Submitted:
16 June 2023
Posted:
19 June 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
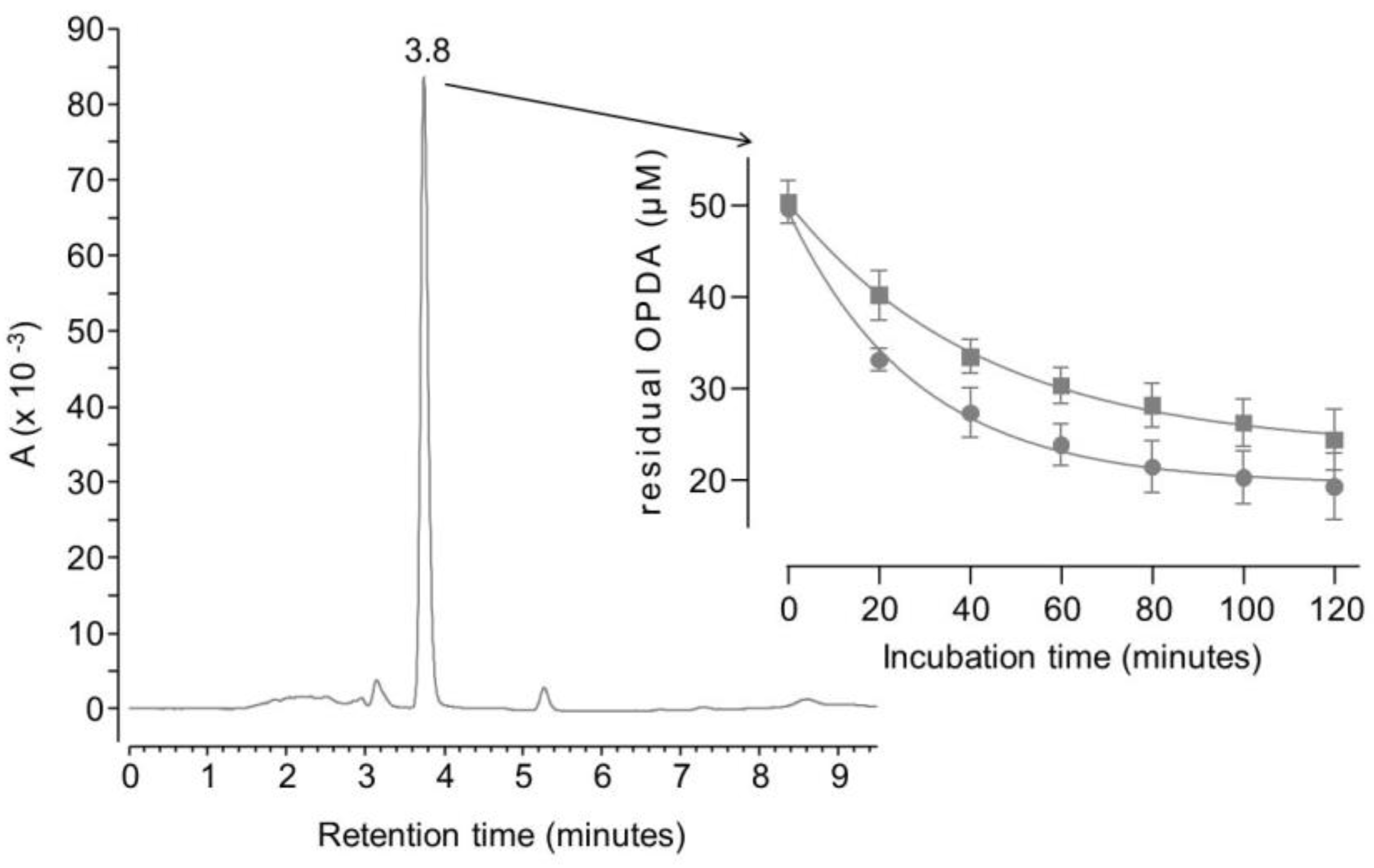
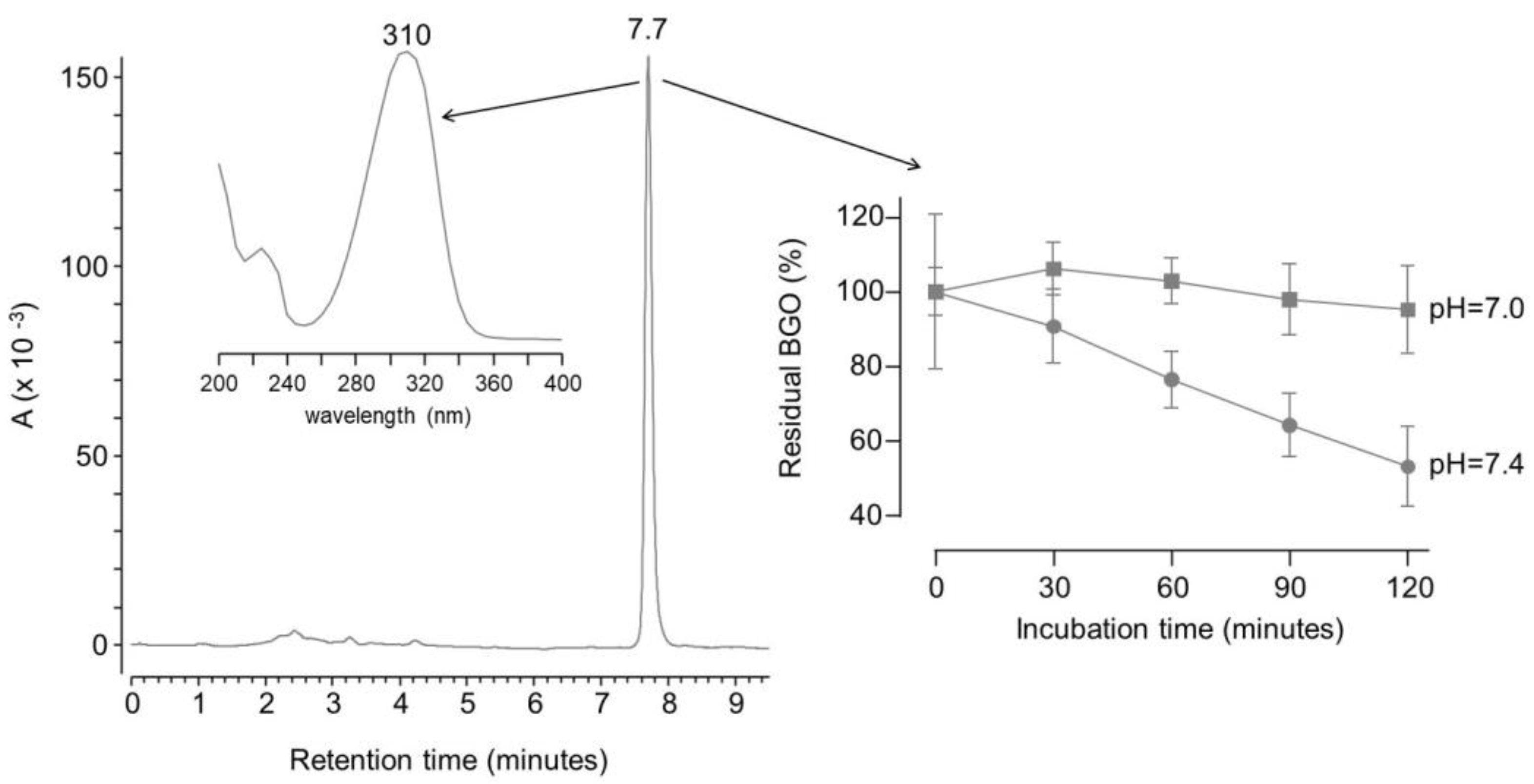
2. Results and discussion
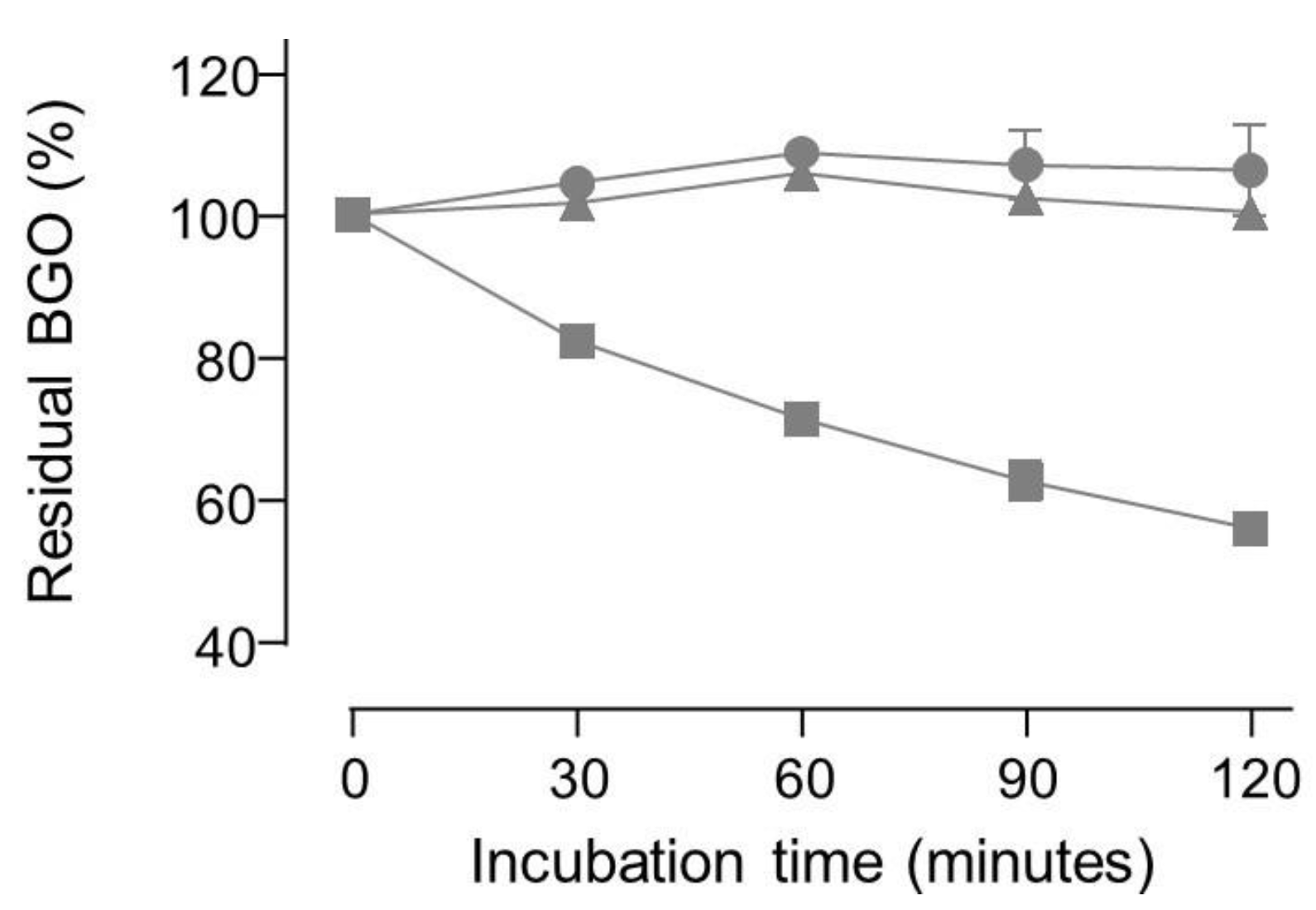
3. Materials and Methods
3.1. Chemicals
3.2. Synthesis of benzylglyoxal dimethylacetal
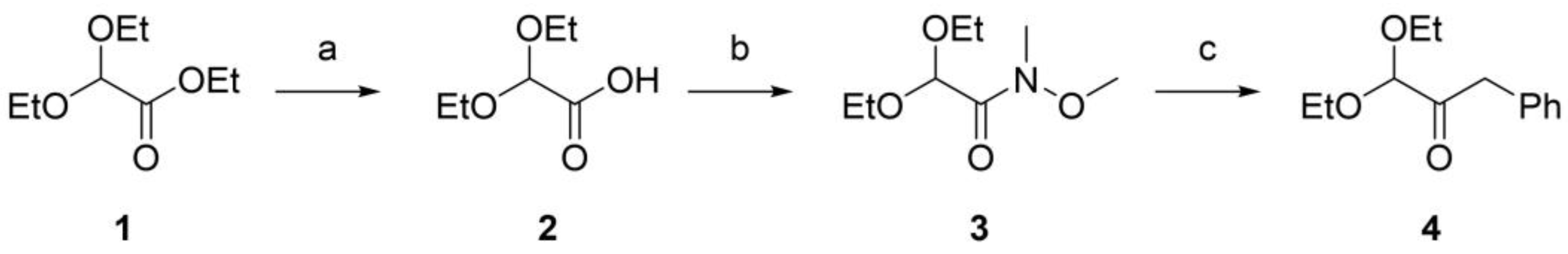
3.2.1. Synthesis of 2,2-diethoxyacetic acid (compound 2, Figure 1)
3.2.2. Synthesis of 2,2-Diethoxy-N-methoxy-N-methyl-acetamide (compound 3, Figure 1)
3.2.3. Synthesis of Benzylglyoxal dimethylacetal (compound 4, Figure 1)
3.3. Stock solutions
3.4. Methods
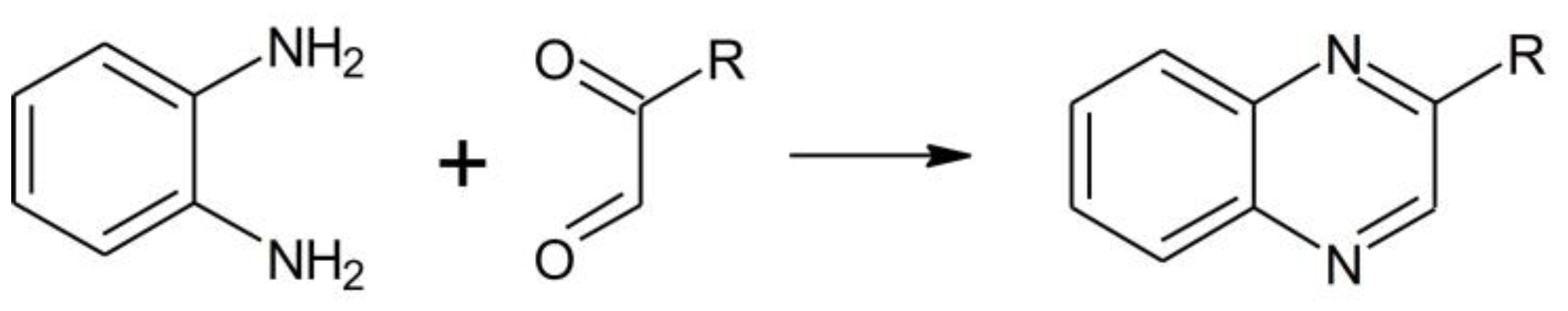
3.4.1. Ortho-phenylenediammine binding kinetics
3.4.2. Direct determination of dicarbonyl binding activity
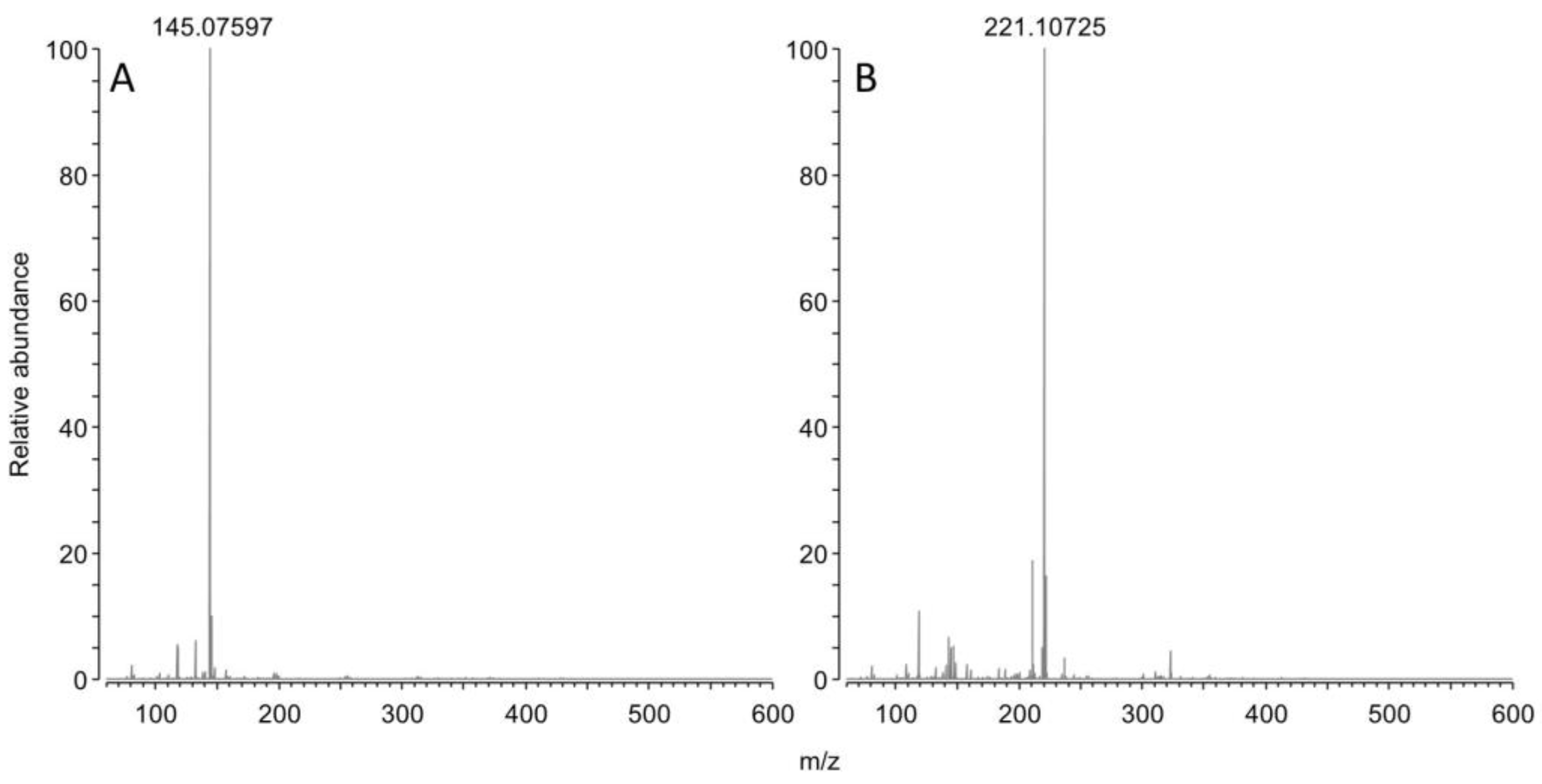
3.4.3. Mass spectrometry
3.4.4. NMR spectroscopy
4. Conclusions
Author Contributions
Data Availability Statement
Conflicts of Interest
References
- Arribas-Lorenzo, G.; Morales, F.J. Analysis, Distribution, and Dietary Exposure of Glyoxal and Methylglyoxal in Cookies and Their Relationship with Other Heat-Induced Contaminants. J. Agric. Food Chem. 2010, 58, 2966–2972. [Google Scholar] [CrossRef]
