Submitted:
26 May 2023
Posted:
29 May 2023
You are already at the latest version
Abstract
Keywords:
Introduction
Results
Activated CD8+ T lymphocytes most strongly stimulate MDA-MB-231 breast cancer cells to pass the BBB
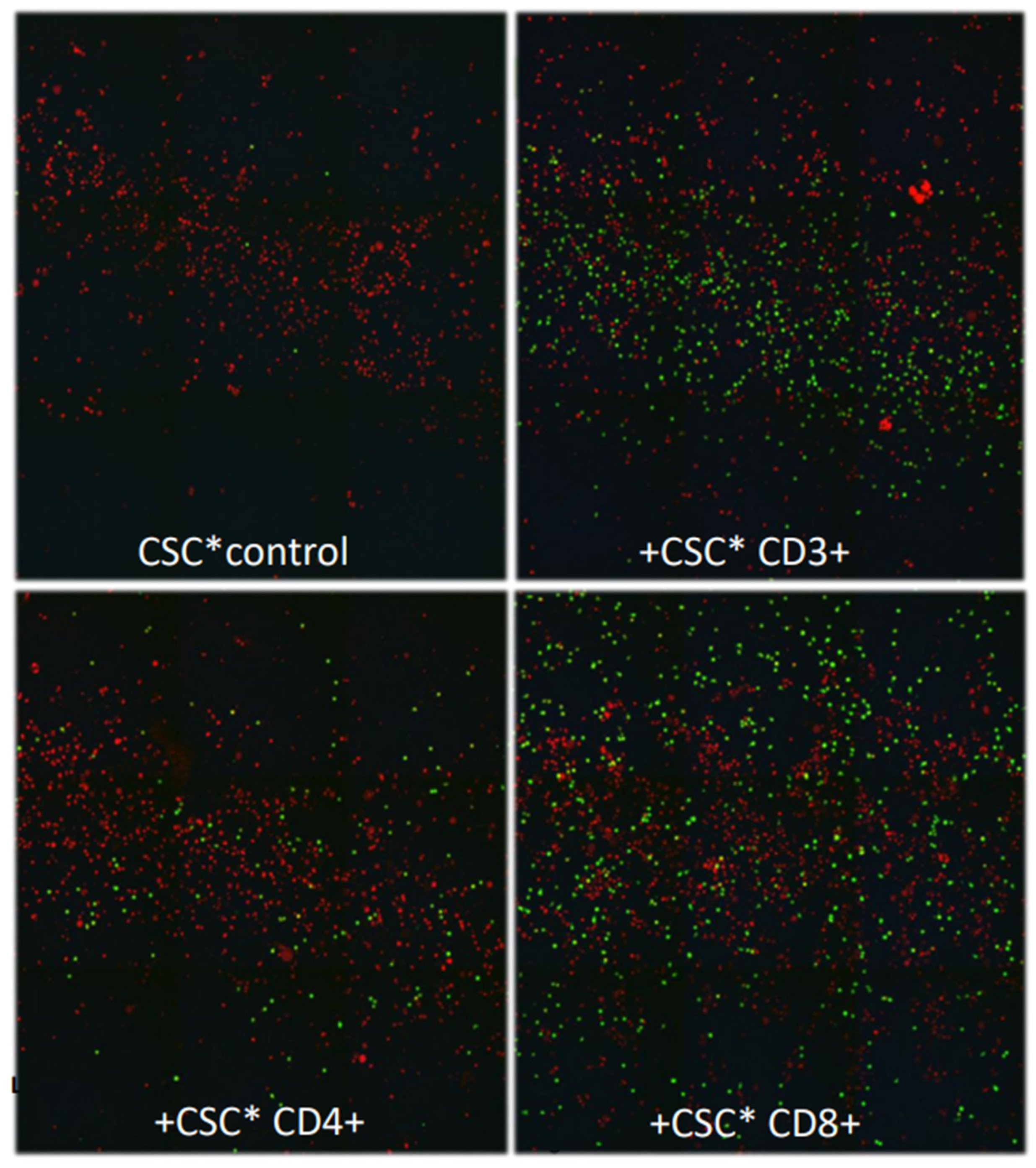
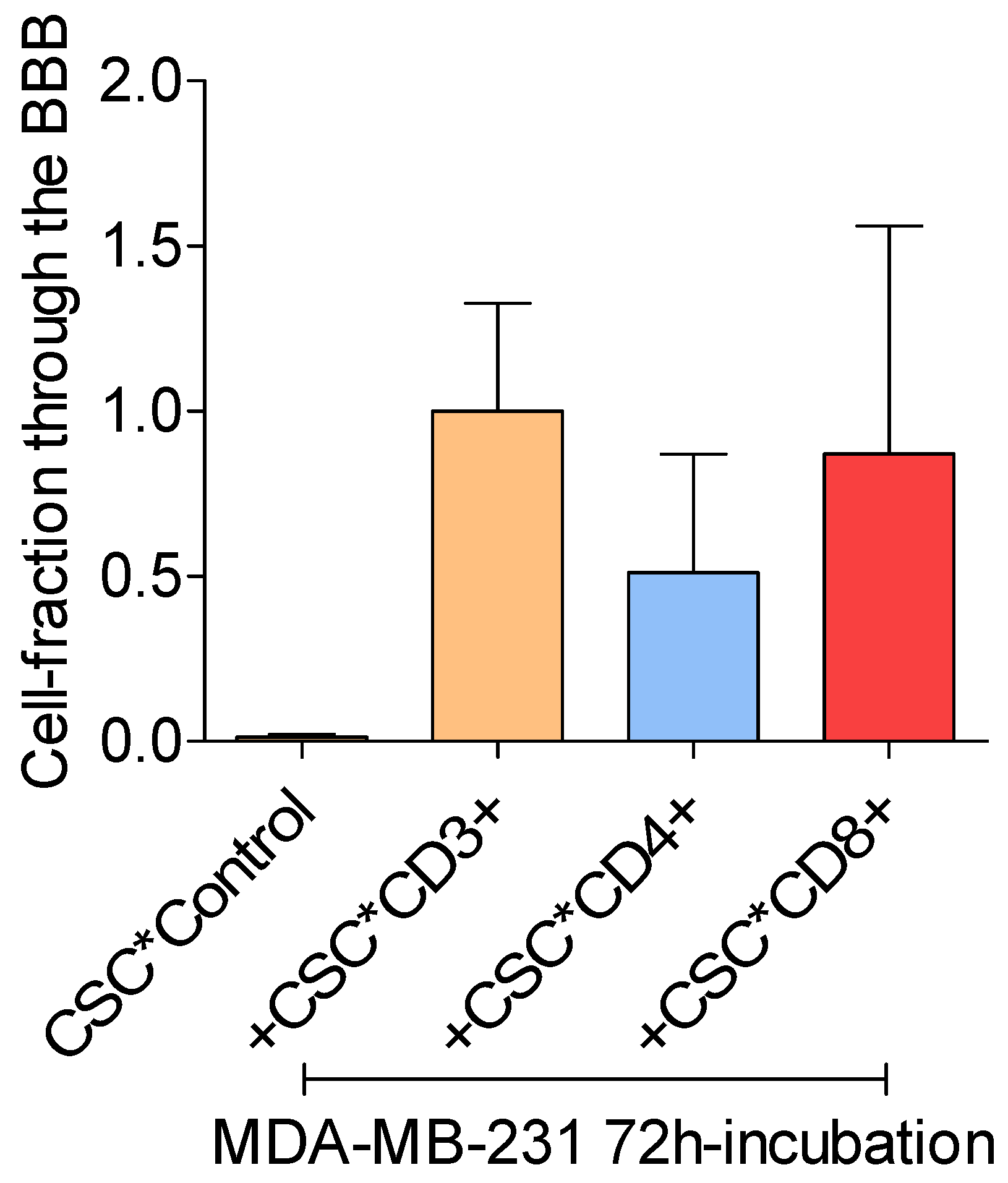
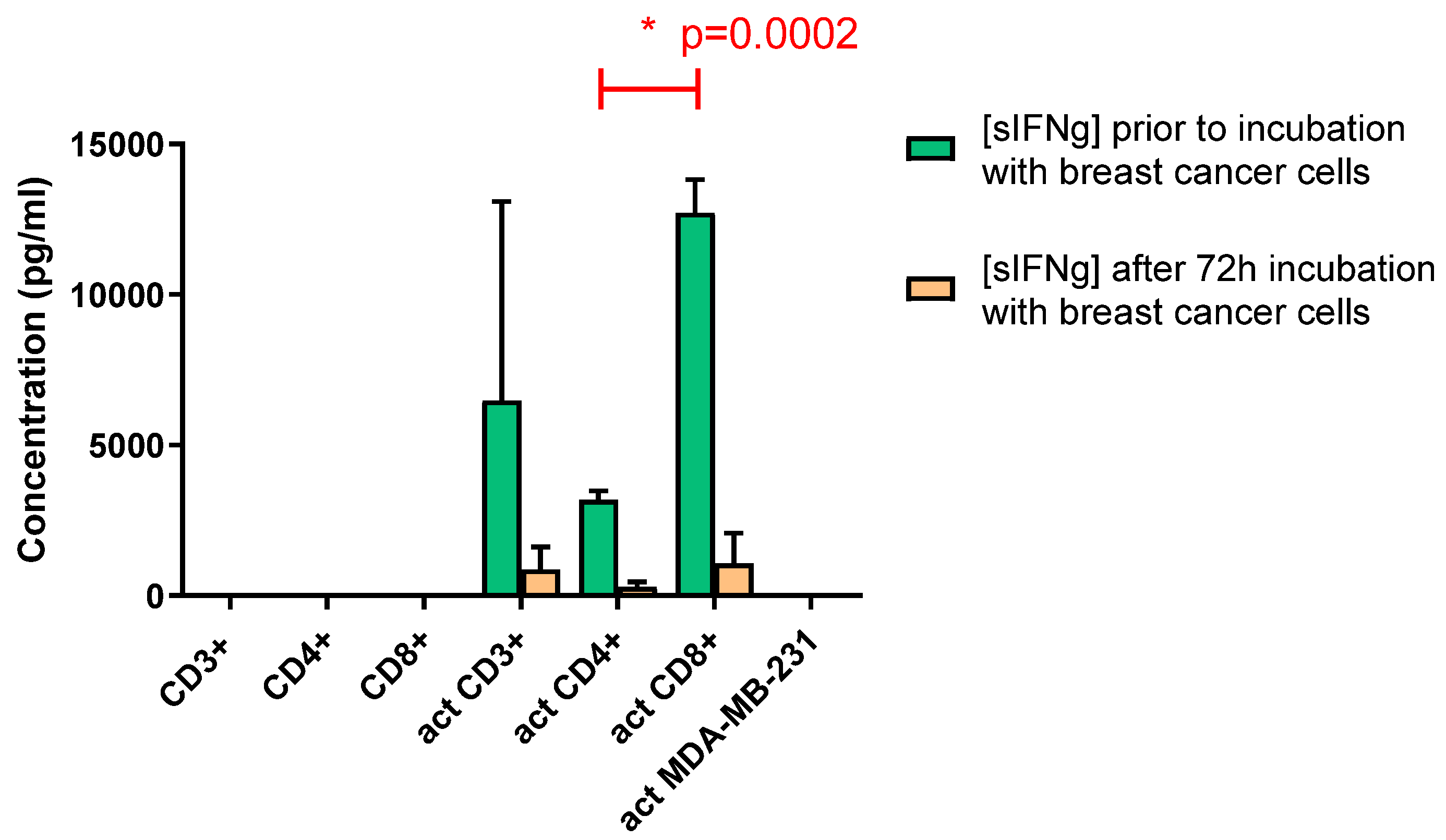
T cell secreted IFN-γ
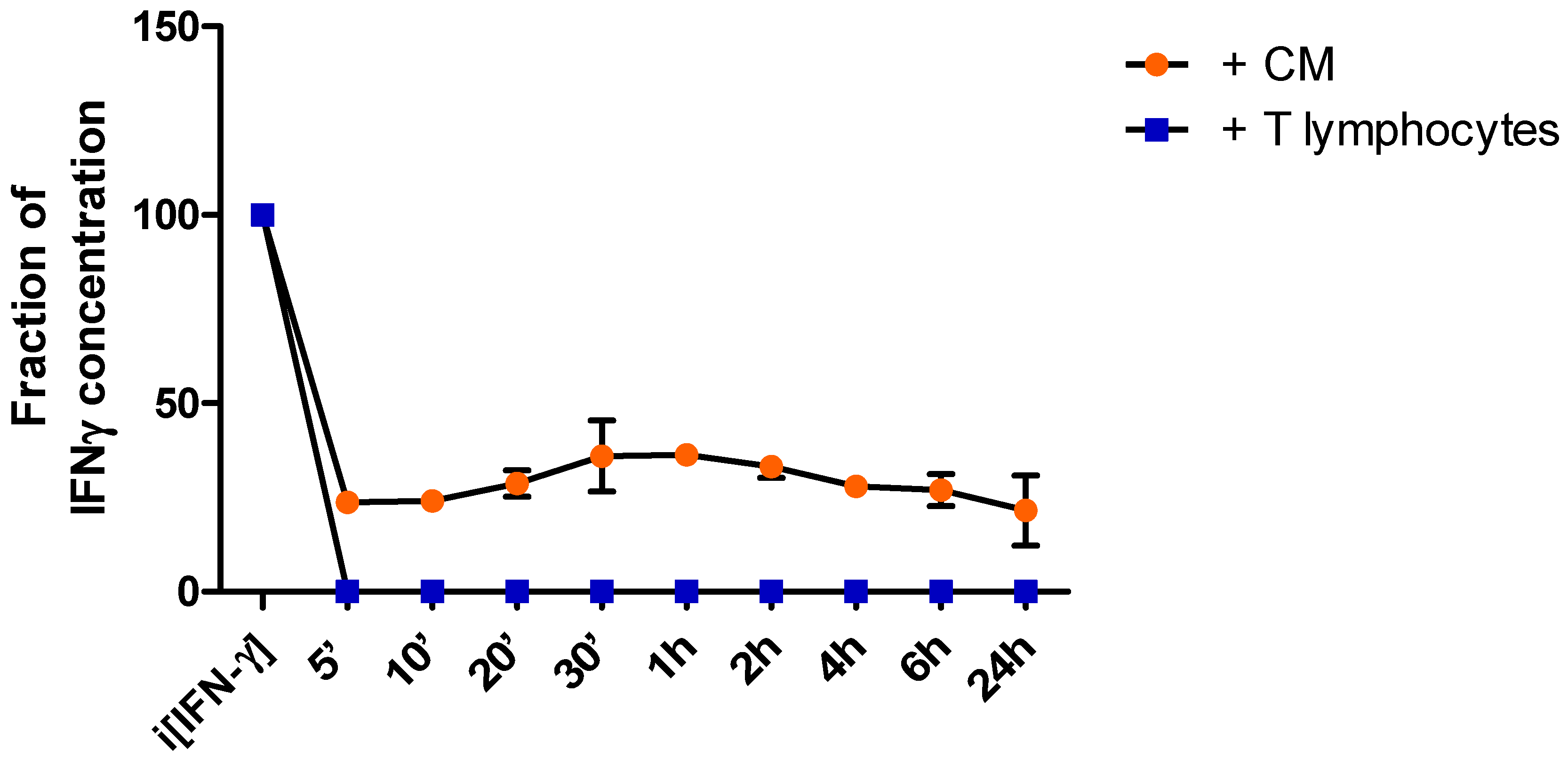
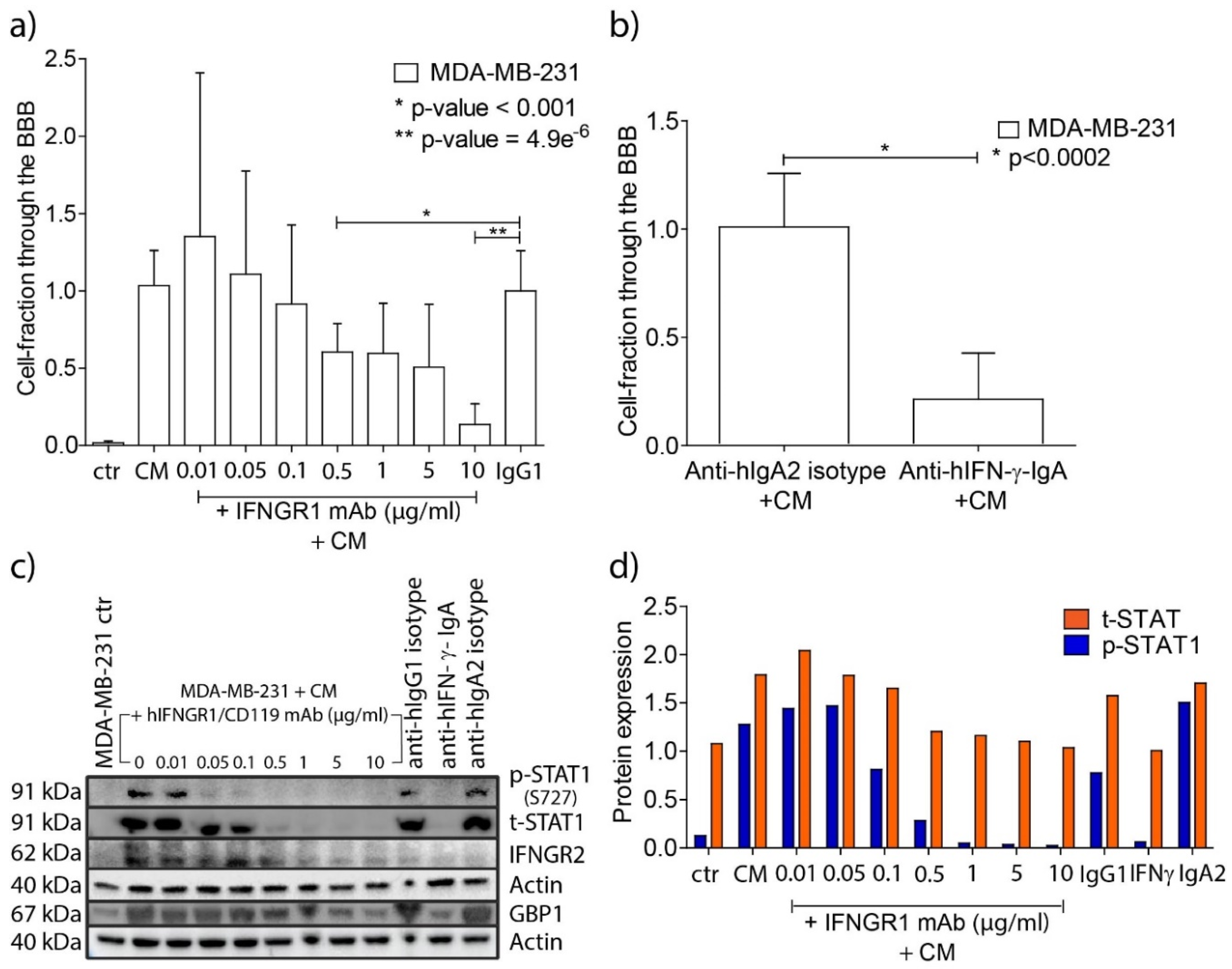
IFN-γ neutralization and IFNGR1 receptor blocking in the breast cancer cells impairs passing the BBB
IFN-γ, CXCL9, -10, -11 and CXCR3 are overexpressed in primary breast cancer samples of patients who developed brain metastasis
Discussion
Materials and Methods
T lymphocyte fluorescence-activated flowcytometric cell sorting
