Submitted:
30 May 2023
Posted:
31 May 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Culturing of A. fumigatus
2.2. Collection of Aspergillus spp. conidia
2.3. Collection and processing of bronchoalveolar lavage fluid (BALF) samples
2.4. Enrichment and isolation of antagonistic bacterial strains from BALF samples
2.5. Confrontation assay on a soft agar overlay assay plate
2.6. High-throughput germination assay
2.7. Identification of the bacterial isolates
2.8. Confrontation on Calu-3 cell cultures on permeable Transwell® inserts
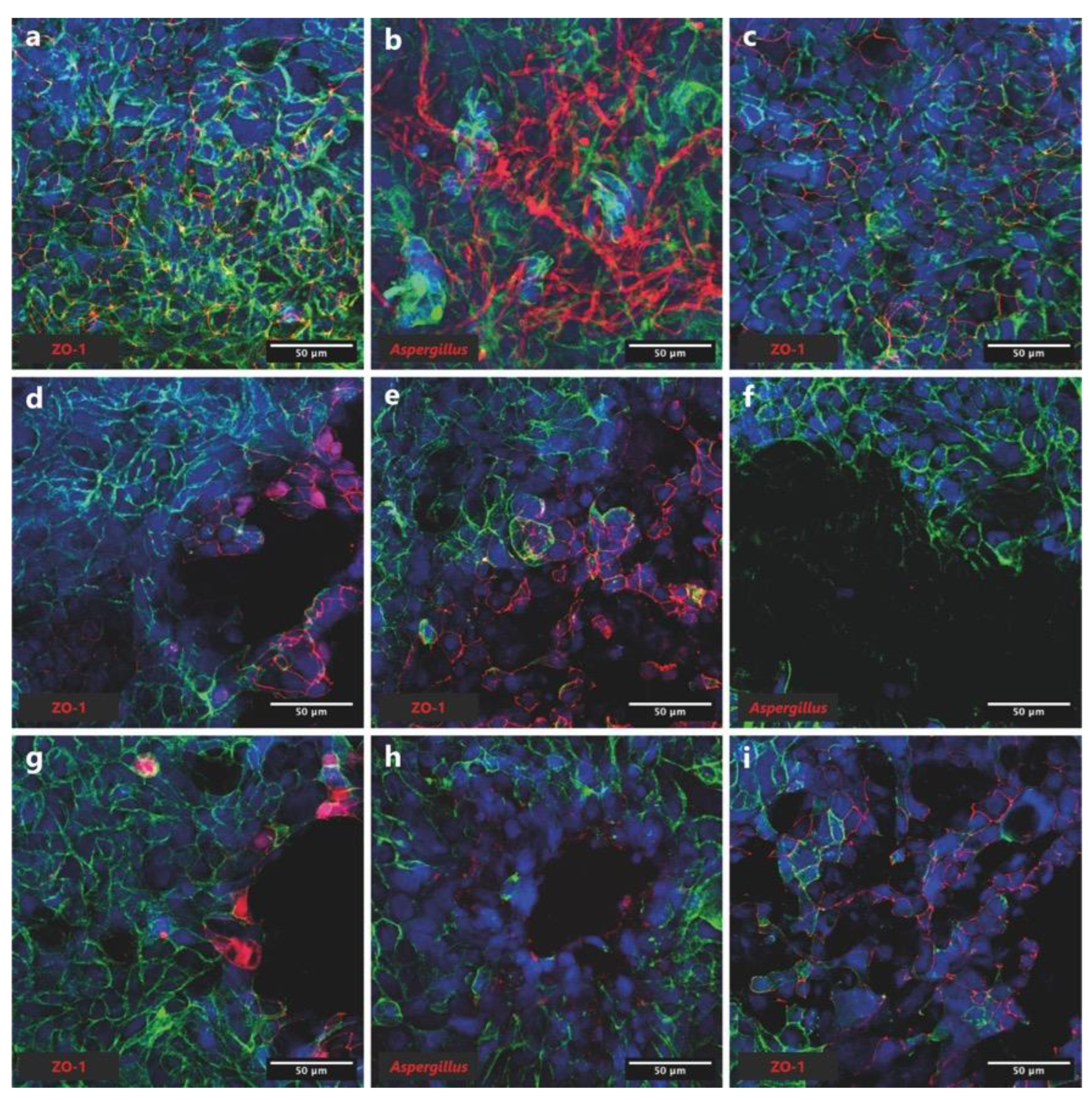
2.9. Immunofluorescence staining and confocal imaging
