Submitted:
01 June 2023
Posted:
02 June 2023
You are already at the latest version
Abstract

Keywords:
1. Introduction
2. Materials and Methods
2.1. Preparation of Ferrate Fe(VI)
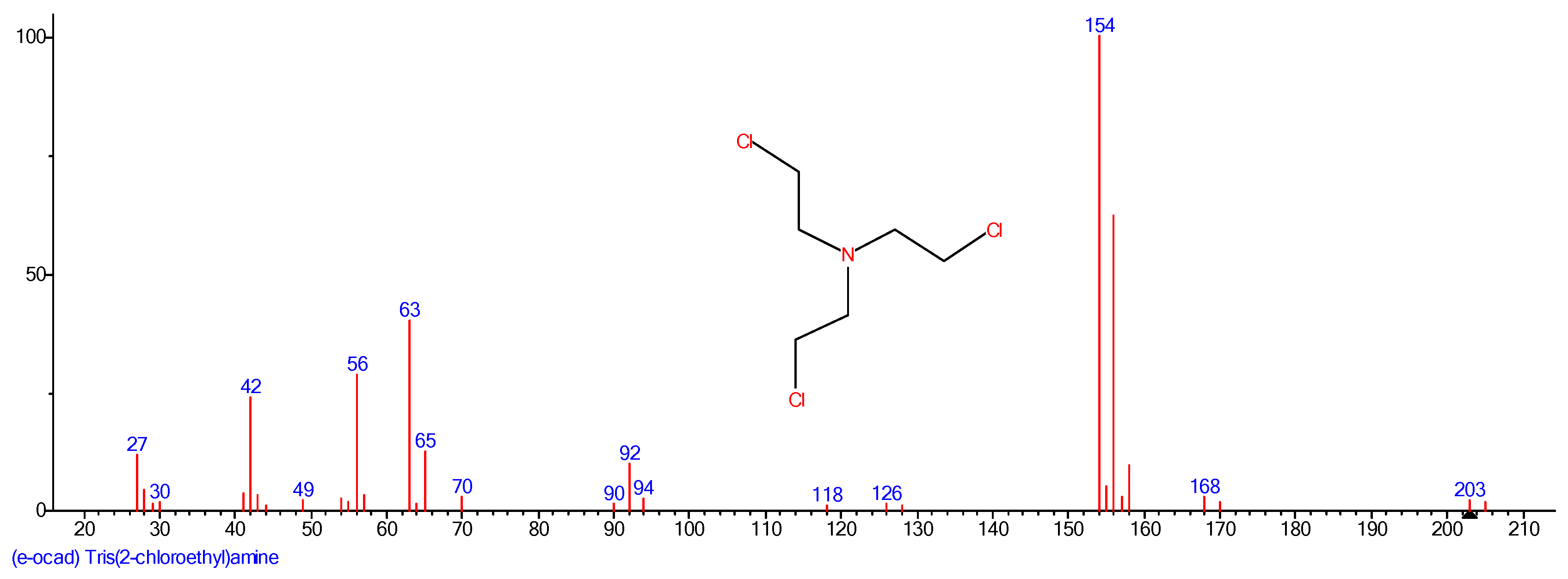
2.2. Preparation of Nitrogen Mustard HN3
2.3. Reaction of Ferrate Fe(VI) with Nitrogen Mustard HN3