Activation and Expansion of T lymphocytes
Cell line culture procedure
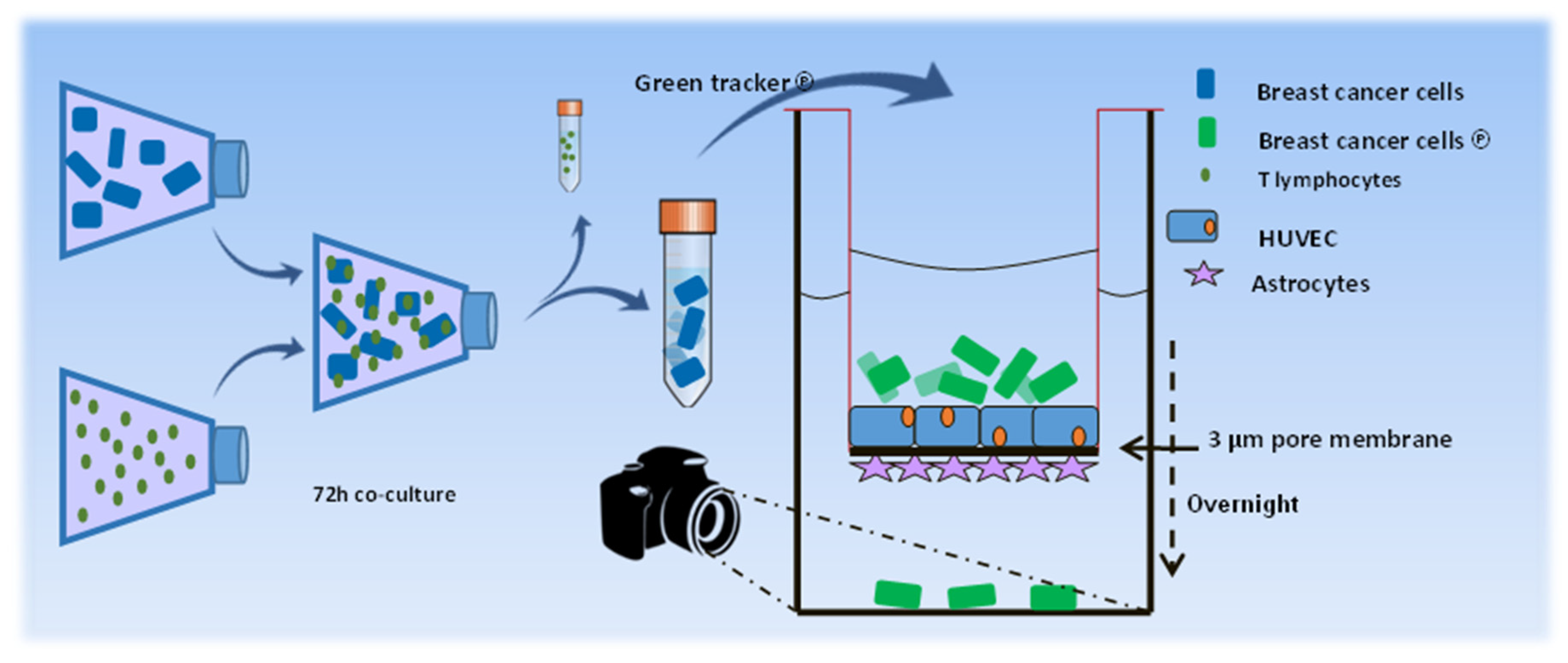
Construction of the in vitro blood-brain-barrier (BBB) model
Measuring the transmigration of breast cancer cells through the in vitro BBB
IFN-γ signaling pathway blocking and inhibition experiments
Reverse transcription-polymerase chain reaction (RT-PCR)
Western blot
Re-analysis of the gene expression profile data
Statistical analysis
Conclusions
Supplementary Materials
Author Contributions
Funding
References
- Leone, J.P. and B.A. Leone, Breast cancer brain metastases: the last frontier. Exp Hematol Oncol, 2015. 4: p. 33. [CrossRef]
