cammarota, N. Personeni, and L. Rimassa, “The Role of Cabozantinib as a Therapeutic Option for Hepatocellular Carcinoma: Current Landscape and Future Challenges,” Journal of Hepatocellular Carcinoma, vol. Volume 8, pp. 177–191, Mar. 2021. [CrossRef]
- N. Personeni, T. Pressiani, and L. Rimassa, “Cabozantinib in patients with hepatocellular carcinoma failing previous treatment with sorafenib,” Future Oncology, vol. 15, no. 21, pp. 2449– 2462, Jul. 2019. [CrossRef]
- T. A. Mannsåker et al., “Cabozantinib Is Effective in Melanoma Brain Metastasis Cell Lines and Affects Key Signaling Pathways,” International Journal of Molecular Sciences, vol. 22, no. 22, p. 12296, Nov. 2021. [CrossRef]
- H. Zhao, R. Nolley, A. Chan, E. B. Rankin, and D. M. Peehl, “Cabozantinib inhibits tumor growth and metastasis of a patient-derived xenograft model of papillary renal cell carcinoma with MET mutation,” Cancer Biology & Therapy, vol. 18, no. 11, pp. 863–871, Aug. 2016. [CrossRef]
- W.-T. Liao et al., “Cost-effectiveness analysis of cabozantinib as second-line therapy in advanced hepatocellular carcinoma,” Liver International, vol. 39, no. 12, pp. 2408–2416, Oct. 2019. [CrossRef]
- P. Y. Wen et al., “Phase II study of cabozantinib in patients with progressive glioblastoma: Subset analysis of patients naive to antiangiogenic therapy,” Neuro-oncology, vol. 20, no. 2, pp. 249–258, Aug. 2017. [CrossRef]
- T. F. Cloughesy et al., “Phase II study of cabozantinib in patients with progressive glioblastoma: Subset analysis of patients with prior antiangiogenic therapy,” Neuro-oncology, vol. 20, no. 2, pp. 259–267, Oct. 2017. [CrossRef]
- P. Schöffski, J.-Y. Blay, and I. Ray-Coquard, “Cabozantinib as an emerging treatment for sarcoma,” Current Opinion in Oncology, vol. 32, no. 4, pp. 321–331, Jun. 2020. [CrossRef]
- P. Maroto et al., “Cabozantinib for the treatment of solid tumors: A systematic review,” Therapeutic Advances in Medical Oncology, vol. 14, p. 175883592211071, Jan. 2022. [CrossRef]
- M. L. B. Peters and R. A. Miksad, “Cabozantinib in the treatment of hepatocellular carcinoma,” Future Oncology, vol. 13, no. 22, pp. 1915–1929, Sep. 2017. [CrossRef]
- M. Santoni, R. Iacovelli, V. Colonna, S. G. Klinz, G. Mauri, and M. Nuti, “Antitumor effects of the multi-target tyrosine kinase inhibitor cabozantinib: A comprehensive review of the preclinical evidence,” Expert Review of Anticancer Therapy, vol. 21, no. 9, pp. 1029–1054, Aug. 2021. [CrossRef]
- T. Graham et al., “Preclinical Evaluation of Imaging Biomarkers for Prostate Cancer Bone Metastasis and Response to Cabozantinib,” Journal of the National Cancer Institute, vol. 106, no. 4, Mar. 2014. [CrossRef]
- T. Graham et al., “Preclinical Evaluation of Imaging Biomarkers for Prostate Cancer Bone Metastasis and Response to Cabozantinib,” Journal of the National Cancer Institute, vol. 106, no. 4, Mar. 2014. [CrossRef]
- E.-K. Song et al., “Potent antitumor activity of cabozantinib, a c-MET and VEGFR2 inhibitor, in a colorectal cancer patient-derived tumor explant model,” International Journal of Cancer, vol. 136, no. 8, pp. 1967–1975, Sep. 2014. [CrossRef]