- Kwak, S.; Choi, Y.S.; Na, H.G.; Bae, C.H.; Song, S.-Y.; Kim, Y.-D. Glyoxal and Methylglyoxal as E-cigarette Vapor Ingredients-Induced Pro-Inflammatory Cytokine and Mucins Expression in Human Nasal Epithelial Cells. Am. J. Rhinol. Allergy 2020, 35, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ho, C.-T. Flavour chemistry of methylglyoxal and glyoxal. Chem. Soc. Rev. 2012, 41, 4140–4149. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Ishida, J.; Xuan, Z.X.; Nakamura, A.; Yoshitake, T. Determination of Glyoxal, Methylglyoxal, Diacethyl, and 2, 3-Pentanedione in Fermented Foods by High-Performance Liquid Chromatography with Fluorescence Detection. J. Liq. Chromatogr. 1994, 17, 203–211. [Google Scholar] [CrossRef]
- Lange, J.N.; Wood, K.D.; Knight, J.; Assimos, D.G.; Holmes, R.P. Glyoxal Formation and Its Role in Endogenous Oxalate Synthesis. Adv. Urol. 2012, 2012, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Thornalley, P.J.; Langborg, A.; Minhas, H.S. Formation of glyoxal, methylglyoxal and 3-deoxyglucosone in the glycation of proteins by glucose. Biochem. J. 1999, 344 Pt 1, 109–116. [Google Scholar] [CrossRef]
- Chakraborty, S.; Karmakar, K.; Chakravortty, D. Cells producing their own nemesis: Understanding methylglyoxal metabolism. IUBMB Life 2014, 66, 667–678. [Google Scholar] [CrossRef]
