Submitted:
15 June 2023
Posted:
15 June 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
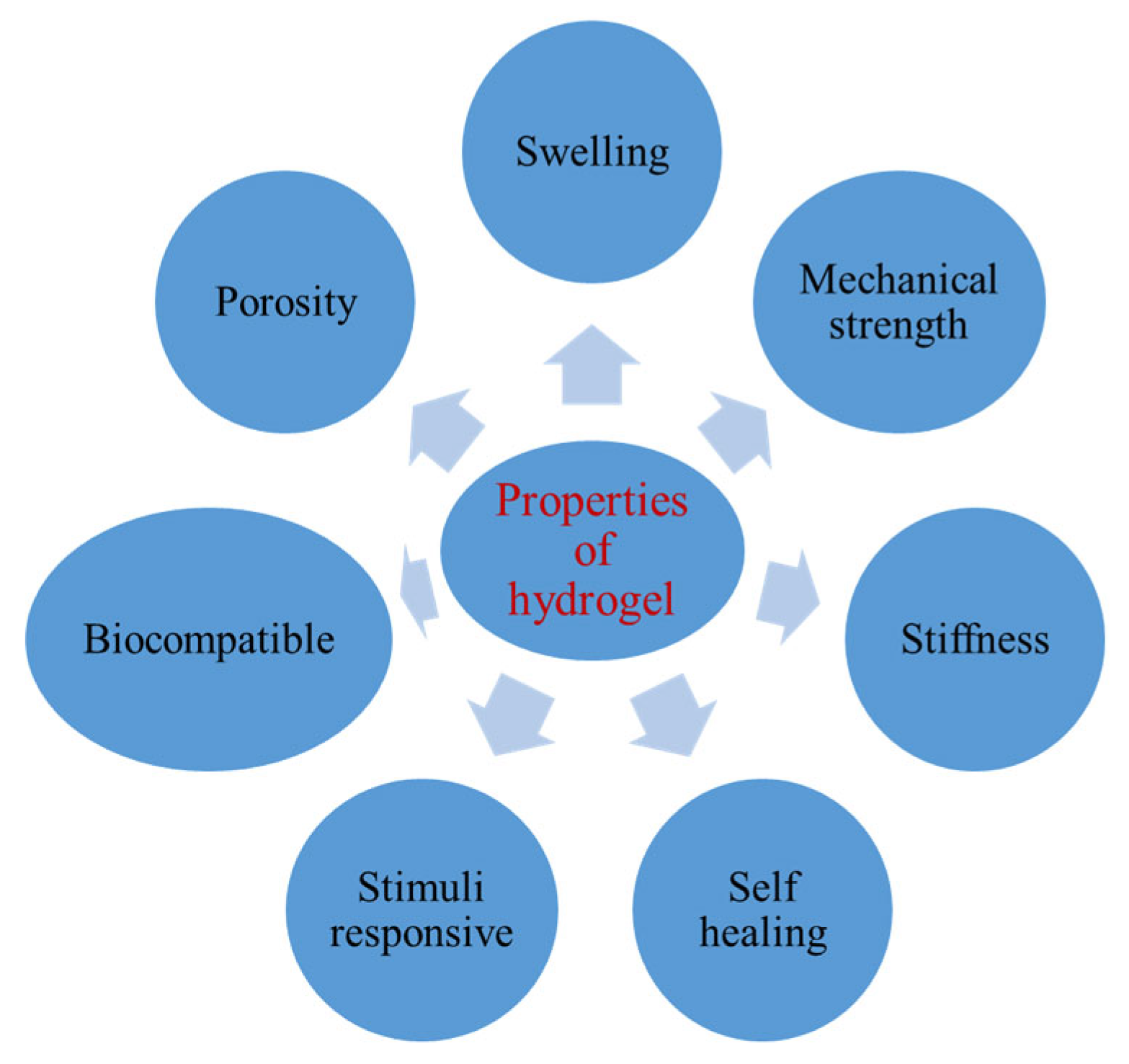
2. Properties of Polymeric Hydrogels
- Solid and liquid-like properties
- Biocompatibility and biodegradability
- Maximum absorption capacity
- Desired porosity and preferred particle size
- Shrink on drying
- Stimuli-responsive
2.1. Swelling
2.2. Mechanical Strength
2.3. Stiffness
2.4. Stress Relaxation
2.5. Self-Healing
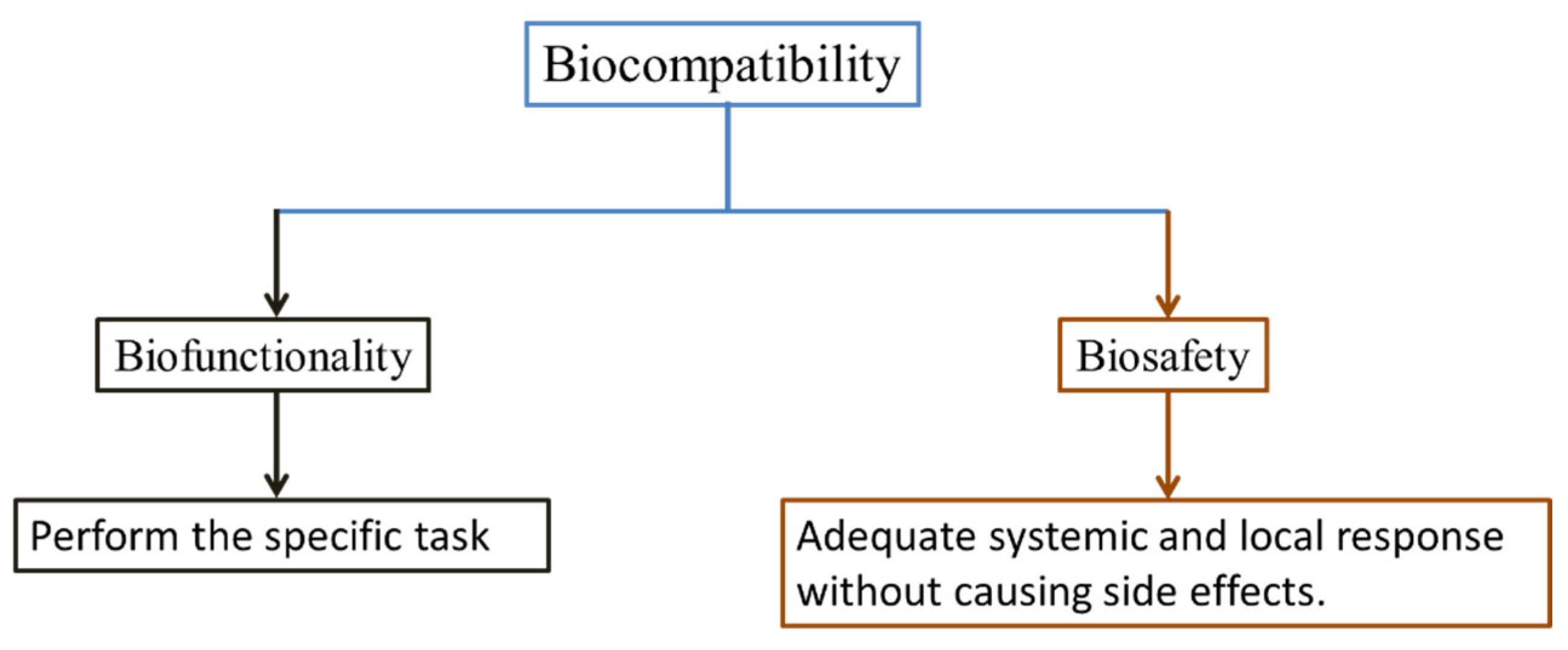
2.6. Biocompatibility
- (i)
- Bio functionality - the ability to perform the specific task for which it is intended.
- (ii)
- Biosafety - the ability to perform adequate systemic and local (the surrounding tissue) host response without causing mutagenesis, cytotoxicity, and carcinogenesis.
2.7. Porosity
- Phase separation by using porogens (chemical additives that generate pores).
- Lyophilization via foam formation and cryogelation.
- The concentration of crosslinkers in polymer chains
- Physical entanglements concentration of the polymer chains
- Polyelectrolytes net charge
3. Advantages of Polymeric Hydrogels [31,32]
- High water content provides flexibility like natural tissue
- Loading and release of therapeutics
- Biocompatible, Biodegradable, and Injectable
- Smart polymeric hydrogels are responsive to external stimuli
- Easy to modify and good transport property
4. Disadvantages of Polymeric Hydrogels [31,32]
- Costly
- Hard to handle.