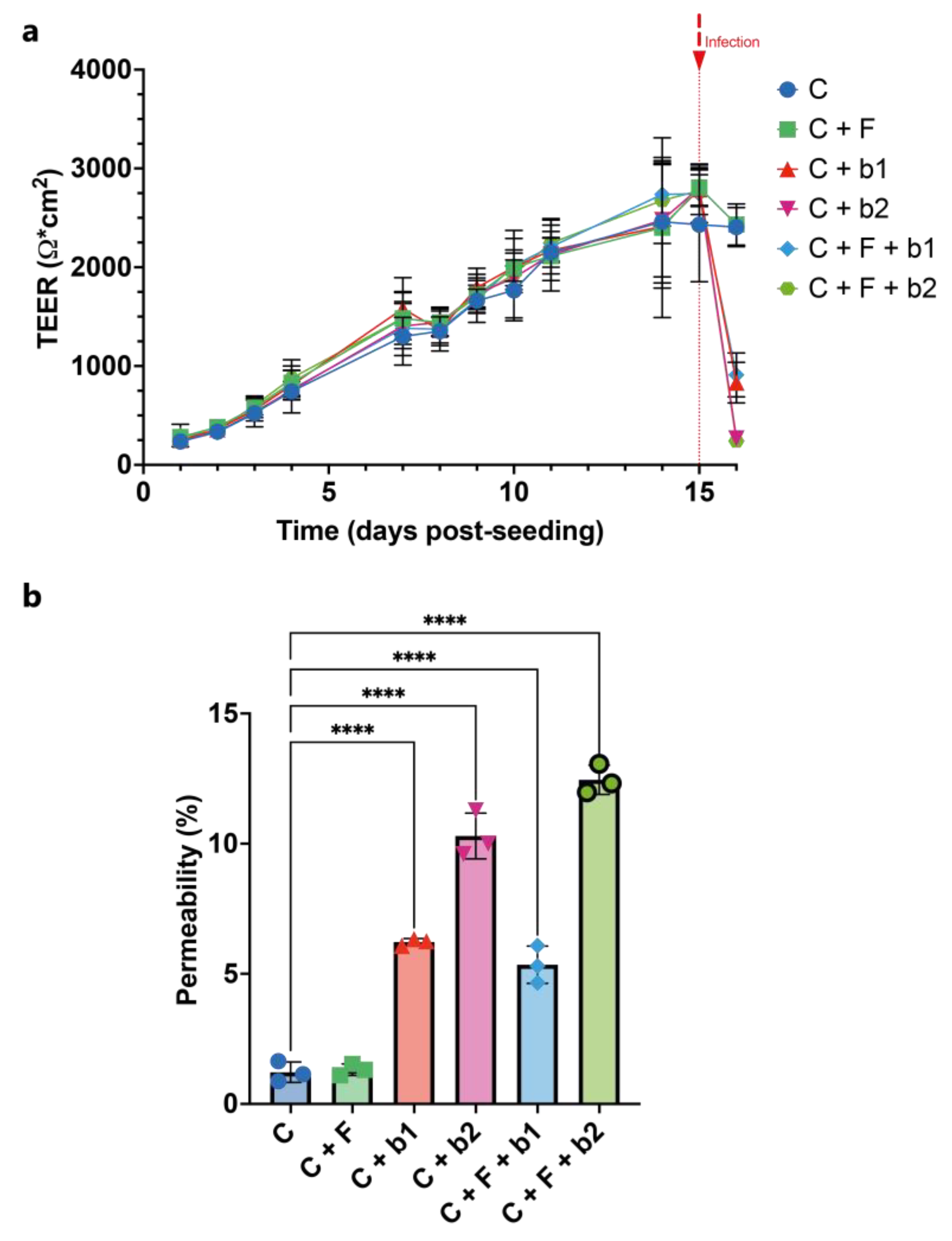
2.10. Assessment of epithelial barrier function: TEER measurements and Lucifer Yellow permeability assay
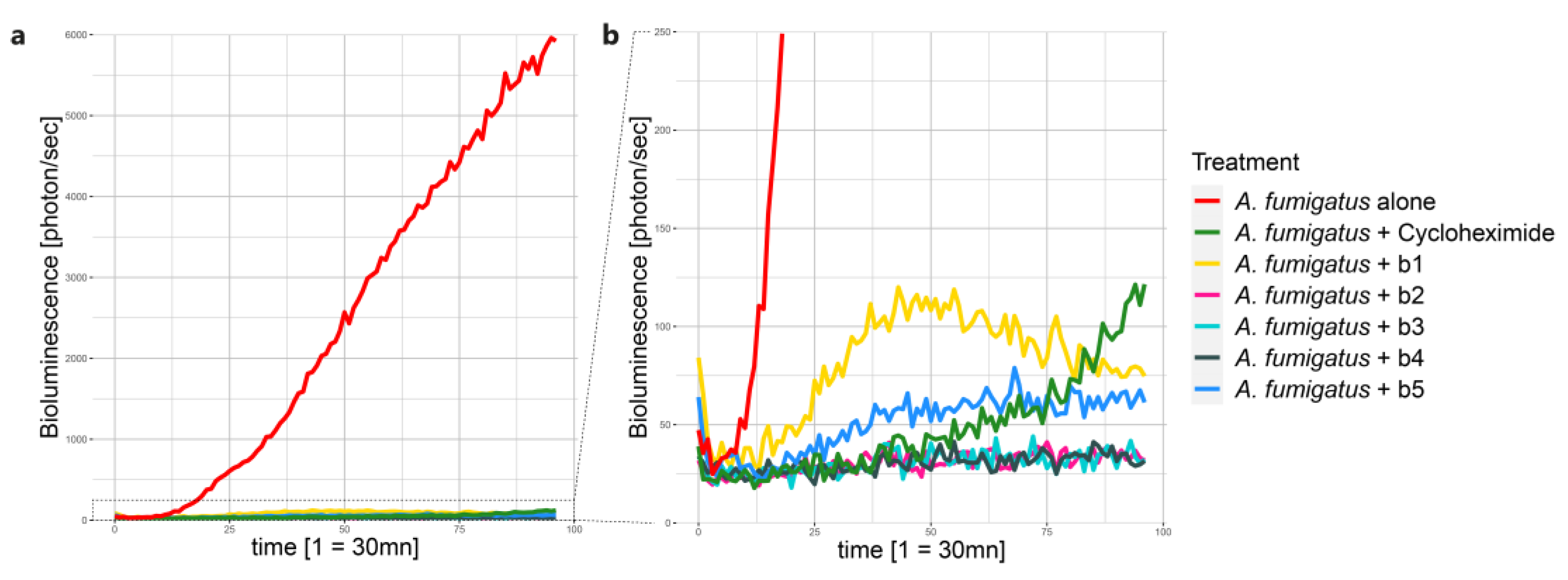
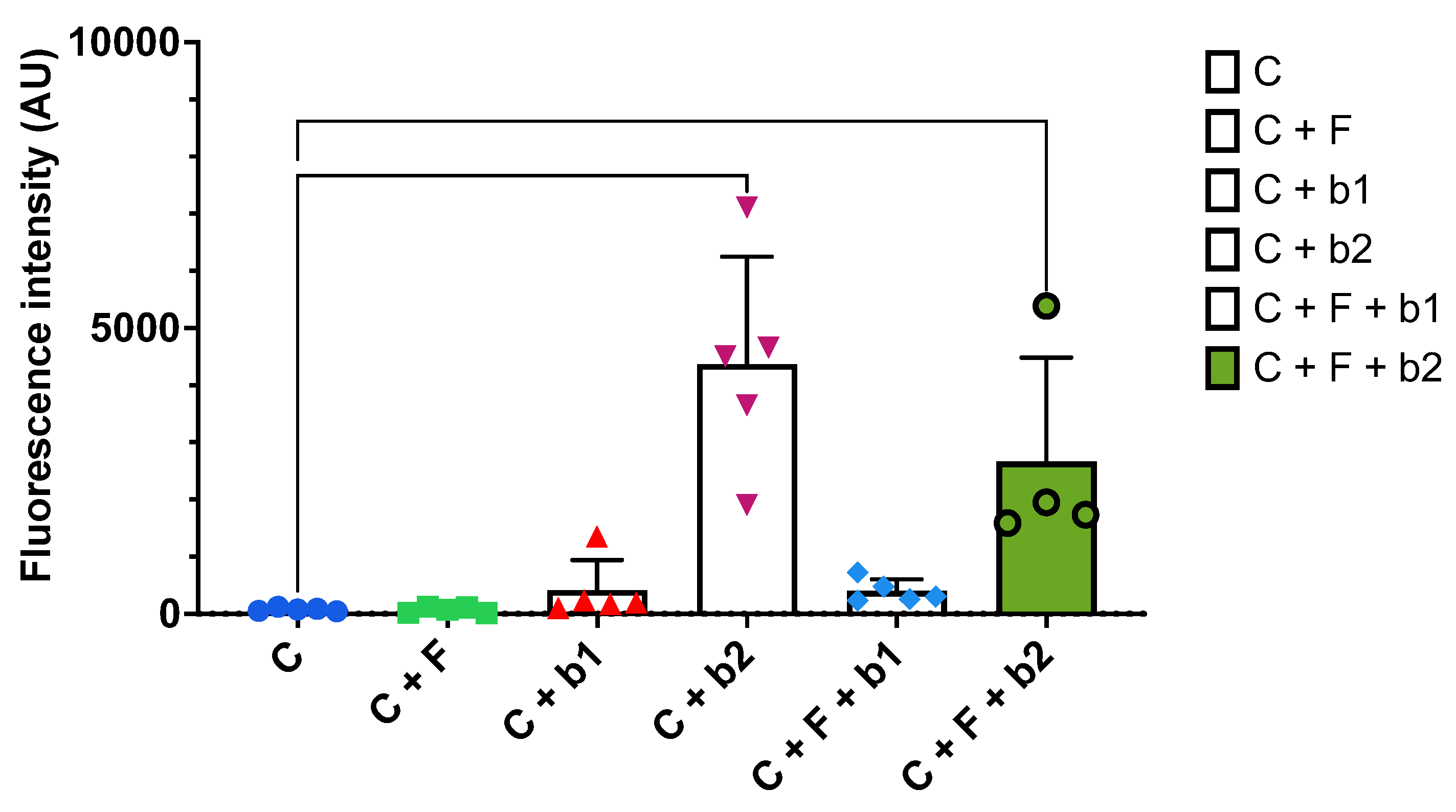
2.11. Bioluminescence imaging
2.12. Cytotoxicity assay