- Phillips, S.A.; Thornalley, P.J. The formation of methylglyoxal from triose phosphates. Investigation using a specific assay for methylglyoxal. JBIC J. Biol. Inorg. Chem. 1993, 212, 101–105. [Google Scholar] [CrossRef]
- Rabbani, N.; Xue, M.; Thornalley, P.J. Dicarbonyls and glyoxalase in disease mechanisms and clinical therapeutics. Glycoconj. J. 2016, 33, 513–525. [Google Scholar] [CrossRef]
- Izaguirre, G.; Kikonyogo, A.; Pietruszko, R. Methylglyoxal as substrate and inhibitor of human aldehyde dehydrogenase: Comparison of kinetic properties among the three isozymes. Comp. Biochem. Physiol. Part B: Biochem. Mol. Biol. 1998, 119, 747–754. [Google Scholar] [CrossRef]
- Jagt, D.L.V.; A Hunsaker, L. Methylglyoxal metabolism and diabetic complications: roles of aldose reductase, glyoxalase-I, betaine aldehyde dehydrogenase and 2-oxoaldehyde dehydrogenase. Chem. Interactions 2003, 143-144, 341–351. [Google Scholar] [CrossRef]
- Vander Jagt, D.L.; Robinson, B.; Taylor, K.K.; Hunsaker, L.A. Reduction of trioses by NADPH-dependent aldo-keto reductases. Aldose reductase, methylglyoxal, and diabetic complications. J. Biol. Chem. 1992, 267, 4364–4369. [Google Scholar] [CrossRef] [PubMed]