- Low mechanical strength.
- Difficult to load hydrophobic therapeutics.
5. Classification of Polymeric Hydrogels
5.1. Based on Their Source and Synthesis
- o Natural polymeric hydrogels
- o Synthetic polymeric hydrogels
- o Hybrid polymeric hydrogels
5.1.1. Natural Polymeric Hydrogels
5.1.2. Synthetic Polymeric Hydrogels
5.1.3. Hybrid Polymeric Hydrogels

5.2. Based on Polymeric Composition
- o Homopolymeric hydrogels
- o Co-polymeric hydrogels
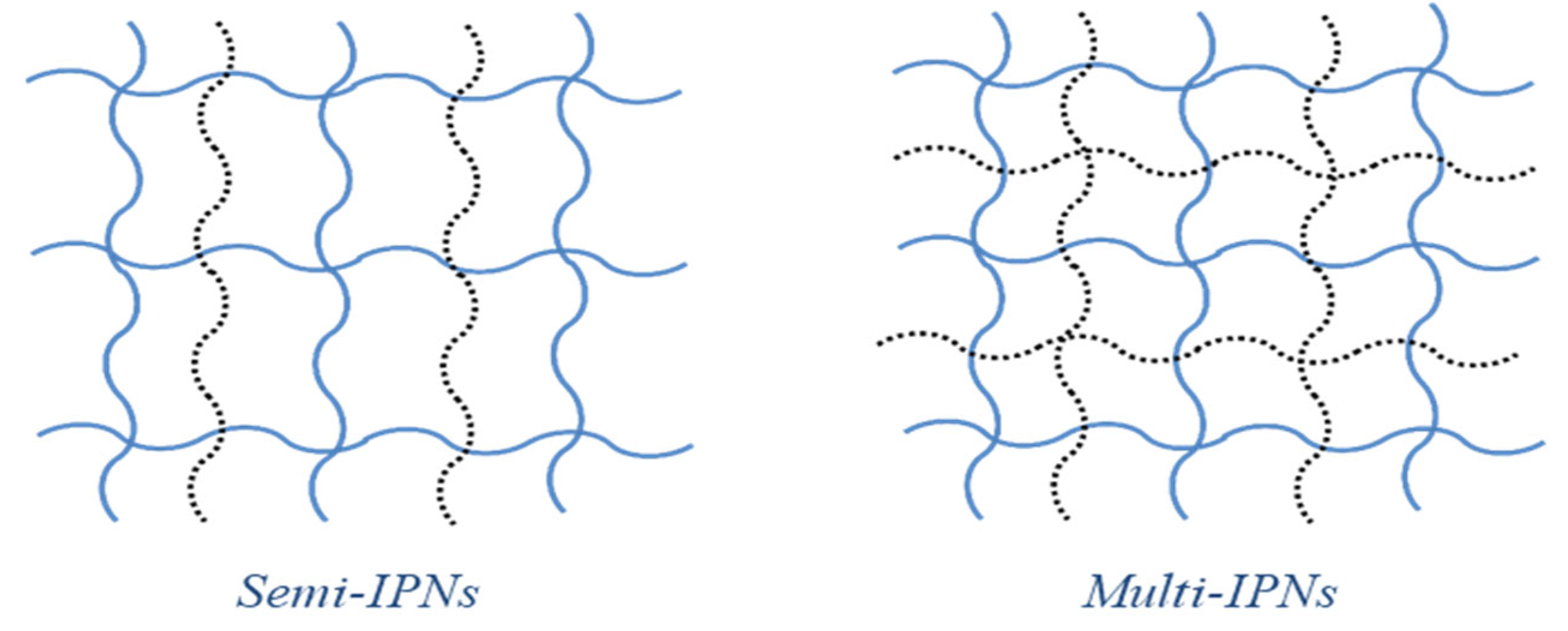
- o Interpenetrating polymeric network hydrogel
- ➢ Semi-interpenetrating polymer network
- ➢ Multi-interpenetrating polymer network
5.2.1. Homopolymeric Hydrogels
5.2.2. Co-polymeric hydrogels
5.2.3. Interpenetrating polymeric network (IPN)
5.2.3.1. Semi-interpenetrating polymeric hydrogel
5.2.3.2. Multi-Interpenetrating Polymeric Hydrogel
Advantages
5.3. Based on Degradability
5.3.1. Biodegradable Hydrogels
5.3.2. Non-Biodegradable Hydrogels
5.4. Based on Configuration
5.4.1. Amorphous Hydrogel
5.4.2. Semi-Crystalline Hydrogel
5.4.3. Crystalline Hydrogels
5.4.4. Hydrocolloid Aggregates
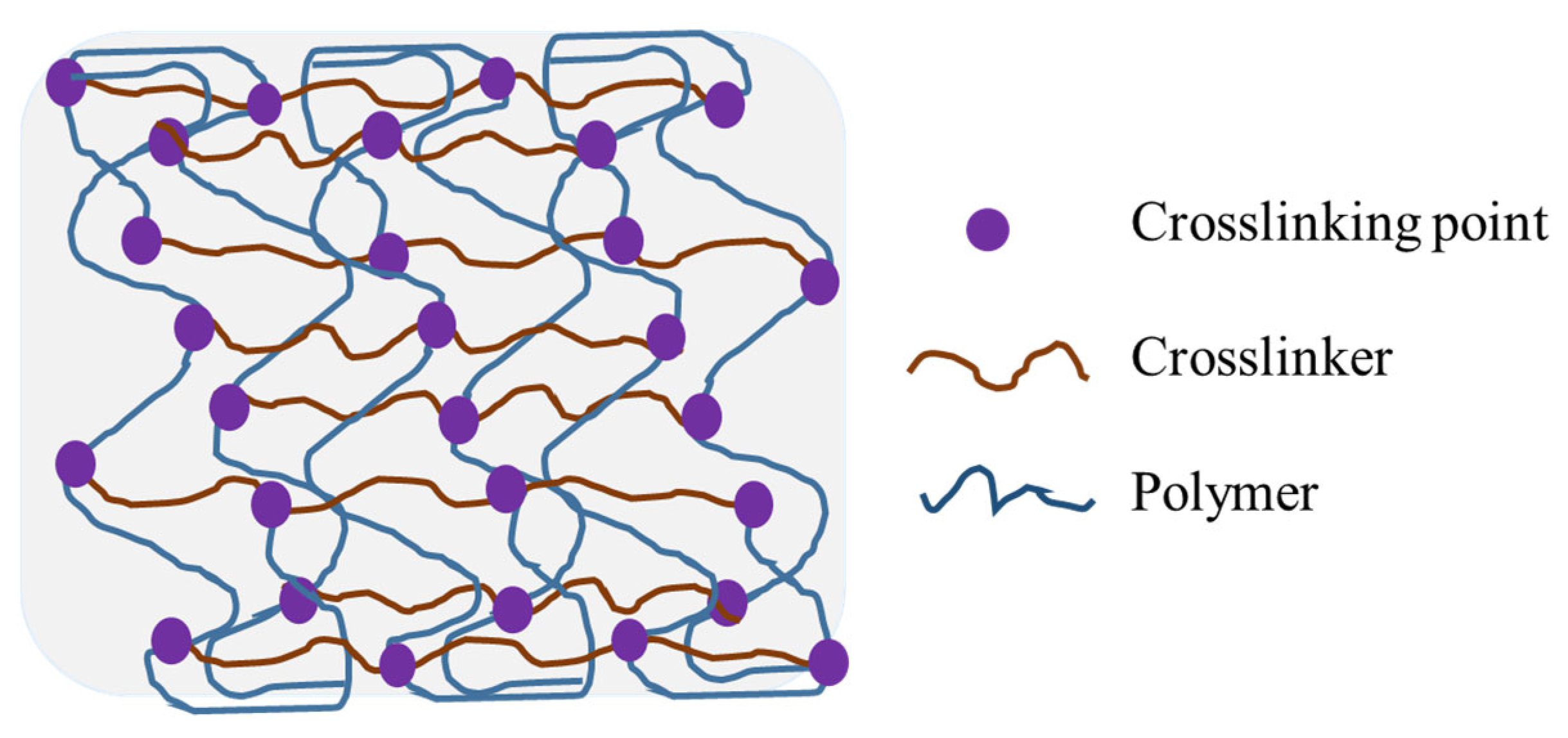
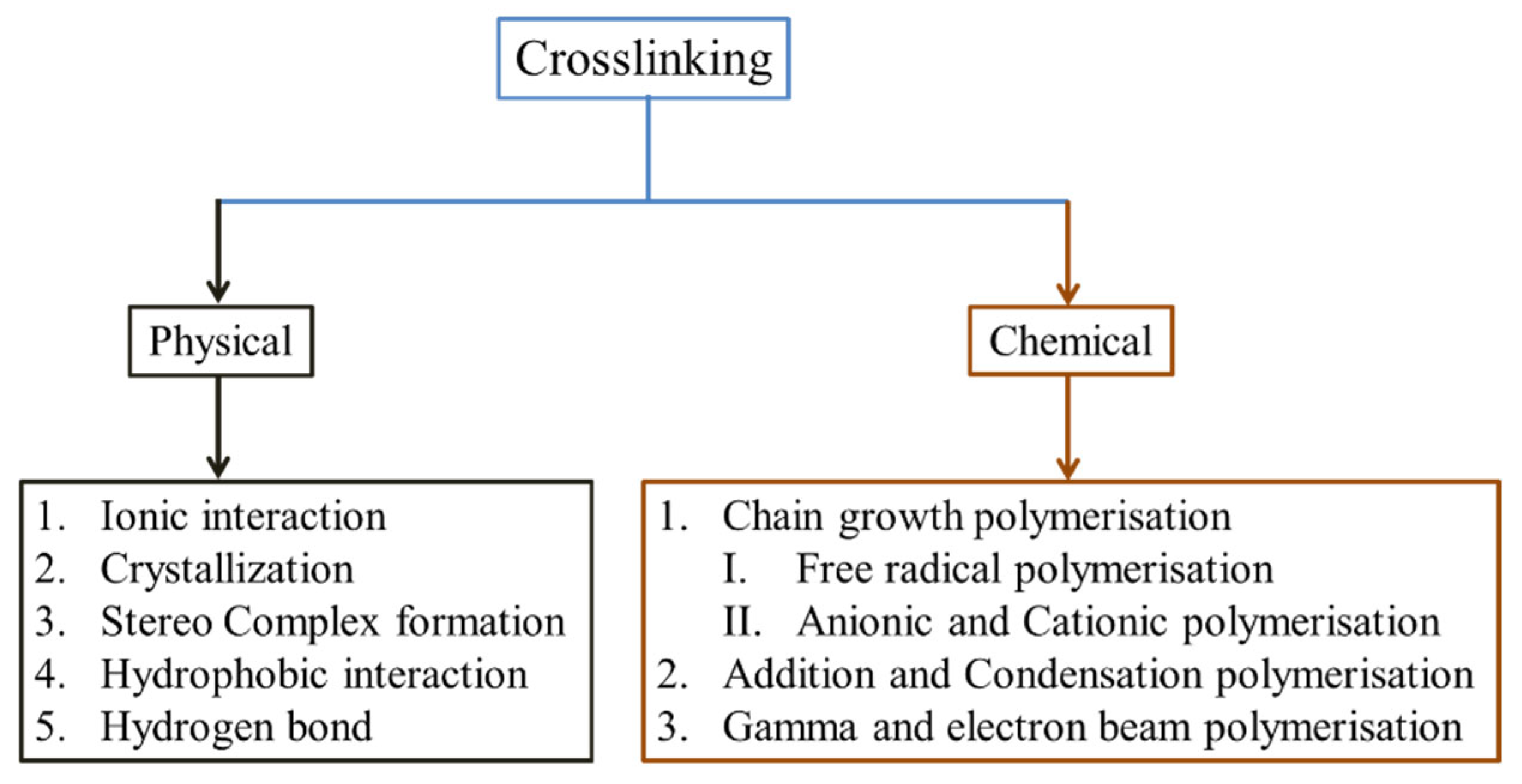
5.5. Based on Type of Crosslinking
5.5.1. Physically Cross-Linked Hydrogels
5.5.2. Chemically Cross-Linked Hydrogels
5.6. Based on Physical Appearance
5.7. Based on Charge
- Neutral (Nonionic)
- Anionic
- Cationic
- Ampholytic
- Zwitter ionic
5.8. Based on Physical Property
5.8.1. Conventional Polymeric Hydrogels
5.8.2. Smart Polymeric Hydrogels
6. Technologies Adopted in Hydrogel Preparation
- Free radicals generation by ionizing radiation that combines as crosslink junctions.
- Linking of polymer chains by chemical crosslinking.
- Linking of polymer chains by physical interactions.
6.1. Bulk Polymerization
6.2. Free Radical Polymerization
6.3. Solution Polymerization or Cross-Linking
