Submitted:
12 May 2023
Posted:
15 May 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental
3. Results and discussion
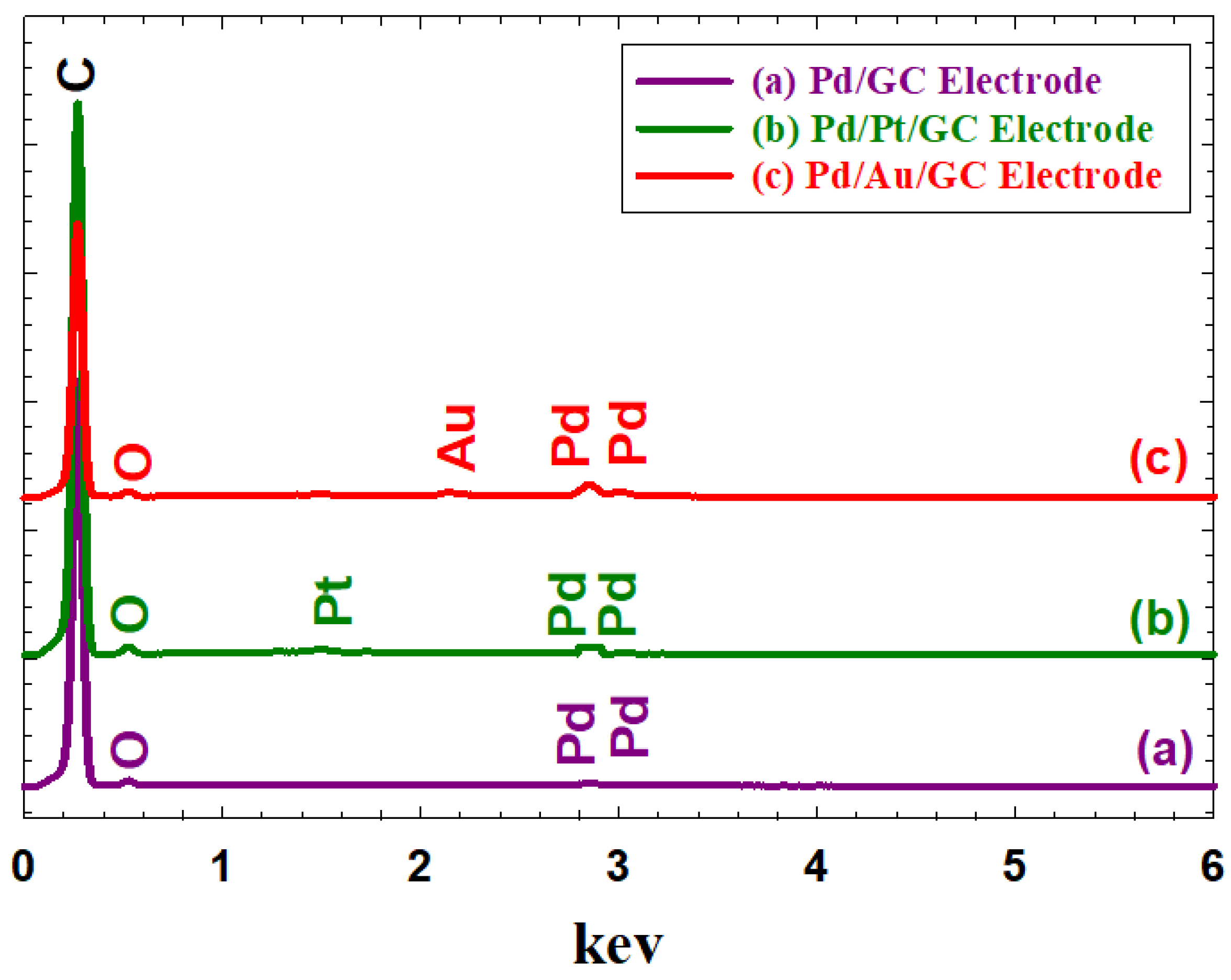
3.1. Electrochemical and Materials characterization
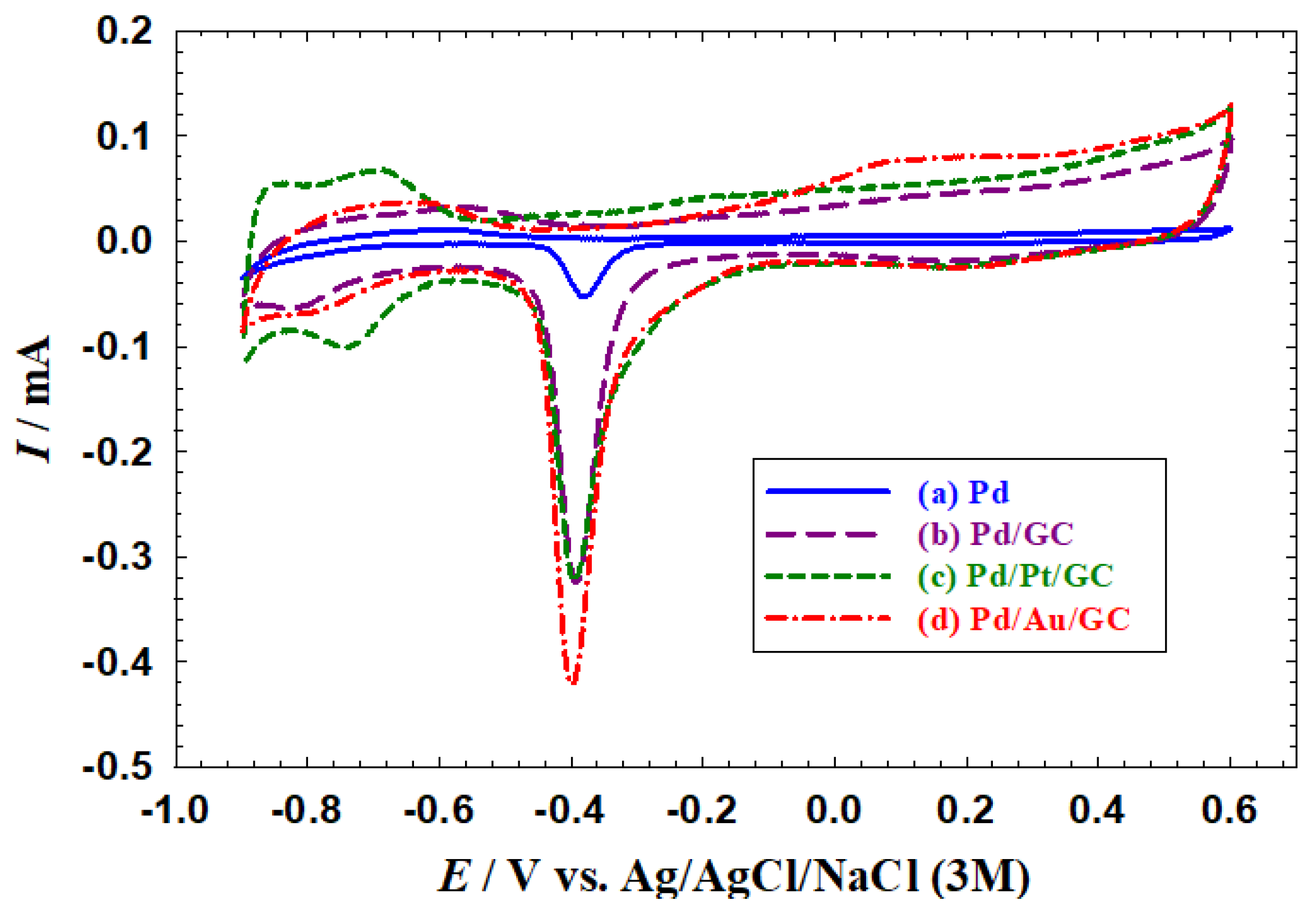
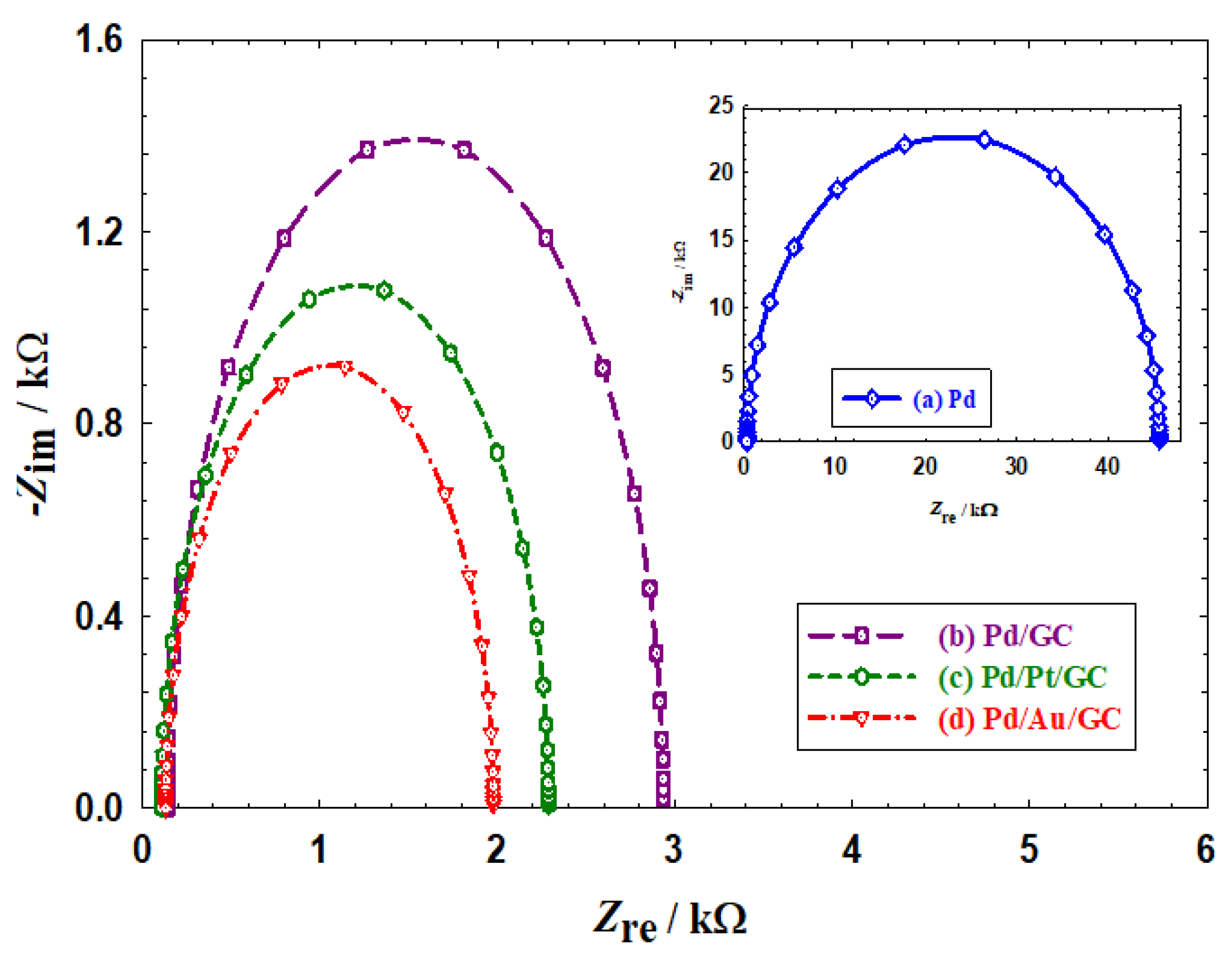
- Compared to the bulk Pd catalyst (Figure 1a), a larger surface area (SA) was obtained at the nanoparticles-modified catalysts (Figure 1b–d). The SA was calculated to be 0.08, 0.52, 0.59, and 0.83 cm2 for the Pd, Pd/GC, Pd/Pt/GC, and Pd/Au/GC catalysts, respectively, based on the charge associated with the PdO reduction peak using a reference value of 420 µC cm−2 [38,39]. . This trend appeared again in the Hads/des region because of the large surface area offered by nanoparticles.
- The Pd/Pt/GC (Figure 1c) and the Pd/Au/GC (Figure 1d) catalysts acquired a broader PdO reduction peak compared to that obtained at the Pd/GC catalyst (Figure 1a). This highlighted the role of adding the Pt and Au surface modifiers in providing diverse Pd−Pd and Pd−O bonding and/or facets’ reconstruction for the Pd surface.
- The large Hads/des peaks at the Pd/Pt/GC catalyst referred to the participation of both Pd and Pt in this reaction [40].
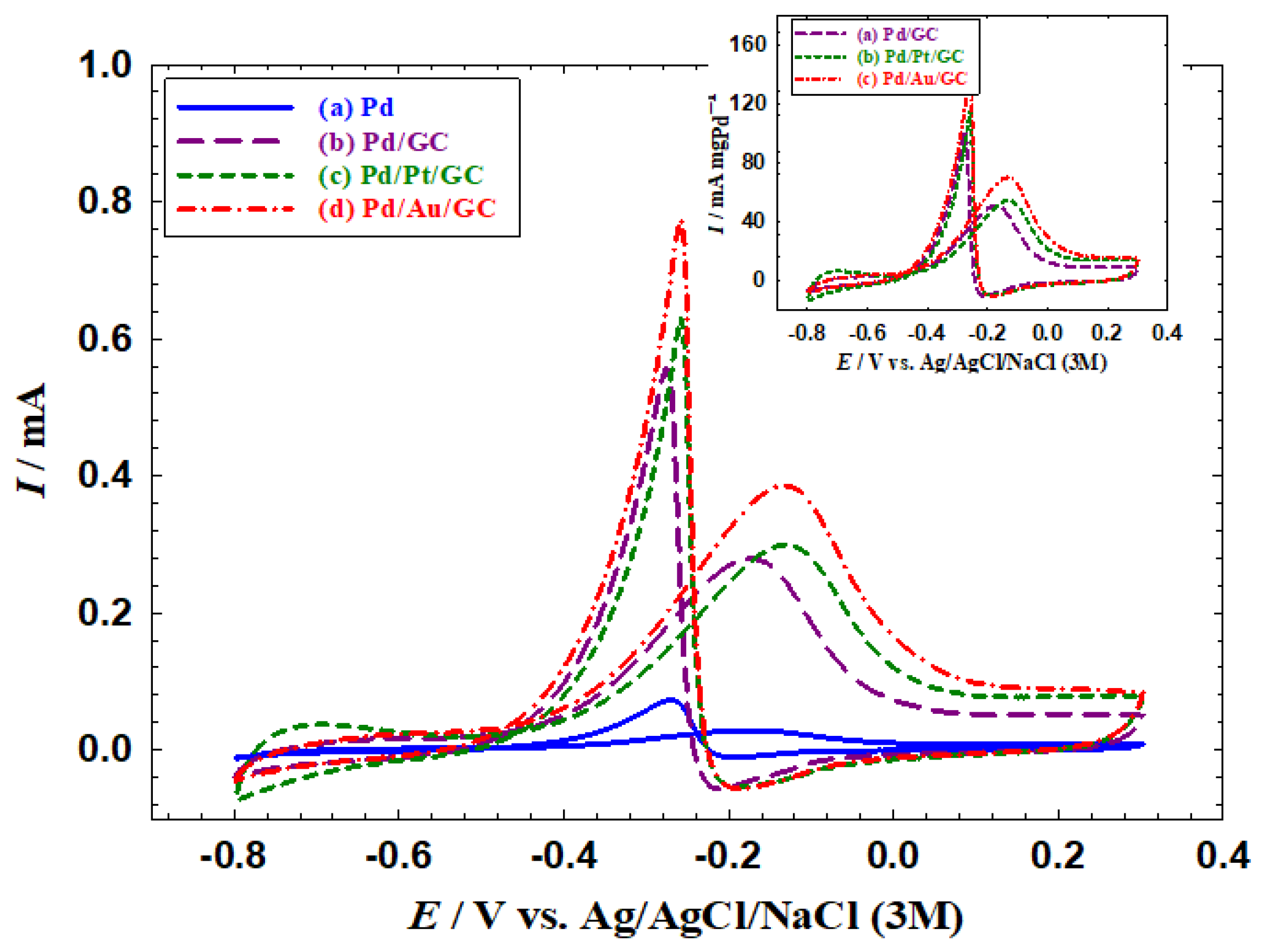
3.2. Electrocatalytic activities of the catalysts toward EOM
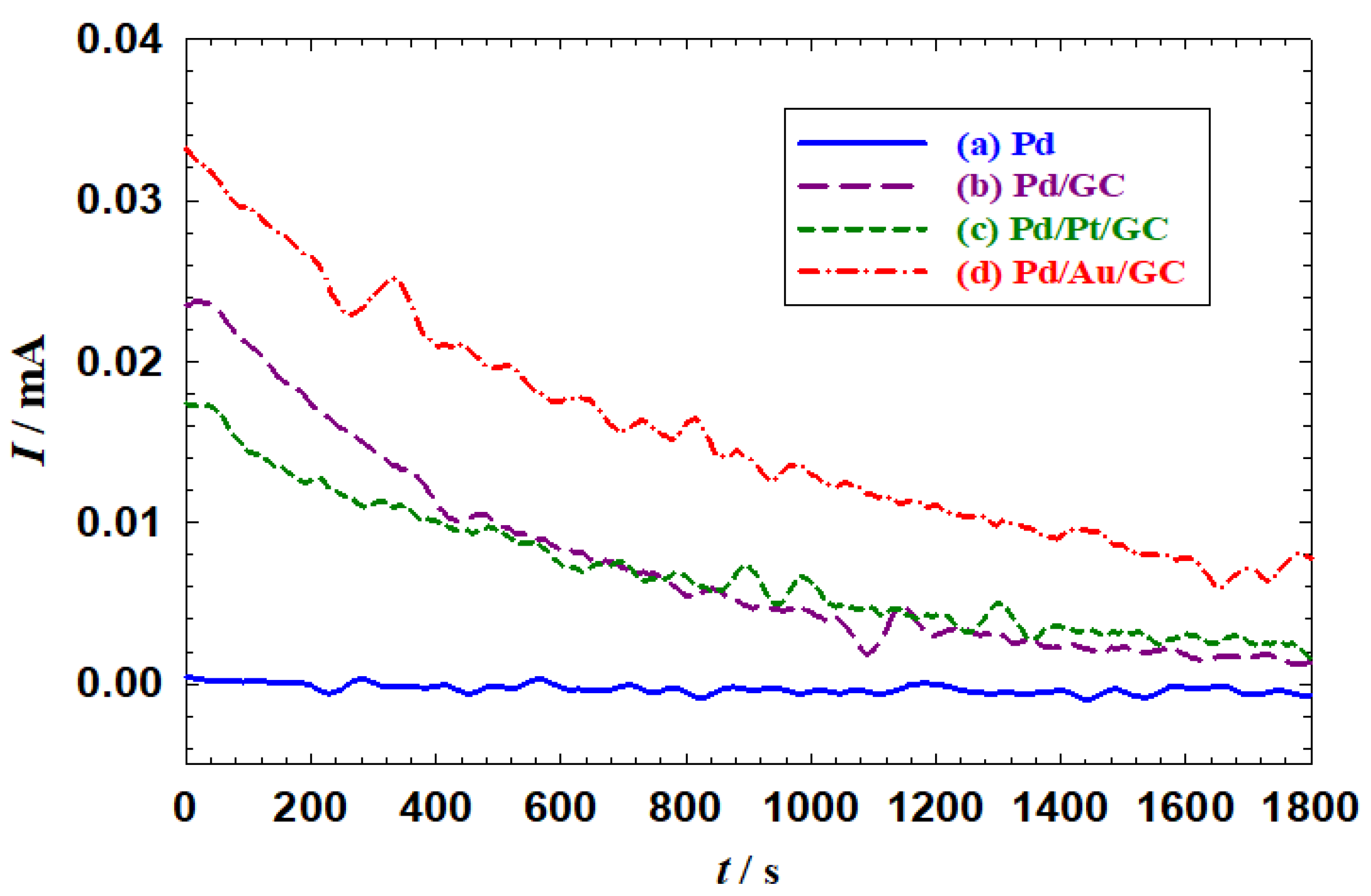
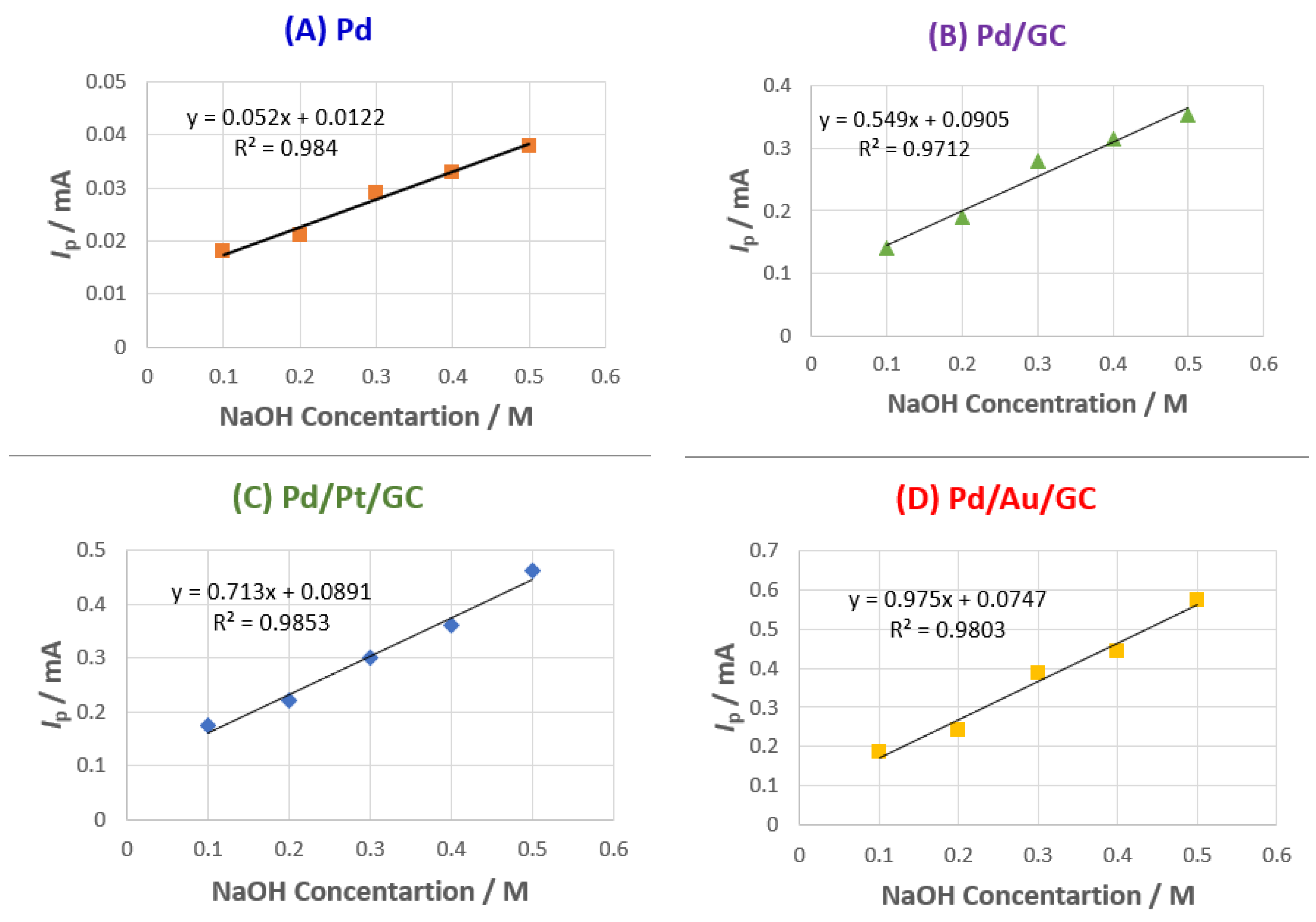
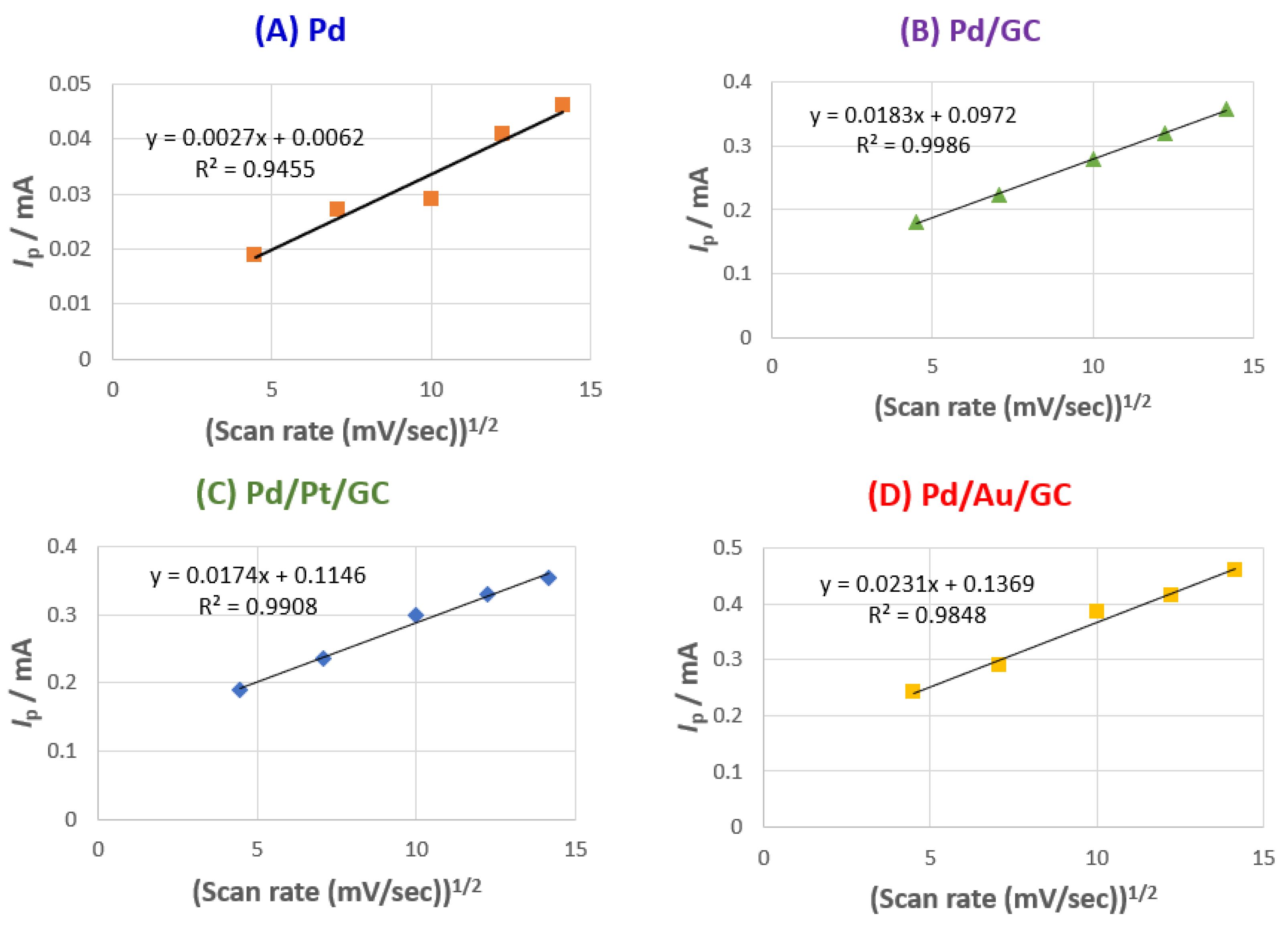
3.3. Parameters affecting 2POR