- Reichard, G.A., Jr.; Skutches, C.L.; Hoeldtke, R.D.; Owen, O.E. Acetone metabolism in humans during diabetic ketoacidosis. Diabetes 1986, 35, 668–674. [Google Scholar] [CrossRef] [PubMed]
- Abordo, E.A.; Minhas, H.S.; Thornalley, P.J. Accumulation of α-oxoaldehydes during oxidative stress: a role in cytotoxicity. Biochem. Pharmacol. 1999, 58, 641–648. [Google Scholar] [CrossRef] [PubMed]
- Lo, T.W.; Westwood, M.E.; McLellan, A.C.; Selwood, T.; Thornalley, P.J. , Binding and modification of proteins by methylglyoxal under physiological conditions. A kinetic and mechanistic study with N alpha-acetylarginine, N alpha-acetylcysteine, and N alpha-acetyllysine, and bovine serum albumin. J. Biol. Chem. 1994, 269, 32299–32305. [Google Scholar] [CrossRef]
- Ahmed, N.; Thornalley, P.J.; Dawczynski, J.; Franke, S.; Strobel, J.; Stein, G.; Haik, G.M. Methylglyoxal-Derived Hydroimidazolone Advanced Glycation End-Products of Human Lens Proteins. Investig. Opthalmology Vis. Sci. 2003, 44, 5287–5292. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Cohenford, M.A.; Dutta, U.; Dain, J.A. The structural modification of DNA nucleosides by nonenzymatic glycation: an in vitro study based on the reactions of glyoxal and methylglyoxal with 2′-deoxyguanosine. Anal. Bioanal. Chem. 2007, 390, 679–688. [Google Scholar] [CrossRef]