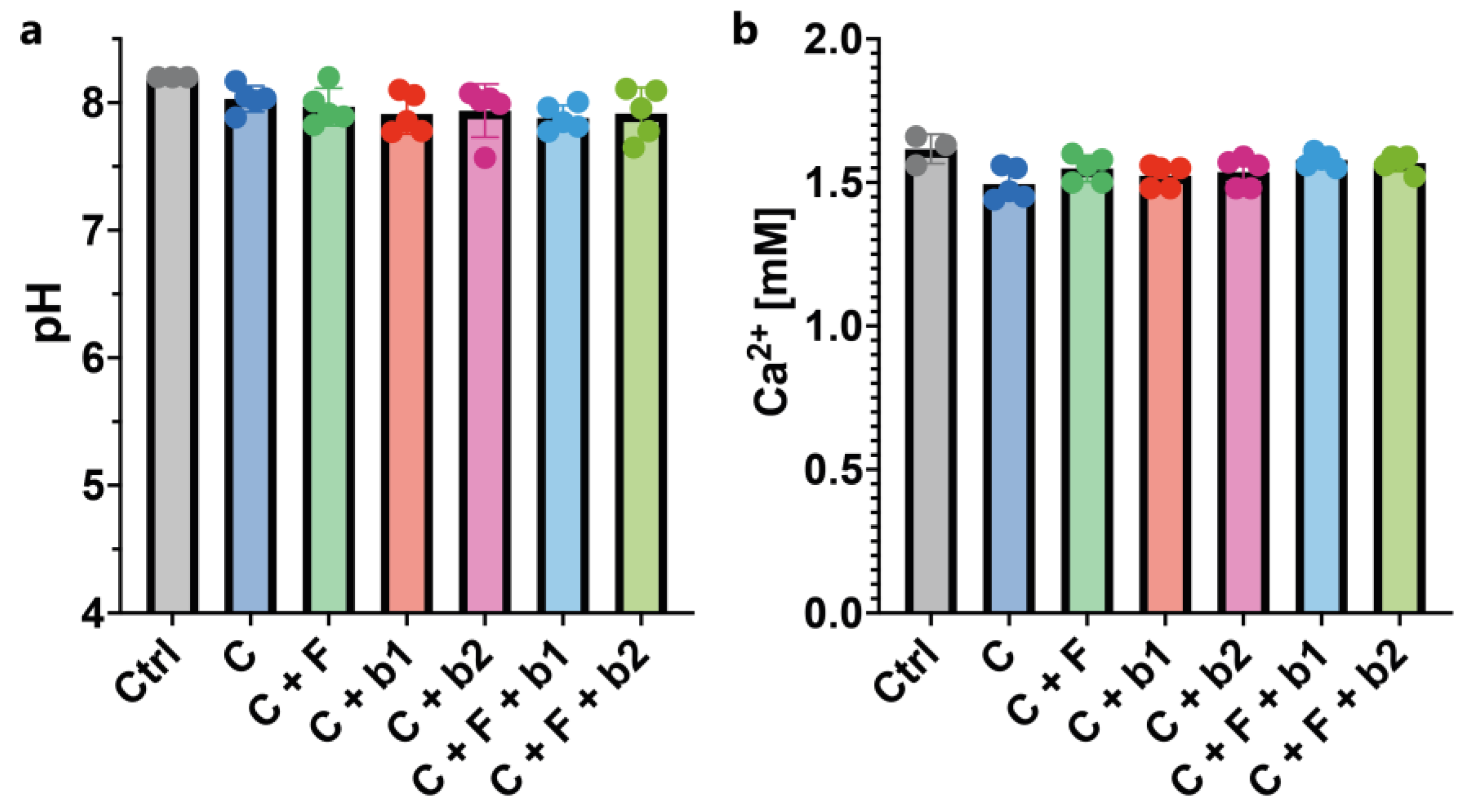
2.13. pH measurements and quantification of calcium
2.14. Statistical analyses
3. Results
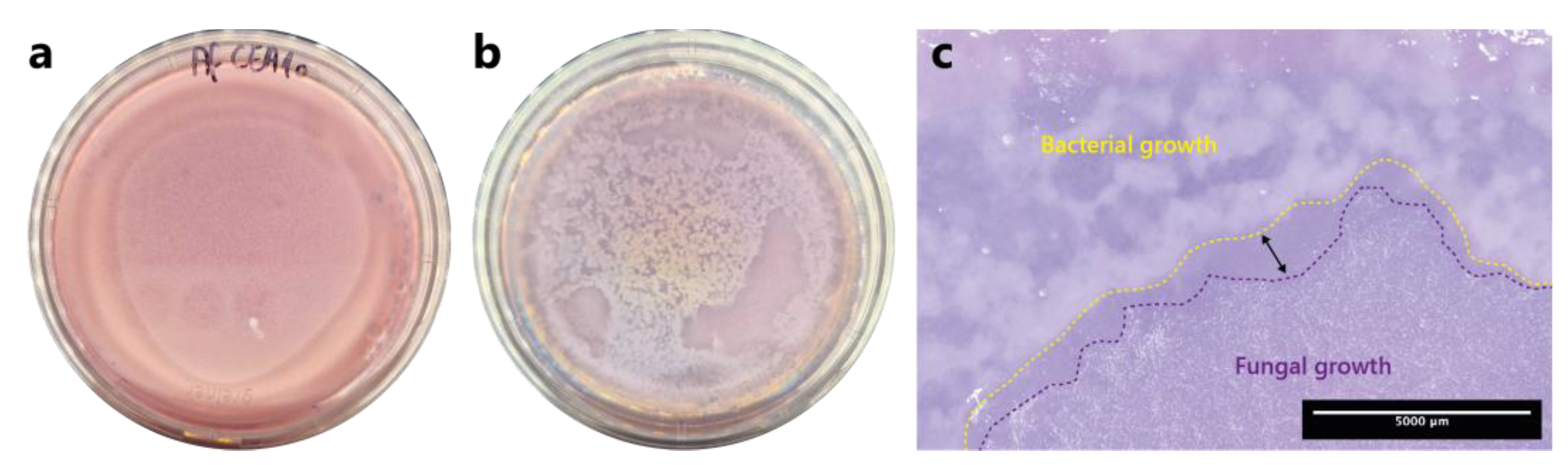
3.1. Enrichment and isolation of lung bacteria inhibiting A. fumigatus conidia germination and growth in one-step sof agar plate assay