4. Conclusion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Pang, L.; Zhu, M.N.; Yu, H. Is green finance really a blessing for green technology and carbon efficiency? Energy Economics 2022, 114, 106272. [Google Scholar] [CrossRef]
- Su, C.-W.; Pang, L.-D.; Qin, M.; Lobonţ, O.-R.; Umar, M. The spillover effects among fossil fuel, renewables and carbon markets: Evidence under the dual dilemma of climate change and energy crises. Energy 2023, 274, 127304. [Google Scholar] [CrossRef]
- Molodtsova, T.; Gorshenkov, M.; Kubrin, S.; Saraev, A.; Ulyankina, A.; Smirnova, N. One-step access to bifunctional γ-Fe2O3/δ-FeOOH electrocatalyst for oxygen reduction reaction and acetaminophen sensing. Journal of the Taiwan Institute of Chemical Engineers 2022, 140, 104569. [Google Scholar] [CrossRef]
- Kaya, S. Synthesis, characterization and 1-propanol electrooxidation application of carbon nanotube supported bimetallic catalysts. International Journal of Hydrogen Energy 2023. [Google Scholar] [CrossRef]
- Farrag, H.H.; Al-Akraa, I.M.; Allam, N.K.; Mohammad, A.M. Amendment of palladium nanocubes with iron oxide nanowires for boosted formic acid electro−oxidation. Arabian Journal of Chemistry 2023, 16. [Google Scholar] [CrossRef]
- Hassan, H.E.; Asal, Y.M.; Mohammad, A.M.; Al-Akraa, I.M. BIODIESEL PRODUCTION FROM CASTOR OIL: MIXING OPTIMIZATION DURING TRANSESTERIFICATION. ARPN Journal of Engineering and Applied Sciences 2022, 17, 844–848. [Google Scholar]