- Lai, S.W.T.; Gonzalez, E.D.J.L.; Zoukari, T.; Ki, P.; Shuck, S.C. Methylglyoxal and Its Adducts: Induction, Repair, and Association with Disease. Chem. Res. Toxicol. 2022, 35, 1720–1746. [Google Scholar] [CrossRef]
- Schalkwijk, C.; Stehouwer, C.D.A. Methylglyoxal, a Highly Reactive Dicarbonyl Compound, in Diabetes, Its Vascular Complications, and Other Age-Related Diseases. Physiol. Rev. 2020, 100, 407–461. [Google Scholar] [CrossRef]
- Wondrak, G.T.; Cervantes-Laurean, D.; Roberts, M.J.; Qasem, J.G.; Kim, M.; Jacobson, E.L.; Jacobson, M.K. Identification of α-dicarbonyl scavengers for cellular protection against carbonyl stress. Biochem. Pharmacol. 2002, 63, 361–373. [Google Scholar] [CrossRef]
- Tan, D.; Wang, Y.; Lo, C.-Y.; Ho, C.-T. Methylglyoxal: its presence and potential scavengers. Asia Pac. J. Clin. Nutr. 2008, 17. [Google Scholar] [CrossRef] [PubMed]
- Löbner, J.; Degen, J.; Henle, T. Creatine Is a Scavenger for Methylglyoxal under Physiological Conditions via Formation of N-(4-Methyl-5-oxo-1-imidazolin-2-yl)sarcosine (MG-HCr). J. Agric. Food Chem. 2015, 63, 2249–2256. [Google Scholar] [CrossRef] [PubMed]
