Submitted:
09 June 2023
Posted:
12 June 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Angiogenesis in Tissue Repair
2.1. Overview of Angiogenic Processes
2.2. Functional Mechanism of Angiogenesis
2.3. Angiogenesis in Acute and Chronic Tissue Repair
3. Role and Interactions of the Extracellular Matrix in Angiogenesis
4. Analytical Background PRP Technology
4.1. PRP in Tissue Repair Mechanisms
4.2. PRP Device Variables and Considerations
4.3. Transitioning from Platelet Concentration to Platelet Dosing
4.4. Gravitational Cellular Density Separation and Platelet Dose
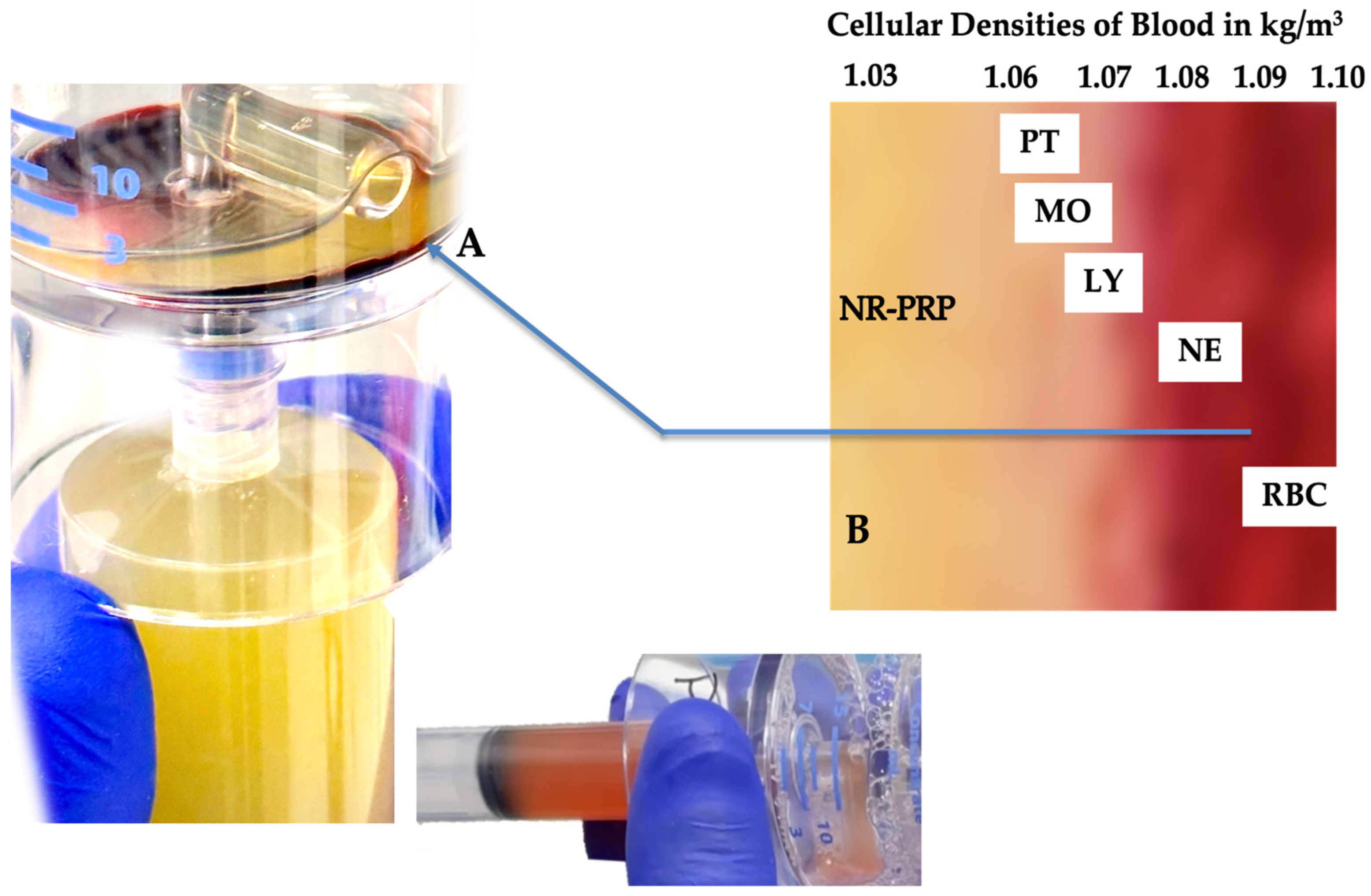
5. Clinical PRP: A Myriad of Angiogenic Stimulators and Actors
5.1. Platelet Dosing and Bioformulation are Important Factors in Angiogenesis Regulation
5.2. Platelets
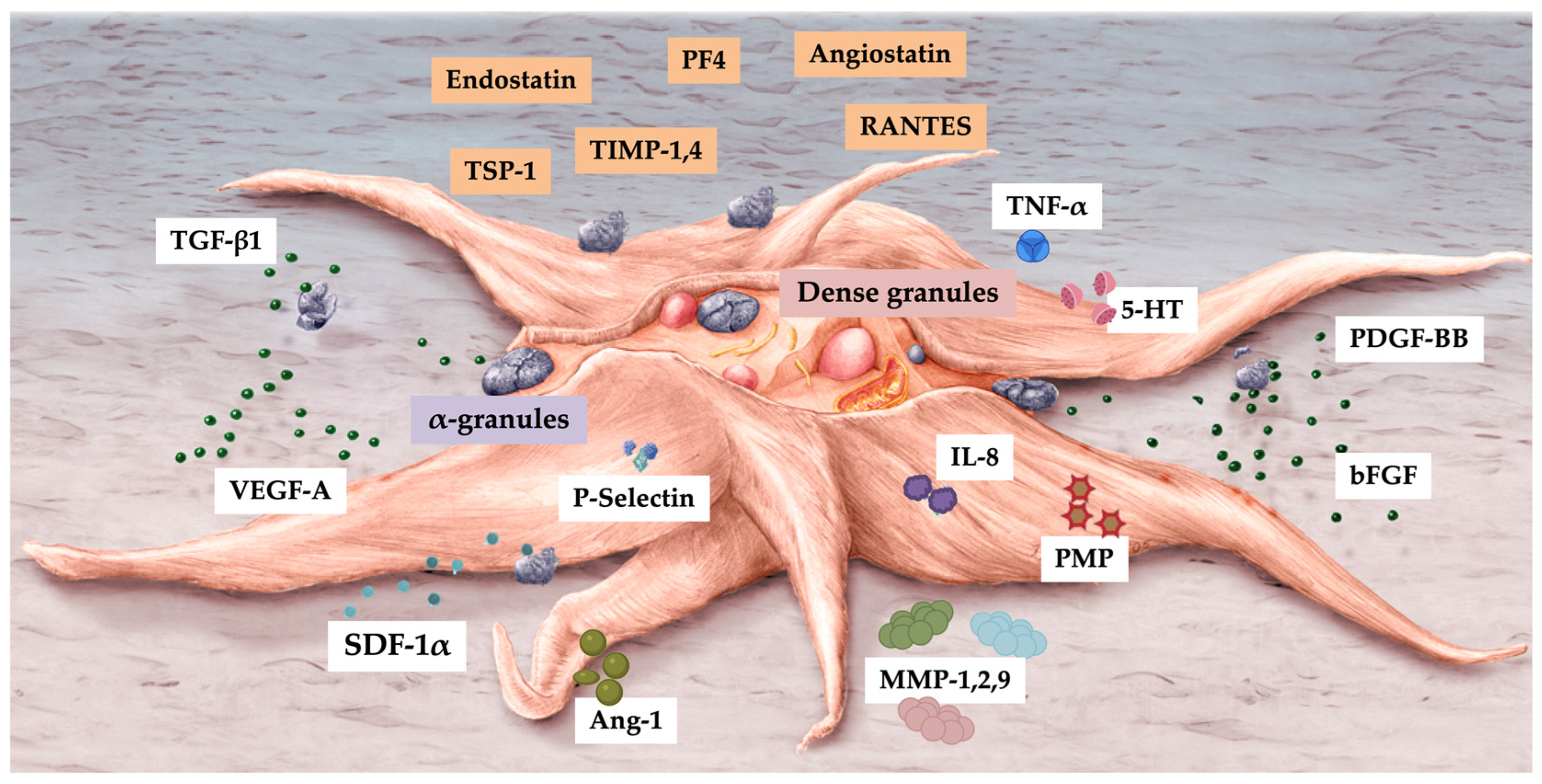
5.3. Platelet α-Granules
5.4. Pro-Angiogenic Platelet Factors
5.4.1. VEGF
5.4.2. Platelet-Derived Growth Factor-BB (PDGF-BB)
5.4.3. Transforming Growth Factor-Beta (TGF-β)
5.4.4. Basic Fibroblast Growth Factor (bFGF)
5.4.5. Angiopoietins (Ang)
5.4.6. Platelet Microparticles (PMPs)
5.4.7. Serotonin (5-HT).
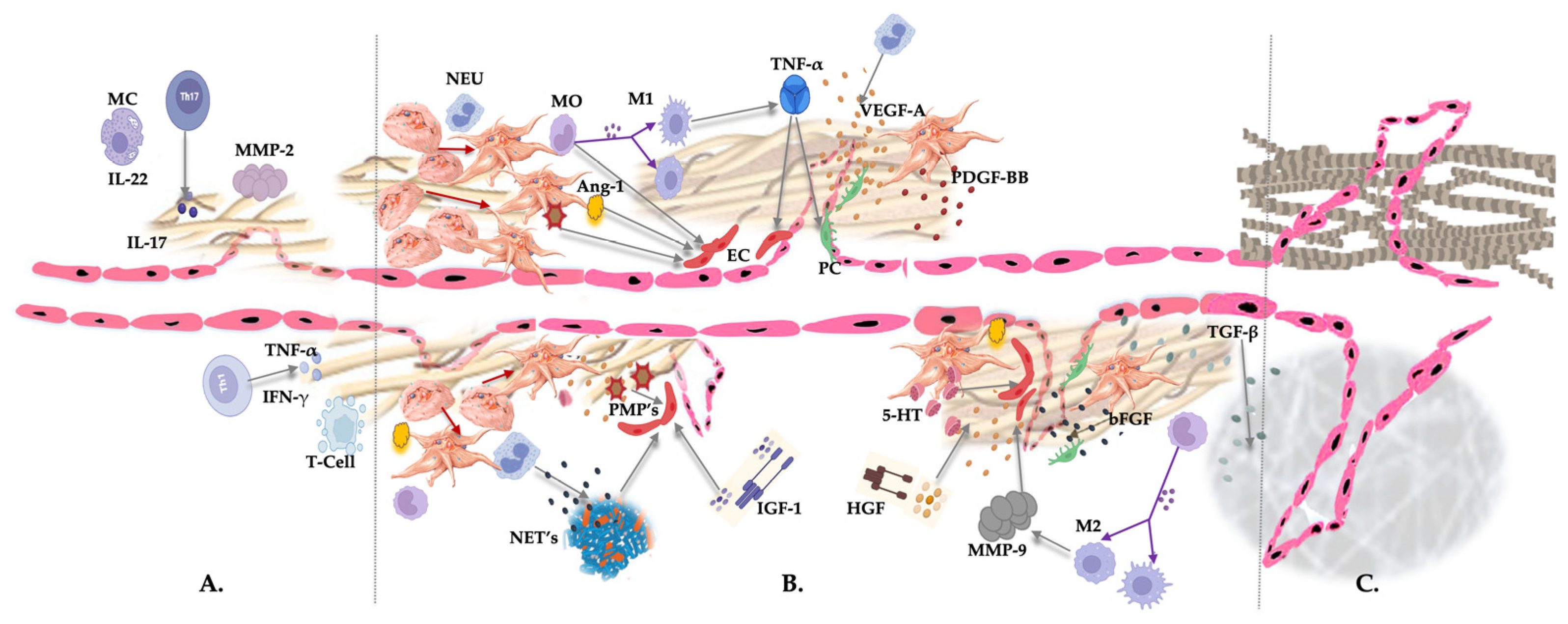
5.5. Pro-Angiogenic Leukocytes
5.5.1. Neutrophils
5.2.2. Monocytes, Macrophages, and TNF-α
5.6. Pro-Angiogenic Plasma-Based Growth Factors
5.6.1. Insulin-Like Growth Factor (IGF-1)
5.6.2. HGF
5.7. In Vitro and Animal Studies
5.7. PRP Classification
5.7.1. Leukocyte-Poor PRP Angiogenic Potential
5.7.2. Leukocyte-Rich PRP Angiogenic Potential
5.7.3. PLs
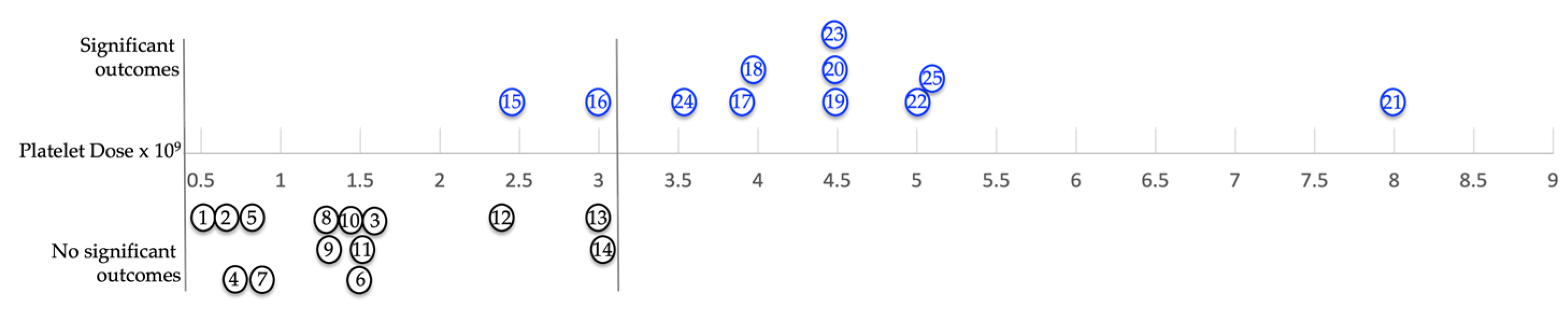
6. The Angiogenic Potential of PRP is Contingent on Platelet Dose and Bioformulation.
| Study Identifier |
PRP Application |
PRP Dose x 109 platelets |
Bioformulation LP / LR |