- Sperduto, P.W., et al., Effect of tumor subtype on survival and the graded prognostic assessment for patients with breast cancer and brain metastases. Int J Radiat Oncol Biol Phys, 2012. 82(5): p. 2111-7. [CrossRef]
- Tham, Y.L., et al., Primary breast cancer phenotypes associated with propensity for central nervous system metastases. Cancer, 2006. 107(4): p. 696-704. [CrossRef]
- Kaplan, M.A., et al., Biological subtypes and survival outcomes in breast cancer patients with brain metastases (study of the Anatolian Society of Medical Oncology). Oncology, 2012. 83(3): p. 141-50. [CrossRef]
- Nam, B.H., et al., Breast cancer subtypes and survival in patients with brain metastases. Breast Cancer Res, 2008. 10(1): p. R20. [CrossRef]
- Sperduto, P.W., et al., The effect of tumor subtype on the time from primary diagnosis to development of brain metastases and survival in patients with breast cancer. J Neurooncol, 2013. 112(3): p. 467-72. [CrossRef]
- Pedrosa, R., et al., Breast cancer brain metastasis: molecular mechanisms and directions for treatment. Neuro Oncol, 2018. 20(11): p. 1439-1449. [CrossRef]
- Mustafa, D.A.M., et al., T lymphocytes facilitate brain metastasis of breast cancer by inducing Guanylate-Binding Protein 1 expression. Acta Neuropathol, 2018. [CrossRef]
- Prakash, B., et al., Structure of human guanylate-binding protein 1 representing a unique class of GTP-binding proteins. Nature, 2000. 403(6769): p. 567-71. [CrossRef]
- Kim, B.H., et al., Interferon-induced guanylate-binding proteins in inflammasome activation and host defense. Nat Immunol, 2016. 17(5): p. 481-9. [CrossRef]