- M. R. Smith et al., “Phase III Study of Cabozantinib in Previously Treated Metastatic Castration-Resistant Prostate Cancer: COMET-1,” Journal of Clinical Oncology, vol. 34, no. 25, pp. 3005–3013, Sep. 2016. [CrossRef]
- J. N. Markowitz and K. S. Fancher, “Cabozantinib: A Multitargeted Oral Tyrosine Kinase Inhibitor,” Pharmacotherapy, vol. 38, no. 3, pp. 357–369, Feb. 2018. [CrossRef]
- M. R. Smith et al., “Cabozantinib in Chemotherapy-Pretreated Metastatic Castration-Resistant Prostate Cancer: Results of a Phase II Nonrandomized Expansion Study,” Journal of Clinical Oncology, vol. 32, no. 30, pp. 3391–3399, Oct. 2014. [CrossRef]
- L. Bertazza et al., “EF24 (a Curcumin Analog) and ZSTK474 Emphasize the Effect of Cabozantinib in Medullary Thyroid Cancer,” Endocrinology, vol. 159, no. 6, pp. 2348–2360, Apr. 2018. [CrossRef]
- M. Tripathi et al., “Modulation of cabozantinib efficacy by the prostate tumor microenvironment,” Oncotarget, vol. 8, no. 50, pp. 87891–87902, Sep. 2017. [CrossRef]
- M. Fioramonti et al., “Cabozantinib targets bone microenvironment modulating human osteoclast and osteoblast functions,” Oncotarget, vol. 8, no. 12, pp. 20113–20121, Feb. 2017. [CrossRef]
- F. M. Yakes et al., “Cabozantinib (XL184), a Novel MET and VEGFR2 Inhibitor, Simultaneously Suppresses Metastasis, Angiogenesis, and Tumor Growth,” Molecular Cancer Therapeutics, vol. 10, no. 12, pp. 2298–2308, Dec. 2011. [CrossRef]
- Grüllich, “Cabozantinib: Multi-kinase Inhibitor of MET, AXL, RET, and VEGFR2,” in Recent results in cancer research, Springer Science+Business Media, 2018, pp. 67–75. [CrossRef]
- D. Hart and R. De Boer, “Profile of cabozantinib and its potential in the treatment of advanced medullary thyroid cancer,” OncoTargets and Therapy, p. 1, Jan. 2013. [CrossRef]
- R. Katayama et al., “Cabozantinib Overcomes Crizotinib Resistance in ROS1 Fusion–Positive Cancer,” Clinical Cancer Research, vol. 21, no. 1, pp. 166–174, Jan. 2015. [CrossRef]
- A. Patnaik et al., “Cabozantinib Eradicates Advanced Murine Prostate Cancer by Activating Antitumor Innate Immunity,” Cancer Discovery, vol. 7, no. 7, pp. 750–765, Jul. 2017. [CrossRef]
- A. Desai and E. J. Small, “Treatment of advanced renal cell carcinoma patients with cabozantinib, an oral multityrosine kinase inhibitor of MET, AXL and VEGF receptors,” Future Oncology, vol. 15, no. 20, pp. 2337–2348, Jul. 2019. [CrossRef]
- M. Santoni, R. Iacovelli, V. Colonna, S. G. Klinz, G. Mauri, and M. Nuti, “Antitumor effects of the multi-target tyrosine kinase inhibitor cabozantinib: A comprehensive review of the preclinical evidence,” Expert Review of Anticancer Therapy, vol. 21, no. 9, pp. 1029–1054, Aug. 2021. [CrossRef]
- R. Kurzrock et al., “Activity of XL184 (Cabozantinib), an Oral Tyrosine Kinase Inhibitor, in Patients With Medullary Thyroid Cancer,” Journal of Clinical Oncology, vol. 29, no. 19, pp. 2660– 2666, Jul. 2011. [CrossRef]
- J. N. Markowitz and K. S. Fancher, “Cabozantinib: A Multitargeted Oral Tyrosine Kinase Inhibitor,” Pharmacotherapy, vol. 38, no. 3, pp. 357–369, Feb. 2018. [CrossRef]