- Ayman, R.; Asal, Y.M.; Mohammad, A.M.; Al-Akraa, I.M. CASTOR OIL CONVERSION TO BIODIESEL: A PROCESS SIMULATION STUDY. ARPN Journal of Engineering and Applied Sciences 2022, 17, 964–968. [Google Scholar]
- Al-Akraa, I.M.; Ohsaka, T.; Mohammad, A.M. A promising amendment for water splitters: Boosted oxygen evolution at a platinum, titanium oxide and manganese oxide hybrid catalyst. Arabian Journal of Chemistry 2019, 12, 897–907. [Google Scholar] [CrossRef]
- Al-Akraa, I.M.; Asal, Y.M.; Khamis, S.D. Assembling of NiOx/MWCNTs-GC anodic nanocatalyst for water electrolysis applications. International Journal of Electrochemical Science 2018, 13, 9712–9720. [Google Scholar] [CrossRef]
- Al-Akraa, I.M.; Asal, Y.M.; Arafa, A.M. Fabrication of MnOx/MWCNTs-GC nanocatalyst for oxygen evolution reaction. International Journal of Electrochemical Science 2018, 13, 8775–8783. [Google Scholar] [CrossRef]
- Al-Akraa, I.M.; Mohammad, A.M.; El-Deab, M.S.; El-Anadouli, B.E. Flower-shaped gold nanoparticles: Preparation, characterization, and electrocatalytic application. Arabian Journal of Chemistry 2017, 10, 877–884. [Google Scholar] [CrossRef]
- Asal, Y.M.; Mohammad, A.M.; Abd El Rehim, S.S.; Al-Akraa, I.M. Co-electrodeposited PtPd anodic catalyst for the direct formic acid fuel cells. Energy Reports 2022, 8, 560–564. [Google Scholar] [CrossRef]
- Al-Qodami, B.A.; Alalawy, H.H.; Sayed, S.Y.; Al-Akraa, I.M.; Allam, N.K.; Mohammad, A.M. Tailor-designed nanowire-structured iron and nickel oxides on platinum catalyst for formic acid electro-oxidation. RSC Advances 2022, 12, 20395–20402. [Google Scholar] [CrossRef] [PubMed]