Reference |
|---|---|---|---|---|
| Non-significant outcomes | ||||
| 1 | Rotator cuff repair | 0.55 | LP | [158] |
| 2 | Lateral epicondylitis | 0.60 | LP | [159] |
| 3 | Rotator cuff repair | 1,518 | LP | [160] |
| 4 | Patella tendinopathy | 0.663 | LP | [158] |
| 5 | Hamstring tendinopathy | 0.750 | LP | [161] |
| 6 | Rotator cuff repair | 0.701 | LP | [162] |
| 7 | Achilles tendinopathy | 0.875 | LP | [163] |
| 8 | Shoulder soft tissue | 1,312 | LR | [164] |
| 9 | Achilles tendinopathy | 1,313 | LP | [165] |
| 10 | Achilles tendinopathy | 1,462 | LR | [166] |
| 11 | Rotator cuff repair | 1,575 | LP | [167] |
| 12 | Achilles tendinopathy | 2,430 | LR | [168] |
| 13 | Lateral epicondylitis | 3,037 | LR | [169] |
| 14 | Achilles tendinopathy | 3,125 | LR | [170] |
| Significant outcomes | ||||
| 15 | Lateral epicondylitis | 2,454 | LR | [171] |
| 16 | Rotator cuff repair | 3,000 | LR | [172] |
| 17 | Lateral epicondylitis | 3,877 | LR | [173] |
| 18 | Ulnar collateral ligament | 3,900 | LR | [174] |
| 19 | Lateral epicondylitis | 4,500 | LR | [175] |
| 20 | Lateral epicondylitis | 4,500 | LR | [176] |
| 21 | Rotator cuff surgical repair | 8,100 | LR | [177] |
| 22 | Patellar tendinopathy | 5,100 | LR | [178] |
| 23 | ACL repair | 4,500 | LR | [179] |
| 24 | Plantar fasciitis | 3,500 | LP | [180] |
| 25 | Rotator cuff repair | 5.275 | LR | [181] |
7. Discussion
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Conflict of Interest
References
- Everts, P.A.; Flanagan, G.; Podesta, L. Autologous Orthobiologics. In Clinical Guide to Musculoskeletal Medicine: A Multidisciplinary Approach; Mostoufi, S.A., George, T.K., Tria Jr., A.J., Eds.; Springer International Publishing: Cham, 2022; pp. 651–679 ISBN 978-3-030-92042-5.
