Submitted:
12 May 2023
Posted:
15 May 2023
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
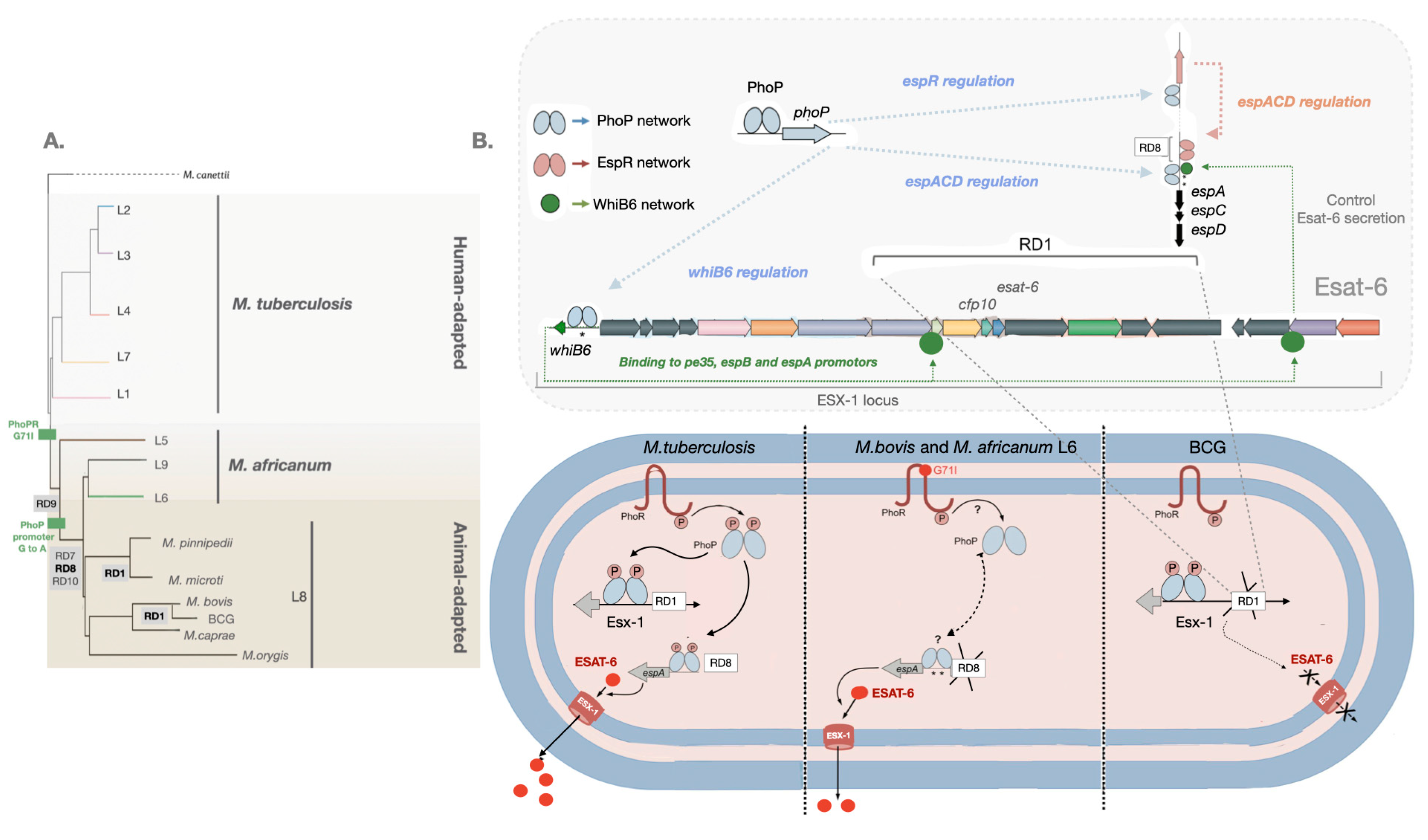
2. Virulence evolution among MTBC
3. ESAT-6 is required for virulence of Mtb
3. ESAT-6 in Mtb pathogenesis
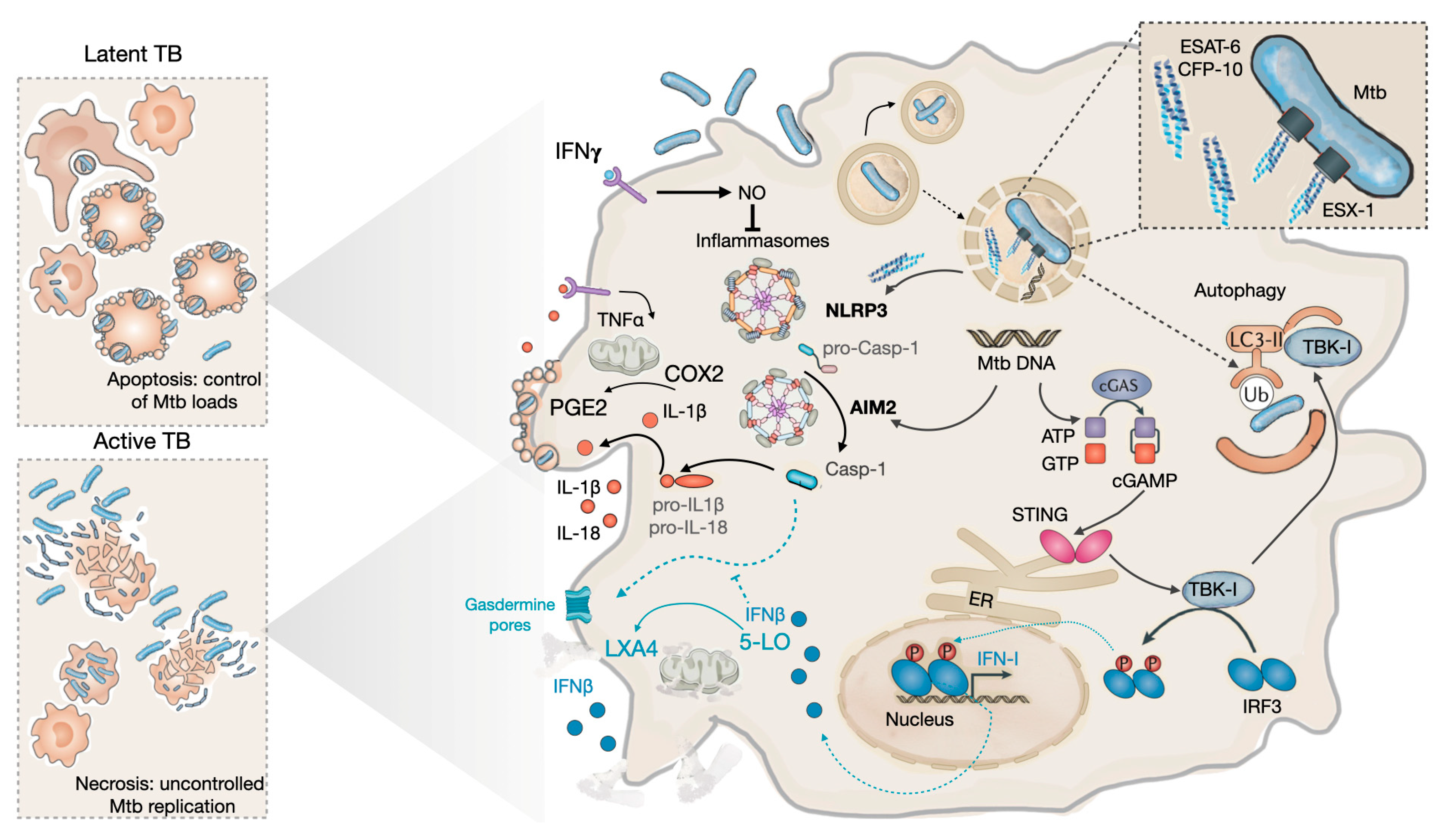
3.1. During the early phases of infection: the innate phase
3.2. During latency
3.2. During progression to disease
4. ESAT-6 from a virulence factor to diagnostic tools and vaccines for TB
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Perrin, P. Human and tuberculosis co-evolution: An integrative view. Tuberculosis 2015, 95, S112–S116. [Google Scholar] [CrossRef] [PubMed]
- Wilson, L.G. Commentary: Medicine, population, and tuberculosis. Int. J. Epidemiol. 2005, 34, 521–524. [Google Scholar] [CrossRef] [PubMed]
- WHO. Global Tuberculosis Report 2022 Factsheet. Available online: https://www.who.int/publications/m/item/global-tuberculosis-report-2022-factsheet (accessed on April 20, 2023).
