Submitted:
17 May 2023
Posted:
18 May 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
2.1. Chemical composition
2.2. Content of phenolic compounds, tannins, flavonoids, flavonols and evaluation of antioxidant activity
2.3. Analysis by LC-DAD
2.4. Analysis by GC-MS
2.5. Toxicity
2.6. Antimicrobial Activity
2.7. Determination of the minimum inhibitory concentration (MIC)
3. Conclusions
4. Experimental Section
4.1. Plant material
4.2. Preparation of extracts
4.3. Phytochemical screening
4.4. Determination of the content of phenolic compounds
4.5. Determination of tannin content
4.6. Determination of flavonoid content
4.7. Determination of flavonols
4.8. Assessment of antioxidant activity
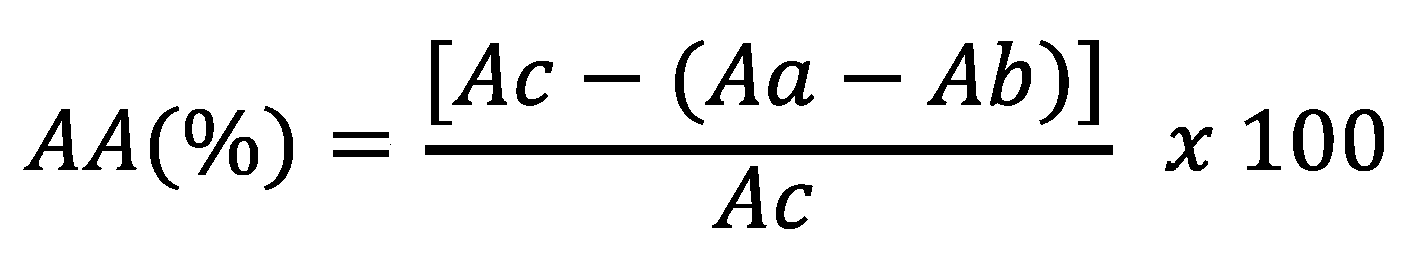
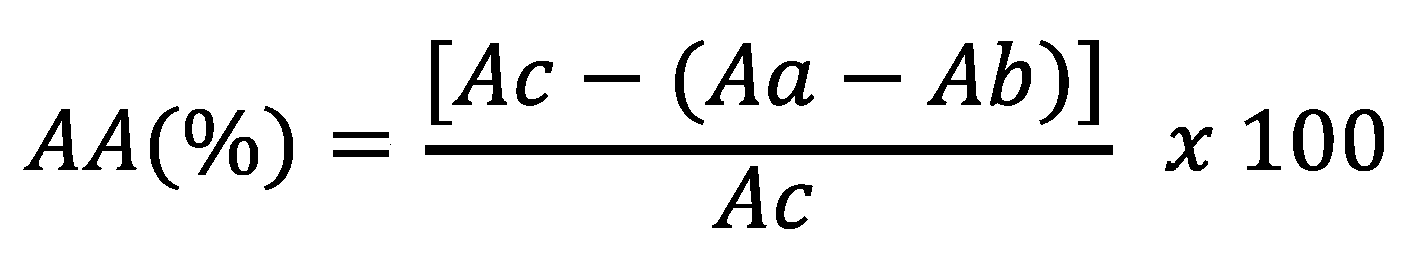
4.9. Analysis by liquid chromatography by diode array detection (LC-DAD)
4.10. Analysis by gas chromatography coupled to mass spectrometry (GC-MS)
4.11. Toxicity evaluation
4.12. Antimicrobial activity evaluation
4.13. Antimicrobial test by the Agar diffusion method (Well)
4.14. Determination of the Minimum Inhibitory Concentration (MIC)
4.15. Statistical analysis
Author Contributions
Acknowledgments
References
- C. Rosa, S. G. Câmara, J. U. Béria, ‘Representações e intenção de uso da fitoterapia na atenção básica à saúde‘. Cien. Saude Colet. 2011, 16, 311–318. [Google Scholar] [CrossRef] [PubMed]
- L. Borzini, R. Condò, P. De Dominicis, A. Casaglia, L. Cerroni, ‘Root canal irrigation: chemical agents and plant extracts against Enterococcus faecalis’. Open. Dent. J. 2016, 10, 692–703. [Google Scholar] [CrossRef] [PubMed]
- N. C. Conde, M. D. S. Vieira Pereira, M. F. Costa Lima Bandeira, G. Naura Venâncio, G. Palma de Oliveira, F. Correia Sampaio, ‘In vitro antimicrobial activity of plants of the Amazon on oral biofilm micro-organisms’. Ver. Odonto Ciênc. 2015, 30, 179–183. [Google Scholar]
- H. Lorenzi, ‘Árvores Brasileiras: Manual de Identificação e Cultivo de Plantas Arbóreas do Brasil’, in ‘Instituto Plantarum de Estudos da Flora LTDA‘, Ed. Nova Odessa, São Paulo, SP, 2000.