- Fadadu, P.P.; Mazzola, A.J.; Hunter, C.W.; Davis, T.T. Review of Concentration Yields in Commercially Available Platelet-Rich Plasma (PRP) Systems: A Call for PRP Standardization. Reg. Anesth. Pain Med. 2019, 44, 652–659. [CrossRef]
- Everts, P.; Onishi, K.; Jayaram, P.; Lana, J.F.; Mautner, and K. Platelet-Rich Plasma: New Performance Understandings and Therapeutic Considerations in 2020; Int. J. Mol. Sci. 2020, 21, 7794. [CrossRef]
- Magalon, J.; Brandin, T.; Francois, P.; Degioanni, C.; De Maria, L.; Grimaud, F.; Veran, J.; Dignat-George, F.; Sabatier, F. Technical and Biological Review of Authorized Medical Devices for Platelets-Rich Plasma Preparation in the Field of Regenerative Medicine. Platelets 2021, 32, 200–208. [CrossRef]
- Gentile, P.; Garcovich, S. Systematic Review—The Potential Implications of Different Platelet-Rich Plasma (PRP) Concentrations in Regenerative Medicine for Tissue Repair. Int. J. Mol. Sci. 2020, 21, 5702. [CrossRef]
- Jain, D.; Goyal, T.; Verma, N.; Paswan, A.K.; Dubey, R.K. Intradiscal Platelet-Rich Plasma Injection for Discogenic Low Back Pain and Correlation with Platelet Concentration: A Prospective Clinical Trial. Pain Med. 2020, 21, 2719–2725. [CrossRef]
- Giusti, I.; D’Ascenzo, S.; Mancò, A.; Di Stefano, G.; Di Francesco, M.; Rughetti, A.; Dal Mas, A.; Properzi, G.; Calvisi, V.; Dolo, V. Platelet Concentration in Platelet-Rich Plasma Affects Tenocyte Behavior In Vitro. BioMed Res. Int. 2014, 2014, 1–12. [CrossRef]
- Li, J.; Zhang, Y.-P.; Kirsner, R.S. Angiogenesis in Wound Repair: Angiogenic Growth Factors and the Extracellular Matrix. Microsc. Res. Tech. 2003, 60, 107–114. [CrossRef]
- Bae, H.; Puranik, A.S.; Gauvin, R.; Edalat, F.; Carrillo-Conde, B.; Peppas, N.A.; Khademhosseini, A. Building Vascular Networks. Sci. Transl. Med. 2012, 4. [CrossRef]
- Guo, S.; DiPietro, L.A. Factors Affecting Wound Healing. J. Dent. Res. 2010, 89, 219–229. [CrossRef]
- Martínez, C.E.; Smith, P.C.; Palma Alvarado, V.A. The Influence of Platelet-Derived Products on Angiogenesis and Tissue Repair: A Concise Update. Front. Physiol. 2015, 6. [CrossRef]
- Everts, P.A.; Sadeghi, P.; Smith, D.R. Basic Science of Autologous Orthobiologics. Phys. Med. Rehabil. Clin. N. Am. 2023, 34, 1–23. [CrossRef]
- Oklu, R.; Walker, T.G.; Wicky, S.; Hesketh, R. Angiogenesis and Current Antiangiogenic Strategies for the Treatment of Cancer. J. Vasc. Interv. Radiol. JVIR 2010, 21, 1791—805; quiz 1806. [CrossRef]
- Li, W.W.; Talcott, K.E.; Zhai, A.W.; Kruger, E.A.; Li, V.W. The Role of Therapeutic Angiogenesis in Tissue Repair and Regeneration. Adv. Skin Wound Care 2005, 18, 491–500. [CrossRef]
- Italiano, J.E.; Richardson, J.L.; Patel-Hett, S.; Battinelli, E.; Zaslavsky, A.; Short, S.; Ryeom, S.; Folkman, J.; Klement, G.L. Angiogenesis Is Regulated by a Novel Mechanism: Pro- and Antiangiogenic Proteins Are Organized into Separate Platelet α Granules and Differentially Released. Blood 2008, 111, 1227–1233. [CrossRef]
- Ucuzian, A.A.; Gassman, A.A.; East, A.T.; Greisler, H.P. Molecular Mediators of Angiogenesis: J. Burn Care Res. 2010, 31, 158–175. [CrossRef]
- Dimmeler, S.; Zeiher, A.M. Endothelial Cell Apoptosis in Angiogenesis and Vessel Regression. [CrossRef]
- Tonnesen, M.; Feng, X.; Clark, R. Angiogenesis in Wound Healing. J. Investig. Dermatol. Symp. Proc. 2000, 5, 40—46. [CrossRef]
- Mohammadi, S.; Nasiri, S.; Mohammadi, M.H.; Malek Mohammadi, A.; Nikbakht, M.; Zahed Panah, M.; Safar, H.; Mostafaei, S.; Norooznezhad, A.H.; Soroosh, A.R.; et al. Evaluation of Platelet-Rich Plasma Gel Potential in Acceleration of Wound Healing Duration in Patients Underwent Pilonidal Sinus Surgery: A Randomized Controlled Parallel Clinical Trial. Transfus. Apher. Sci. 2017, 56, 226–232. [CrossRef]
- Honnegowda, T.M.; Kumar, P.; Udupa, E.G.P.; Kumar, S.; Kumar, U.; Rao, P. Role of Angiogenesis and Angiogenic Factors in Acute and Chronic Wound Healing. 2015, 2, 7. [CrossRef]
- Folkman, J. Clinical Applications of Research on Angiogenesis. N. Engl. J. Med. 1995, 333, 1757–1763. [CrossRef]
- Raziyeva, K.; Kim, Y.; Zharkinbekov, Z.; Kassymbek, K.; Jimi, S.; Saparov, A. Immunology of Acute and Chronic Wound Healing. Biomolecules 2021, 11, 700. [CrossRef]
- Dean, B.J.F.; Gettings, P.; Dakin, S.G.; Carr, A.J. Are Inflammatory Cells Increased in Painful Human Tendinopathy? A Systematic Review. Br. J. Sports Med. 2016, 50, 216–220. [CrossRef]
- Frey, C.; Shereff, M.; Greenidge, N. Vascularity of the Posterior Tibial Tendon. JBJS 1990, 72, 884–888.
- Everts, P.A.; Mazzola, T.; Mautner, K.; Randelli, P.S.; Podesta, L. Modifying Orthobiological PRP Therapies Are Imperative for the Advancement of Treatment Outcomes in Musculoskeletal Pathologies. Biomedicines 2022, 10, 2933. [CrossRef]
- Nishio, H.; Saita, Y.; Kobayashi, Y.; Takaku, T.; Fukusato, S.; Uchino, S.; Wakayama, T.; Ikeda, H.; Kaneko, K. Platelet-Rich Plasma Promotes Recruitment of Macrophages in the Process of Tendon Healing. Regen. Ther. 2020, 14, 262–270. [CrossRef]
- Schulze-Tanzil, G.G.; Cáceres, M.D.; Stange, R.; Wildemann, B.; Docheva, D. Tendon Healing: A Concise Review on Cellular and Molecular Mechanisms with a Particular Focus on the Achilles Tendon. Bone Jt. Res. 2022, 11, 561–574. [CrossRef]
- Clements, J.R. Achilles Tendon Injuries. In Tendon and Ligament Injuries of the Foot and Ankle: An Evidence-Based Approach; Springer, 2022; pp. 265–280.
- Factor, D.; Dale, B. Current Concepts of Rotator Cuff Tendinopathy. Int. J. Sports Phys. Ther. 2014, 9, 274.
- Tempfer, H.; Traweger, A. Tendon Vasculature in Health and Disease. Front. Physiol. 2015, 6, 330. [CrossRef]
- Hynes, R.O. The Extracellular Matrix: Not Just Pretty Fibrils. Science 2009, 326, 1216–1219. [CrossRef]
- Martino, M.M.; Brkic, S.; Bovo, E.; Burger, M.; Schaefer, D.J.; Wolff, T.; Gürke, L.; Briquez, P.S.; Larsson, H.M.; Gianni-Barrera, R.; et al. Extracellular Matrix and Growth Factor Engineering for Controlled Angiogenesis in Regenerative Medicine. Front. Bioeng. Biotechnol. 2015, 3. [CrossRef]
- Bauer, A.L.; Jackson, T.L.; Jiang, Y. Topography of Extracellular Matrix Mediates Vascular Morphogenesis and Migration Speeds in Angiogenesis. PLoS Comput. Biol. 2009, 5, e1000445. [CrossRef]
- Héroult, M.; Reiss, Y.; Augustin, H.G. Angiogenesis and Vascular Morphogenesis. In Encyclopedia of Molecular Pharmacology; Offermanns, S., Rosenthal, W., Eds.; Springer Berlin Heidelberg: Berlin, Heidelberg, 2008; pp. 80–88 ISBN 978-3-540-38918-7.
- Arroyo, A.G.; Iruela-Arispe, M.L. Extracellular Matrix, Inflammation, and the Angiogenic Response. Cardiovasc. Res. 2010, 86, 226–235. [CrossRef]
- Lee, S.; Jilani, S.M.; Nikolova, G.V.; Carpizo, D.; Iruela-Arispe, M.L. Processing of VEGF-A by Matrix Metalloproteinases Regulates Bioavailability and Vascular Patterning in Tumors. J. Cell Biol. 2005, 169, 681–691. [CrossRef]
- Chen, T.T.; Luque, A.; Lee, S.; Anderson, S.M.; Segura, T.; Iruela-Arispe, M.L. Anchorage of VEGF to the Extracellular Matrix Conveys Differential Signaling Responses to Endothelial Cells. J. Cell Biol. 2010, 188, 595–609. [CrossRef]
- Everts, P.A.; Devilee, R.J.J.; Brown Mahoney, C.; van Erp, A.; Oosterbos, C.J.M.; Stellenboom, M.; Knape, J.T.A.; van Zundert, A. Exogenous Application of Platelet-Leukocyte Gel during Open Subacromial Decompression Contributes to Improved Patient Outcome. Eur. Surg. Res. 2008, 40, 203–210. [CrossRef]
- Andia, I.; Maffulli, N. A Contemporary View of Platelet-Rich Plasma Therapies: Moving toward Refined Clinical Protocols and Precise Indications. Regen. Med. 2018, 13, 717–728. [CrossRef]
- Puzzitiello, R.N.; Patel, B.H.; Forlenza, E.M.; Nwachukwu, B.U.; Allen, A.A.; Forsythe, B.; Salzler, M.J. Adverse Impact of Corticosteroids on Rotator Cuff Tendon Health and Repair: A Systematic Review of Basic Science Studies. Arthrosc. Sports Med. Rehabil. 2020, 2, e161–e169. [CrossRef]
- Filardo, G.; Di Matteo, B.; Kon, E.; Merli, G.; Marcacci, M. Platelet-Rich Plasma in Tendon-Related Disorders: Results and Indications. Knee Surg. Sports Traumatol. Arthrosc. 2018, 26, 1984–1999. [CrossRef]