- S. Deng, A. Solinas, and D. F. Calvisi, “Cabozantinib for HCC Treatment, From Clinical Back to Experimental Models,” Frontiers in Oncology, vol. 11, Oct. 2021. [CrossRef]
- S. Osanto and T. Van Der Hulle, “Cabozantinib in the treatment of advanced renal cell carcinoma in adults following prior vascular endothelial growth factor targeted therapy: Clinical trial evidence and experience,” Therapeutic Advances in Urology, vol. 10, no. 3, pp. 109–123, Jan. 2018. [CrossRef]
- M. R. Smith et al., “Phase III Study of Cabozantinib in Previously Treated Metastatic Castration-Resistant Prostate Cancer: COMET-1,” Journal of Clinical Oncology, vol. 34, no. 25, pp. 3005–3013, Sep. 2016. [CrossRef]
- M. Tripathi et al., “Modulation of cabozantinib efficacy by the prostate tumor microenvironment,” Oncotarget, vol. 8, no. 50, pp. 87891–87902, Sep. 2017. [CrossRef]
- T. K. Choueiri et al., “Cabozantinib versus Everolimus in Advanced Renal-Cell Carcinoma,” The New England Journal of Medicine, vol. 373, no. 19, pp. 1814–1823, Nov. 2015. [CrossRef]
- F. M. Yakes et al., “Cabozantinib (XL184), a Novel MET and VEGFR2 Inhibitor, Simultaneously Suppresses Metastasis, Angiogenesis, and Tumor Growth,” Molecular Cancer Therapeutics, vol. 10, no. 12, pp. 2298–2308, Dec. 2011. [CrossRef]
- Grüllich, “Cabozantinib: Multi-kinase Inhibitor of MET, AXL, RET, and VEGFR2,” in Recent results in cancer research, Springer Science+Business Media, 2018, pp. 67–75. [CrossRef]
- N. Rathi, B. L. Maughan, N. Agarwal, and U. Swami, “Mini-Review: Cabozantinib in the Treatment of Advanced Renal Cell Carcinoma and Hepatocellular Carcinoma,” Cancer Management and Research, vol. Volume 12, pp. 3741–3749, May 2020. [CrossRef]
- Q. Xiang et al., “Cabozantinib Suppresses Tumor Growth and Metastasis in Hepatocellular Carcinoma by a Dual Blockade of VEGFR2 and MET,” Clinical Cancer Research, vol. 20, no. 11, pp. 2959–2970, May 2014. [CrossRef]
- Small, “Treatment of advanced renal cell carcinoma patients with cabozantinib, an oral multityrosine kinase inhibitor of MET, AXL and VEGF receptors,” Future Oncology, vol. 15, no. 20, pp. 2337–2348, Jul. 2019. [CrossRef]
- Grüllich, “Cabozantinib: A MET, RET, and VEGFR2 Tyrosine Kinase Inhibitor,” in Springer eBooks, 2014, pp. 207–214. [CrossRef]
- J. Trojan, “Cabozantinib for the Treatment of Advanced Hepatocellular Carcinoma: Current Data and Future Perspectives,” Drugs, vol. 80, no. 12, pp. 1203–1210, Jul. 2020. [CrossRef]
- F. M. Yakes et al., “Cabozantinib (XL184), a Novel MET and VEGFR2 Inhibitor, Simultaneously Suppresses Metastasis, Angiogenesis, and Tumor Growth,” Molecular Cancer Therapeutics, vol. 10, no. 12, pp. 2298–2308, Dec. 2011. [CrossRef]
- Q. Xiang et al., “Cabozantinib Suppresses Tumor Growth and Metastasis in Hepatocellular Carcinoma by a Dual Blockade of VEGFR2 and MET,” Clinical Cancer Research, vol. 20, no. 11, pp. 2959–2970, May 2014. [CrossRef]
- F. Bentzien et al., “In Vitro and In Vivo Activity of Cabozantinib (XL184), an Inhibitor of RET, MET, and VEGFR2, in a Model of Medullary Thyroid Cancer,” Thyroid, vol. 23, no. 12, pp. 1569–1577, Dec. 2013. [CrossRef]