- N. K. Kaushik, A. Bagavan, A. A. Rahuman, A. A. Zahir, C. Kamaraj, G. Elango, C. Jayaseelan, A. V. Kirthi, T. Santhoshkumar, S. Marimuthu, G. Rajakumar, S. K. Tiwari, D. Sahal, ‘Evaluation of antiplasmodial activity of medicinal plants from North Indian Buchpora and South Indian Eastern Ghats’. Malar. J. 2015, 14. [Google Scholar]
- J. Sanz-Biset, J. Campos-de-la-Cruz, M. A. Epiquién-Rivera, S. Canigueral, ‘A first survey on the medicinal plants of the Chazuta valley (Peruvian Amazon)’. J. Ethnopharmacol. 2009, 122, 333–362. [Google Scholar] [CrossRef] [PubMed]
- T. Akther, M. S. Khan, S. Hemalatha, ‘Extraction of flavonoid from various parts of Couroupita guianensis and its efficacy against pathogenic bacteria. Asian J. Pharm.Clin. Res. 2017, 10, 354–358. [Google Scholar] [CrossRef]
- I. A. Freires, S. M. De Alencar, P. L. Rosalen, ‘A pharmacological perspective on the use of brazilian red propolis and its isolated compounds against human diseases’. Eur J. Med. Chem. 2016, 110, 267–79. [Google Scholar] [CrossRef]
- C. Chen, H. C. CHANG, H. W. YANG, G. L. CHEN, ‘Evaluation of total antioxidant activity of several popular vegetables and chinese herbs: a fast approach with ABTS/H2O2/HRP system in microplates’. J. Food Drug Anal. 2004, 12, 29–33. [Google Scholar]
- J. R. Brunet, M. D. Echevarría, F. M. Luzardo, E. N. Romero, L. M. V. Guerra, J. L. S. Romero, ‘Inhibición de la replicación del virus de inmunodeficiencia humana por extractos de taninos de Pinus caribaea Morelet’. Rev. Cubana Farm. 2003, 37, 0–0. [Google Scholar]
- S. Kaiser, C. Pavei, G. G. Ortega, ‘Estudo da relação estrutura-atividade de saponinas hemolíticas e/ou imunoadjuvantes mediante uso de análise multivariada‘. Rev. Bras. Farmacogn. 2010, 20, 300–309. [Google Scholar] [CrossRef]
- C. Aleluia, V. de Cássia Procópio, M. T. G. Oliveira, P. G. S. Furtado, J. F. G. Giovannini, S. M. S. de Mendonça, ‘Fitoterápicos na odontologia’. Rev. Odont. Univers. Cidade Sao Paulo 2017, 27, 126–134. [Google Scholar]
- J. I. Achika, R.G. Ayo, J.D. Habila, A.O. Oyewale, ‘Terpenes with antimicrobial and antioxidant activities from Lannea humilis (Oliv.). Sci. Afri. 2020, 10, e00552. [Google Scholar]
- R. Singh, N. Kumari, M. Gangwar, G. Nath, ‘Qualitative characterization of phytochemicals and in vitro antimicrobial evaluation of leaf extract of Couroupita guianensis aubl. - a threatened medicinal tree’. Int. J. Pharm. Pharm. Sci. 2015, 7, 212–215. [Google Scholar]
- M. Sirisha, V. Jaishree, ‘Phytochemical screening, antioxidant and antiproliferative activities of successive extracts of Couroupita guianensis Aubl. Plant’. Ind. J. Nat. Prod. Res. 2018, 9, 22–27. [Google Scholar]
- P. Pandurangan, M. Sahadeven, S. Sunkar, S. K. N. M. Dhana, ‘Comparative analysis of biochemical compounds of leaf, flower and fruit of Couroupita guianensis and synthesis of silver nanoparticles’. Pharmacogn. J. 2018, 10, 315–323. [Google Scholar] [CrossRef]
- A. Elumalai, K. Bargavi, S. Krishna, M. C. Eswaraiah, ‘Evaluation of anti-oxidant and hepatoprotective activity of Couroupita guianensis leaves’. J. Cell. Tissue Res. 2013, 13, 3745. [Google Scholar]
- F. R. P. Silva, S. S. M. F. R. P. Silva, S. S. M. Almeida, ‘Análise fitoquímica e microbiológica da atividade do extrato bruto etanólico da andiroba, Carapa guianensis aubl’. Biota Amazônia 2014, 4, 10–14. [Google Scholar] [CrossRef]
- C. Duque. Available online: https://bv.fapesp.br/pt/bolsas/186095/efeito-antimicrobiano-e-anti-inflamatorio-de-acidos-fenolicos-isolados-combinados-e-incorporados-em/ (accessed on 17 May 2022).