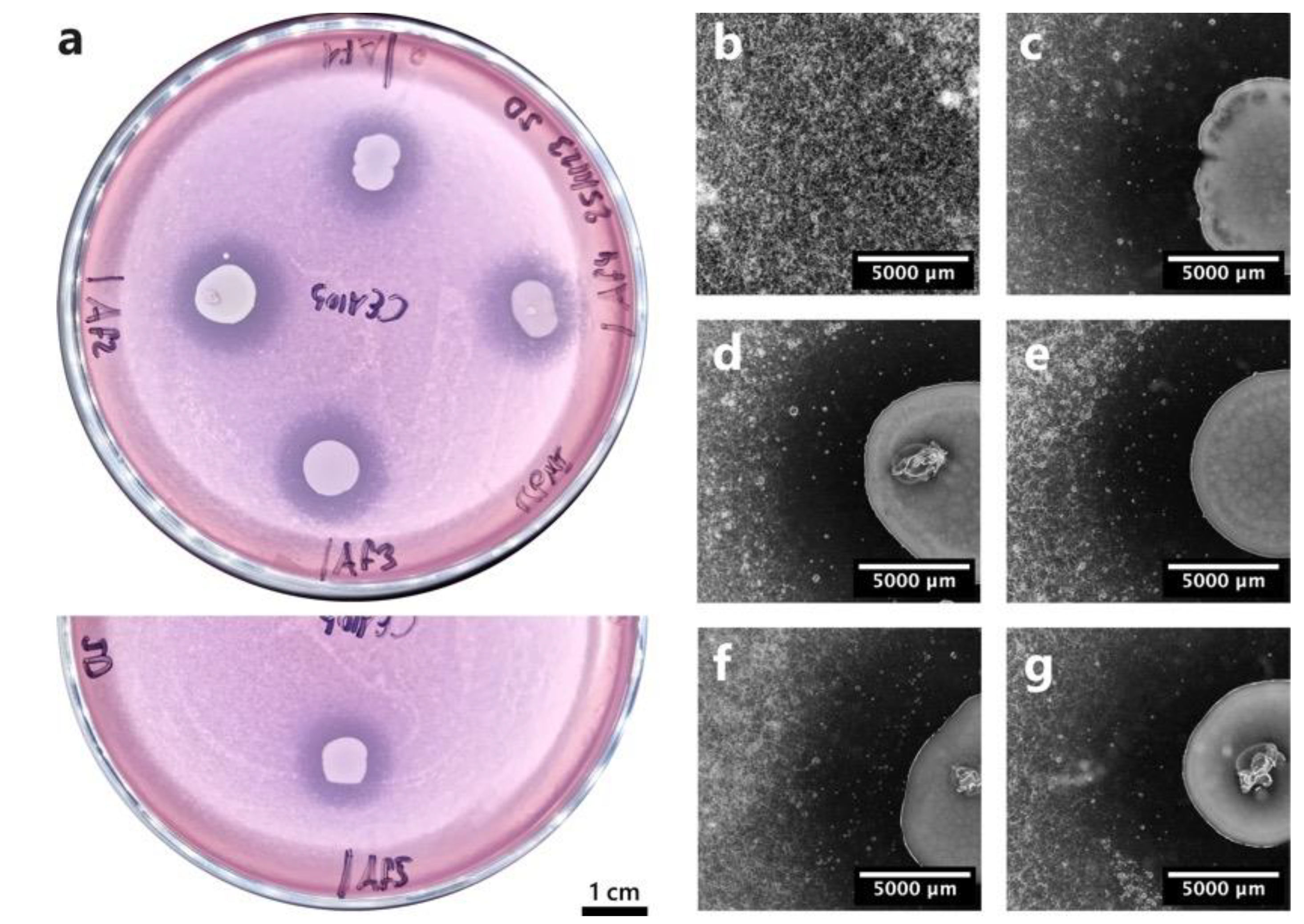
3.2. Soft agar inhibition assay
3.3. High-Troughput screening assay
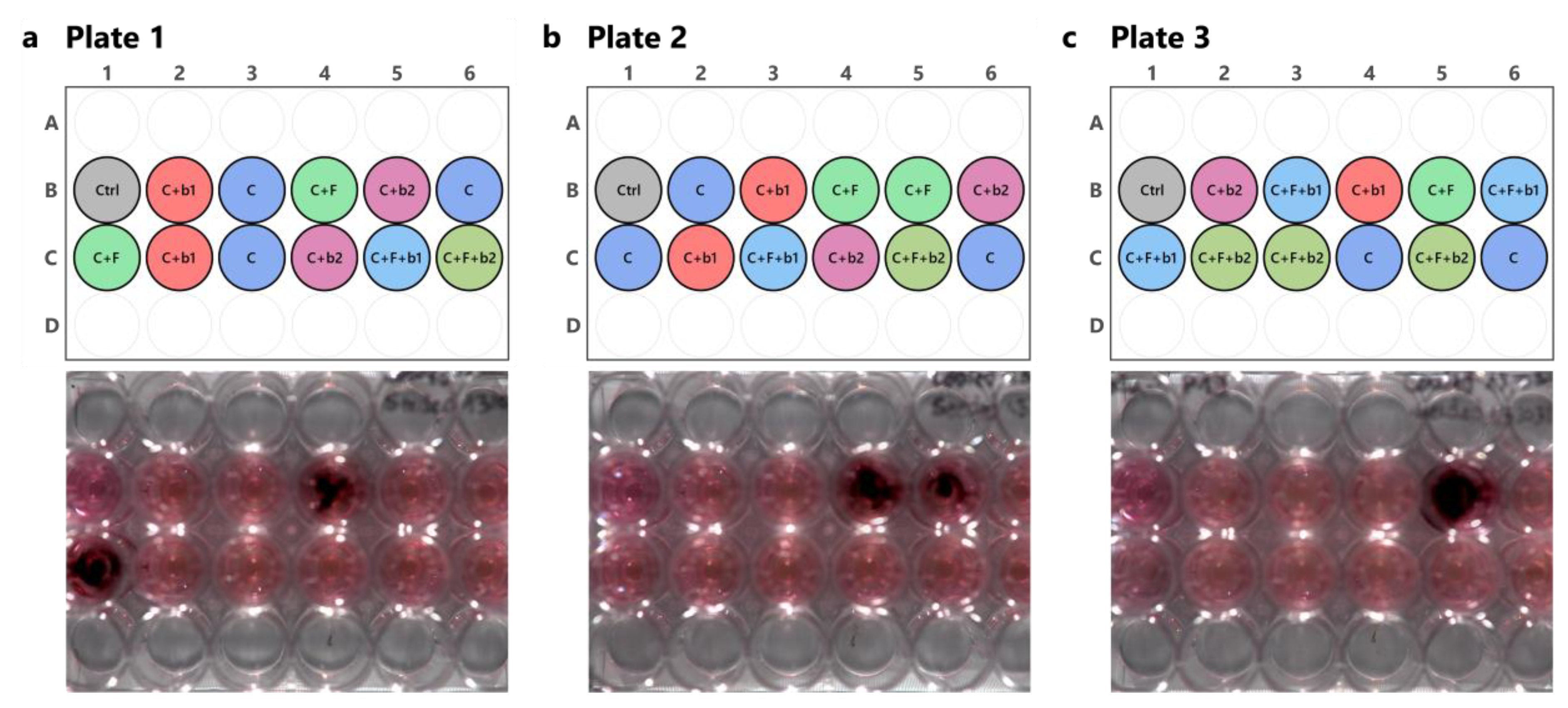
3.4. In-vitro confrontation of A. fumigatus with BALF P. aeruginosa isolates on Calu-3 cell monolayers
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bongomin, F., et al., Global and multi-national prevalence of fungal diseases—estimate precision. Journal of Fungi, 2017. 3. [CrossRef]
- Paulussen, C., et al., Ecology of aspergillosis: insights into the pathogenic potency of Aspergillus fumigatus and some other Aspergillus species. Microbial Biotechnology, 2017. 10(2): p. 296-322.