- Belk, J.W.; Kraeutler, M.J.; Houck, D.A.; Goodrich, J.A.; Dragoo, J.L.; McCarty, E.C. Platelet-Rich Plasma Versus Hyaluronic Acid for Knee Osteoarthritis: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Am. J. Sports Med. 2020, 036354652090939. [CrossRef]
- Xuan, Z.; Yu, W.; Dou, Y.; Wang, T. Efficacy of Platelet-Rich Plasma for Low Back Pain: A Systematic Review and Meta-Analysis. J. Neurol. Surg. Part Cent. Eur. Neurosurg. 2020. [CrossRef]
- Miroshnychenko, O.; Chalkley, R.J.; Leib, R.D.; Everts, P.A.; Dragoo, J.L. Proteomic Analysis of Platelet-Rich and Platelet-Poor Plasma. Regen. Ther. 2020, 15, 226–235. [CrossRef]
- Hersant, B.; Sid-Ahmed, M.; Braud, L.; Jourdan, M.; Baba-Amer, Y.; Meningaud, J.-P.; Rodriguez, A.-M. Platelet-Rich Plasma Improves the Wound Healing Potential of Mesenchymal Stem Cells through Paracrine and Metabolism Alterations. Stem Cells Int. 2019, 2019, 1–14. [CrossRef]
- Mammoto, T.; Jiang, A.; Jiang, E.; Mammoto, A. Platelet-Rich Plasma Extract Prevents Pulmonary Edema through Angiopoietin-Tie2 Signaling. Am. J. Respir. Cell Mol. Biol. 2015, 52, 56–64. [CrossRef]
- Berndt, S.; Carpentier, G.; Turzi, A.; Borlat, F.; Cuendet, M.; Modarressi, A. Angiogenesis Is Differentially Modulated by Platelet-Derived Products. Biomedicines 2021, 9, 251. [CrossRef]
- Cherian, C.; Malanga, G.; Mautner, K. OPTIMIZING PLATELET-RICH PLASMA (PRP) INJECTIONS: A NARRATIVPEroceedings. 2, 17. [CrossRef]
- Beitzel, K.; Allen, D.; Apostolakos, J.; Russell, R.; McCarthy, M.; Gallo, G.; Cote, M.; Mazzocca, A. US Definitions, Current Use, and FDA Stance on Use of Platelet-Rich Plasma in Sports Medicine. J. Knee Surg. 2014, 28, 029–034. [CrossRef]
- Magalon, J.; Chateau, A.L.; Bertrand, B.; Louis, M.L.; Silvestre, A.; Giraudo, L.; Veran, J.; Sabatier, F. DEPA Classification: A Proposal for Standardising PRP Use and a Retrospective Application of Available Devices. BMJ Open Sport Exerc. Med. 2016, 2, e000060. [CrossRef]
- Lin, M.-T.; Wei, K.-C.; Wu, C.-H. Effectiveness of Platelet-Rich Plasma Injection in Rotator Cuff Tendinopathy: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Diagnostics 2020, 10, 189. [CrossRef]
- Lutz, C.; Cheng, J.; Prysak, M.; Zukofsky, T.; Rothman, R.; Lutz, G. Clinical Outcomes Following Intradiscal Injections of Higher-Concentration Platelet-Rich Plasma in Patients with Chronic Lumbar Discogenic Pain. Int. Orthop. 2022. [CrossRef]
- Piao, L.; Park, H.; Jo, C.H. Theoretical Prediction and Validation of Cell Recovery Rates in Preparing Platelet-Rich Plasma through a Centrifugation. PLOS ONE 2017, 12, e0187509. [CrossRef]
- Verhaegen, F.; Brys, P.; Debeer, P. Rotator Cuff Healing after Needling of a Calcific Deposit Using Platelet-Rich Plasma Augmentation: A Randomized, Prospective Clinical Trial. J. Shoulder Elbow Surg. 2016, 25, 169—173. [CrossRef]
- Fu, C.-J.; Sun, J.-B.; Bi, Z.-G.; Wang, X.-M.; Yang, C.-L. Evaluation of Platelet-Rich Plasma and Fibrin Matrix to Assist in Healing and Repair of Rotator Cuff Injuries: A Systematic Review and Meta-Analysis. Clin. Rehabil. 2017, 31, 158–172. [CrossRef]
- Nguyen, P.A.; Pham, T.A.V. Effects of Platelet-Rich Plasma on Human Gingival Fibroblast Proliferation and Migration in Vitro. J. Appl. Oral Sci. 2018, 26. [CrossRef]
- Miron, R.J.; Chai, J.; Fujioka-Kobayashi, M.; Sculean, A.; Zhang, Y. Evaluation of 24 Protocols for the Production of Platelet-Rich Fibrin. BMC Oral Health 2020, 20, 310. [CrossRef]
- Carmeliet, P. Mechanisms of Angiogenesis and Arteriogenesis. Nat. Med. 2000, 6, 389–395. [CrossRef]
- Etulain, J.; Mena, H.A.; Meiss, R.P.; Frechtel, G.; Gutt, S.; Negrotto, S.; Schattner, M. An Optimised Protocol for Platelet-Rich Plasma Preparation to Improve Its Angiogenic and Regenerative Properties. Sci. Rep. 2018, 8, 1513. [CrossRef]
- Browder, T.; Folkman, J.; Pirie-Shepherd, S. The Hemostatic System as a Regulator of Angiogenesis. J. Biol. Chem. 2000, 275, 1521—1524. [CrossRef]
- Eisinger, F.; Patzelt, J.; Langer, H.F. The Platelet Response to Tissue Injury. Front. Med. 2018, 5, 317. [CrossRef]
- Collins, T.; Alexander, D.; Barkatali, B. Platelet-Rich Plasma: A Narrative Review. EFORT Open Rev. 2021, 6, 225–235. [CrossRef]
- Battinelli, E.M.; Markens, B.A.; Italiano, J.E. Release of Angiogenesis Regulatory Proteins from Platelet Alpha Granules: Modulation of Physiologic and Pathologic Angiogenesis. Blood 2011, 118, 1359–1369. [CrossRef]
- Giusti, I.; Rughetti, A.; D’Ascenzo, S.; Millimaggi, D.; Pavan, A.; Dell’Orso, L.; Dolo, V. Identification of an Optimal Concentration of Platelet Gel for Promoting Angiogenesis in Human Endothelial Cells. Transfusion (Paris) 2009, 49, 771–778. [CrossRef]
- Mackman, N. Role of Tissue Factor in Hemostasis, Thrombosis, and Vascular Development. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1015–1022. [CrossRef]
- Huang, J.; Swieringa, F.; Solari, F.A.; Provenzale, I.; Grassi, L.; De Simone, I.; Baaten, C.C.F.M.J.; Cavill, R.; Sickmann, A.; Frontini, M.; et al. Assessment of a Complete and Classified Platelet Proteome from Genome-Wide Transcripts of Human Platelets and Megakaryocytes Covering Platelet Functions. Sci. Rep. 2021, 11, 12358. [CrossRef]
- Blair, P. Platelet α–Granules: Basic Biology and Clinical Correlates. 2010, 29. [CrossRef]
- Kisucka, J.; Butterfield, C.E.; Duda, D.G.; Eichenberger, S.C.; Saffaripour, S.; Ware, J.; Ruggeri, Z.M.; Jain, R.K.; Folkman, J.; Wagner, D.D. Platelets and Platelet Adhesion Support Angiogenesis While Preventing Excessive Hemorrhage. Proc. Natl. Acad. Sci. 2006, 103, 855–860. [CrossRef]
- Baumgartner, H.R.; Muggli, R.; Tschopp, T.B.; Turitto, V.T. Platelet Adhesion, Release and Aggregation in Flowing Blood: Effects of Surface Properties and Platelet Function. Thromb. Haemost. 1976, 35, 124–138.
- Brill, A. Differential Role of Platelet Granular Mediators in Angiogenesis. Cardiovasc. Res. 2004, 63, 226–235. [CrossRef]
- Shibuya, M. Vascular Endothelial Growth Factor (VEGF) and Its Receptor (VEGFR) Signaling in Angiogenesis: A Crucial Target for Anti- and Pro-Angiogenic Therapies. Genes Cancer 2011, 2, 1097–1105. [CrossRef]
- Everts, P.A.M.; Hoffmann, J.; Weibrich, G.; Mahoney, C.B.; Schönberger, J.P.A.M.; van Zundert, A.; Knape, J.T.A. Differences in Platelet Growth Factor Release and Leucocyte Kinetics during Autologous Platelet Gel Formation. Transfus. Med. 2006, 16, 363–368. [CrossRef]
- Kushida, S.; Kakudo, N.; Morimoto, N.; Hara, T.; Ogawa, T.; Mitsui, T.; Kusumoto, K. Platelet and Growth Factor Concentrations in Activated Platelet-Rich Plasma: A Comparison of Seven Commercial Separation Systems. J. Artif. Organs 2014, 17, 186–192. [CrossRef]
- Baria, M.; Vasileff, W.K.; Miller, M.; Borchers, J.; Flanigan, D.C.; Durgam, S.S. Cellular Components and Growth Factor Content of Platelet-Rich Plasma With a Customizable Commercial System. Am. J. Sports Med. 2019, 47, 1216–1222. [CrossRef]
- Chen, K.; Kolls, J.K. T Cell–Mediated Host Immune Defenses in the Lung. Annu. Rev. Immunol. 2013, 31, 605–633. [CrossRef]
- White, S.J.; Chong, J.J.H. Growth Factor Therapy for Cardiac Repair: An Overview of Recent Advances and Future Directions. Biophys. Rev. 2020, 12, 805–815. [CrossRef]
- Edelberg, J.M.; Lee, S.H.; Kaur, M.; Tang, L.; Feirt, N.M.; McCabe, S.; Bramwell, O.; Wong, S.C.; Hong, M.K. Platelet-Derived Growth Factor-AB Limits the Extent of Myocardial Infarction in a Rat Model: Feasibility of Restoring Impaired Angiogenic Capacity in the Aging Heart. Circulation 2002, 105, 608—613. [CrossRef]
- Chen, Y.; Jiang, L.; Lyu, K.; Lu, J.; Long, L.; Wang, X.; Liu, T.; Li, S. A Promising Candidate in Tendon Healing Events—PDGF-BB. Biomolecules 2022, 12, 1518. [CrossRef]
- Kovacevic, D.; Rodeo, S.A. Biological Augmentation of Rotator Cuff Tendon Repair. Clin. Orthop. 2008, 466, 622–633.
- Guerrero, P.A.; McCarty, J.H. TGF-β Activation and Signaling in Angiogenesis. In Physiologic and Pathologic Angiogenesis - Signaling Mechanisms and Targeted Therapy; Simionescu, D., Simionescu, A., Eds.; InTech, 2017 ISBN 978-953-51-3023-9.