- G. Sathishkumar, K. J. Pradeep, V. Vignesh, C. Rajkuberan, M. Jeyaraj, M. Selvakumar, J. Rakhi, S. Sivaramakrishnan, ‘Cannonball fruit (Couroupita guianensis, Aubl.) extract mediated synthesis of gold nanoparticles and evaluation of its antioxidant activity’. J. Mol. Liq. 2016, 215, 229–236. [Google Scholar] [CrossRef]
- S. Bhuvaneswari, K. R. Aravind, B. Ramkumar, N. V. Raja, A. Neelakandan, P. M. Kumar, N. K. U. Prakash, ‘Studies on the phytochemistry and bioactivity of leaves of trees in Chennai’. Int. J. Chem. Tech. Res. 2014, 6, 4078–4083. [Google Scholar]
- M. M..G. Pinheiro, S. B. O. Fernandes, C. E. Fingolo, F. Boylan, P. D. Fernandes, ‚Anti-inflammatory activity of ethanol extract and fractions from Couroupita guianensis Aublet leaves‘. J. Ethnopharm. 2013, 146, 324–330. [Google Scholar] [CrossRef]
- H. L. Raghavendra, T. R. P. Kekuda, D. Pushpavathi, M. Shilpa, T. Petkar, A. Siddiqha, ‚Antimicrobial, radical scavenging, and insecticidal activity of leaf and flower extracts of Couroupita guianensis Aubl’. Int. J.Green Pharm. 2017, 11, 171–179.
- D. M. Oliveira, D. H. M. Bastos, ‘Biodisponibilidade de ácidos fenólicos‘. Quim. Nova 2014, 34, 1051–1056. [Google Scholar]
- L. M. N. Oliveira, L. M. R. Silva, A. C. S. Lima, R. R. Almeida, N. M. P. S. Ricardo, E. A. T. Figueiredo, R. W. Figueiredo, ‘Characterization of rutin, phenolic compounds and antioxidant capacity of pulps and by-products of tropical fruits’. Res. Soc. Dev. 2020, 9, e42942812. [Google Scholar] [CrossRef]
- D. Stojković, J. Petrović, M. Soković, J. Glamočlija, J. Kukić-Marković, S. Petrović, ‘In situ antioxidant and antimicrobial activities of naturally occurring caffeic acid, p-coumaric acid and rutin, using food systems’. J. Sci. Food Agric. 2013, 93, 3205–3208. [Google Scholar] [CrossRef] [PubMed]
- B. Gupta, B. Huang, ‘Mechanism of salinity tolerance in plants: physiological, biochemical, and molecular characterization’. Int. J. Genomics 2014, 2014, 1–18. [Google Scholar]
- J. H. G. Lago, A. T. Ito, C. M. Fernandes, M. C. M. Young, M. J. Kato, ‘Secondary metabolites isolated from Piper chimonanti folium and their antifungal activity’. Nat. Prod. Res. 2012, 26, 770–773. [Google Scholar] [CrossRef]
- V. A. Facundo, A. R. Pollli, R. V. Rodrigues, J. S. T. Militão, R. G. Stabelli, C. T. Cardoso, ‘Constituintes químicos fixos e voláteis dos talos e frutos de Piper tuberculatum Jacq. e das raízes de P. hispidum HBK’. Acta Amazon. 2008, 38, 743–748. [Google Scholar] [CrossRef]
- H. D. S. Alves, G. E. D. Oliveira, M. D. G. Zoghbi, M. C. D. O. Chaves, ‘Flavonóides de Piper carniconnectivum C. DC.. Piperaceae’. Rev. Bras. Farmacogn. 2010, 20, 160–164. [Google Scholar]
- M. Araujo, M. C. O. Pinheiro, I. E. A. Z. Teixeira, L. G. Riachi, C. B. Rocha, C. A. De Maria, R. F. A. Moreira, ‘Volatile and semi-volatile composition of the ripe Brazilian Couroupita guianensis fruit’. Nat. Product. J. 2014, 4, 280–289. [Google Scholar]
- A. Venkatraman, L. A. Sheba, ‘Antioxidant potential and chromatographic profiling of Couroupita guianensis fruit pulp‘. J. Adv. Sci. Res. 2022, 13, 286–293. [Google Scholar]
- R. Lavanya, S. A. John, ‘Investigation of secondary metabolites from Couroupita guianensis through GC-MS’. Int. J. Phytopharm. 2015, 5, 81–85.