- Latgé, J.P. and G. Chamilos, Aspergillus fumigatus and Aspergillosis in 2019. Clin Microbiol Rev, 2019. 33(1).
- Gago, S., D.W. Denning, and P. Bowyer, Pathophysiological aspects of Aspergillus colonization in disease. Medical Mycology, 2019. 57(2): p. S219-S227.
- WHO fungal priority pathogens list to guide research, development and public health action. 2022.
- Arastehfar, A., et al., Aspergillus fumigatus and aspergillosis: From basics to clinics. Studies in Mycology, 2021. 100: p. 100115.
- Palmieri, F., et al., Recent Advances in Fungal Infections: From Lung Ecology to Therapeutic Strategies With a Focus on Aspergillus spp. Frontiers in Medicine, 2022. 9. [CrossRef]
- Ortiz, S.C., et al., Novel Insights into Aspergillus fumigatus Pathogenesis and Host Response from State-of-the-Art Imaging of Host-Pathogen Interactions during Infection. Journal of Fungi, 2022. 8(3): p. 264. [CrossRef]
- Barda, O., et al., The pH-Responsive Transcription Factor PacC Governs Pathogenicity and Ochratoxin A Biosynthesis in Aspergillus carbonarius. Frontiers in Microbiology, 2020. 11.
- Bertuzzi, M., et al., The pH-responsive PacC transcription factor of Aspergillus fumigatus governs epithelial entry and tissue invasion during pulmonary aspergillosis. PLoS Pathogens, 2014. 10(10): p. e1004413.
- Bignell, E., et al., The Aspergillus pH-responsive transcription factor PacC regulates virulence. Molecular Microbiology, 2005. 55(4): p. 1072-84.
- Blatzer, M., et al., SidL, an Aspergillus fumigatus transacetylase involved in biosynthesis of the siderophores ferricrocin and hydroxyferricrocin. Applied and Environmental Microbiology, 2011. 77(14): p. 4959-66. [CrossRef]
- Haas, H., M. Eisendle, and B.G. Turgeon, Siderophores in fungal physiology and virulence. Annual Review of Phytopathology, 2008. 46: p. 149-87. [CrossRef]
- Gresnigt, M.S., et al., Reducing hypoxia and inflammation during invasive pulmonary aspergillosis by targeting the Interleukin-1 receptor. Scientific Reports, 2016. 6(1): p. 26490. [CrossRef]
- Hérivaux, A., et al., Lung microbiota predict invasive pulmonary aspergillosis and its outcome in immunocompromised patients. Thorax, 2021. [CrossRef]
- Ao, Z., et al., Clinical characteristics, diagnosis, outcomes and lung microbiome analysis of invasive pulmonary aspergillosis in the community-acquired pneumonia patients. BMJ Open Respir Res, 2023. 10(1). [CrossRef]
- Kolwijck, E. and F.L. van de Veerdonk, The potential impact of the pulmonary microbiome on immunopathogenesis of Aspergillus-related lung disease. European Journal of Immunology, 2014. 44(11): p. 3156-3165.
- Bowyer, P., et al., Telomere-to-telomere genome sequence of the model mould pathogen Aspergillus fumigatus. Nature Communications, 2022. 13(1): p. 5394. [CrossRef]
- Resendiz-Sharpe, A., et al., Longitudinal multimodal imaging-compatible mouse model of triazole-sensitive and -resistant invasive pulmonary aspergillosis. Dis Model Mech, 2022. 15(3). [CrossRef]
- Das, S., et al., A prevalent and culturable microbiota links ecological balance to clinical stability of the human lung after transplantation. 2020: p. 0-3. [CrossRef]
- Brock, M., et al., Bioluminescent Aspergillus fumigatus, a new tool for drug efficiency testing and in vivo monitoring of invasive aspergillosis. Applied and Environmental Microbiology, 2008. 74(22): p. 7023-7035. [CrossRef]
- Muyzer, G., et al., Phylogenetic relationships of Thiomicrospira species and their identification in deep-sea hydrothermal vent samples by denaturing gradient gel-electrophoresis of 16S rDNA fragments. Archives of Microbiology, 1995. 164(3): p. 165-172.
