Submitted:
24 May 2023
Posted:
25 May 2023
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and methods
Animals
Collection of oocytes and cumulus cells
Blood and tissue collection
Telomere length measurement
Quantitative reverse-transcription polymerase chain reaction (qRT-PCR)
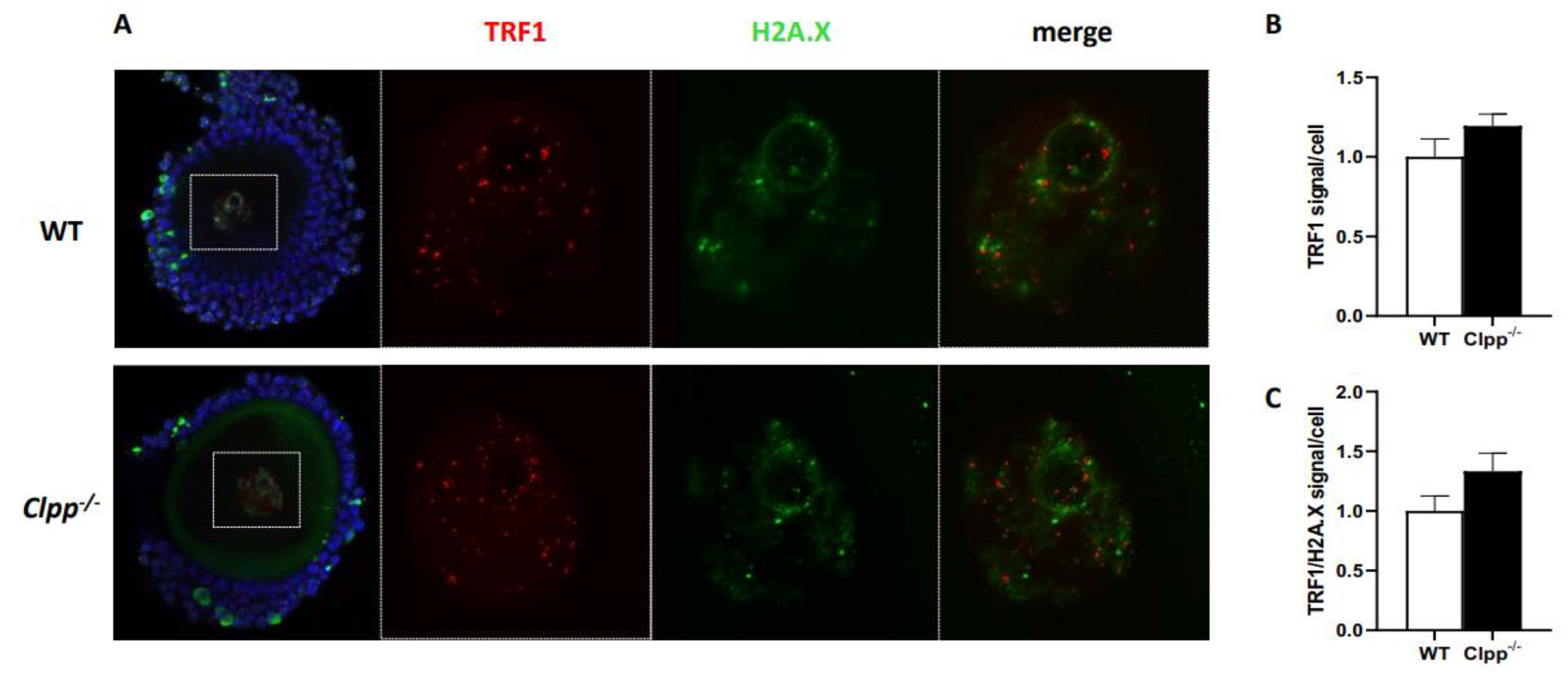
Immunofluorescent staining
Statistical analysis
Results
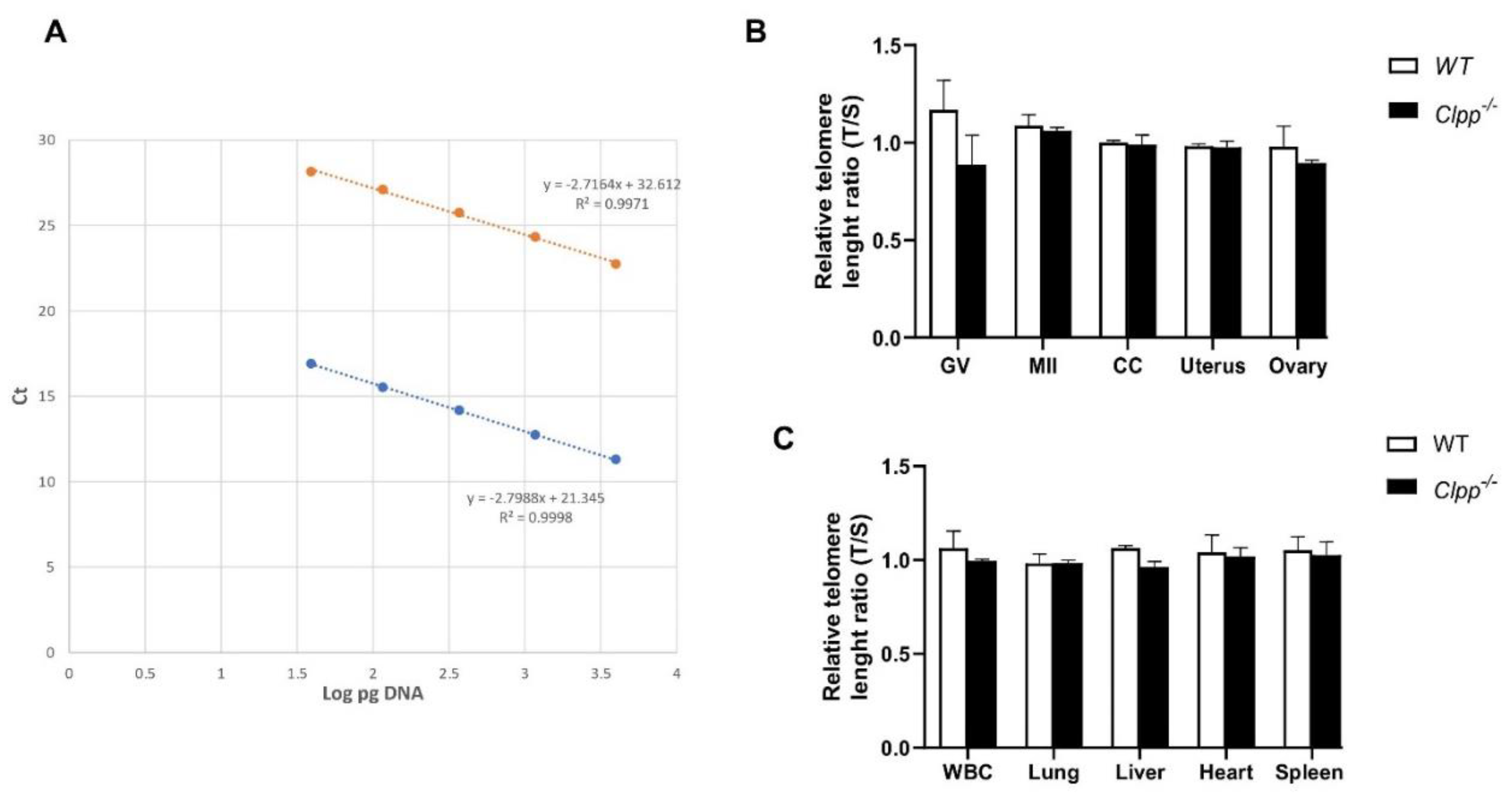
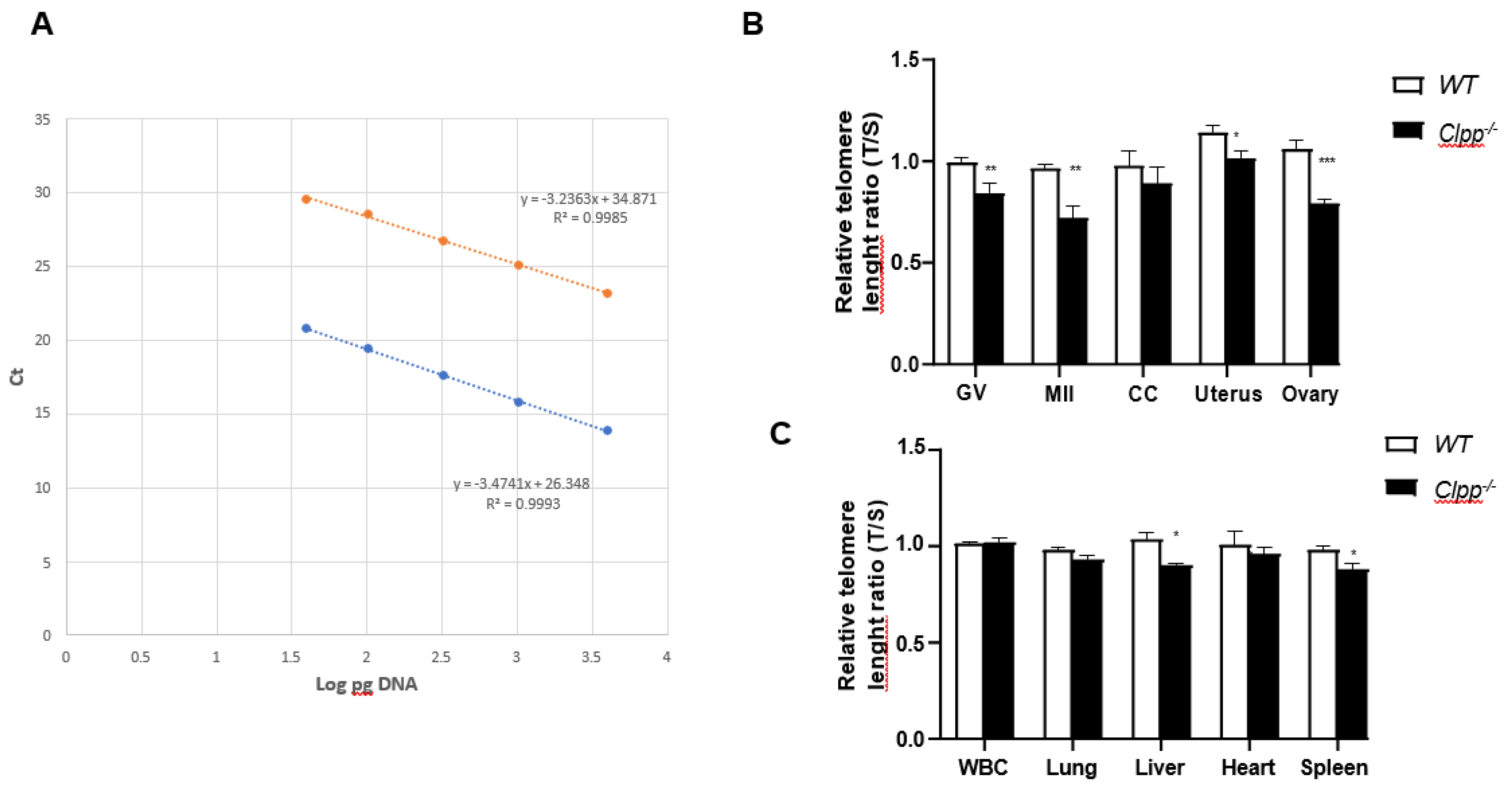
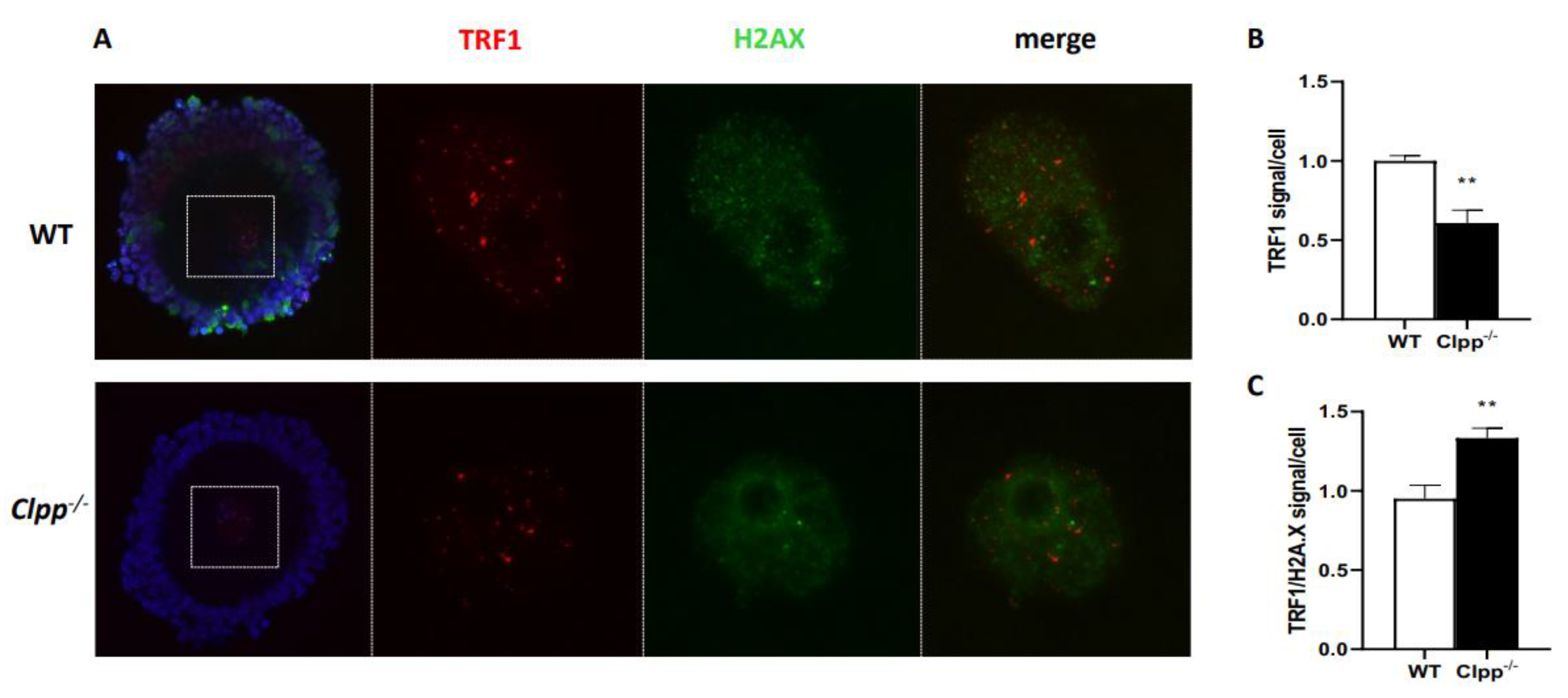
Telomere length in Clpp-/- mice oocytes, ovaries and somatic cells and tissues
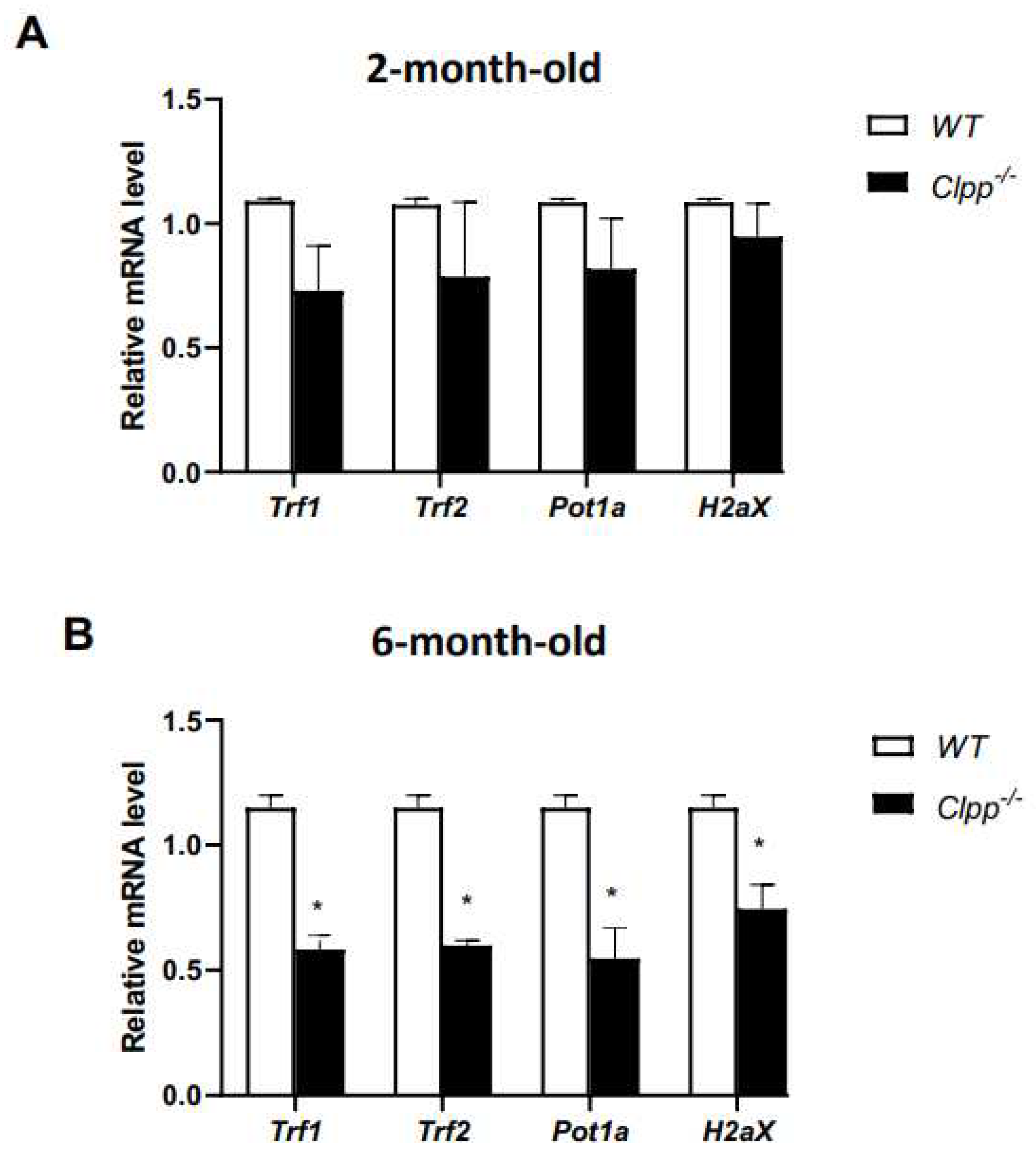
Expression of telomere-associated genes in Clpp -/- oocytes
Discussion
Funding
Conflicts of Interest
References
- Esencan, E.; Simsek, B.; Seli, E. Analysis of female demographics in the United States: life expectancy, education, employment, family building decisions, and fertility service utilization. Curr Opin Obstet Gynecol 2021, 33, 170–177. [Google Scholar] [CrossRef]
- Sweeney, M.M.; Raley, R.K. Race, ethnicity, and the changing context of childbearing in the United States. Annu Rev Sociol 2014, 40, 539–558. [Google Scholar] [CrossRef]
- Bernadotte, A.; V. M., M.; Spivak, I.M. Markers of cellular senescence. Telomere shortening as a marker of cellular senescence. Aging 2016, 8, 3–11. [Google Scholar] [CrossRef]
- Kasapoğlu, I.; Seli, E. Mitochondrial dysfunction and ovarian aging. Endocrinology 2020, 161. [Google Scholar] [CrossRef]