7. Method of Crosslinking
7.1. Physical Crosslinking
- Heating or Cooling: In this method, hydrogels are formed because of the intramolecular coil formation and association between the coils by applying heat. Hydrogels formed by carrageenan or gelatin are an example of this method.
- Ionic Interactions: In this method, hydrogels are formed by the addition of counter ions as a crosslinker. Hydrogels formed by chitosan-glycerol phosphate salt and chitosan-polylysine are examples of this method.
- Hydrogen Bonding: In this method, hydrogels are formed by hydrogen bonding that, involves reducing the pH of carboxyl groups containing polymer solutions. The hydrogel formed by CMC hydrogel is an example of this method.
- Freeze Thawing: The principle behind this method is the microcrystal formation after freeze-thawing. The polymeric hydrogel formed by the cryogelation of xanthan is an example of this method.
7.2. Chemical Cross-Linking
- Chemical cross-linkers- In this technique, a new molecule is added as a chemical cross-linking agent such as glutaraldehyde and epichlorohydrin for cross-linking of polymer chains to synthesize hydrogels.
- Grafting- Grafting is a technique in which a monomer polymerizes on a preformed polymer support. There are two types of grafting: chemical grafting and radiation grafting.
- Chemical grafting- In this method polymer chains are activated by chemical reagents such as N-vinyl-2-pyrrolidone to graft starch with acrylic acid.
- Radiation grafting- This method involves the formation of free radicals by exposing high-energy radiation onto the polymeric chain. There are three ways to perform this method: simultaneous or direct, pre-irradiation and pre-irradiation oxidative.
Advantages
- I.
- Does not require the use of catalyst nor additives to initiate the reaction
- II.