- Al-Qodami, B.A.; Alalawy, H.H.; Al-Akraa, I.M.; Sayed, S.Y.; Allam, N.K.; Mohammad, A.M. Surface engineering of nanotubular ferric oxyhydroxide “goethite” on platinum anodes for durable formic acid fuel cells. International Journal of Hydrogen Energy 2022, 47, 264–275. [Google Scholar] [CrossRef]
- Al-Akraa, I.M.; Mohammad, A.M. A spin-coated TiOx-modified Pt anodic catalyst for the direct methanol fuel cells. Energy Reports 2022, 8, 438–442. [Google Scholar] [CrossRef]
- Al-Akraa, I.M.; Salama, A.E.; Asal, Y.M.; Mohammad, A.M. Boosted performance of NiOx/Pt nanocatalyst for the electro-oxidation of formic acid: A substrate’s functionalization with multi-walled carbon nanotubes. Arabian Journal of Chemistry 2021, 14. [Google Scholar] [CrossRef]
- Rubio, G.A.; Agila, W.E. A Fuzzy Model to Manage Water in Polymer Electrolyte Membrane Fuel Cells. Processes 2021, 9. [Google Scholar] [CrossRef]
- Shyu, J.-C.; Hung, S.-H. Flow Field Effect on the Performance of Direct Formic Acid Membraneless Fuel Cells: A Numerical Study. Processes 2021, 9. [Google Scholar] [CrossRef]
- Siwal, S.S.; Thakur, S.; Zhang, Q.B.; Thakur, V.K. Electrocatalysts for electrooxidation of direct alcohol fuel cell: chemistry and applications. Materials Today Chemistry 2019, 14, 100182. [Google Scholar] [CrossRef]
- Berretti, E.; Longhi, M.; Atanassov, P.; Sebastián, D.; Lo Vecchio, C.; Baglio, V.; Serov, A.; Marchionni, A.; Vizza, F.; Santoro, C.; et al. Platinum group metal-free Fe-based (FeNC) oxygen reduction electrocatalysts for direct alcohol fuel cells. Current Opinion in Electrochemistry 2021, 29, 100756. [Google Scholar] [CrossRef]