- Wang, T.; Babayev, E.; Jiang, Z.; Li, G.; Zhang, M.; Esencan, E.; Horvath, T.; Seli, E. Mitochondrial unfolded protein response gene Clpp is required to maintain ovarian follicular reserve during aging, for oocyte competence, and development of pre-implantation embryos. Aging Cell 2018, 30, e12784. [Google Scholar] [CrossRef]
- Barnes, R.P.; Fouquerel, E.; Opresko, P.L. The impact of oxidative DNA damage and stress on telomere homeostasis. Mech Ageing Dev 2019, 177, 37–45. [Google Scholar] [CrossRef]
- Victorelli, S.; Passos, J.F. Telomeres and Cell Senescence - Size Matters Not. EBioMedicine 2017, 21, 14–20. [Google Scholar] [CrossRef]
- Derevyanko, A.; Skowronska, A.; Skowronski, M.T.; Kordowitzki, P. The Interplay between telomeres, mitochondria, and chronic stress exposure in the aging egg. Cells 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- van der Reest, J.; Nardini Cecchino, G.; Haigis, M.C.; Kordowitzki, P. Mitochondria: Their relevance during oocyte ageing. Ageing Res Rev 2021, 70, 101378. [Google Scholar] [CrossRef] [PubMed]
- Babayev, E.; Seli, E. Oocyte mitochondria function and reproduction. Curr Opin Obstet Gynecol 2015, 27, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Seli, E.; Wang, T.; Horvath, T.L. Mitochondrial unfolded protein response: a stress response with implications for fertility and reproductive aging. Fertil Steril 