- Colzani, M.; De Maddis, D.; Casali, G.; Carini, M.; Vistoli, G.; Aldini, G. Reactivity, Selectivity, and Reaction Mechanisms of Aminoguanidine, Hydralazine, Pyridoxamine, and Carnosine as Sequestering Agents of Reactive Carbonyl Species: A Comparative Study. Chemmedchem 2016, 11, 1778–1789. [Google Scholar] [CrossRef] [PubMed]
- Vistoli, G.; Orioli, M.; Pedretti, A.; Regazzoni, L.; Canevotti, R.; Negrisoli, G.; Carini, M.; Aldini, G. Design, Synthesis, and Evaluation of Carnosine Derivatives as Selective and Efficient Sequestering Agents of Cytotoxic Reactive Carbonyl Species. Chemmedchem 2009, 4, 967–975. [Google Scholar] [CrossRef]
- Fritzsche, S.; Billig, S.; Rynek, R.; Abburi, R.; Tarakhovskaya, E.; Leuner, O.; Frolov, A.; Birkemeyer, C. Derivatization of Methylglyoxal for LC-ESI-MS Analysis—Stability and Relative Sensitivity of Different Derivatives. Molecules 2018, 23, 2994. [Google Scholar] [CrossRef]
- Li, P.; Zhu, Y.; He, S.; Fan, J.; Hu, Q.; Cao, Y. Development and Validation of a High-Performance Liquid Chromatography Method for the Determination of Diacetyl in Beer Using 4-Nitro-o-phenylenediamine as the Derivatization Reagent. J. Agric. Food Chem. 2012, 60, 3013–3019. [Google Scholar] [CrossRef]