- M. Santoni, R. Iacovelli, V. Colonna, S. G. Klinz, G. Mauri, and M. Nuti, “Antitumor effects of the multi-target tyrosine kinase inhibitor cabozantinib: A comprehensive review of the preclinical evidence,” Expert Review of Anticancer Therapy, vol. 21, no. 9, pp. 1029–1054, Aug. 2021. [CrossRef]
- Grüllich, “Cabozantinib: Multi-kinase Inhibitor of MET, AXL, RET, and VEGFR2,” in Recent results in cancer research, Springer Science+Business Media, 2018, pp. 67–75. [CrossRef]
- A. Desai and E. J. Small, “Treatment of advanced renal cell carcinoma patients with cabozantinib, an oral multityrosine kinase inhibitor of MET, AXL and VEGF receptors,” Future Oncology, vol. 15, no. 20, pp. 2337–2348, Jul. 2019. [CrossRef]
- A. Varkaris et al., “Integrating Murine and Clinical Trials with Cabozantinib to Understand Roles of MET and VEGFR2 as Targets for Growth Inhibition of Prostate Cancer,” Clinical Cancer Research, vol. 22, no. 1, pp. 107–121, Jan. 2016. [CrossRef]
- F. Koinis et al., “Resistance to MET/VEGFR2 Inhibition by Cabozantinib Is Mediated by YAP/TBX5-Dependent Induction of FGFR1 in Castration-Resistant Prostate Cancer,” Cancers, vol. 12, no. 1, p. 244, Jan. 2020. [CrossRef]
- Grüllich, “Cabozantinib: Multi-kinase Inhibitor of MET, AXL, RET, and VEGFR2,” in Recent results in cancer research, Springer Science+Business Media, 2018, pp. 67–75. [CrossRef]
- G. Bergers and D. Hanahan, “Modes of resistance to anti-angiogenic therapy,” Nature Reviews Cancer, vol. 8, no. 8, pp. 592–603, Aug. 2008. [CrossRef]
- E. Daudigeos-Dubus et al., “Dual inhibition using cabozantinib overcomes HGF/MET signaling mediated resistance to pan-VEGFR inhibition in orthotopic and metastatic neuroblastoma tumors,” International Journal of Oncology, vol. 50, no. 1, pp. 203–211, Dec. 2016. [CrossRef]
- O. Casanovas, D. J. Hicklin, G. Bergers, and D. Hanahan, “Drug resistance by evasion of antiangiogenic targeting of VEGF signaling in late-stage pancreatic islet tumors,” Cancer Cell, vol. 8, no. 4, pp. 299–309, Oct. 2005. [CrossRef]
- Grüllich, “Cabozantinib: A MET, RET, and VEGFR2 Tyrosine Kinase Inhibitor,” in Springer eBooks, 2014, pp. 207–214. [CrossRef]
- Q. Xiang et al., “Cabozantinib Suppresses Tumor Growth and Metastasis in Hepatocellular Carcinoma by a Dual Blockade of VEGFR2 and MET,” Clinical Cancer Research, vol. 20, no. 11, pp. 2959–2970, May 2014. [CrossRef]
- M. Yakes et al., “Cabozantinib (XL184), a Novel MET and VEGFR2 Inhibitor, Simultaneously Suppresses Metastasis, Angiogenesis, and Tumor Growth,” Molecular Cancer Therapeutics, vol. 10, no. 12, pp. 2298–2308, Dec. 2011. [CrossRef]
- B. Sennino et al., “Suppression of Tumor Invasion and Metastasis by Concurrent Inhibition of c-Met and VEGF Signaling in Pancreatic Neuroendocrine Tumors,” Cancer Discovery, vol. 2, no. 3, pp. 270–287, Mar. 2012. [CrossRef]
- R. Shang et al., “Cabozantinib-based combination therapy for the treatment of hepatocellular carcinoma,” Gut, vol. 70, no. 9, pp. 1746–1757, Nov. 2020. [CrossRef]