- Dheda, K.; Barry, C.E., 3rd; Maartens, G. Tuberculosis. The Lancet 2016, 387, 1211–1226. [Google Scholar] [CrossRef] [PubMed]
- Dheda, K.; Gumbo, T.; Maartens, G.; Dooley, K.E.; McNerney, R.; Murray, M.; Furin, J.; Nardell, E.A.; London, L.; Lessem, E.; et al. The epidemiology, pathogenesis, transmission, diagnosis, and management of multidrug-resistant, extensively drug-resistant, and incurable tuberculosis. The Lancet Respiratory Medicine 2017, 5, 291–360. [Google Scholar] [CrossRef] [PubMed]
- Cambier, C.J.; Falkow, S.; Ramakrishnan, L. Host evasion and exploitation schemes of Mycobacterium tuberculosis. Cell 2014, 159, 1497–1509. [Google Scholar] [CrossRef]
- de Jong, B.C.; Antonio, M.; Gagneux, S. Mycobacterium africanum—Review of an Important Cause of Human Tuberculosis in West Africa. PLoS Negl. Trop. Dis. 2010, 4, e744. [Google Scholar] [CrossRef]
- de Jong, B.C.; Hill, P.C.; Aiken, A.; Awine, T.; Martin, A.; Adetifa, I.M.; Jackson-Sillah, D.J.; Fox, A.; Kathryn, D.; Gagneux, S.; et al. Progression to Active Tuberculosis, but Not Transmission, Varies by Mycobacterium tuberculosis Lineage in The Gambia. The Journal of Infectious Diseases 2008, 198, 1037–1043. [Google Scholar] [CrossRef]
- Mostowy, S.; Onipede, A.; Gagneux, S.; Niemann, S.; Kremer, K.; Desmond Edward, P.; Kato-Maeda, M.; Behr, M. Genomic Analysis Distinguishes Mycobacterium africanum. J. Clin. Microbiol. 2004, 42, 3594–3599. [Google Scholar] [CrossRef]
- Silva, M.L.; Cá, B.; Osório, N.S.; Rodrigues, P.N.S.; Maceiras, A.R.; Saraiva, M. Tuberculosis caused by Mycobacterium africanum: Knowns and unknowns. PLOS Pathogens 2022, 18, e1010490. [Google Scholar] [CrossRef]
- Müller, B.; Dürr, S.; Alonso, S.; Hattendorf, J.; Laisse, C.J.M.; Parsons, S.D.C.; van Helden, P.; Zinsstag, J. Zoonotic Mycobacterium bovis–induced Tuberculosis in Humans. Emerging Infectious Disease journal 2013, 19, 899. [Google Scholar] [CrossRef]
- Fabre, M.; Hauck, Y.; Soler, C.; Koeck, J.-L.; van Ingen, J.; van Soolingen, D.; Vergnaud, G.; Pourcel, C. Molecular characteristics of “Mycobacterium canettii” the smooth Mycobacterium tuberculosis bacilli. Infection, Genetics and Evolution 2010, 10, 1165–1173. [Google Scholar] [CrossRef] [PubMed]
- Gagneux, S. Ecology and evolution of Mycobacterium tuberculosis. Nature Reviews Microbiology 2018, 16, 202–213. [Google Scholar] [CrossRef] [PubMed]
- Gonzalo-Asensio, J.; Malaga, W.; Pawlik, A.; Astarie-Dequeker, C.; Passemar, C.; Moreau, F.; Laval, F.; Daffé, M.; Martin, C.; Brosch, R.; et al. Evolutionary history of tuberculosis shaped by conserved mutations in the PhoPR virulence regulator. Proceedings of the National Academy of Sciences 2014, 111, 11491–11496. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, N.D.; Dunavan, C.P.; Diamond, J. Origins of major human infectious diseases. Nature 2007, 447, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Comas, I.; Coscolla, M.; Luo, T.; Borrell, S.; Holt, K.E.; Kato-Maeda, M.; Parkhill, J.; Malla, B.; Berg, S.; Thwaites, G.; et al. Out-of-Africa migration and Neolithic coexpansion of Mycobacterium tuberculosis with modern humans. Nat. Genet. 2013, 45, 1176–1182. [Google Scholar] [CrossRef] [PubMed]