- B. J. Young, N. I. Riera, M. E. Beily, P. A. Bres, D. C. Crespo, A. E. Ronco, ‘Toxicity of the effluent from an anaerobic bioreactor treating cereal residues on Lactuca sativa’. Ecotoxicol. Environ. Saf. 2021, 76, 82–186.
- G. Sivapragasam, V. Soundararajan, C. Yeng, S. L. Ngit, A. W. Habibah, F. Hariri, S. Subramaniam, S. Sreenivasan, ‘In vitro and in vivo-scientific evaluation on cytotoxicity and genotoxicity of traditional medicinal plant Couroupita guianensis aubl. Flower’. Pharmacol. online 2019, 2, 24–38. [Google Scholar]
- S. H. Patel, J. V. Suthar, R. K. Patel, U. S. Zankharia, V. R. Jani, K. N. Gajjar, ‘Antimicrobial activity investigation of Aegle marmelos, Couroupita guianesis, Manilkara hexandra, cow urine and dung’. Res. J. Pharm. Biol. Chem. Sci. 2015, 6, 1014–1022. [Google Scholar]
- I. M. Soares, M. F. Ribeiro, O. J. Costa, A. E. Souza, A. A. Aguiar, R. S. Barbosa, T. C. Alvim, S. D. Ascêncio, R. W. S. Aguiar, ‘Application of a degreasing process and sequential ultrasound-assisted extraction to obtain phenolic compounds and elucidate of the potential antioxidant of Siparuna guianensis Aublet’. J. Med. Plant. Res. 2017, 11, 357–366. [Google Scholar]
- L. C. F. Saraiva, W. M. N. Maia, F. R. Leal, A. L. M. Maia Filho, C. M. Feitosa, ‘Triagem fitoquímica das folhas de Moringa oleifera’. Boletim Informativo Geum 2018, 9, 12–19.
- C. M. O. Simões, E. P. Schenkel, J. C. P. de Mello, L. A. Mentz, P. R. Petrovick, ‘Farmacognosia: do produto natural ao medicamento’. Ed. Artmed, Porto Alegre, RS, 2016.
- L. C. O. Silva, R. A. Lima, ‘Identificação das classes de metabólitos segundários no extrato etanólico dos frutos e folhas de Eugenia uniflora L’, Ed Reget, Porto Velho, RO, 2016, 20, 381 – 288.
- E. L. Amorim, V. T. N. A. Castro, J. Melo, A. Corrêa, T. J. S. Peixoto Sobrinho, ‘Standard operating procedures (SOP) for the spectrophotometric determination of phenolic compounds contained in plant sample’, In ‘Latest Research into Quality Control’, Ed. Intechopen, Rijeka, Croatia, 2012, 1, 47 – 66.