- Logsdon, J.E.; Loke, R.A. Isopropyl Alcohol. In Kirk-Othmer Encyclopedia of Chemical Technology; 2000.
- Nacef, M.; Lelièvre-Desmas, M.; Drider, D.; Flahaut, C.; Chollet, S. Artisanal and industrial Maroilles cheeses: Are they different? Comparison using sensory, physico-chemical and microbiological approaches. International Dairy Journal 2019, 89, 42–52. [Google Scholar] [CrossRef]
- Qi, Z.; Kaufman, A. Performance of 2-propanol in direct-oxidation fuel cells. Journal of Power Sources 2002, 112, 121–129. [Google Scholar] [CrossRef]
- Wang, Y.; Li, L.; Hu, L.; Zhuang, L.; Lu, J.; Xu, B. A feasibility analysis for alkaline membrane direct methanol fuel cell: thermodynamic disadvantages versus kinetic advantages. Electrochemistry Communications 2003, 5, 662–666. [Google Scholar] [CrossRef]
- Lu, J.; Lu, S.; Wang, D.; Yang, M.; Liu, Z.; Xu, C.; Jiang, S.P. Nano-structured PdxPt1−x/Ti anodes prepared by electrodeposition for alcohol electrooxidation. Electrochimica Acta 2009, 54, 5486–5491. [Google Scholar] [CrossRef]
- Liu, Y.; Zeng, Y.; Liu, R.; Wu, H.; Wang, G.; Cao, D. Poisoning of acetone to Pt and Au electrodes for electrooxidation of 2-propanol in alkaline medium. Electrochimica Acta 2012, 76, 174–178. [Google Scholar] [CrossRef]