2019, 111, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Houtkooper, R.H.; Mouchiroud, L.; Ryu, D.; Moullan, N.; Katsyuba, E.; Knott, G.; Williams, R.W.; Auwerx, J. Mitonuclear protein imbalance as a conserved longevity mechanism. Nature 2013, 497, 451–457. [Google Scholar] [CrossRef]
- Durieux, J.; Wolff, S.; Dillin, A. The cell-non-autonomous nature of electron transport chain-mediated longevity. Cell.
- Moehle, E.A.; Shen, K.; Dillin, A. Mitochondrial proteostasis in the context of cellular and organismal health and aging. J Biol Chem 2019, 294, 5396–5407. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Bener, M.B.; Jiang, Z.; Wang, T.; Esencan, E. ; Scott R III; Horvath, T. ; Seli, E. Mitofusin 1 is required for female fertility and to maintain ovarian follicular reserve Cell Death Dis 2019, 10, 560. [Google Scholar] [PubMed]
- Zhang, M.; Bener, M.B.; Jiang, Z.; Wang, T.; Esencan, E.; Scott, R., III; Horvath, T.; Seli, E. Mitofusin 2 plays a role in oocyte and follicle development, and is required to maintain ovarian follicular reserve during reproductive aging. Aging 2019, 11, 3919–3938. [Google Scholar] [CrossRef]
- Zhao, Q.; Wang, J.; Levichkin, I.V.; Stasinopoulos, S.; Ryan, M.T.; Hoogenraad, N.J. A mitochondrial specific stress response in mammalian cells. Embo J 2002, 21, 4411–4419. [Google Scholar] [CrossRef] [PubMed]