- Altschul, S.F., et al., Basic local alignment search tool. J Mol Biol, 1990. 215(3): p. 403-10.
- Arefin, A., et al., Micromachining of Polyurethane Membranes for Tissue Engineering Applications. ACS Biomaterials Science and Engineering, 2018. 4(10): p. 3522-3533. [CrossRef]
- Quast, J., ggplate: Create Layout Plots of Biological Culture Plates and Microplates. 2023.
- Anith, K.N., N.S. Nysanth, and C. Natarajan, Novel and rapid agar plate methods for in vitro assessment of bacterial biocontrol isolates’ antagonism against multiple fungal phytopathogens. Letters in Applied Microbiology, 2021. 73(2): p. 229-236. [CrossRef]
- Muñoz, C.Y., et al., Biocontrol properties from phyllospheric bacteria isolated from Solanum lycopersicum and Lactuca sativa and genome mining of antimicrobial gene clusters. BMC Genomics, 2022. 23(1): p. 152. [CrossRef]
- Estoppey, A., et al., Improved methods to assess the effect of bacteria on germination of fungal spores. FEMS Microbiology Letters, 2022. 369(1). [CrossRef]
- Verburg, K., et al., Novel Treatment Approach for Aspergilloses by Targeting Germination. J Fungi (Basel), 2022. 8(8). [CrossRef]
- Stjärne Aspelund, A., et al., Microbiological findings in bronchoalveolar lavage fluid from lung transplant patients in Sweden. Transpl Infect Dis, 2018. 20(6): p. e12973. [CrossRef]
- Zeglen, S., et al., Frequency of Pseudomonas aeruginosa colonizations/infections in lung transplant recipients. Transplant Proc, 2009. 41(8): p. 3222-4. [CrossRef]
- Okamoto, K. and C.A.Q. Santos, Management and prophylaxis of bacterial and mycobacterial infections among lung transplant recipients. Ann Transl Med, 2020. 8(6): p. 413. [CrossRef]
- Patel, T.S., et al., Antifungal Prophylaxis in Lung Transplant Recipients. Transplantation, 2016. 100(9): p. 1815-26. [CrossRef]
- Moore, G.E., R.E. Gerner, and H.A. Franklin, Culture of Normal Human Leukocytes. JAMA, 1967. 199(8): p. 519-524.
- Ruhluel, D., et al., Development of liquid culture media mimicking the conditions of sinuses and lungs in cystic fibrosis and health [version 2; peer review: 2 approved]. F1000Research, 2022. 11(1007).
- Sass, G., et al., Aspergillus-Pseudomonas interaction, relevant to competition in airways. Med Mycol, 2019. 57(Supplement_2): p. S228-S232.
- Margalit, A., J.C. Carolan, and K. Kavanagh, Bacterial Interactions with Aspergillus fumigatus in the Immunocompromised Lung. Microorganisms, 2021. 9(2). [CrossRef]
- Keown, K., et al., Coinfection with Pseudomonas aeruginosa and Aspergillus fumigatus in cystic fibrosis. European Respiratory Review, 2020. 29(158): p. 200011.
- Wood, S.J., et al., Pseudomonas aeruginosa Cytotoxins: Mechanisms of Cytotoxicity and Impact on Inflammatory Responses. Cells, 2023. 12(1). [CrossRef]
- Li, J., et al., Pseudomonas aeruginosa Exoprotein-Induced Barrier Disruption Correlates With Elastase Activity and Marks Chronic Rhinosinusitis Severity. Frontiers in Cellular and Infection Microbiology, 2019. 9. [CrossRef]
- Srinivasan, B., et al., TEER Measurement Techniques for In Vitro Barrier Model Systems. Journal of Laboratory Automation, 2015. 20(2): p. 107-126. [CrossRef]
| ID | Strain name | Reference |
|---|---|---|
| CEA10 | Aspergillus fumigatus CEA10, WT | [18] |
| CEA10b | Aspergillus fumigatus CEA10, red-shifted bioluminescent strain | [19] |
| ID | Antibacterial exposure | Antibacterial/Antifungal drugs administered |
|---|---|---|
| BAL1 | yes | Colifin (colistimethate) |
| BAL2 | yes | Bactrim (sulfamethoxazole + trimethoprim) |
| BAL3 | yes | Colifin (colistimethate) |
| BAL4 | no | Ambisome (amphotericin B) |
| BAL5 | no | Ambisome (amphotericin B) |
| Strain code | Species | Query cover | Per. ident |
|---|---|---|---|
| b1 | Pseudomonas aeruginosa | 100% | 99.21% |
| b2 | Pseudomonas aeruginosa | 100% | 97.40% |
| b3 | Pseudomonas aeruginosa | 100% | 98.64% |
| b4 | Pseudomonas aeruginosa | 100% | 98.72% |
| b5 | Pseudomonas aeruginosa | 100% | 99.48% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