- Barros, A.; Rodrigues, J.A.; Almeida, P.J.; Oliva-Teles, M.T. Determination of glyoxal, methylglyoxal, and diacetyl in selected beer and wine, by hplc with uv spectrophotometric detection, after derivatization with o-phenylenediamine. J. Liq. Chromatogr. Relat. Technol. 1999, 22, 2061–2069. [Google Scholar] [CrossRef]
- Brun, N.; González-Sánchez, J.M.; Demelas, C.; Clément, J.-L.; Monod, A. A fast and efficient method for the analysis of α-dicarbonyl compounds in aqueous solutions: Development and application. Chemosphere 2023, 319, 137977. [Google Scholar] [CrossRef] [PubMed]
- Evans, T.W.; Dehn, W.M. The Benzilic Acid Rearrangement. J. Am. Chem. Soc. 1930, 52, 252–254. [Google Scholar] [CrossRef]
- Thornalley, P.J. Glutathione-dependent detoxification of α-oxoaldehydes by the glyoxalase system: involvement in disease mechanisms and antiproliferative activity of glyoxalase I inhibitors. Chem. Interactions 1998, 111-112, 137–151. [Google Scholar] [CrossRef]
- Keller, B.O.; Sui, J.; Young, A.B.; Whittal, R.M. Interferences and contaminants encountered in modern mass spectrometry. Anal. Chim. Acta 2008, 627, 71–81. [Google Scholar] [CrossRef] [PubMed]
| Time (minutes) | 10 mM NH4Cl | CH3CN |
|---|---|---|
| 0.00 | 70% | 30% |
| 1.00 | 70% | 30% |
| 6.00 | 40% | 60% |
| 6.00 | 70% | 30% |
| 9.50 | 70% | 30% |
| 22.00 | 95% | 5% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