- Haynes, C.M.; Petrova, K.; Benedetti, C.; Yang, Y.; Ron, D. ClpP mediates activation of a mitochondrial unfolded protein response in C. elegans. Dev Cell 2007, 13, 467–480. [Google Scholar] [CrossRef]
- Al-Furoukh, N.; Ianni, A.; Nolte, H.; Hölper, S.; Krüger, M.; Wanrooij, S.; al. , e. ClpX stimulates the mitochondrial unfolded protein response (UPRmt) in mammalian cells. Biochim Biophys Acta 2015, 1853, 2580–2591. [Google Scholar] [CrossRef]
- Nargund, A.M.; Fiorese, C.J.; Pellegrino, M.W.; Deng, P.; Haynes, C.M. Mitochondrial and nuclear accumulation of the transcription factor ATFS-1 promotes OXPHOS recovery during the UPR(mt). Mol Cell 2015, 58, 123–133. [Google Scholar] [CrossRef]
- Gispert, S.; Parganlija, D.; Klinkenberg, M.; Dröse, S.; Wittig, I.; Mittelbronn, M.; Grzmil, P.; Koob, S.; Hamann, A.; Walter, M. , et al. Loss of mitochondrial peptidase Clpp leads to infertility, hearing loss plus growth retardation via accumulation of CLPX, mtDNA and inflammatory factors. Hum Mol Genet 2013, 22, 4871–4887. [Google Scholar] [CrossRef]
- Seli, E.; Lalioti, M.D.; Flaherty, S.M.; Sakkas, D.; Terzi, N.; Steitz, J.A. An embryonic poly(A)-binding protein (ePAB) is expressed in mouse oocytes and early preimplantation embryos. Proc Natl Acad Sci USA 2005, 102, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Martínez, P.; Flores, J.M.; Blasco, M.A. 53BP1 deficiency combined with telomere dysfunction activates ATR-dependent DNA damage response. J Cell Biol 2012, 197, 283–300. [Google Scholar] [CrossRef] [PubMed]