3. Results and Discussion
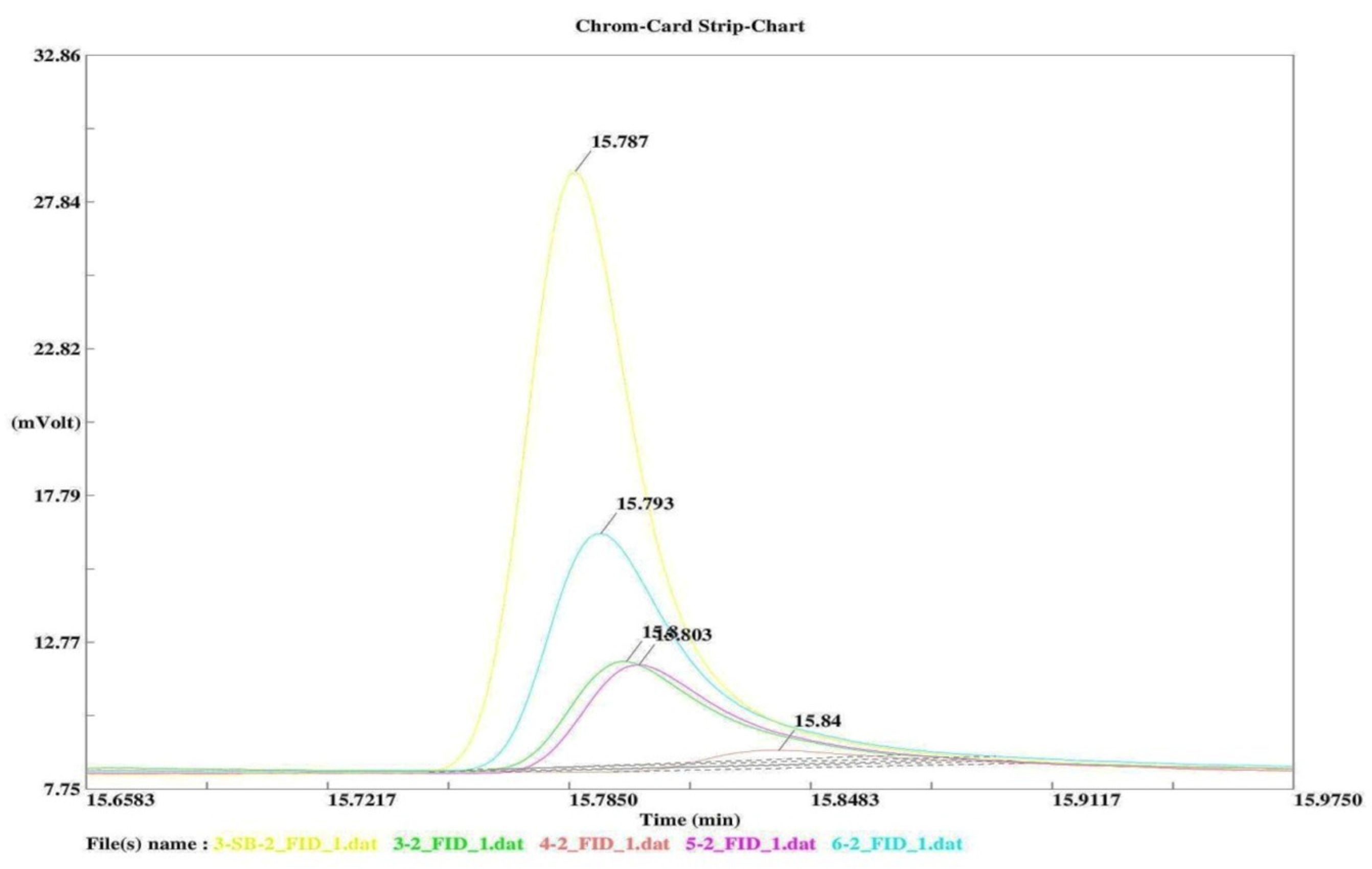
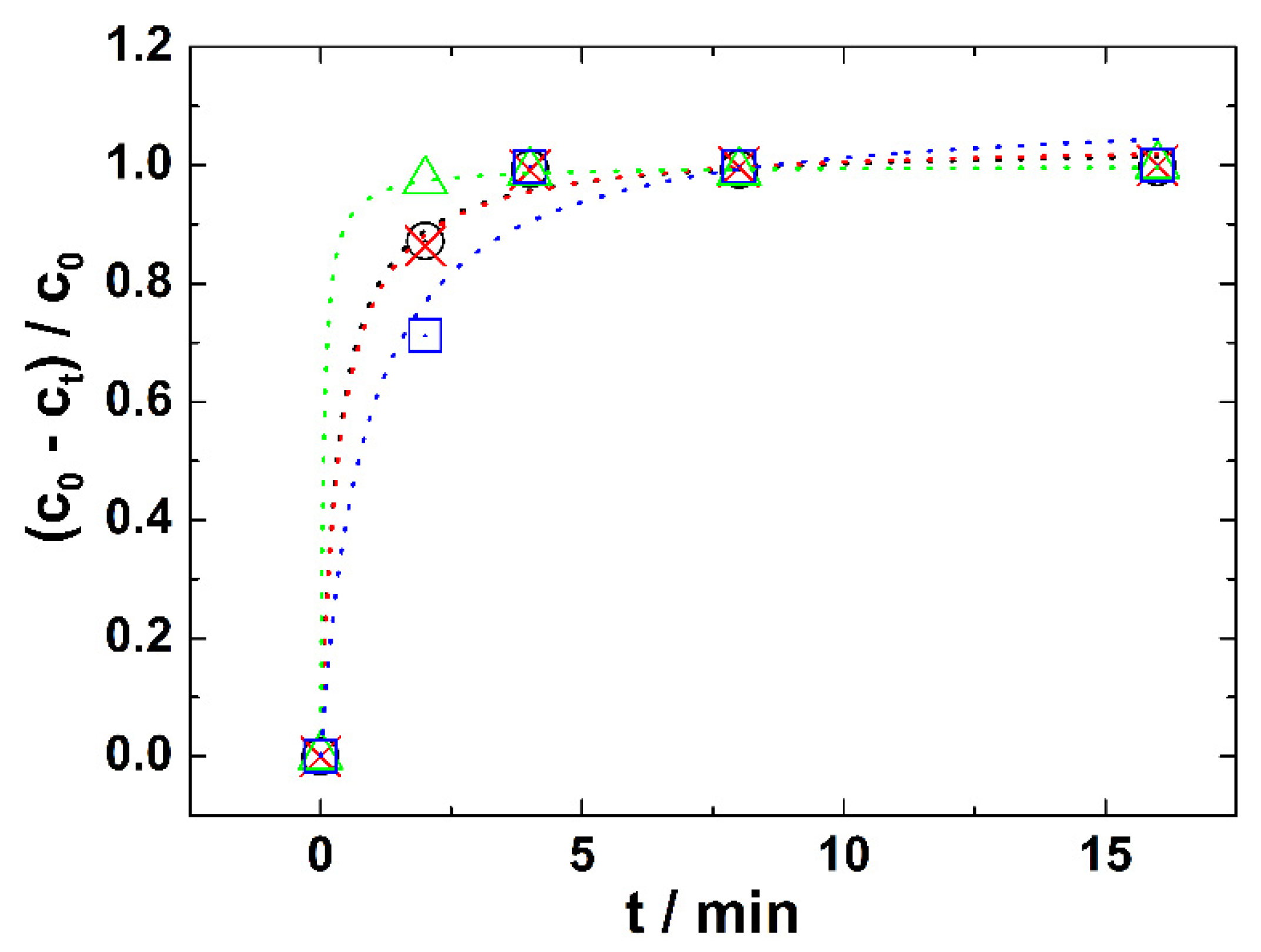
3.1. Degradation of Nitrogen Mustard
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Prentiss, A.M. Classification of Chemical Agents, A Treatise on Chemical Warfare. New York : McGraw Hill, 1937.
- Chauhan S, Chauhan S, D’Cruz R, Faruqi S, Singh KK, Varma S, Singh, M., Karthik, V. Chemical warfare agents. Environ Toxicol Pharmacol. 2008, Vol. 26, 2.
- Ganesan, K., Raza, S.K., & Vijayaraghavan, R. Chemical warfare agents. Journal of pharmacy & bioallied sciences. 2010, Vol. 2, 3.