- M. Santoni, R. Iacovelli, V. Colonna, S. G. Klinz, G. Mauri, and M. Nuti, “Antitumor effects of the multi-target tyrosine kinase inhibitor cabozantinib: A comprehensive review of the preclinical evidence,” Expert Review of Anticancer Therapy, vol. 21, no. 9, pp. 1029–1054, Aug. 2021. [CrossRef]
- S. Deng, A. Solinas, and D. F. Calvisi, “Cabozantinib for HCC Treatment, From Clinical Back to Experimental Models,” Frontiers in Oncology, vol. 11, Oct. 2021. [CrossRef]
- S. Deng, A. Solinas, and D. F. Calvisi, “Cabozantinib for HCC Treatment, From Clinical Back to Experimental Models,” Frontiers in Oncology, vol. 11, Oct. 2021. [CrossRef]
- J. Szymczyk, K. D. Sluzalska, I. Materla, Ł. Opaliński, J. Otlewski, and M. Zakrzewska, “FGF/FGFR-Dependent Molecular Mechanisms Underlying Anti-Cancer Drug Resistance,” Cancers, vol. 13, no. 22, p. 5796, Nov. 2021. [CrossRef]
- R. Katayama et al., “Cabozantinib Overcomes Crizotinib Resistance in ROS1 Fusion–Positive Cancer,” Clinical Cancer Research, vol. 21, no. 1, pp. 166–174, Jan. 2015. [CrossRef]
- N. L. Komarova and D. Wodarz, “Drug resistance in cancer: Principles of emergence and prevention,” Proceedings of the National Academy of Sciences of the United States of America, vol. 102, no. 27, pp. 9714–9719, Jun. 2005. [CrossRef]
- R. B. Corcoran, J. Settleman, and J. A. Engelman, “Potential Therapeutic Strategies to Overcome Acquired Resistance to BRAF or MEK Inhibitors in BRAF Mutant Cancers,” Oncotarget, vol. 2, no. 4, pp. 336–346, Apr. 2011. [CrossRef]
- E. Daudigeos-Dubus et al., “Dual inhibition using cabozantinib overcomes HGF/MET signaling mediated resistance to pan-VEGFR inhibition in orthotopic and metastatic neuroblastoma tumors,” International Journal of Oncology, vol. 50, no. 1, pp. 203–211, Dec. 2016. [CrossRef]
- R. C. O’Connor, “A Review of Mechanisms of Circumvention and Modulation of Chemotherapeutic Drug Resistance,” Current Cancer Drug Targets, vol. 9, no. 3, pp. 273–280, May 2009. [CrossRef]
- S. Crystal et al., “Patient-derived models of acquired resistance can identify effective drug combinations for cancer,” Science, vol. 346, no. 6216, pp. 1480–1486, Dec. 2014. [CrossRef]
- N. L. Komarova and D. Wodarz, “Drug resistance in cancer: Principles of emergence and prevention,” Proceedings of the National Academy of Sciences of the United States of America, vol. 102, no. 27, pp. 9714–9719, Jun. 2005. [CrossRef]
- R. B. Corcoran, J. Settleman, and J. A. Engelman, “Potential Therapeutic Strategies to Overcome Acquired Resistance to BRAF or MEK Inhibitors in BRAF Mutant Cancers,” Oncotarget, vol. 2, no. 4, pp. 336–346, Apr. 2011. [CrossRef]
- E. Daudigeos-Dubus et al., “Dual inhibition using cabozantinib overcomes HGF/MET signaling mediated resistance to pan-VEGFR inhibition in orthotopic and metastatic neuroblastoma tumors,” International Journal of Oncology, vol. 50, no. 1, pp. 203–211, Dec. 2016. [CrossRef]
- R. C. O’Connor, “A Review of Mechanisms of Circumvention and Modulation of Chemotherapeutic Drug Resistance,” Current Cancer Drug Targets, vol. 9, no. 3, pp. 273–280, May 2009. [CrossRef]
- J. Qi et al., “Multiple Mutations and Bypass Mechanisms Can Contribute to Development of Acquired Resistance to MET Inhibitors,” Cancer Research, vol. 71, no. 3, pp. 1081–1091, Feb. 2011. [CrossRef]