- Markiewicz, M.E.P.; Bergens, S.H. Electro-oxidation of 2-propanol and acetone over platinum, platinum–ruthenium, and ruthenium nanoparticles in alkaline electrolytes. Journal of Power Sources 2008, 185, 222–225. [Google Scholar] [CrossRef]
- Santasalo-Aarnio, A.; Kwon, Y.; Ahlberg, E.; Kontturi, K.; Kallio, T.; Koper, M.T.M. Comparison of methanol, ethanol and iso-propanol oxidation on Pt and Pd electrodes in alkaline media studied by HPLC. Electrochemistry Communications 2011, 13, 466–469. [Google Scholar] [CrossRef]
- Zhou, W.; Wang, C.; Xu, J.; Du, Y.; Yang, P. Enhanced electrocatalytic performance for isopropanol oxidation on Pd–Au nanoparticles dispersed on poly(p-phenylene) prepared from biphenyl. Materials Chemistry and Physics 2010, 123, 390–395. [Google Scholar] [CrossRef]
- Santasalo, A.; Vidal-Iglesias, F.J.; Solla-Gullón, J.; Berná, A.; Kallio, T.; Feliu, J.M. Electrooxidation of methanol and 2-propanol mixtures at platinum single crystal electrodes. Electrochimica Acta 2009, 54, 6576–6583. [Google Scholar] [CrossRef]
- Liu, J.; Ye, J.; Xu, C.; Jiang, S.P.; Tong, Y. Electro-oxidation of methanol, 1-propanol and 2-propanol on Pt and Pd in alkaline medium. Journal of Power Sources 2008, 177, 67–70. [Google Scholar] [CrossRef]
- Asal, Y.M.; Mohammad, A.M.; El Rehim, S.S.A.; Al-Akraa, I.M. Preparation of Co-electrodeposited Pd-Au Nanocatalyst for Methanol Electro-oxidation. International Journal of Electrochemical Science 2021, 16, 1–11. [Google Scholar] [CrossRef]
- Al-Akraa, I.M.; Mohammad, A.M.; El-Deab, M.S.; El-Anadouli, B.E. Self-assembling of gold nanoparticles array for electro-sensing applications. International Journal of Electrochemical Science 2013, 8, 458–466. [Google Scholar] [CrossRef]