- Szinicz, L. History of chemical and biological warfare agents. Toxicology. 2005, Vol. 214, 3. [CrossRef]
- GUPTA, Ramesh, C. Handbook of toxicology of chemical warfare agents. s.l. : Academic Press, 2015. ISBN: 978–0-12–800159–2.
- Marrs, Timothy, T., Robert, L. Maynard, and Frederick Sidell, eds. Chemical warfare agents: Toxicology and treatment. s.l. : John Wiley & Sons, 2007. ISBN 978–0-470–01359–5.
- Romano, J.A. Jr., Lukey, B.J., Salem, H.,. Chemical Warfare Agents: Chemistry, Pharmacology, Toxicology, and Therapeutics. s.l. : CRC Taylor & Francis, 2007. ISBN13: 978–1-4200–4661–8.
- Small, M.J. Compounds Formed from the Chemical Decontamination of HD, GB, and VX and Their Environmental Fate. Fort Detrick, Frederick MD; : U.S. Army Medical Bioengineering Research and Development Laboratory, 1984. Technical Report 8304, AD A149515.
- Trapp, R. The Detoxification and Natural Degradation of Chemical Warfare Agents. . Stockholm, Sweden : Stockholm International Peace Institute (SIPRI),, 1985. [CrossRef]
- Clark, D.N. Review of Reactions of Chemical Agents in Water. Battelle, Columbus, OH. : Defense Technical Information Center, Ft. Belvoir, VA, 1989. AD-A213 287.
- MacNaughton, M.G. and Brewer, J.H.,. Environmental Chemistry and Fate of Chemical Warfare Agents. Final report.,. San Antonio, TX : Southwest Research Institute, 1994. SwRI Project 01–5864.
- Munro, N.B.; et al. The Sources, Fate, and Toxicity of Chemical Warfare Agent Degradation Products. Environ. Health Persp.,. 1999, Vol. 107, 933. [CrossRef]
- Talmage, S.S., Watson, A.P., Hauschild, V., Munro, N.B., King, J.,. Chemical Warfare Agent Degradation and Decontamination. Current Organic Chemistry,. 2007, Vol. 11. [CrossRef]
- Snelson, A., Taylor, K., O’Neill, H.J.,. Reaction of CW agents simulants on surfaces in the presence of O3, UV and O3+UV. J. Environ. Sci. Health. 1984, Vol. A19, 7. [CrossRef]
- Popiel, S., Nalepa, T., Dzierziak, D., Stankiewicz, R., Witkiewicz, Z.,. Rate of dibutylsulfide decomposition by ozonation and the O3/H2O2 advanced oxidation process. Journal of Hazardous Materials. 2009, Vol. 164. [CrossRef]
- Popiel, S., Witkiewicz, Z., Chrzanowski, M.,. Sulfur mustard destruction using ozone, UV, hydrogen peroxide and their combination. Journal of Hazardous Materials. 2007, Vol. 153. [CrossRef]
- Liu. Y., Howarth, A.J., Vermeulen, N.A., Moon, S.-Y.,. Catalytic degradation of chemical warfare agents and their simulants by metal-organic frameworks. Coordination Chemistry Reviews. 2017, Vol. 346. [CrossRef]
- Tang, J.,Li, P.,Islamoglu, T., Li, S., Zhang, X.,Son, F.A., Chen, Z.,. Micropore environment regulation of zirconium MOFs for instantaneous hydrolysis of an organophosphorus chemical. Cell Reports Physical Science. 2, 2021. [CrossRef]
- Shen, Z., Zhong, J.-Y., Han, X.-Y., Wanga, L.-Y., Cui, Y., Chen, L.-K.,. Decontamination of Chemical Warfare Agents on sensitive equipment materials using Zr4+ and Ge4+ co-doped TiO2 and hydrofluoroether suspension. Chemical Engineering Journal. 2016, Vol. 302. [CrossRef]
- Bromberg, L., Schreuder-Gibson, H., Creasy, W.R., McGarvey, D.J., Fry, R.A., Hatton, T.A.,. Degradation of Chemical Warfare Agents by Reactive Polymers. Ind. Eng. Chem. Res. 2009, Vol. 48. [CrossRef]
- Iwai, T., Inoue, H., Kakegawa, K., Ohrui, Y., Nagoya, T., Nagashima, H., Miyahara, H., Chiba, K., Seto, Y., Okino, A.,. Development of a High-Efficiency Decomposition Technology for Volatile Chemical Warfare Agent Sarin Using Dielectric Barrier Discharge. Plasma Chemistry and Plasma Processing. 2020, Vol. 40. [CrossRef]
- Ohmori, T., Kawahara, K., Nakayama, K., Shioda, A., Ishikawa, S., Kanamori-Kataoka, M., Kishi, S., Komano, A., Seto, Y.,. Decontamination of nerve agents by immobilized organophosphorus hydrolase. Forensic Toxicology. 2013, Vol. 31. [CrossRef]
- Organization, Science and Technology Organization North Atlantic Treaty. Long-Term Scientific Study on CBRN Defence. Neuilly-sur-Seine Cedex, France: NATO Science and Technology organization, TR-HFM-273, 2021. ISBN 978–92–837–2327–1.