- Ferrari, G.; Pintucci, G.; Seghezzi, G.; Hyman, K.; Galloway, A.C.; Mignatti, P. VEGF, a Prosurvival Factor, Acts in Concert with TGF-Β1 to Induce Endothelial Cell Apoptosis. Proc. Natl. Acad. Sci. 2006, 103, 17260–17265. [CrossRef]
- Pepper, M.S. Transforming Growth Factor-Beta: Vasculogenesis, Angiogenesis, and Vessel Wall Integrity. Cytokine Growth Factor Rev. 1997, 8, 21–43. [CrossRef]
- Cao, R.; Brakenhielm, E.; Wahlestedt, C.; Thyberg, J.; Cao, Y. Leptin Induces Vascular Permeability and Synergistically Stimulates Angiogenesis with FGF-2 and VEGF. Proc. Natl. Acad. Sci. 2001, 98, 6390–6395. [CrossRef]
- Vlodavsky, I.; Bar-Shavit, R.; Ishar-Michael, R.; Bashkin, P.; Fuks, Z. Extracellular Sequestration and Release of Fibroblast Growth Factor: A Regulatory Mechanism? Trends Biochem. Sci. 1991, 16, 268–271. [CrossRef]
- Christiaens, V.; Lijnen, H. Angiogenesis and Development of Adipose Tissue. Mol. Cell. Endocrinol. 2010, 318, 2–9. [CrossRef]
- Cross, M.J.; Claesson-Welsh, L. FGF and VEGF Function in Angiogenesis: Signalling Pathways, Biological Responses and Therapeutic Inhibition. Trends Pharmacol. Sci. 2001, 22, 201–207. [CrossRef]
- van Meurs, M.; Kümpers, P.; Ligtenberg, J.J.; Meertens, J.H.; Molema, G.; Zijlstra, J.G. Bench-to-Bedside Review: Angiopoietin Signalling in Critical Illness–a Future Target? Crit. Care 2009, 13, 1–13. [CrossRef]
- De Mast, Q.; Faradz, S.M.H.; Griffioen, A.W.; Michels, M.; Sebastian, S.; De Groot, P.G.; Fijnheer, R.; Djamiatun, K.; Van Der Ven, A.J.A.M. Imbalance of Angiopoietin-1 and Angiopoetin-2 in Severe Dengue and Relationship with Thrombocytopenia, Endothelial Activation, and Vascular Stability. Am. J. Trop. Med. Hyg. 2012, 87, 943–946. [CrossRef]
- Nachman, R.L.; Rafii, S. Platelets, Petechiae, and Preservation of the Vascular Wall. N. Engl. J. Med. 2008, 359, 1261–1270. [CrossRef]
- Koblizek, T.I.; Weiss, C.; Yancopoulos, G.D.; Deutsch, U.; Risau, W. Angiopoietin-1 Induces Sprouting Angiogenesis in Vitro. Curr. Biol. 1998, 8, 529–532. [CrossRef]
- Fiedler, U.; Reiss, Y.; Scharpfenecker, M.; Grunow, V.; Koidl, S.; Thurston, G.; Gale, N.W.; Witzenrath, M.; Rosseau, S.; Suttorp, N. Angiopoietin-2 Sensitizes Endothelial Cells to TNF-α and Has a Crucial Role in the Induction of Inflammation. Nat. Med. 2006, 12, 235–239. [CrossRef]
- Brill, A.; Dashevsky, O.; Rivo, J.; Gozal, Y.; Varon, D. Platelet-Derived Microparticles Induce Angiogenesis and Stimulate Post-Ischemic Revascularization. Cardiovasc. Res. 2005, 67, 30–38. [CrossRef]
- Kim, H.K.; Song, K.S.; Chung, J.-H.; Lee, K.R.; Lee, S.-N. Platelet Microparticles Induce Angiogenesis in Vitro: Platelet Microparticles Induce Angiogenesis in Vitro. Br. J. Haematol. 2004, 124, 376–384. [CrossRef]
- Mause, S.F.; Ritzel, E.; Liehn, E.A.; Hristov, M.; Bidzhekov, K.; Müller-Newen, G.; Soehnlein, O.; Weber, C. Platelet Microparticles Enhance the Vasoregenerative Potential of Angiogenic Early Outgrowth Cells After Vascular Injury. Circulation 2010, 122, 495–506. [CrossRef]
- Yoon, J.P.; Yoon, J.W.; Kim, H.; Oh, J.H. Selective Serotonin Reuptake Inhibitor Promotes Bone-Tendon Interface Healing in a Rotator Cuff Tear Rat Model. Tissue Eng. Regen. Med. 2022, 19, 853–860. [CrossRef]
- Herr, N.; Bode, C.; Duerschmied, D. The Effects of Serotonin in Immune Cells. Front. Cardiovasc. Med. 2017, 4, 48. [CrossRef]
- Arreola, R.; Becerril-Villanueva, E.; Cruz-Fuentes, C.; Velasco-Velázquez, M.A.; Garcés-Alvarez, M.E.; Hurtado-Alvarado, G.; Quintero-Fabian, S.; Pavón, L. Immunomodulatory Effects Mediated by Serotonin. J. Immunol. Res. 2015, 2015, 1–21. [CrossRef]
- Pakala, R.; Willerson, J.T.; Benedict, C.R. Mitogenic Effect of Serotonin on Vascular Endothelial Cells. Circulation 1994, 90, 1919–1926. [CrossRef]
- Matsusaka, S.; Wakabayashi, I. 5-Hydroxytryptamine as a Potent Migration Enhancer of Human Aortic Endothelial Cells. FEBS Lett. 2005, 579, 6721–6725. [CrossRef]
- Zamani, A.; Qu, Z. Serotonin Activates Angiogenic Phosphorylation Signaling in Human Endothelial Cells. FEBS Lett. 2012, 586, 2360–2365. [CrossRef]
- Kreuger, J.; Phillipson, M. Targeting Vascular and Leukocyte Communication in Angiogenesis, Inflammation and Fibrosis. Nat. Rev. Drug Discov. 2016, 15, 125–142. [CrossRef]
- Taichman, N.S.; Young, S.; Cruchley, A.T.; Taylor, P.; Paleolog, E. Human Neutrophils Secrete Vascular Endothelial Growth Factor. J. Leukoc. Biol. 1997, 62, 397–400. [CrossRef]
- Tang, Y.-Q.; Yeaman, M.R.; Selsted, M.E. Antimicrobial Peptides from Human Platelets. Infect. Immun. 2002, 70, 6524–6533. [CrossRef]
- Moojen, D.J.F.; Schure, R.-M.; Overdevest, E.P.; van Zundert, A.; Knape, J.T.A.; Castelein, R.M.; Creemers, L.B.; Dhert, W.J.A. Antimicrobial Activity of Platelet-Leukocyte Gel AgainstStaphylococcus Aureus. J. Orthop. Res. 2008, 26, 404–410. [CrossRef]
- Vasina, E.M.; Cauwenberghs, S.; Feijge, M.A.H.; Heemskerk, J.W.M.; Weber, C.; Koenen, R.R. Microparticles from Apoptotic Platelets Promote Resident Macrophage Differentiation. Cell Death Dis. 2011, 2, e211–e211. [CrossRef]
- Aldabbous, L.; Abdul-Salam, V.; McKinnon, T.; Duluc, L.; Pepke-Zaba, J.; Southwood, M.; Ainscough, A.J.; Hadinnapola, C.; Wilkins, M.R.; Toshner, M.; et al. Neutrophil Extracellular Traps Promote Angiogenesis: Evidence From Vascular Pathology in Pulmonary Hypertension. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 2078–2087. [CrossRef]
- Fetz, A.E.; Radic, M.Z.; Bowlin, G.L. Neutrophils in Biomaterial-Guided Tissue Regeneration: Matrix Reprogramming for Angiogenesis. Tissue Eng. Part B Rev. 2021, 27, 95–106. [CrossRef]
- Wang, J. Neutrophils in Tissue Injury and Repair. Cell Tissue Res. 2018, 371, 531–539. [CrossRef]
- Shantsila, E.; Watson, T.; Lip, G.Y.H. Endothelial Progenitor Cells in Cardiovascular Disorders. J. Am. Coll. Cardiol. 2007, 49, 741–752. [CrossRef]
- Shantsila, E.; Lip, G.Y.H. Monocytes in Acute Coronary Syndromes. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 1433—1438. [CrossRef]
- Romagnani, P.; Annunziato, F.; Liotta, F.; Lazzeri, E.; Mazzinghi, B.; Frosali, F.; Cosmi, L.; Maggi, L.; Lasagni, L.; Scheffold, A.; et al. CD14+CD34 low Cells With Stem Cell Phenotypic and Functional Features Are the Major Source of Circulating Endothelial Progenitors. Circ. Res. 2005, 97, 314–322. [CrossRef]
- Brown, B.N.; Valentin, J.E.; Stewart-Akers, A.M.; McCabe, G.P.; Badylak, S.F. Macrophage Phenotype and Remodeling Outcomes in Response to Biologic Scaffolds with and without a Cellular Component. Biomaterials 2009, 30, 1482–1491. [CrossRef]
- Spiller, K.L.; Anfang, R.R.; Spiller, K.J.; Ng, J.; Nakazawa, K.R.; Daulton, J.W.; Vunjak-Novakovic, G. The Role of Macrophage Phenotype in Vascularization of Tissue Engineering Scaffolds. Biomaterials 2014, 35, 4477–4488. [CrossRef]
- Roh, J.D.; Sawh-Martinez, R.; Brennan, M.P.; Jay, S.M.; Devine, L.; Rao, D.A.; Yi, T.; Mirensky, T.L.; Nalbandian, A.; Udelsman, B. Tissue-Engineered Vascular Grafts Transform into Mature Blood Vessels via an Inflammation-Mediated Process of Vascular Remodeling. Proc. Natl. Acad. Sci. 2010, 107, 4669–4674. [CrossRef]
- Stein, M.; Keshav, S.; Harris, N.; Gordon, S. Interleukin 4 Potently Enhances Murine Macrophage Mannose Receptor Activity: A Marker of Alternative Immunologic Macrophage Activation. J. Exp. Med. 1992, 176, 287–292.