- Unchanged mechanical properties concerning the pristine polymeric matrix
7.3. Radiation Crosslinking
8. Characterization of Hydrogels
8.1. Morphological Characterization
8.1.1. Direct Imaging
Drawback:
8.1.2. Indirect Imaging
Drawback:
8.2. Mechanical Property of Hydrogels
8.2.1. Yield Stress of Hydrogel
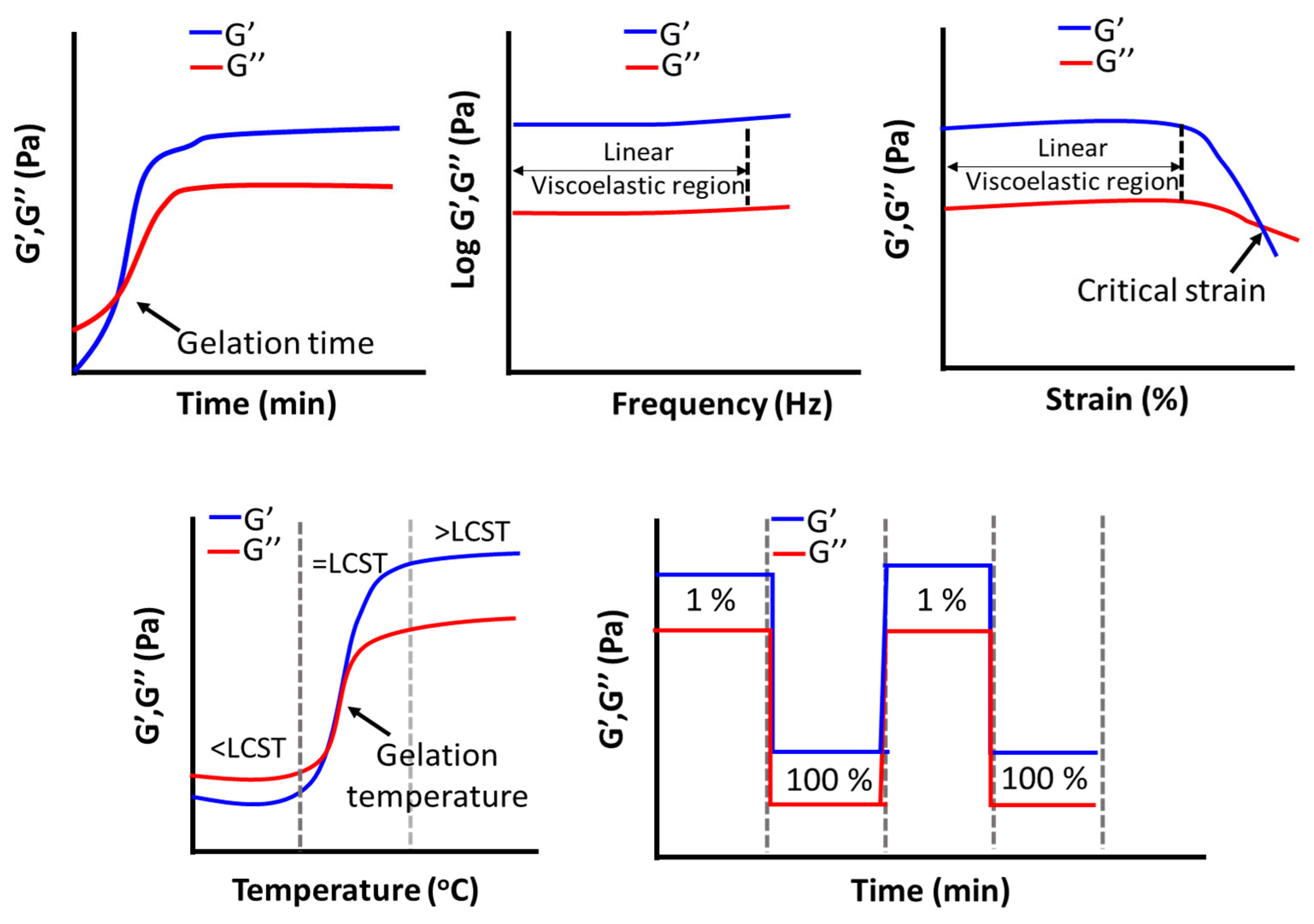
Shear-Thinning Hydrogels
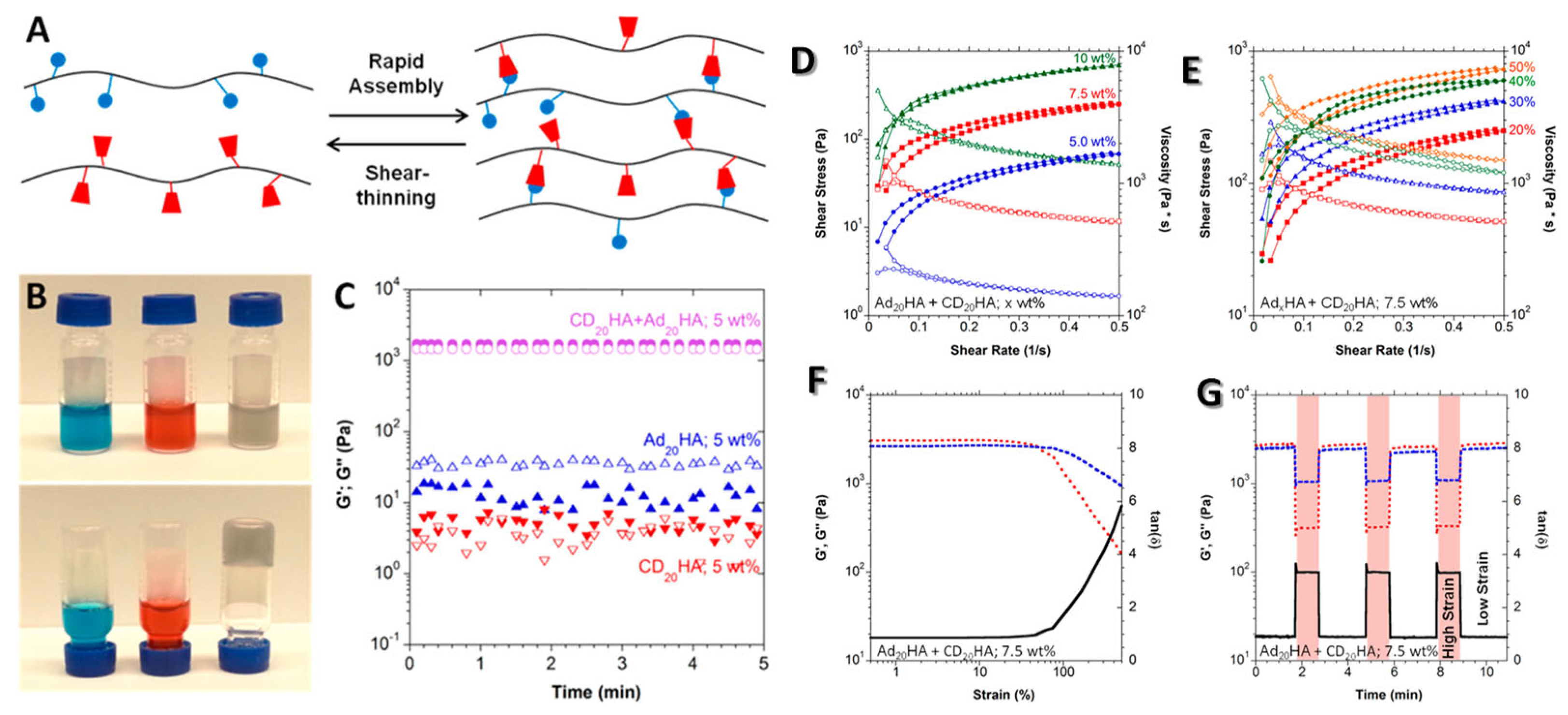

8.2.2. Determination of Thixotropic Behavior of Hydrogels
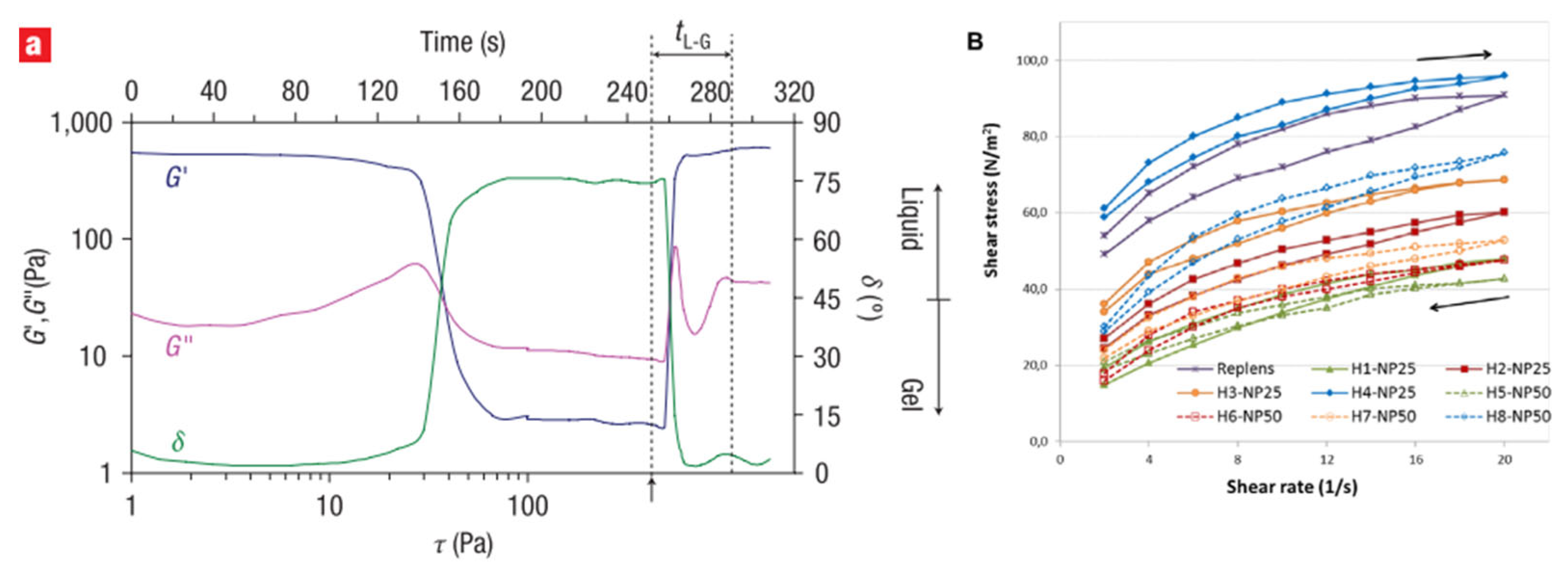
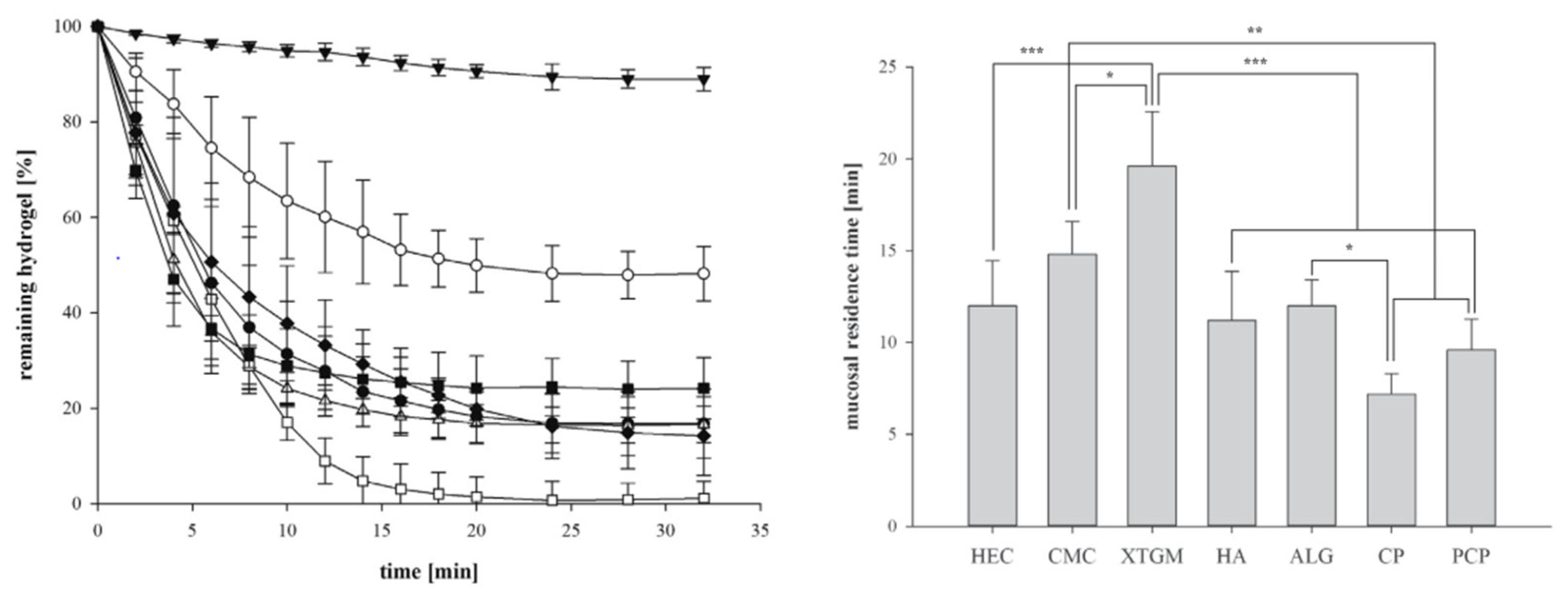
8.2.3. Determination of Mucoadhesive Property of Hydrogels
8.3. Swelling Behavior and Crosslinking Density
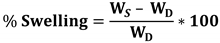
9. Applications of Hydrogels
9.1. Drug Delivery
9.2. Tissue Engineering
9.3. Contact Lenses
9.4. Biosensor
9.5. Wound Dressing
10. Current Status Concerning their Synthesis and Formulations of Polymeric Hydrogels
11. Limitations of Hydrogels
12. Strategies to Overcome the Mechanical Limitations
- i.
- Uneven distribution of covalent bonds causes damage in network structure that can be reduced by changing the covalent cross-linking points by active cross-linking sites.
- i.
- ii. The strength and flexibility of polymeric hydrogels can be improved by the introduction of another network in a certain network system (e.g., double network polymeric hydrogels) [71].
- i.
- iii. The physical adsorption or chemical bonding between the polymer chains and nanoparticles as multifunctional crosslinking points can improve the mechanical properties of hydrogels by dissipating energy; on the other hand, the hydrogels are strengthened by the high surface area and modulus of nanoparticles.
- i.
- iv. Strong, robust, and stimuli-responsive hydrogels made from polymers prepared through non-covalent interactions and supramolecular self-assembling structures [99].
13. Future Perspectives
14. Conclusions
Author Contributions
Funding
References
- Wichterle, O.; LÍM, D. Hydrophilic Gels for Biological Use. Nature 1960, 185, 117–118. [Google Scholar] [CrossRef]
- Maitra, J.; Shukla, V.K. Cross-linking in Hydrogels - A Review. 2014.
- Ahmed, E.M. Hydrogel: Preparation, characterization, and applications: A review. Journal of Advanced Research 2015, 6, 105–121. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Sun, Y.; Kaplan, J.A.; Grinstaff, M.W.; Parquette, J.R. Photo-crosslinking of a self-assembled coumarin-dipeptide hydrogel. New Journal of Chemistry 2015, 39, 3225–3228. [Google Scholar] [CrossRef]