- Zboril, R., Andrle, M., Oplustil, F., Machala, L., Tucek, J., Filip, J., Marusak, Z., Sharma, V.K. Treatment of chemical warfare agents by zero-valent iron nanoparticles and ferrate(VI)/(III) composite. Journal of Hazardous Materials. 2012, Vols. 211–212. [CrossRef]
- Bartlett, P.D., Ross, S.D., Swain, C.G.,. Kinetics and Mechanism of the Reactions of Tertiary β-Chloroethylamines in Solution. III. β-Chloroethyldiethylamine and tris-β-Chloroethylamine. J. Am. Chem. Soc. 1947, Vol. 71, 4. [CrossRef]
- Czölderova, M., Behúl, M., Filip, J., Zajíček, P., Grabic, R., Vojs-Staňová, A.,Gál, M., Kerekeš, K., Híveš, J., Ryba, J., Rybanská, M., Brandeburová, P., Mackuľak, T.,. 3D printed polyvinyl alcohol ferrate(VI) capsules: Effective means for the removal of pharmaceuticals and illicit drugs from wastewater,. Chemical Engineering Journal,. 2018, Vol. 349. [CrossRef]
- Sharma, V.K. Oxidation of nitrogen-containing pollutants by novel ferrate (VI) technology: A review. Journal of Environmental Science and Health Part, A. 2010, Vol. 45, 6, pp. 645–667. [CrossRef]