- Sainson, R.C.A.; Johnston, D.A.; Chu, H.C.; Holderfield, M.T.; Nakatsu, M.N.; Crampton, S.P.; Davis, J.; Conn, E.; Hughes, C.C.W. TNF Primes Endothelial Cells for Angiogenic Sprouting by Inducing a Tip Cell Phenotype. Blood 2008, 111, 4997–5007. [CrossRef]
- Delafontaine, P.; Song, Y.-H.; Li, Y. Expression, Regulation, and Function of IGF-1, IGF-1R, and IGF-1 Binding Proteins in Blood Vessels. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 435–444. [CrossRef]
- Nakao-Hayashi, J.; Ito, H.; Kanayasu, T.; Morita, I.; Murota, S. Stimulatory Effects of Insulin and Insulin-like Growth Factor I on Migration and Tube Formation by Vascular Endothelial Cells. Atherosclerosis 1992, 92, 141–149. [CrossRef]
- Beitia, M.; Delgado, D.; Mercader, J.; Sánchez, P.; López De Dicastillo, L.; Sánchez, M. Action of Platelet-Rich Plasma on In Vitro Cellular Bioactivity: More than Platelets. Int. J. Mol. Sci. 2023, 24, 5367. [CrossRef]
- Smith, L.E.; Shen, W.; Perruzzi, C.; Soker, S.; Kinose, F.; Xu, X.; Robinson, G.; Driver, S.; Bischoff, J.; Zhang, B. Regulation of Vascular Endothelial Growth Factor-Dependent Retinal Neovascularization by Insulin-like Growth Factor-1 Receptor. Nat. Med. 1999, 5, 1390–1395. [CrossRef]
- Kluge, A.; Zimmermann, R.; Weihrauch, D.; Mohri, M.; Sack, S.; Schaper, J.; Schaper, W. Coordinate Expression of the Insulin-like Growth Factor System after Microembolisation in Porcine Heart. Cardiovasc. Res. 1997, 33, 324–331. [CrossRef]
- Fukushima, T.; Uchiyama, S.; Tanaka, H.; Kataoka, H. Hepatocyte Growth Factor Activator: A Proteinase Linking Tissue Injury with Repair. Int. J. Mol. Sci. 2018, 19, 3435. [CrossRef]
- Siedlecki, Z.; Grzyb, S.; Rość, D.; Śniegocki, M.; 1 Department of Neurosurgery and Neurotraumatology and Pediatric Neurosurgery, Collegium Medicum in Bydgoszcz of Nicolaus Copernicus University in Toruń, Poland; 2 Department of Pathophysiology, Collegium Medicum in Bydgoszcz of Nicolaus Copernicus University in Toruń, Poland Plasma HGF Concentration in Patients with Brain TumorsRunning Headline: HGF in Patients with Brain Tumors. AIMS Neurosci. 2020, 7, 107–119. [CrossRef]
- Zhang, Z.; Li, Y.; Zhang, T.; Shi, M.; Song, X.; Yang, S.; Liu, H.; Zhang, M.; Cui, Q.; Li, Z. Hepatocyte Growth Factor-Induced Tendon Stem Cell Conditioned Medium Promotes Healing of Injured Achilles Tendon. Front. Cell Dev. Biol. 2021, 9, 654084. [CrossRef]
- Bertrand-Duchesne, M.; Grenier, D.; Gagnon, G. Epidermal Growth Factor Released from Platelet-rich Plasma Promotes Endothelial Cell Proliferation in Vitro. J. Periodontal Res. 2010, 45, 87–93. [CrossRef]
- Li, X.; Hou, J.; Wu, B.; Chen, T.; Luo, A. Effects of Platelet-Rich Plasma and Cell Coculture on Angiogenesis in Human Dental Pulp Stem Cells and Endothelial Progenitor Cells. J. Endod. 2014, 40, 1810–1814.
- Anitua, E.; Prado, R.; Sánchez, M.; Orive, G. Platelet-Rich Plasma: Preparation and Formulation. Oper. Tech. Orthop. 2012, 22, 25–32. [CrossRef]
- Calabriso, N.; Stanca, E.; Rochira, A.; Damiano, F.; Giannotti, L.; Di Chiara Stanca, B.; Massaro, M.; Scoditti, E.; Demitri, C.; Nitti, P.; et al. Angiogenic Properties of Concentrated Growth Factors (CGFs): The Role of Soluble Factors and Cellular Components. Pharmaceutics 2021, 13, 635. [CrossRef]
- Zhou, Y.; Wang, J.H.-C. PRP Treatment Efficacy for Tendinopathy: A Review of Basic Science Studies. BioMed Res. Int. 2016, 2016, 1–8. [CrossRef]
- Notodihardjo, P.V.; Morimoto, N.; Kakudo, N.; Matsui, M.; Sakamoto, M.; Liem, P.H.; Suzuki, K.; Tabata, Y.; Kusumoto, K. Gelatin Hydrogel Impregnated with Platelet-Rich Plasma Releasate Promotes Angiogenesis and Wound Healing in Murine Model. J. Artif. Organs 2015, 18, 64–71. [CrossRef]
- Roy, S.; Driggs, J.; Elgharably, H.; Biswas, S.; Findley, M.; Khanna, S.; Gnyawali, U.; Bergdall, V.K.; Sen, C.K. Platelet-rich Fibrin Matrix Improves Wound Angiogenesis via Inducing Endothelial Cell Proliferation. Wound Repair Regen. 2011, 19, 753–766. [CrossRef]
- Lyras, D.N.; Kazakos, K.; Verettas, D.; Polychronidis, A.; Tryfonidis, M.; Botaitis, S.; Agrogiannis, G.; Simopoulos, C.; Kokka, A.; Patsouris, E. The Influence of Platelet-Rich Plasma on Angiogenesis During the Early Phase of Tendon Healing. Foot Ankle Int. 2009, 30, 1101–1106. [CrossRef]
- Chahla, J.; Cinque, M.E.; Piuzzi, N.S.; Mannava, S.; Geeslin, A.G.; Murray, I.R.; Dornan, G.J.; Muschler, G.F.; LaPrade, R.F. A Call for Standardization in Platelet-Rich Plasma Preparation Protocols and Composition Reporting: A Systematic Review of the Clinical Orthopaedic Literature. J. Bone Jt. Surg. 2017, 99, 1769–1779. [CrossRef]
- Ehrenfest, D.M.D.; Andia, I.; Zumstein, M.A.; Zhang, C.-Q.; Pinto, N.R.; Bielecki, T. Classification of Platelet Concentrates (Platelet-Rich Plasma-PRP, Platelet-Rich Fibrin-PRF) for Topical and Infiltrative Use in Orthopedic and Sports Medicine: Current Consensus, Clinical Implications and Perspectives. 7.
- Rossi, L.A.; Murray, I.R.; Chu, C.R.; Muschler, G.F.; Rodeo, S.A.; Piuzzi, N.S. Classification Systems for Platelet-Rich Plasma. Bone Jt. J. 2019, 101-B, 891–896. [CrossRef]
- Gupta, A.; Jeyaraman, M.; Potty, A. Leukocyte-Rich vs. Leukocyte-Poor Platelet-Rich Plasma for the Treatment of Knee Osteoarthritis. Biomedicines 2023, 11, 141. [CrossRef]
- Muchedzi, T.A.; Roberts, S.B. A Systematic Review of the Effects of Platelet Rich Plasma on Outcomes for Patients with Knee Osteoarthritis and Following Total Knee Arthroplasty. The Surgeon 2018, 16, 250–258. [CrossRef]
- D’Addona, A.; Maffulli, N.; Formisano, S.; Rosa, D. Inflammation in Tendinopathy. The Surgeon 2017, 15, 297–302. [CrossRef]
- Yuan, Z.; Wang, Y.; Li, Y.; Lin, C.; Wang, S.; Wang, J.; Ma, C.; Wu, S. Comparison of Leukocyte-Rich and Leukocyte-Poor Platelet-Rich Plasma on Pressure Ulcer in a Rat Model. J. Burn Care Res. 2023, irac191. [CrossRef]
- Lana, J.F. Leukocyte-Rich PRP versus Leukocyte-Poor PRP - The Role of Monocyte/Macrophage Function in the Healing Cascade. J. Clin. Orthop. Trauma 2019, 6. [CrossRef]
- Kobayashi, Y.; Saita, Y.; Nishio, H.; Ikeda, H.; Takazawa, Y.; Nagao, M.; Takaku, T.; Komatsu, N.; Kaneko, K. Leukocyte Concentration and Composition in Platelet-Rich Plasma (PRP) Influences the Growth Factor and Protease Concentrations. J. Orthop. Sci. 2016, 21, 683–689. [CrossRef]
- Pifer, M.A.; Maerz, T.; Baker, K.C.; Anderson, K. Matrix Metalloproteinase Content and Activity in Low-Platelet, Low-Leukocyte and High-Platelet, High-Leukocyte Platelet Rich Plasma (PRP) and the Biologic Response to PRP by Human Ligament Fibroblasts. Am. J. Sports Med. 2014, 42, 1211–1218. [CrossRef]
- Hohmann, E.; Tetsworth, K.; Glatt, V. Is Platelet-Rich Plasma Effective for the Treatment of Knee Osteoarthritis? A Systematic Review and Meta-Analysis of Level 1 and 2 Randomized Controlled Trials. Eur. J. Orthop. Surg. Traumatol. 2020. [CrossRef]
- Jaipersad, A.S.; Lip, G.Y.H.; Silverman, S.; Shantsila, E. The Role of Monocytes in Angiogenesis and Atherosclerosis. J. Am. Coll. Cardiol. 2014, 63, 1–11. [CrossRef]
- Korntner, S.; Lehner, C.; Gehwolf, R.; Wagner, A.; Grütz, M.; Kunkel, N.; Tempfer, H.; Traweger, A. Limiting Angiogenesis to Modulate Scar Formation. Adv. Drug Deliv. Rev. 2019, 146, 170–189. [CrossRef]
- Gimeno-LLuch, I.; Benito-Jardón, M.; Guerrero-Barberà, G.; Burday, N.; Costell, M. The Role of the Fibronectin Synergy Site for Skin Wound Healing. Cells 2022, 11, 2100. [CrossRef]
- Meftahpour, V.; Malekghasemi, S.; Baghbanzadeh, A.; Aghebati-Maleki, A.; Pourakbari, R.; Fotouhi, A.; Aghebati-Maleki, L. Platelet Lysate: A Promising Candidate in Regenerative Medicine. Regen. Med. 2021, 16, 71–85. [CrossRef]