- Riaz, M., et al., Growth and metastatic behavior of molecularly well-characterized human breast cancer cell lines in mice. Breast Cancer Res Treat, 2014. 148(1): p. 19-31. [CrossRef]
- Eugenin, E.A. and J.W. Berman, Chemokine-dependent mechanisms of leukocyte trafficking across a model of the blood-brain barrier. Methods, 2003. 29(4): p. 351-61. [CrossRef]
- Farrar, M.A. and R.D. Schreiber, The molecular cell biology of interferon-gamma and its receptor. Annu Rev Immunol, 1993. 11: p. 571-611. [CrossRef]
- Bach, E.A., M. Aguet, and R.D. Schreiber, The IFN gamma receptor: a paradigm for cytokine receptor signaling. Annu Rev Immunol, 1997. 15: p. 563-91. [CrossRef]
- Barnholt, K.E., et al., Adenosine blocks IFN-gamma-induced phosphorylation of STAT1 on serine 727 to reduce macrophage activation. J Immunol, 2009. 183(10): p. 6767-77. [CrossRef]
- Decker, T. and P. Kovarik, Serine phosphorylation of STATs. Oncogene, 2000. 19(21): p. 2628-37. [CrossRef]
- Aaltomaa, S., et al., Lymphocyte infiltrates as a prognostic variable in female breast cancer. Eur J Cancer, 1992. 28A(4-5): p. 859-64. [CrossRef]
- Carlomagno, C., et al., Prognostic significance of necrosis, elastosis, fibrosis and inflammatory cell reaction in operable breast cancer. Oncology, 1995. 52(4): p. 272-7. [CrossRef]
- Lee, A.H., et al., Different patterns of inflammation and prognosis in invasive carcinoma of the breast. Histopathology, 2006. 48(6): p. 692-701. [CrossRef]
- Marrogi, A.J., et al., Study of tumor infiltrating lymphocytes and transforming growth factor-beta as prognostic factors in breast carcinoma. Int J Cancer, 1997. 74(5): p. 492-501. [CrossRef]
- Mahmoud, S.M., et al., Tumor-infiltrating CD8+ lymphocytes predict clinical outcome in breast cancer. J Clin Oncol, 2011. 29(15): p. 1949-55. [CrossRef]
- Hammerl, D., et al., Breast cancer genomics and immuno-oncological markers to guide immune therapies. Seminars in Cancer Biology, 2018. 52: p. 178-188. [CrossRef]
- Morvan, P.Y., et al., Distinct pattern of IL-2 and IFN-gamma gene expression in CD4 and CD8 T cells: cytofluorometric analysis at a single cell level using non-radioactive probes. Cell Mol Biol (Noisy-le-grand), 1995. 41(7): p. 945-57.