- Sun, Y.; Kaplan, J.A.; Shieh, A.; Sun, H.-L.; Croce, C.M.; Grinstaff, M.W.; Parquette, J.R. Self-assembly of a 5-fluorouracil-dipeptide hydrogel. Chemical Communications 2016, 52, 5254–5257. [Google Scholar] [CrossRef] [PubMed]
- Verhulsel, M.; Vignes, M.; Descroix, S.; Malaquin, L.; Vignjevic, D.M.; Viovy, J.L. A review of microfabrication and hydrogel engineering for micro-organs on chips. Biomaterials 2014, 35, 1816–1832. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Tang, W.; Wang, X.; Zhao, X.; Chen, C.; Zhu, Z. Applications of Hydrogels with Special Physical Properties in Biomedicine. Polymers (Basel) 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Saroia, J.; Yan-en, W.; Wei, Q.; Zhang, K.; Lu, T.; Zhang, B. A review on biocompatibility nature of hydrogels with 3D printing techniques, tissue engineering application and its future prospective. Bio-Design and Manufacturing 2018, 1, 265–279. [Google Scholar] [CrossRef]
- Sun, Z.; Song, C.; Wang, C.; Hu, Y.; Wu, J. Hydrogel-Based Controlled Drug Delivery for Cancer Treatment: A Review. Molecular Pharmaceutics 2020, 17, 373–391. [Google Scholar] [CrossRef]
- Liang, K.; Bae, K.H.; Kurisawa, M. Recent advances in the design of injectable hydrogels for stem cell-based therapy. Journal of Materials Chemistry B 2019, 7, 3775–3791. [Google Scholar] [CrossRef]
- Tanaka, M.; Nakahata, M.; Linke, P.; Kaufmann, S. Stimuli-responsive hydrogels as a model of the dynamic cellular microenvironment. Polymer Journal 2020, 52, 861–870. [Google Scholar] [CrossRef]
- Chen, F.M.; Liu, X. Advancing biomaterials of human origin for tissue engineering. Prog Polym Sci 2016, 53, 86–168. [Google Scholar] [CrossRef]
- Ćorković, I.; Pichler, A.; Šimunović, J.; Kopjar, M. Hydrogels: Characteristics and Application as Delivery Systems of Phenolic and Aroma Compounds. Foods 2021, 10. [Google Scholar] [CrossRef]
- W. L. Hukins, D.; C. Leahy, J.; J. Mathias, K. Biomaterials: defining the mechanical properties of natural tissues and selection of replacement materials. Journal of Materials Chemistry 1999, 9, 629–636. [Google Scholar] [CrossRef]
- Darnell, M.C.; Sun, J.Y.; Mehta, M.; Johnson, C.; Arany, P.R.; Suo, Z.; Mooney, D.J. Performance and biocompatibility of extremely tough alginate/polyacrylamide hydrogels. Biomaterials 2013, 34, 8042–8048. [Google Scholar] [CrossRef]
- Fariba, G.; Farahani, S.V.; Faraahani, E.V. THEORETICAL DESCRIPTION OF HYDROGEL SWELLING: A REVIEW. 2010.