- Doucet, C.; Ernou, I.; Zhang, Y.; Llense, J.; Begot, L.; Holy, X.; Lataillade, J. Platelet Lysates Promote Mesenchymal Stem Cell Expansion: A Safety Substitute for Animal Serum in Cell-based Therapy Applications. J. Cell. Physiol. 2005, 205, 228–236. [CrossRef]
- Chiara Barsotti, M.; Losi, P.; Briganti, E.; Sanguinetti, E.; Magera, A.; Al Kayal, T.; Feriani, R.; Di Stefano, R.; Soldani, G. Effect of Platelet Lysate on Human Cells Involved in Different Phases of Wound Healing. PLoS One 2013, 8, e84753. [CrossRef]
- Dale, D.C.; Boxer, L.; Liles, W.C. The Phagocytes: Neutrophils and Monocytes. Blood 2008, 112, 935–945. [CrossRef]
- Fedorova, N.V.; Ksenofontov, A.L.; Serebryakova, M.V.; Stadnichuk, V.I.; Gaponova, T.V.; Baratova, L.A.; Sud’ina, G.F.; Galkina, S.I. Neutrophils Release Metalloproteinases during Adhesion in the Presence of Insulin, but Cathepsin G in the Presence of Glucagon. Mediators Inflamm. 2018, 2018, 1–9. [CrossRef]
- Peterson, J.E.; Zurakowski, D.; Italiano, J.E.; Michel, L.V.; Fox, L.; Klement, G.L.; Folkman, J. Normal Ranges of Angiogenesis Regulatory Proteins in Human Platelets. Am. J. Hematol. 2010, 85, 487–493. [CrossRef]
- Upputuri, P.K.; Sivasubramanian, K.; Mark, C.S.K.; Pramanik, M. Recent Developments in Vascular Imaging Techniques in Tissue Engineering and Regenerative Medicine. BioMed Res. Int. 2015, 2015, 783983. [CrossRef]
- Oh, J.H.; Kim, W.; Park, K.U.; Roh, Y.H. Comparison of the Cellular Composition and Cytokine-Release Kinetics of Various Platelet-Rich Plasma Preparations. Am. J. Sports Med. 2015, 43, 3062–3070. [CrossRef]
- Bielecki, T.; M. Dohan Ehrenfest, D.; A. Everts, P.; Wiczkowski, A. The Role of Leukocytes from L-PRP/L-PRF in Wound Healing and Immune Defense: New Perspectives. Curr. Pharm. Biotechnol. 2012, 13, 1153–1162. [CrossRef]
- Kuffler, D. Variables Affecting the Potential Efficacy of PRP in Providing Chronic Pain Relief. J. Pain Res. 2018, Volume 12, 109–116. [CrossRef]
- Magalon, J.; Bausset, O.; Serratrice, N.; Giraudo, L.; Aboudou, H.; Veran, J.; Magalon, G.; Dignat-Georges, F.; Sabatier, F. Characterization and Comparison of 5 Platelet-Rich Plasma Preparations in a Single-Donor Model. Arthrosc. J. Arthrosc. Relat. Surg. 2014, 30, 629–638. [CrossRef]
- Rodeo, S.A.; Delos, D.; Williams, R.J.; Adler, R.S.; Pearle, A.; Warren, R.F. The Effect of Platelet-Rich Fibrin Matrix on Rotator Cuff Tendon Healing: A Prospective, Randomized Clinical Study. Am. J. Sports Med. 2012, 40, 1234–1241. [CrossRef]
- Linnanmäki, L.; Kanto, K.; Karjalainen, T.; Leppänen, O.V.; Lehtinen, J. Platelet-Rich Plasma or Autologous Blood Do Not Reduce Pain or Improve Function in Patients with Lateral Epicondylitis: A Randomized Controlled Trial. Clin. Orthop. 2020, 478, 1892–1900. [CrossRef]
- Walsh, M.R.; Nelson, B.J.; Braman, J.P.; Yonke, B.; Obermeier, M.; Raja, A.; Reams, M. Platelet-Rich Plasma in Fibrin Matrix to Augment Rotator Cuff Repair: A Prospective, Single-Blinded, Randomized Study with 2-Year Follow-Up. J. Shoulder Elbow Surg. 2018, 27, 1553–1563. [CrossRef]
- Levy, G.M.; Lucas, P.; Hope, N. Efficacy of a Platelet-Rich Plasma Injection for the Treatment of Proximal Hamstring Tendinopathy: A Pilot Study. J. Sci. Med. Sport 2019, 22, 247–252. [CrossRef]
- Longo, U.G.; Castricini, R.; De Benedetto, M.; Panfoli, N.; Pirani, P.; Zini, R.; Maffulli, N.; Denaro, V. Paper # 117: Platelet-Rich Fibrin Matrix Augmentation for Arthroscopic Rotator Cuff Repair: A Randomized Controlled Trial. Arthrosc. J. Arthrosc. Relat. Surg. 2011, 27, e145–e146. [CrossRef]
- Boesen, A.P.; Hansen, R.; Boesen, M.I.; Malliaras, P.; Langberg, H. Effect of High-Volume Injection, Platelet-Rich Plasma, and Sham Treatment in Chronic Midportion Achilles Tendinopathy: A Randomized Double-Blinded Prospective Study. Am. J. Sports Med. 2017, 45, 2034–2043. [CrossRef]
- EL-Hakeim, E.H.; Selim, Z.I.; Omran, E.A.H.; Hosney, M.; Abd-Elkader, A.S. Platelet Rich Plasma versus Hyaluronic Acid Injection in Chronic Painful Shoulder: Randomized Blind Clinical Trial. Indian J. Rheumatol. 2022, 17. [CrossRef]
- Thermann, H.; Fischer, R.; Gougoulias, N.; Cipollaro, L.; Maffulli, N. Endoscopic Debridement for Non-Insertional Achilles Tendinopathy with and without Platelet-Rich Plasma. J. Sport Health Sci. 2023, 12, 275–280. [CrossRef]
- Kearney, R.S.; Ji, C.; Warwick, J.; Parsons, N.; Brown, J.; Harrison, P.; Young, J.; Costa, M.L.; ATM Trial Collaborators; Dasari, K.; et al. Effect of Platelet-Rich Plasma Injection vs Sham Injection on Tendon Dysfunction in Patients With Chronic Midportion Achilles Tendinopathy: A Randomized Clinical Trial. JAMA 2021, 326, 137. [CrossRef]
- Bergeson, A.G.; Tashjian, R.Z.; Greis, P.E.; Crim, J.; Stoddard, G.J.; Burks, R.T. Effects of Platelet-Rich Fibrin Matrix on Repair Integrity of At-Risk Rotator Cuff Tears. Am. J. Sports Med. 2012, 40, 286–293. [CrossRef]
- Krogh, T.P.; Ellingsen, T.; Christensen, R.; Jensen, P.; Fredberg, U. Ultrasound-Guided Injection Therapy of Achilles Tendinopathy With Platelet-Rich Plasma or Saline: A Randomized, Blinded, Placebo-Controlled Trial. Am. J. Sports Med. 2016, 44, 1990–1997. [CrossRef]
- Krogh, T.P.; Fredberg, U.; Stengaard-Pedersen, K.; Christensen, R.; Jensen, P.; Ellingsen, T. Treatment of Lateral Epicondylitis With Platelet-Rich Plasma, Glucocorticoid, or Saline: A Randomized, Double-Blind, Placebo-Controlled Trial. Am. J. Sports Med. 2013, 41, 625–635. [CrossRef]
- Keene, D.J.; Alsousou, J.; Harrison, P.; Hulley, P.; Wagland, S.; Parsons, S.R.; Thompson, J.Y.; O’Connor, H.M.; Schlüssel, M.M.; Dutton, S.J.; et al. Platelet Rich Plasma Injection for Acute Achilles Tendon Rupture: PATH-2 Randomised, Placebo Controlled, Superiority Trial. BMJ 2019, l6132. [CrossRef]
- Raeissadat, S.A.; Ghorbani, E.; Sanei Taheri, M.; Soleimani, R.; Rayegani, S.M.; Babaee, M.; Payami, S. MRI Changes After Platelet Rich Plasma Injection in Knee Osteoarthritis (Randomized Clinical Trial). J. Pain Res. 2020, Volume 13, 65–73. [CrossRef]
- Jo, C.H.; Lee, S.Y.; Yoon, K.S.; Shin, S. Effects of Platelet-Rich Plasma With Concomitant Use of a Corticosteroid on Tenocytes From Degenerative Rotator Cuff Tears in Interleukin 1β–Induced Tendinopathic Conditions. Am. J. Sports Med. 2017, 45, 1141–1150. [CrossRef]
- Morrey, B.F. Platelet-Rich Plasma Versus Autologous Whole Blood for the Treatment of Chronic Lateral Elbow Epicondylitis: A Randomized Controlled Clinical Trial. Yearb. Orthop. 2012, 2012, 197–199. [CrossRef]
- Podesta, L.; Crow, S.A.; Volkmer, D.; Bert, T.; Yocum, L.A. Treatment of Partial Ulnar Collateral Ligament Tears in the Elbow With Platelet-Rich Plasma. Am. J. Sports Med. 2013, 41, 1689–1694. [CrossRef]
- Mishra, A.K.; Skrepnik, N.V.; Edwards, S.G.; Jones, G.L.; Sampson, S.; Vermillion, D.A.; Ramsey, M.L.; Karli, D.C.; Rettig, A.C. Efficacy of Platelet-Rich Plasma for Chronic Tennis Elbow: A Double-Blind, Prospective, Multicenter, Randomized Controlled Trial of 230 Patients. Am. J. Sports Med. 2014, 42, 463–471. [CrossRef]
- Peerbooms, J.C.; Sluimer, J.; Bruijn, D.J.; Gosens, T. Positive Effect of an Autologous Platelet Concentrate in Lateral Epicondylitis in a Double-Blind Randomized Controlled Trial: Platelet-Rich Plasma Versus Corticosteroid Injection with a 1-Year Follow-Up. Am. J. Sports Med. 2010, 38, 255–262. [CrossRef]
- Randelli, P.; Arrigoni, P.; Ragone, V.; Aliprandi, A.; Cabitza, P. Platelet Rich Plasma in Arthroscopic Rotator Cuff Repair: A Prospective RCT Study, 2-Year Follow-Up. J. Shoulder Elbow Surg. 2011, 20, 518–528. [CrossRef]
- Kaux, J.-F.; Bouvard, M.; Lecut, C.; Oury, C.; Gothot, A.; Sanchez, M.; Crielaard, J.-M. Reflections about the Optimisation of the Treatment of Tendinopathies with PRP. 4.