- Markiewicz, M.E.P.; Hebert, D.M.; Bergens, S.H. Electro-oxidation of 2-propanol on platinum in alkaline electrolytes. Journal of Power Sources 2006, 161, 761–767. [Google Scholar] [CrossRef]
- Xu, C.; Tian, Z.; Chen, Z.; Jiang, S.P. Pd/C promoted by Au for 2-propanol electrooxidation in alkaline media. Electrochemistry Communications 2008, 10, 246–249. [Google Scholar] [CrossRef]
- Cheng, Y.; Liu, Y.; Cao, D.; Wang, G.; Gao, Y. Effects of acetone on electrooxidation of 2-propanol in alkaline medium on the Pd/Ni-foam electrode. Journal of Power Sources 2011, 196, 3124–3128. [Google Scholar] [CrossRef]
- Asal, Y.M.; Mohammad, A.M.; Abd El Rehim, S.S.; Al-Akraa, I.M. Augmented formic acid electro-oxidation at a co-electrodeposited Pd/Au nanoparticle catalyst. Journal of Saudi Chemical Society 2022, 26, 101508. [Google Scholar] [CrossRef]
- Farrag, H.H.; Al-Akraa, I.M.; Allam, N.K.; Mohammad, A.M. Amendment of palladium nanocubes with iron oxide nanowires for boosted formic acid electro−oxidation. Arabian Journal of Chemistry 2023, 16, 104524. [Google Scholar] [CrossRef]
- Al-Akraa, I.M.; Asal, Y.M.; Darwish, S.A.; Fikry, R.M.; Mahmoud, R.H.; Hassan, M.; Mohammad, A.M. Effect of Palladium Loading on Catalytic Properties of Pd/GCE for the Electro-oxidation of Methanol, Formic Acid, and Ethylene Glycol. International Journal of Electrochemical Science 2022, 17, 220455. [Google Scholar] [CrossRef]