- E. L. Amorim, J. E. Nascimento, J. M. Monteiro, T. J. S. Peixoto Sobrinho, T. A. Araújo, U. P. Albuquerque, ‘A simple and accurate procedure for the determination of tannin and flavonoid levels and some applications in ethnobotany and ethnopharmacology’. Functional Ecosystems and Communities 2008, 2, 88–94. [Google Scholar]
- G. Miliauskas, P. R. Venskutonis, T. A. Van Beek, ‘Screening of radical scavenging activity of some medicinal and aromatic plant extracts’. Food. Chem. 2004, 85, 231–237. [Google Scholar] [CrossRef]
- T. J. P. Sobrinho, V. T. Castro, A. M. Saraiva, D. M. Almeida, E. A. Tavares, E. L. Amorim, ‘Phenolic content and antioxidant capacity of four Cnidoscolus species (Euphorbiaceae) used as ethnopharmacologicals in Caatinga, Brazil’. African J. Pharm. Pharmacol. 2011, 5, 2310–2316. [Google Scholar]
- Y. Chen, B. Huang, J. He, L. Han, Y. Zhan, Y. Wang, ‘In vitro and in vivo antioxidant effects of the ethanolic extract of Swertia chirayita’. J. Ethnopharmacol. 2011, 136, 309–315. [Google Scholar] [CrossRef]
- S. V. Rabêlo, M. M. D. Costa, R. C. Libório, J. R. G. D. S. Almeida, ‘Antioxidant and antimicrobial activity of extracts from atemoia (Annona cherimola Mill. x A. squamosa L.)’. Rev. Bras. Fruticult. 2014, 36, 265–271. [Google Scholar] [CrossRef]
- D. U. Meneguetti, F. C. da Silva, R. A. Zan, L. J. Ramos, ‘Adaptation of the micronucleus technique in Allium cepa, for mutagenicity analysis of the Jamari river valley, western Amazon, Brazil’. J. Environ. Anal. Toxicol. 2012, 2, 2161–0525. [Google Scholar]
- J. L. McLaughlin, L. L. Rogers, J. E. Anderson, ‘The use of biological assays to evaluate botanicals’. Drug Inf. J. 1998, 32, 513–524. [Google Scholar] [CrossRef]
- J. M. Nguta, J. M. Mbaria, P. K. Gathumbi, J. D. Kabasa, S. G. Kiama, ‘Biological screening of Kenya medicinal plants using Artemia salina (ARTEMIIDAE)’. Pharmacologyonline 2011, 2, 458–478. [Google Scholar]
- A. I. T. D. Oliveira, T. S. Mahmoud, G. N. L. D. Nascimento, J. F. M. D. Silva, R. S. Pimenta, P. B. D. Morais, ‘Chemical composition and antimicrobial potential of palm leaf extracts from Babaçu (Attalea speciosa), Buriti (Mauritia flexuosa), and Macaúba (Acrocomia aculeata)’. Sci. World J. 2016, 2016. [Google Scholar]
- I. I. T. Oliveira, J. B. Cabral, T. S. Mahmoud, G. N. L. Do Nascimento, J. F. M. Da Silva, R. S. Pimenta, P. B. De Morais, ‘In vitro antimicrobial activity and fatty acid composition through gas chromatography-mass spectrometry (GC-MS) of ethanol extracts of Mauritia flexuosa (Buriti) fruits’. J. Med. Plants Res. 2017, 11, 635–641. [Google Scholar]
- M. A. Wikler, ‘Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically: approved standard’, In ‘National Committee for Clinical Laboratory Standards (NCCLS)’, Wayne, PA, 2000, Vol. 26, pp. M7 – M5.
- J. C. Palomino, A. Martin, M. Camacho, H. Guerra, J. Swings, F. Portaels, ‘Resazurin microtiter assay plate: simple and inexpensive method for detection of drug resistance in Mycobacterium tuberculosis’. Antimicrob. Agents Chemother. 2002, 46, 2720–2722. [Google Scholar] [CrossRef]