- Everhart, J.S.; Cavendish, P.A.; Eikenberry, A.; Magnussen, R.A.; Kaeding, C.C.; Flanigan, D.C. Platelet-Rich Plasma Reduces Failure Risk for Isolated Meniscal Repairs but Provides No Benefit for Meniscal Repairs With Anterior Cruciate Ligament Reconstruction. Am. J. Sports Med. 2019, 47, 1789–1796. [CrossRef]
- Vahdatpour, B.; Kianimehr, L.; Ahrar, M. Autologous Platelet-Rich Plasma Compared with Whole Blood for the Treatment of Chronic Plantar Fasciitis; a Comparative Clinical Trial. Adv. Biomed. Res. 2016, 5, 84. [CrossRef]
- Holtby, R.; Christakis, M.; Maman, E.; MacDermid, J.C.; Dwyer, T.; Athwal, G.S.; Faber, K.; Theodoropoulos, J.; Woodhouse, L.J.; Razmjou, H. Impact of Platelet-Rich Plasma on Arthroscopic Repair of Small- to Medium-Sized Rotator Cuff Tears: A Randomized Controlled Trial. Orthop. J. Sports Med. 2016, 4, 232596711666559. [CrossRef]
- Deppermann, C.; Kubes, P. Start a Fire, Kill the Bug: The Role of Platelets in Inflammation and Infection. Innate Immun. 2018, 24, 335–348. [CrossRef]
- Reed, M.J.; Edelberg, J.M. Impaired Angiogenesis in the Aged. Sci. Aging Knowl. Environ. 2004, 2004, pe7–pe7. [CrossRef]
- Dobke, M.; Peterson, D.R.; Mattern, R.-H.; Arm, D.M.; William W Li Microvascular Tissue as a Platform Technology to Modify the Local Microenvironment and Influence the Healing Cascade. Regen. Med. 2020, 15, 1313–1328. [CrossRef]
- Lopez-Vidriero, E.; Goulding, K.A.; Simon, D.A.; Sanchez, M.; Johnson, D.H. The Use of Platelet-Rich Plasma in Arthroscopy and Sports Medicine: Optimizing the Healing Environment. Arthrosc. J. Arthrosc. Relat. Surg. 2010, 26, 269–278. [CrossRef]
- Lansdown, D.A.; Fortier, L.A. Platelet-Rich Plasma: Formulations, Preparations, Constituents, and Their Effects. Oper. Tech. Sports Med. 2017, 25, 7–12. [CrossRef]
- Haller, H.L.; Sander, F.; Nischwitz, S.P.; Popp, D.; Rapp, M.; Hartmann, B.; Demircan, M.; Kamolz, L.P. Oxygen, PH, Lactate, and Metabolism- How Old Knowledge and New Insights Might Be Combined for New Wound Treatment; MEDICINE & PHARMACOLOGY, 2021; [CrossRef]
- Diegelmann, R., F. Wound Healing: An Overview of Acute, Fibrotic and Delayed Healing. Front. Biosci. 2004, 9, 283. [CrossRef]
- Liu, X.; Zhang, R.; Zhu, B.; Li, Y.; Liu, X.; Guo, S.; Wang, C.; Wang, D.; Li, S. Effects of Leukocyte- and Platelet-rich Plasma on Tendon Disorders Based on in Vitro and in Vivo Studies (Review). Exp. Ther. Med. 2021, 21, 639. [CrossRef]
- Andia, I.; Sanchez, M.; Maffulli, N. Tendon Healing and Platelet-Rich Plasma Therapies. Expert Opin. Biol. Ther. 2010, 10, 1415–1426. [CrossRef]
- Dittadi, R.; Meo, S.; Fabris, F.; Gasparini, G.; Contri, D.; Medici, M.; Gion, M. Validation of Blood Collection Procedures for the Determination of Circulating Vascular Endothelial Growth Factor (VEGF) in Different Blood Compartments. Int. J. Biol. Markers 2001, 16, 87–96. [CrossRef]
- Vogrin, M.; Rupreht, M.; Dinevski, D.; Hašpl, M.; Kuhta, M.; Jevsek, M.; Knežević, M.; Rožman, P. Effects of a Platelet Gel on Early Graft Revascularization after Anterior Cruciate Ligament Reconstruction: A Prospective, Randomized, Double-Blind, Clinical Trial. Eur. Surg. Res. 2010, 45, 77–85. [CrossRef]
- Rupreht, M.; Jevtič, V.; Serša, I.; Vogrin, M.; Jevšek, M. Evaluation of the Tibial Tunnel after Intraoperatively Administered Platelet-rich Plasma Gel during Anterior Cruciate Ligament Reconstruction Using Diffusion Weighted and Dynamic Contrast-enhanced MRI. J. Magn. Reson. Imaging 2013, 37, 928–935. [CrossRef]
- Rhee, J.-S.; Black, M.; Silvia, U.; Fischer, S.; Morgenstern, E.; Hammes, H.-P.; Preissner, K. The Functional Role of Blood Platelet Components in Angiogenesis. Thromb. Haemost. 2004, 92, 394–402. [CrossRef]
- Avraham-Davidi, I.; Yona, S.; Grunewald, M.; Landsman, L.; Cochain, C.; Silvestre, J.S.; Mizrahi, H.; Faroja, M.; Strauss-Ayali, D.; Mack, M. On-Site Education of VEGF-Recruited Monocytes Improves Their Performance as Angiogenic and Arteriogenic Accessory Cells. J. Exp. Med. 2013, 210, 2611–2625. [CrossRef]
- Grunewald, M.; Avraham, I.; Dor, Y.; Bachar-Lustig, E.; Itin, A.; Yung, S.; Chimenti, S.; Landsman, L.; Abramovitch, R.; Keshet, E. VEGF-Induced Adult Neovascularization: Recruitment, Retention, and Role of Accessory Cells. Cell 2006, 124, 175–189. [CrossRef]
- Ohki, Y.; Heissig, B.; Sato, Y.; Akiyama, H.; Zhu, Z.; Hicklin, D.J.; Shimada, K.; Ogawa, H.; Daida, H.; Hattori, K. Granulocyte Colony-stimulating Factor Promotes Neovascularization by Releasing Vascular Endothelial Growth Factor from Neutrophils. FASEB J. 2005, 19, 2005–2007. [CrossRef]
- Ardi, V.C.; Kupriyanova, T.A.; Deryugina, E.I.; Quigley, J.P. Human Neutrophils Uniquely Release TIMP-Free MMP-9 to Provide a Potent Catalytic Stimulator of Angiogenesis. Proc. Natl. Acad. Sci. 2007, 104, 20262–20267. [CrossRef]
- Heissig, B.; Hattori, K.; Friedrich, M.; Rafii, S.; Werb, Z. Angiogenesis: Vascular Remodeling of the Extracellular Matrix Involves Metalloproteinases. Curr. Opin. Hematol. 2003, 10, 136–141. [CrossRef]
- Neve, A.; Cantatore, F.P.; Maruotti, N.; Corrado, A.; Ribatti, D. Extracellular Matrix Modulates Angiogenesis in Physiological and Pathological Conditions. BioMed Res. Int. 2014, 2014. [CrossRef]
- Bir, S.C.; Esaki, J.; Marui, A.; Sakaguchi, H.; Kevil, C.G.; Ikeda, T.; Komeda, M.; Tabata, Y.; Sakata, R. Therapeutic Treatment with Sustained-Release Platelet-Rich Plasma Restores Blood Perfusion by Augmenting Ischemia-Induced Angiogenesis and Arteriogenesis in Diabetic Mice. J. Vasc. Res. 2011, 48, 195–205. [CrossRef]

| Pro Angiogenetic Stimulator | Presence in LR-PRP | Anti-Angiogenic Inhibitors | Presence in LR-PRP |
| 5-HT | * | Angiostatin | * |
| Ang | * | Endostatin | * |
| bFGF | * | Heparinases | |
| G-CSF | IL-10,12 | ||
| HGF | Interferon α/β/γ | ||
| IGF-1 | */- | PA | |
| IL-3, 8 | PF-4 | * | |
| Macrophage | RANTES | * | |
| MMP-2 | * | TGF-β | * |
| MMP-9 | * | Thrombospondin-1 | * |
| Monocyte | * | TIMP1-4 | * |
| Neutrophil | * | TSP-1 | * |
| PDGF-BB | * | Vasculostatin | |
| PMP | * | ||
| Progranulin | |||
| TGF-β | * | ||
| Thrombin | |||
| TNF-α | |||
| VEGF | * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