- Al-Akraa, I.M.; Asal, Y.M.; Mohammad, A.M. Surface engineering of Pt surfaces with Au and cobalt oxide nanostructures for enhanced formic acid electro-oxidation. Arabian Journal of Chemistry 2022, 15, 103965. [Google Scholar] [CrossRef]
- Mohammad, A.M.; Al-Akraa, I.M.; El-Deab, M.S. Superior electrocatalysis of formic acid electro-oxidation on a platinum, gold and manganese oxide nanoparticle-based ternary catalyst. International Journal of Hydrogen Energy 2018, 43, 139–149. [Google Scholar] [CrossRef]
- Al-Akraa, I.M.; Mohammad, A.M.; El-Deab, M.S.; El-Anadouli, B.E. Electrocatalysis by design: Synergistic catalytic enhancement of formic acid electro-oxidation at core–shell Pd/Pt nanocatalysts. International Journal of Hydrogen Energy 2015, 40, 1789–1794. [Google Scholar] [CrossRef]
- Hassan, K.M.; Hathoot, A.A.; Maher, R.; Abdel Azzem, M. Electrocatalytic oxidation of ethanol at Pd, Pt, Pd/Pt and Pt/Pd nano particles supported on poly 1,8-diaminonaphthalene film in alkaline medium. RSC Advances 2018, 8, 15417–15426. [Google Scholar] [CrossRef] [PubMed]
- Raoof, J.B.; Ojani, R.; Hosseini, S.R. An Electrochemical Investigation of Methanol Oxidation on Nickel Hydroxide Nanoparticles. South African Journal of Chemistry 2013, 66, 47–53. [Google Scholar]
- Danaee, I.; Jafarian, M.; Mirzapoor, A.; Gobal, F.; Mahjani, M.G. Electrooxidation of methanol on NiMn alloy modified graphite electrode. Electrochimica Acta 2010, 55, 2093–2100. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