- Smogorzewska, A.; Steensel, B.; Bianchi, A.; Oelmann, S.; Schaefer, M.R.; Schnapp, G.; Lange, T. Control of Human Telomere Length by TRF1 and TRF2. Mol Cell Biol 2000, 20, 1659–1668. [Google Scholar] [CrossRef] [PubMed]
- Bonner, W.M.; West, M.H.; Stedman, J.D. Two-dimensional gel analysis of histones in acid extracts of nuclei, cells, and tissues. Eur J Biochem 1980, 109, 17–23. [Google Scholar] [CrossRef]
- Celeste, A.; Petersen, S.; Romanienko, P.J.; Fernandez-Capetillo, O.; Tang Chen, H.; Sedelnikova, O.A.; Reina-San-Martin, B.; Coppola, V.; Meffre, E.; Difilippantonio, M.J. , et al. Genomic instability in mice lacking histone H2AX. Science 2002, 296, 922–927. [Google Scholar] [CrossRef]
- Rogakou, E.P.; Pilch, D.R.; Orr, A.H.; Ivanova, V.S.; Bonner, W.M. DNA double-stranded breaks induce histone H2AX phosphorylation on serine 139. J Biol Chem 1998, 273, 5858–5868. [Google Scholar] [CrossRef]
- von Zglinicki, T. Oxidative stress shortens telomeres. Trends Biochem Sci 2002, 27, 339–344. [Google Scholar] [CrossRef]
- Kordowitzki, P.; Hamdi, M.; Derevyanko, A.; Rizos, D.; Blasco, M. The effect of rapamycin on bovine oocyte maturation success and metaphase telomere length maintenance. Aging 2020, 12, 7576–7584. [Google Scholar] [CrossRef]
- Chang, A.C.Y., Ong. Telomere shortening and metabolic compromise underlie dystrophic cardiomyopathy. Proc Natl Acad Sci U S A 2016, 113, 13120–13125. [Google Scholar] [CrossRef]