- M.J.A.D., Z.M.; Kabiri, K. Superabsorbent Polymer Materials: A Review. 2008.
- Li, X.; Sun, Q.; Li, Q.; Kawazoe, N.; Chen, G. Functional Hydrogels With Tunable Structures and Properties for Tissue Engineering Applications. Frontiers in Chemistry 2018, 6. [Google Scholar] [CrossRef]
- Haq, M.A.; Su, Y.; Wang, D. Mechanical properties of PNIPAM based hydrogels: A review. Materials Science and Engineering: C 2017, 70, 842–855. [Google Scholar] [CrossRef] [PubMed]
- Heo, J.; Koh, R.H.; Shim, W.; Kim, H.D.; Yim, H.G.; Hwang, N.S. Riboflavin-induced photo-crosslinking of collagen hydrogel and its application in meniscus tissue engineering. Drug Deliv Transl Res 2016, 6, 148–158. [Google Scholar] [CrossRef] [PubMed]
- Dragan, E.S. Design and applications of interpenetrating polymer network hydrogels. A review. Chemical Engineering Journal 2014, 243, 572–590. [Google Scholar] [CrossRef]
- Slaughter, B.V.; Khurshid, S.S.; Fisher, O.Z.; Khademhosseini, A.; Peppas, N.A. Hydrogels in Regenerative Medicine. Advanced Materials 2009, 21, 3307–3329. [Google Scholar] [CrossRef] [PubMed]
- Cui, K.; Ye, Y.N.; Yu, C.; Li, X.; Kurokawa, T.; Gong, J.P. Stress Relaxation and Underlying Structure Evolution in Tough and Self-Healing Hydrogels. ACS Macro Letters 2020, 9, 1582–1589. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Hsu, S.-h. Synthesis and Biomedical Applications of Self-healing Hydrogels. Frontiers in Chemistry 2018, 6. [Google Scholar] [CrossRef]
- Krogsgaard, M.; Behrens, M.A.; Pedersen, J.S.; Birkedal, H. Self-Healing Mussel-Inspired Multi-pH-Responsive Hydrogels. Biomacromolecules 2013, 14, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Yang, J.H.; Zhou, J.; Xu, F.; Zrínyi, M.; Dussault, P.H.; Osada, Y.; Chen, Y.M. Self-healing gels based on constitutional dynamic chemistry and their potential applications. Chemical Society Reviews 2014, 43, 8114–8131. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-L.; Chuo, T.-W. Self-healing polymers based on thermally reversible Diels–Alder chemistry. Polymer Chemistry 2013, 4, 2194–2205. [Google Scholar] [CrossRef]
- Ikada, Y. Section 11 - Biocompatibility of hydrogels. In Gels Handbook, Osada, Y., Kajiwara, K., Fushimi, T., Irasa, O., Hirokawa, Y., Matsunaga, T., Shimomura, T., Wang, L., Ishida, H., Eds.; Academic Press: Burlington, 2001; pp. 388–407. [Google Scholar]
- Annabi, N.; Nichol, J.W.; Zhong, X.; Ji, C.; Koshy, S.; Khademhosseini, A.; Dehghani, F. Controlling the porosity and microarchitecture of hydrogels for tissue engineering. Tissue Eng Part B Rev 2010, 16, 371–383. [Google Scholar] [CrossRef] [PubMed]
- Mastropietro, D.J.; Omidian, H.; Park, K. Drug delivery applications for superporous hydrogels. Expert Opin Drug Deliv 2012, 9, 71–89. [Google Scholar] [CrossRef]
- Ghasemiyeh, P.; Mohammadi-Samani, S. Hydrogels as Drug Delivery Systems; Pros and Cons. Trends in Pharmaceutical Sciences 2019, 5, 7–24. [Google Scholar] [CrossRef]
- Alonso, J.M.; Andrade Del Olmo, J.; Perez Gonzalez, R.; Saez-Martinez, V. Injectable Hydrogels: From Laboratory to Industrialization. Polymers (Basel) 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- M Khansari, M.; Sorokina, L.; Mukherjee, P.; Mukhtar, F.; Rezazadeh Shirdar, M.; Shahidi, M.; Shokuhfar, T. Classification of Hydrogels Based on Their Source: A Review and Application in Stem Cell Regulation. JOM 2017. [Google Scholar] [CrossRef]
- Catoira, M.C.; Fusaro, L.; Di Francesco, D.; Ramella, M.; Boccafoschi, F. Overview of natural hydrogels for regenerative medicine applications. Journal of Materials Science: Materials in Medicine 2019, 30, 115. [Google Scholar] [CrossRef]
- Vieira, S.; da Silva Morais, A.; Silva-Correia, J.; Oliveira, J.M.; Reis, R.L. Natural-Based Hydrogels: From Processing to Applications. In Encyclopedia of Polymer Science and Technology; pp. 1-27.
- Kumar, A.C.; Erothu, H. Synthetic Polymer Hydrogels. In Biomedical Applications of Polymeric Materials and Composites; 2016; pp. 141-162.
- Cai, M.-H.; Chen, X.-Y.; Fu, L.-Q.; Du, W.-L.; Yang, X.; Mou, X.-Z.; Hu, P.-Y. Design and Development of Hybrid Hydrogels for Biomedical Applications: Recent Trends in Anticancer Drug Delivery and Tissue Engineering. Frontiers in Bioengineering and Biotechnology 2021, 9. [Google Scholar] [CrossRef]
- Du, F.; Qiao, B.; Nguyen, T.D.; Vincent, M.P.; Bobbala, S.; Yi, S.; Lescott, C.; Dravid, V.P.; Olvera de la Cruz, M.; Scott, E.A. Homopolymer self-assembly of poly(propylene sulfone) hydrogels via dynamic noncovalent sulfone–sulfone bonding. Nature Communications 2020, 11, 4896. [Google Scholar] [CrossRef] [PubMed]
- Begam, T.; Nagpal, A.; Singhal, R. A study on copolymeric hydrogels based on acrylamide-methacrylate and its modified vinyl-amine-containing derivative. Designed Monomers and Polymers - DES MONOMERS POLYM 2004, 7, 311–330. [Google Scholar] [CrossRef]
- Dragan, E.S. Advances in interpenetrating polymer network hydrogels and their applications. Pure and Applied Chemistry 2014, 86, 1707–1721. [Google Scholar] [CrossRef]
- Kamath, K.R.; Park, K. Biodegradable hydrogels in drug delivery. Advanced Drug Delivery Reviews 1993, 11, 59–84. [Google Scholar] [CrossRef]
- Imazato, S.; Kitagawa, H.; Tsuboi, R.; Kitagawa, R.; Thongthai, P.; Sasaki, J.I. Non-biodegradable polymer particles for drug delivery: A new technology for "bio-active" restorative materials. Dent Mater J 2017, 36, 524–532. [Google Scholar] [CrossRef] [PubMed]
- Agren, M.S. An amorphous hydrogel enhances epithelialisation of wounds. Acta Derm Venereol 1998, 78, 119–122. [Google Scholar] [CrossRef]
- Okay, O. Semicrystalline physical hydrogels with shape-memory and self-healing properties. Journal of Materials Chemistry B 2019, 7, 1581–1596. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wang, G.; Marquez, M.; Hu, Z. The formation of crystalline hydrogel films by self-crosslinking microgels. Soft Matter 2009, 5, 820–826. [Google Scholar] [CrossRef]
- Burey, P.; Bhandari, B.; Howes, T.; Gidley, M. Hydrocolloid Gel Particles: Formation, Characterization, and Application. Critical reviews in food science and nutrition 2008, 48, 361–377. [Google Scholar] [CrossRef]
- Parhi, R. Cross-Linked Hydrogel for Pharmaceutical Applications: A Review. Adv Pharm Bull 2017, 7, 515–530. [Google Scholar] [CrossRef]
- Ren, T.; Gan, J.; Zhou, L.; Chen, H. Physically Crosslinked Hydrogels Based on Poly (Vinyl Alcohol) and Fish Gelatin for Wound Dressing Application: Fabrication and Characterization. Polymers 2020, 12, 1729. [Google Scholar] [CrossRef]
- Akhtar, M.F.; Hanif, M.; Ranjha, N.M. Methods of synthesis of hydrogels … A review. Saudi Pharmaceutical Journal 2016, 24, 554–559. [Google Scholar] [CrossRef]
- Hoffman, A.S. Conventional and Environmentally-Sensitive Hydrogels for Medical and Industrial Uses: A Review Paper. In Polymer Gels: Fundamentals and Biomedical Applications, DeRossi, D., Kajiwara, K., Osada, Y., Yamauchi, A., Eds.; Springer US: Boston, MA, 1991; pp. 289–297. [Google Scholar]
- Samal, S.K.; Dash, M.; Dubruel, P.; Van Vlierberghe, S. 8 - Smart polymer hydrogels: properties, synthesis and applications. In Smart Polymers and their Applications, Aguilar, M.R., San Román, J., Eds.; Woodhead Publishing: 2014; pp. 237-270.