- J. A. Engelman et al., “MET Amplification Leads to Gefitinib Resistance in Lung Cancer by Activating ERBB3 Signaling,” Science, vol. 316, no. 5827, pp. 1039–1043, May 2007. [CrossRef]
- L. V. Sequist et al., “Genotypic and Histological Evolution of Lung Cancers Acquiring Resistance to EGFR Inhibitors,” Science Translational Medicine, vol. 3, no. 75, Mar. 2011. [CrossRef]
- R. Katayama et al., “Therapeutic strategies to overcome crizotinib resistance in non-small cell lung cancers harboring the fusion oncogene EML4-ALK,” Proceedings of the National Academy of Sciences of the United States of America, vol. 108, no. 18, pp. 7535–7540, Apr. 2011. [CrossRef]
- M. E. Gorre et al., “Clinical Resistance to STI-571 Cancer Therapy Caused by BCR-ABL Gene Mutation or Amplification,” Science, vol. 293, no. 5531, pp. 876–880, Aug. 2001. [CrossRef]
- M. Santoni, R. Iacovelli, V. Colonna, S. G. Klinz, G. Mauri, and M. Nuti, “Antitumor effects of the multi-target tyrosine kinase inhibitor cabozantinib: A comprehensive review of the preclinical evidence,” Expert Review of Anticancer Therapy, vol. 21, no. 9, pp. 1029–1054, Aug. 2021. [CrossRef]
- M. Guix et al., “Acquired resistance to EGFR tyrosine kinase inhibitors in cancer cells is mediated by loss of IGF-binding proteins,” Journal of Clinical Investigation, Jun. 2008. [CrossRef]
- R. Kotecha et al., “Regression of Intracranial Meningiomas Following Treatment with Cabozantinib,” Current Oncology, vol. 28, no. 2, pp. 1537–1543, Apr. 2021. [CrossRef]
- R. Goldbrunner et al., “EANO guidelines for the diagnosis and treatment of meningiomas,” Lancet Oncology, vol. 17, no. 9, pp. e383–e391, Sep. 2016. [CrossRef]
- Y. Sheikh, E. B. Siqueira, and F. A. Dayel, “Meningioma in children: A report of nine cases and review of the literature,” Surgical Neurology, vol. 45, no. 4, pp. 328–335, Apr. 1996. [CrossRef]
- L. Kim, “A narrative review of targeted therapies in meningioma,” Chinese Clinical Oncology, vol. 9, no. 6, p. 76, Dec. 2020. [CrossRef]
- C. Ogasawara, B. D. Philbrick, and D. C. Adamson, “Meningioma: A Review of Epidemiology, Pathology, Diagnosis, Treatment, and Future Directions,” Biomedicines, vol. 9, no. 3, p. 319, Mar. 2021. [CrossRef]
- E. Lou et al., “Bevacizumab therapy for adults with recurrent/progressive meningioma: A retrospective series,” Journal of Neuro-oncology, vol. 109, no. 1, pp. 63–70, Apr. 2012. [CrossRef]
- T. Garzon-Muvdi, D. D. Bailey, M. N. Pernik, and E. Pan, “Basis for Immunotherapy for Treatment of Meningiomas,” Frontiers in Neurology, vol. 11, Aug. 2020. [CrossRef]
- T. Garzon-Muvdi, D. D. Bailey, M. N. Pernik, and E. Pan, “Basis for Immunotherapy for Treatment of Meningiomas,” Frontiers in Neurology, vol. 11, Aug. 2020. [CrossRef]
- N. Wang and M. Osswald, “Meningiomas: Overview and New Directions in Therapy,” Seminars in Neurology, vol. 38, no. 01, pp. 112–120, Feb. 2018. [CrossRef]
- V. E. Clark et al., “Genomic Analysis of Non- NF2 Meningiomas Reveals Mutations in TRAF7, KLF4, AKT1, and SMO,” Science, vol. 339, no. 6123, pp. 1077–1080, Mar. 2013. [CrossRef]