- Amano, H.; Chaudhury, A.; Rodriguez-Aguayo, C.; Lu, L.; Akhanov, V.; Catic, A.; Popov, Y.V.; Verdin, E.; Johnson, H.; Stossi, F. , et al. Telomere dysfunction induces sirtuin repression that drives telomere-dependent disease. Cell Metab 2019, 29, 1274–1290. [Google Scholar] [CrossRef]
- Butts, S.; Riethman, H.; Ratcliffe, S.; Shaunik, A.; Coutifaris, C.; Barnhart, K. Correlation of telomere length and telomerase activity with occult ovarian insufficiency. J Clinl Endocrinol Metab 2009, 94, 4835–4843. [Google Scholar] [CrossRef] [PubMed]
- Hande, M.; Samper, E.; Lansdorp, P.; Blasco, M.T. Telomere length dynamics and chromosomal instability in cells derived from telomerase null mice. J Cell Biol 1999, 144, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Garcia, C.K.; Wright, W.E.; Shay, J.W. Human diseases of telomerase dysfunction: insights into tissue aging. . Nucleic Acids Res 2007, 35, 7406–7416. [Google Scholar] [CrossRef]
- Calado, R.T.; Young, N.S. Telomere diseases. N Engl J Med 2009, 361, 2353–2365. [Google Scholar] [CrossRef] [PubMed]
- Carulli, L. Telomere shortening as genetic risk factor of liver cirrhosis. World J Gastroenterol 2015, 21, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Calado, R.T.; Brudno, J.; Mehta, P.; Kovacs, J.J.; Wu, C.; Zago, M.A.; Chanock, S.J.; Boyer, T.D.; Young, N.S. Constitutional telomerase mutations are genetic risk factors for cirrhosis. Hepatology 2011, 53, 1600–1607. [Google Scholar] [CrossRef]