- Ferrate (VI) and ferrate (V) oxidation of organic compounds: Kinetics and mechanism. Coordination Chemistry Reviews. 2013, Vol. 257, 2, pp. 495–510. [CrossRef]
- Sarma, R.; et al. Studies of the di-iron (VI) intermediate in ferrate-dependent oxygen evolution from water. Journal of the American Chemical Society. 2012, Vol. 134, 37, pp. 15371–15386. [CrossRef]
- Luo, C., Feng, M., Sharma, V.K., Huang, Ch-H. Revelation of ferrate(VI) unimolecular decay under alkaline conditions: Investigation of involvement of Fe(IV) and Fe(V) species. Chemical Engineering Journal. 2020, Vol. 338, pp. 124–134. [CrossRef]
- J.; Híves, J., Benova, M., Bouzek, K., Sitek, J., Sharma, V.K. The cyclic voltammetric study of ferrate(VI) formation in a molten Na/K hydroxide mixture. Electrochimica Acta. 2008, Vol. 54. [CrossRef]
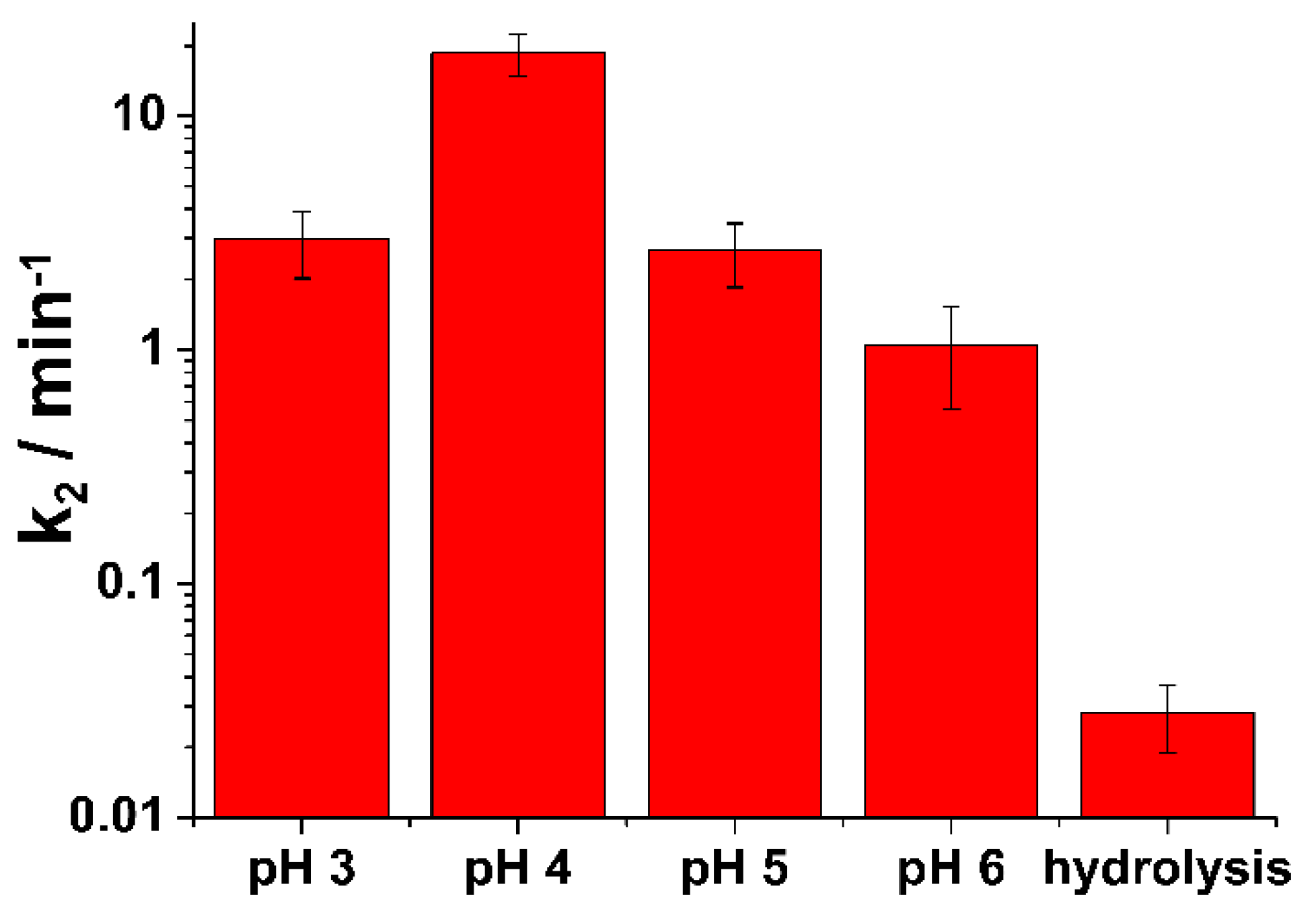
| pH | k2 (min−1) | χ2 | Adjusted R-Squared |
|---|---|---|---|
| 3 | 2.961±0.942 | 6.48×10−4 | 0.997 |
| 4 | 18.596±3.750 | 1.16×10−5 | 0.999 |
| 5 | 2.658±0.808 | 6.87×10−4 | 0.996 |
| 6 | 1.054±0.498 | 4.59×10−3 | 0.975 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