- T. Kaley et al., “Phase II trial of sunitinib for recurrent and progressive atypical and anaplastic meningioma,” Neuro-oncology, vol. 17, no. 1, pp. 116–121, Aug. 2014. [CrossRef]
- T. A. Wilson et al., “Review of Atypical and Anaplastic Meningiomas: Classification, Molecular Biology, and Management,” Frontiers in Oncology, vol. 10, Nov. 2020. [CrossRef]
- M. Abedalthagafi et al., “Oncogenic PI3K mutations are as common asAKT1andSMOmutations in meningioma,” Neuro-oncology, vol. 18, no. 5, pp. 649–655, Jan. 2016. [CrossRef]
- C. A. Dasanu, Y. Samara, I. Codreanu, F. M. Limonadi, O. Hamid, and J. Alvarez-Argote, “Systemic therapy for relapsed/refractory meningioma: Is there potential for antiangiogenic agents?,” Journal of Oncology Pharmacy Practice, vol. 25, no. 3, pp. 638–647, Sep. 2018. [CrossRef]
- T. Graillon, E. Tabouret, and O. Chinot, “Chemotherapy and targeted therapies for meningiomas: What is the evidence?,” Current Opinion in Neurology, vol. 34, no. 6, pp. 857–867, Oct. 2021. [CrossRef]
- B. Ansmann, A. Hecht, D. K. Henn, S. Leptien, and H. G. Stelzer, “The future of monitoring in clinical research - a holistic approach: Linking risk-based monitoring with quality management principles.,” PubMed, Jan. 2013. [CrossRef]
- J. A. Usher-Smith, J. Emery, W. Hamilton, S. J. Griffin, and F. M. Walter, “Risk prediction tools for cancer in primary care,” British Journal of Cancer, vol. 113, no. 12, pp. 1645–1650, Dec. 2015. [CrossRef]
- S. Hirschfeld and A. Zajicek, “What Could the Future of Safety Monitoring Look Like?,” Therapeutic Innovation & Regulatory Science, Sep. 2019. [CrossRef]
- D. Levêque and G. Becker, “The role of therapeutic drug monitoring in the management of safety of anticancer agents: A focus on 3 cytotoxics,” Expert Opinion on Drug Safety, vol. 18, no. 11, pp. 1009–1015, Sep. 2019. [CrossRef]
- G. Trifirò and J. Sultana, “A New Era of Pharmacovigilance: Future Challenges and Opportunities,” Frontiers in Drug Safety and Regulation, vol. 2, Feb. 2022. [CrossRef]
- B. Yao, L. Zhu, Q. Jiang, and H. R. Xia, “Safety Monitoring in Clinical Trials,” Pharmaceutics, vol. 5, no. 4, pp. 94–106, Jan. 2013. [CrossRef]
- J. P. Walker, S. Licqurish, P. P.-C. Chiang, M. Pirotta, and J. P. Emery, “Cancer Risk Assessment Tools in Primary Care: A Systematic Review of Randomized Controlled Trials,” Annals of Family Medicine, vol. 13, no. 5, pp. 480–489, Sep. 2015. [CrossRef]
- A. Galpin and W. J. Evans, “Therapeutic drug monitoring in cancer management,” Clinical Chemistry, vol. 39, no. 11, pp. 2419–2430, Nov. 1993. [CrossRef]
- J. Hippisley-Cox and C. Coupland, “Development and validation of risk prediction algorithms to estimate future risk of common cancers in men and women: Prospective cohort study,” BMJ Open, vol. 5, no. 3, p. e007825, Mar. 2015. [CrossRef]
- S. Bhagat et al., “An Industry Perspective on Risks and Mitigation Strategies Associated with Post Conduct Phase of Clinical Trial,” Reviews on Recent Clinical Trials, vol. 15, no. 1, pp. 28–33, Feb. 2020. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