- D. F. Ferreira, ‘Sisvar: um programa para análises e ensino de estatística’. Rev. Cient. Symposium 2008, 6, 36–41. [Google Scholar]
| Chemical Group | Extract | ||
| CUE | CSE | ESE | |
| Flavonoids | + | + | + |
| Tannins | + | + | + |
| Phytosterols | - | - | - |
| Triterpenoids | + | + | + |
| Quinones | - | - | - |
| Saponins | + | + | + |
| Alkaloids | - | + | + |
| Extract | Control | ||||
| CUE Mean ± SD |
CSE Mean ± SD |
ESE Mean ± SD |
Gallic acid Mean ± SD |
Rutin Mean ± SD |
|
| Phenolic compounds (mg GAE/g) |
85.58 ± 0.51a | 90.19 ± 0.29a | 92.31 ± 0.38a | - | - |
| Tannins (mg GAE/g) |
20.96 ± 0.62a | 17.69 ± 0.29a | 19.62 ± 0.88a | - | - |
| Flavonoids (mg RE/g) |
307.21 ± 1.05a | 101.07 ± 1.99b | 65.63 ± 0.53c | - | |
| Flavonols (mg RE/g) |
100.89 ± 1.05a | 88.61 ± 0.80b | 56.33 ± 0.80c | - | - |
| DPPH• IC50 (µg/mL) |
59.51 ± 0.26c | 31.13 ± 0.55b | 2.98 ± 0.96a | - | 11.92 ± 0.47 |
| ABTS•+ IC50 (µg/mL) |
30.32 ± 1.60c | 15.74 ± 1.45b | 4.93 ± 0.90a | 6.75 ± 0.01 | - |
| Compound | Concentration (mg/g) | ||
| CUE Mean ± SD |
CSE Mean ± SD |
ESE Mean ± SD |
|
| Cafeic acid | 37.6 ± 0.1a | 39.4 ± 0.2a | 38.5 ± 0.2a |
| Sinapic acid | 37.1 ± 0.1a | 38.6 ± 0.1a | 38.1 ± 0.1a |
| Rutin | 124.1 ± 0.4a | 138.1 ± 0.6a | 129.2 ± 0.5a |
| Quercetin | 169.8 ± 0.6a | 181.4 ± 0.7a | 177.3 ± 0.4a |
| Luteolin | 100.1± 0.2a | 103.2 ± 0.4a | 101.8 ± 0.3a |
| Kampefrol | 94.8 ± 0.2a | 97.4 ± 0.3a | 96.3 ± 0.2a |
| Apigenin | 75.2 ± 0.1a | 78.8 ± 0.2a | 3.4 ± 0.1b |
| Compound | Concentration (mg/g) | ||
| CUE Mean ± SD |
CSE Mean ± SD |
ESE Mean ± SD |
|
| Stigmasterol | 69.7 ± 0.1b | 85.1 ± 0.2a | -* |
| β-sitosterol | 80.3 ± 0.2a | 91.9 ± 0.3a | -* |
| Samples | Concentration |
ARL ± DP (mm) Mean ± SD |
ICR | Effect | GR (%) |
| Control | 43.08 ± 5.8 | 1 | 100 | ||
| CUE | 50µg/mL | 58.44 ± 1.29 | 1.36 | GS | 135.6 |
| 250µg/mL | 41.11 ± 11.72 | 0.95 | SCE | 95.4 | |
| 750µg/mL | 19.24 ± 1.77 | 0.45 | GI | 44.7 | |
| CSE | 50µg/mL | 47.40 ± 11.40 | 1.10 | SCE | 110 |
| 250µg/mL | 34.83 ± 7.41 | 0.81 | SCE | 80.8 | |
| 750µg/mL | 20.64 ± 3.06 | 0.48 | GI | 47.9 | |
| ESE | 50µg/mL | 60.29 ± 1.70 | 1.40 | GS | 139.9 |
| 250µg/mL | 44.49 ± 11.20 | 1.03 | SCE | 103.2 | |
| 750µg/mL | 32.34 ± 2.00 | 0.75 | GI | 75 |
| Extract | IC50 (µg/mL) |
Toxicity |
| CUE | 2.318 | Non toxic |
| CSE | 2.308 | Non toxic |
| ESE | 1.478 | Non toxic |
| Extrats | Concentration (mg) |
Microorganisms | |
|---|---|---|---|
|
S. aureus Mean ± SD |
S. mutans Mean ± SD |
||
| Zone of inibition (mm) | |||
| CUE | 50 | 10.50 ± 0.70b | 7.83 ± 0.49b |
| 100 | 10.44 ± 1.75b | 7.87 ± 0.15b | |
| 200 | 12.21 ± 1.23a | 10.31 ± 0.17a | |
| CSE | 50 | 8.09 ± 0.22c | 4.48 ± 2.89b |
| 100 | 9.61 ± 0.15b | 9.16 ± 0.39a | |
| 200 | 10.29 ± 0.04a | 9.99 ± 1.39a | |
| ESE | 50 | 7.72 ± 2.83b | 8.13 ± 0.15b |
| 100 | 12.57 ± 0.74a | 8.69 ± 0.62b | |
| 200 | 13.41 ± 1.76a | 9.06 ± 0.55a | |
| Chlorhexidine (+) | 10.82 ± 0.81a | 12.37 ± 0.61a | |
| DMSO (-) | 0.00 ± 0.00 | 0.00 ± 0.00 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