- Kordowitzki, P. Oxidative stress induces telomere dysfunction and shortening in human oocytes of advanced age donors. Cells 2021, 10, 1866. [Google Scholar] [CrossRef] [PubMed]
- de La Roche Saint-André, C. Alternative ends: telomeres and meiosis. Biochimie 2008, 90, 181–189. [Google Scholar] [CrossRef]
- Morell, M.A.; Werling, U.; Edelmann, W.; Roberson, M.S.; Cohen, P.E. Analysis of meiotic prophase I in live mouse spermatocytes. Chromosome Res 2008, 6, 743–760. [Google Scholar] [CrossRef]
- Liu, L.; Franco, S.; Spyropoulos, B.; Moens, P.B.; Blasco, M.A.; Keefe, D.L. Irregular telomeres impair meiotic synapsis and recombination in mice. Proc Natl Acad Sci U S A 2004, 101, 6496–6501. [Google Scholar] [CrossRef]
- Keefe, D.L.; Franco, S.; Liu, L.; Trimarchi, J.; Cao, B.; Weitzen, S.; Agarwal, S.; Blasco, M.A. Telomere length predicts embryo fragmentation after in vitro fertilization in women--toward a telomere theory of reproductive aging in women. Am J Obstet Gynecol 2005, 192, 1256–1260. [Google Scholar] [CrossRef] [PubMed]
- Keefe, D.L.; Liu, L.; Marquard, K. Telomeres and aging-related meiotic dysfunction in women. Cell Mol Life Sci 2007, 64, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Keefe, D.L. Telomeres, reproductive aging, and genomic instability during early development. Reprod Sci 2016, 23, 1612–1615. [Google Scholar] [CrossRef] [PubMed]