- Elsayed, M.M. Hydrogel Preparation Technologies: Relevance Kinetics, Thermodynamics and Scaling up Aspects. Journal of Polymers and the Environment 2019, 27, 871–891. [Google Scholar] [CrossRef]
- Weerasundara, L.; Gabriele, B.; Figoli, A.; Ok, Y.S.; Bundschuh, J. Hydrogels: Novel materials for contaminant removal in water—A review. Critical Reviews in Environmental Science and Technology 2020, 51, 1–45. [Google Scholar] [CrossRef]
- Mantha, S.; Pillai, S.; Khayambashi, P.; Upadhyay, A.; Zhang, Y.; Tao, O.; Pham, H.; Tran, S. Smart Hydrogels in Tissue Engineering and Regenerative Medicine. Materials 2019, 12, 3323. [Google Scholar] [CrossRef] [PubMed]
- Raghuwanshi, V.S.; Garnier, G. Characterisation of hydrogels: Linking the nano to the microscale. Advances in Colloid and Interface Science 2019, 274, 102044. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.S.; Islam, M.M.; Islam, M.S.; Zaman, A.; Ahmed, T.; Biswas, S.; Sharmeen, S.; Rashid, T.U.; Rahman, M.M. Morphological Characterization of Hydrogels. In Cellulose-Based Superabsorbent Hydrogels, Mondal, M.I.H., Ed.; Springer International Publishing: Cham, 2019; pp. 819–863. [Google Scholar]
- Kisley, L.; Miller, K.A.; Guin, D.; Kong, X.; Gruebele, M.; Leckband, D.E. Direct Imaging of Protein Stability and Folding Kinetics in Hydrogels. ACS Applied Materials & Interfaces 2017, 9, 21606–21617. [Google Scholar] [CrossRef]
- Barker, E.C.; Goh, C.Y.; Jones, F.; Mocerino, M.; Skelton, B.W.; Becker, T.; Ogden, M.I. Investigating hydrogel formation using in situ variable-temperature scanning probe microscopy. Chemical Science 2015, 6, 6133–6138. [Google Scholar] [CrossRef] [PubMed]
- Marapureddy, S.G.; Thareja, P. Structure and Rheology of Hydrogels: Applications in Drug Delivery. In Biointerface Engineering: Prospects in Medical Diagnostics and Drug Delivery, Chandra, P., Pandey, L.M., Eds.; Springer Singapore: Singapore, 2020; pp. 75–99. [Google Scholar]
- Chen, L.; Raeburn, J.; Sutton, S.; Spiller, D.G.; Williams, J.; Sharp, J.S.; Griffiths, P.C.; Heenan, R.K.; King, S.M.; Paul, A.; et al. Tuneable mechanical properties in low molecular weight gels. Soft Matter 2011, 7, 9721–9727. [Google Scholar] [CrossRef]
- Zheng, L.-Y.; Shi, J.-M.; Chi, Y.-H. Tannic Acid Physically Cross-Linked Responsive Hydrogel. Macromolecular Chemistry and Physics 2018, 219, 1800234. [Google Scholar] [CrossRef]
- Naé, H.N.; Reichert, W.W. Rheological properties of lightly crosslinked carboxy copolymers in aqueous solutions. Rheologica Acta 1992, 31, 351–360. [Google Scholar] [CrossRef]
- Fresno Contreras, M.J.; Ramírez Diéguez, A.; Jiménez Soriano, M.M. Rheological characterization of hydroalcoholic gels--15% ethanol--of Carbopol Ultrez 10. Farmaco 2001, 56, 437–441. [Google Scholar] [CrossRef]
- Barnes, H. A Brief History of the Yield Stress. Applied Rheology 2019, 9, 262–266. [Google Scholar] [CrossRef]
- Islam, M.T.; Rodríguez-Hornedo, N.; Ciotti, S.; Ackermann, C. Rheological characterization of topical carbomer gels neutralized to different pH. Pharm Res 2004, 21, 1192–1199. [Google Scholar] [CrossRef]
- Madhavikutty, A.S.; Ohta, S.; Chandel, A.K.S.; Qi, P.; Ito, T. Analysis of endoscopic injectability and post-ejection dripping of yield stress fluids: Laponite, Carbopol and Xanthan Gum. Journal of Chemical Engineering of Japan 2021, 54, 500–511. [Google Scholar] [CrossRef]
- Kröger, M.; Vermant, J. The Structure and Rheology of Complex Fluids. Applied Rheology 2019, 10, 110–111. [Google Scholar] [CrossRef]
- Nutan, B.; Chandel, A.K.S.; Biswas, A.; Kumar, A.; Yadav, A.; Maiti, P.; Jewrajka, S.K. Gold Nanoparticle Promoted Formation and Biological Properties of Injectable Hydrogels. Biomacromolecules 2020, 21, 3782–3794. [Google Scholar] [CrossRef]
- Kumar, A.; Anuradha; Biswas, A. ; Jewrajka, S.K. Injectable amphiphilic hydrogel systems from the self-assembly of partially alkylated poly(2-dimethyl aminoethyl) methacrylate with inherent antimicrobial property and sustained release behaviour. European Polymer Journal 2022, 179, 111559. [Google Scholar] [CrossRef]
- Nutan, B.; Chandel, A.K.S.; Jewrajka, S.K. Liquid Prepolymer-Based in Situ Formation of Degradable Poly(ethylene glycol)-Linked-Poly(caprolactone)-Linked-Poly(2-dimethylaminoethyl)methacrylate Amphiphilic Conetwork Gels Showing Polarity Driven Gelation and Bioadhesion. ACS Applied Bio Materials 2018, 1, 1606–1619. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Nutan, B.; Jewrajka, S.K. Modulation of Properties through Covalent Bond Induced Formation of Strong Ion Pairing between Polyelectrolytes in Injectable Conetwork Hydrogels. ACS Applied Bio Materials 2021, 4, 3374–3387. [Google Scholar] [CrossRef]
- Singh Chandel, A.K.; Kannan, D.; Nutan, B.; Singh, S.; Jewrajka, S.K. Dually crosslinked injectable hydrogels of poly(ethylene glycol) and poly[(2-dimethylamino)ethyl methacrylate]-b-poly(N-isopropyl acrylamide) as a wound healing promoter. Journal of Materials Chemistry B 2017, 5, 4955–4965. [Google Scholar] [CrossRef] [PubMed]
- Rodell, C.B.; Kaminski, A.L.; Burdick, J.A. Rational design of network properties in guest-host assembled and shear-thinning hyaluronic acid hydrogels. Biomacromolecules 2013, 14, 4125–4134. [Google Scholar] [CrossRef]
- Shin, M.; Lee, H. Gallol-Rich Hyaluronic Acid Hydrogels: Shear-Thinning, Protein Accumulation against Concentration Gradients, and Degradation-Resistant Properties. Chemistry of Materials 2017, 29. [Google Scholar] [CrossRef]
- Salome Amarachi, C.; Attama, A.; Kenechukwu, F. Nanoemulsions — Advances in Formulation, Characterization and Applications in Drug Delivery. 2014.