- Seder, R.A., et al., CD8+ T cells can be primed in vitro to produce IL-4. J Immunol, 1992. 148(6): p. 1652-6. [CrossRef]
- Erard, F., et al., Switch of CD8 T cells to noncytolytic CD8-CD4- cells that make TH2 cytokines and help B cells. Science, 1993. 260(5115): p. 1802-5. [CrossRef]
- Pearce, E.L., et al., Control of effector CD8+ T cell function by the transcription factor Eomesodermin. Science, 2003. 302(5647): p. 1041-3. [CrossRef]
- Schroder, K., et al., Interferon-gamma: an overview of signals, mechanisms and functions. J Leukoc Biol, 2004. 75(2): p. 163-89. [CrossRef]
- Ni, C., et al., Accelerated tumour metastasis due to interferon-gamma receptor-mediated dissociation of perivascular cells from blood vessels. J Pathol, 2017. 242(3): p. 334-346. [CrossRef]
- Xiao, M., et al., IFNgamma promotes papilloma development by up-regulating Th17-associated inflammation. Cancer Res, 2009. 69(5): p. 2010-7. [CrossRef]
- Singh, S., et al., Loss of ELF5-FBXW7 stabilizes IFNGR1 to promote the growth and metastasis of triple-negative breast cancer through interferon-gamma signalling. Nat Cell Biol, 2020. 22(5): p. 591-602. [CrossRef]
- Der, S.D., et al., Identification of genes differentially regulated by interferon alpha, beta, or gamma using oligonucleotide arrays. Proc Natl Acad Sci U S A, 1998. 95(26): p. 15623-8. [CrossRef]
- Rossi, D. and A. Zlotnik, The biology of chemokines and their receptors. Annu Rev Immunol, 2000. 18: p. 217-42. [CrossRef]
- Lew, D.J., T. Decker, and J.E. Darnell, Jr., Alpha interferon and gamma interferon stimulate transcription of a single gene through different signal transduction pathways. Mol Cell Biol, 1989. 9(12): p. 5404-11. [CrossRef]
- Darnell, J.E., Jr., I.M. Kerr, and G.R. Stark, Jak-STAT pathways and transcriptional activation in response to IFNs and other extracellular signaling proteins. Science, 1994. 264(5164): p. 1415-21. [CrossRef]
- Zhao, J., et al., Oncogenic Role of Guanylate Binding Protein 1 in Human Prostate Cancer. Front Oncol, 2019. 9: p. 1494. [CrossRef]
- Bai, S., T. Chen, and X. Deng, Guanylate-Binding Protein 1 Promotes Migration and Invasion of Human Periodontal Ligament Stem Cells. Stem Cells Int, 2018. 2018: p. 6082956. [CrossRef]
- Salmaggi, A., et al., Expression and modulation of IFN-gamma-Inducible chemokines (IP-10, Mig, and I-TAC) in human brain endothelium and astrocytes: Possible relevance for the immune invasion of the central nervous system and the pathogenesis of multiple sclerosis. Journal of Interferon and Cytokine Research, 2002. 22(6): p. 631-640. [CrossRef]
- Banisadr, G., et al., Chemokines and brain functions. Curr Drug Targets Inflamm Allergy, 2005. 4(3): p. 387-99. [CrossRef]
- Muller, M., et al., Review: The chemokine receptor CXCR3 and its ligands CXCL9, CXCL10 and CXCL11 in neuroimmunity--a tale of conflict and conundrum. Neuropathol Appl Neurobiol, 2010. 36(5): p. 368-87. [CrossRef]
- Ramesh, G., A.G. MacLean, and M.T. Philipp, Cytokines and Chemokines at the Crossroads of Neuroinflammation, Neurodegeneration, and Neuropathic Pain. Mediators of Inflammation, 2013. [CrossRef]
- Weenink, B., et al., Low-grade glioma harbors few CD8 T cells, which is accompanied by decreased expression of chemo-attractants, not immunogenic antigens. Sci Rep, 2019. 9(1): p. 14643. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