- Esencan, E.; Cozzolino, M.; Imamoglu, G.; Seli, E. Mitochondrial stress response gene Clpp is not required for granulosa cell function. Antioxidants 2020, 10. [Google Scholar] [CrossRef]
- Morin, S.J.; Tao, X.; Marin, D.; Zhan, Y.; Landis, J.; Bedard, J.; Scott, R.T.; Seli, E. DNA methylation-based age prediction and telomere length in white blood cells and cumulus cells of infertile women with normal or poor response to ovarian stimulation. Aging 2018, 10, 3761–3773. [Google Scholar] [CrossRef]
- Hanson, B.M.; Tao, X.; Zhan, Y.; Jenkins, T.G.; Morin, S.J.; Scott, R.T.; Seli, E.U. Young women with poor ovarian response exhibit epigenetic age acceleration based on evaluation of white blood cells using a DNA methylation-derived age prediction model. Hum Reprod 2020 35, 2579–2588. [CrossRef]
- Hanson, B.M.; Tao, X.; Zhan, Y.; Kim, J.G.; Klimczak, A.M.; Herlihy, N.S.; Scott, R.T.J.; Seli, E. Shorter telomere length of white blood cells is associated with higher rates of aneuploidy among infertile women undergoing in vitro fertilization. Fertil Steril 2021, 115, 957–965. [Google Scholar] [CrossRef]
- Lin, X.; Gu, J.; Lu, C.; Spitz, M.R.; Wu, X. Expression of telomere-associated genes as prognostic markers for overall survival in patients with non–small cell lung cancer. Clin Cancer Res 2006, 12, 5720–5725. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