- Pek, Y.S.; Wan, A.C.; Shekaran, A.; Zhuo, L.; Ying, J.Y. A thixotropic nanocomposite gel for three-dimensional cell culture. Nat Nanotechnol 2008, 3, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Szymańska, E.; Orłowski, P.; Winnicka, K.; Tomaszewska, E.; Bąska, P.; Celichowski, G.; Grobelny, J.; Basa, A.; Krzyżowska, M. Multifunctional Tannic Acid/Silver Nanoparticle-Based Mucoadhesive Hydrogel for Improved Local Treatment of HSV Infection: In Vitro and In Vivo Studies. Int J Mol Sci 2018, 19. [Google Scholar] [CrossRef] [PubMed]
- Sreedevi Madhavikutty, A.; Singh Chandel, A.K.; Tsai, C.C.; Inagaki, N.F.; Ohta, S.; Ito, T. pH responsive cationic guar gum-borate self-healing hydrogels for muco-adhesion. Sci Technol Adv Mater 2023, 24, 2175586. [Google Scholar] [CrossRef] [PubMed]
- Baus, R.A.; Zahir-Jouzdani, F.; Dünnhaupt, S.; Atyabi, F.; Bernkop-Schnürch, A. Mucoadhesive hydrogels for buccal drug delivery: In vitro-in vivo correlation study. Eur J Pharm Biopharm 2019, 142, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Tavakoli, S.; Klar, A.S. Advanced Hydrogels as Wound Dressings. Biomolecules 2020, 10, 1169. [Google Scholar] [CrossRef]
- Bera, A.; Singh Chandel, A.K.; Uday Kumar, C.; Jewrajka, S.K. Degradable/cytocompatible and pH responsive amphiphilic conetwork gels based on agarose-graft copolymers and polycaprolactone. Journal of Materials Chemistry B 2015, 3, 8548–8557. [Google Scholar] [CrossRef]
- Chandel, A.K.S.; Nutan, B.; Raval, I.H.; Jewrajka, S.K. Self-Assembly of Partially Alkylated Dextran-graft-poly[(2-dimethylamino)ethyl methacrylate] Copolymer Facilitating Hydrophobic/Hydrophilic Drug Delivery and Improving Conetwork Hydrogel Properties. Biomacromolecules 2018, 19, 1142–1153. [Google Scholar] [CrossRef]
- Nutan, B.; Chandel, A.K.S.; Bhalani, D.V.; Jewrajka, S.K. Synthesis and tailoring the degradation of multi-responsive amphiphilic conetwork gels and hydrogels of poly (β-amino ester) and poly (amido amine). Polymer 2017, 111, 265–274. [Google Scholar] [CrossRef]
- Caló, E.; Khutoryanskiy, V.V. Biomedical applications of hydrogels: A review of patents and commercial products. European Polymer Journal 2015, 65, 252–267. [Google Scholar] [CrossRef]
- Shoukat, H.; Buksh, K.; Noreen, S.; Pervaiz, F.; Maqbool, I. Hydrogels as potential drug-delivery systems: network design and applications. Therapeutic Delivery 2021, 12, 375–396. [Google Scholar] [CrossRef]
- Bhalani, D.V.; Nutan, B.; Kumar, A.; Singh Chandel, A.K. Bioavailability Enhancement Techniques for Poorly Aqueous Soluble Drugs and Therapeutics. Biomedicines 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Mantha, S.; Pillai, S.; Khayambashi, P.; Upadhyay, A.; Zhang, Y.; Tao, O.; Pham, H.M.; Tran, S.D. Smart Hydrogels in Tissue Engineering and Regenerative Medicine. Materials (Basel) 2019, 12, 3323. [Google Scholar] [CrossRef] [PubMed]
- Michalek, J.; Hobzova, R.; Pradny, M.; Duskova-Smrckova, M. Hydrogels Contact Lenses. 2010; pp. 303-315.
- Herrmann, A.; Haag, R.; Schedler, U. Hydrogels and Their Role in Biosensing Applications. Advanced Healthcare Materials 2021, 10, 2100062. [Google Scholar] [CrossRef] [PubMed]
- Shrivastav, A.M.; Cvelbar, U.; Abdulhalim, I. A comprehensive review on plasmonic-based biosensors used in viral diagnostics. Communications Biology 2021, 4, 70. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Marchant, R.E. Design properties of hydrogel tissue-engineering scaffolds. Expert Rev Med Devices 2011, 8, 607–626. [Google Scholar] [CrossRef]
- Li, J.; Wu, C.; Chu, P.K.; Gelinsky, M. 3D printing of hydrogels: Rational design strategies and emerging biomedical applications. Materials Science and Engineering: R: Reports 2020, 140, 100543. [Google Scholar] [CrossRef]
- Billiet, T.; Vandenhaute, M.; Schelfhout, J.; Van Vlierberghe, S.; Dubruel, P. A review of trends and limitations in hydrogel-rapid prototyping for tissue engineering. Biomaterials 2012, 33, 6020–6041. [Google Scholar] [CrossRef] [PubMed]
- Yazici, I.; Okay, O. Spatial inhomogeneity in poly(acrylic acid) hydrogels. Polymer 2005, 46, 2595–2602. [Google Scholar] [CrossRef]
- Ikada, Y. Challenges in tissue engineering. J R Soc Interface 2006, 3, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Galante, R.; Pinto, T.; Colaço, R.; Serro, A. Sterilization of hydrogels for biomedical applications: A review: STERILIZATION OF HYDROGELS. Journal of Biomedical Materials Research Part B: Applied Biomaterials 2017, 106. [Google Scholar] [CrossRef]
- Paidikondala, M.; Wang, S.; Hilborn, J.; Larsson, S.; Varghese, O.P. Impact of Hydrogel Cross-Linking Chemistry on the in Vitro and in Vivo Bioactivity of Recombinant Human Bone Morphogenetic Protein-2. ACS Applied Bio Materials 2019, 2, 2006–2012. [Google Scholar] [CrossRef]
- Wang, K.; Hao, Y.; Wang, Y.; Chen, J.; Mao, L.; Deng, Y.; Chen, J.; Yuan, S.; Zhang, T.; Ren, J.; et al. Functional Hydrogels and Their Application in Drug Delivery, Biosensors, and Tissue Engineering. International Journal of Polymer Science 2019, 2019, 1–14. [Google Scholar] [CrossRef]
- Kumar, A.; Nutan, B.; Jewrajka, S.K. Stability and acidic pH-mediated leakage of guest molecules from self-assembly of poly(amidoamine)-graft-alkyl copolymers. Polymer 2019, 183, 121894. [Google Scholar] [CrossRef]
- Madl, C.M.; Heilshorn, S.C.; Blau, H.M. Bioengineering strategies to accelerate stem cell therapeutics. Nature 2018, 557, 335–342. [Google Scholar] [CrossRef]
- Nutan, B.; Kumar, A.; Jewrajka, S.K. Library of Derivatizable Multiblock Copolymers by Nucleophilic Substitution Polymerization and Targeting Specific Properties. Biomacromolecules 2020, 21, 5029–5043. [Google Scholar] [CrossRef]
- Bellofatto, K.; Lebreton, F.; Wassmer, C.-H.; Hasany, M.; Hanna, R.; Khatri, R.; Mar Fonseca, L.d.; Perez, L.; Cottet-Dumoulin, D.; Parnaud, G.; et al. 402.7: Homemade Hydrogel From Human Amniotic Membrane Improves Islet Transplantation Outcomes in Diabetic Immunodeficient Mice. Transplantation 2021, 105. [Google Scholar] [CrossRef]
- Mantha, S.; Pillai, S.; Khayambashi, P.; Upadhyay, A.; Zhang, Y.; Tao, O.; Pham, H.M.; Tran, S.D. Smart Hydrogels in Tissue Engineering and Regenerative Medicine. Materials (Basel) 2019, 12. [Google Scholar] [CrossRef] [PubMed]
- Chandel, A.K.S.; Shimizu, A.; Hasegawa, K.; Ito, T. Advancement of Biomaterial-Based Postoperative Adhesion Barriers. Macromolecular Bioscience 2021, 21, 2000395. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, S.; Okada, R.; Kushioka, J.; Kodama, J.; Tsukazaki, H.; Bal, Z.; Tateiwa, D.; Ukon, Y.; Hirai, H.; Makino, T.; et al. Effects of rhBMP-2-loaded hydroxyapatite granules/beta-tricalcium phosphate hydrogel (HA/β-TCP/hydrogel) composite on a rat model of caudal intervertebral fusion. Scientific Reports 2022, 12, 7906. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