- Azevedo-Pereira, J.M.; Pires, D.; Calado, M.; Mandal, M.; Santos-Costa, Q.; Anes, E. HIV/Mtb Co-Infection: From the Amplification of Disease Pathogenesis to an “Emerging Syndemic”. Microorganisms 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Barry, C.E.; Boshoff, H.I.; Dartois, V.; Dick, T.; Ehrt, S.; Flynn, J.; Schnappinger, D.; Wilkinson, R.J.; Young, D. The spectrum of latent tuberculosis: rethinking the biology and intervention strategies. Nature Reviews Microbiology 2009, 7, 845–855. [Google Scholar] [CrossRef] [PubMed]
- Blaser, M.J.; Kirschner, D. The equilibria that allow bacterial persistence in human hosts. Nature 2007, 449, 843–849. [Google Scholar] [CrossRef]
- Brites, D.; Gagneux, S. Old and new selective pressures on Mycobacterium tuberculosis. Infection, Genetics and Evolution 2012, 12, 678–685. [Google Scholar] [CrossRef]
- Anes, E.; Pires, D.; Mandal, M.; Azevedo-Pereira, J.M. Spatial localization of cathepsins: Implications in immune activation and resolution during infections. Front. Immunol. 2022, 13, 955407. [Google Scholar] [CrossRef]
- Armstrong, J.A.; Hart, P.D. Phagosome-lysosome interactions in cultured macrophages infected with virulent tubercle bacilli. Reversal of the usual nonfusion pattern and observations on bacterial survival. J. Exp. Med. 1975, 142, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Pires, D.; Bernard, E.M.; Pombo, J.P.; Carmo, N.; Fialho, C.; Gutierrez, M.G.; Bettencourt, P.; Anes, E. Mycobacterium tuberculosis Modulates miR-106b-5p to Control Cathepsin S Expression Resulting in Higher Pathogen Survival and Poor T-Cell Activation. Front. Immunol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Pires, D.; Calado, M.; Velez, T.; Mandal, M.; Catalão, M.J.; Neyrolles, O.; Lugo-Villarino, G.; Vérollet, C.; Azevedo-Pereira, J.M.; Anes, E. Modulation of Cystatin C in Human Macrophages Improves Anti-Mycobacterial Immune Responses to Mycobacterium tuberculosis Infection and Coinfection With HIV. Front. Immunol. 2021, 12, 4693. [Google Scholar] [CrossRef] [PubMed]
- Pires, D.; Mandal, M.; Pinho, J.; Catalão, M.J.; Almeida, A.J.; Azevedo-Pereira, J.M.; Gaspar, M.M.; Anes, E. Liposomal Delivery of Saquinavir to Macrophages Overcomes Cathepsin Blockade by Mycobacterium tuberculosis and Helps Control the Phagosomal Replicative Niches. Int. J. Mol. Sci. 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Pires, D.; Marques, J.; Pombo, J.P.; Carmo, N.; Bettencourt, P.; Neyrolles, O.; Lugo-Villarino, G.; Anes, E. Role of Cathepsins in Mycobacterium tuberculosis Survival in Human Macrophages. Sci. Rep. 2016, 6, 32247. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.B.; Gern, B.H.; Delahaye, J.L.; Adams, K.N.; Plumlee, C.R.; Winkler, J.K.; Sherman, D.R.; Gerner, M.Y.; Urdahl, K.B. Alveolar Macrophages Provide an Early Mycobacterium tuberculosis Niche and Initiate Dissemination. Cell Host & Microbe 2018, 24, 439–446.e434. [Google Scholar] [CrossRef]
- Mishra, B.B.; Moura-Alves, P.; Sonawane, A.; Hacohen, N.; Griffiths, G.; Moita, L.F.; Anes, E. Mycobacterium tuberculosis protein ESAT-6 is a potent activator of the NLRP3/ASC inflammasome. Cell. Microbiol. 2010, 12, 1046–1063. [Google Scholar] [CrossRef]
- Aguilo, J.I.; Alonso, H.; Uranga, S.; Marinova, D.; Arbués, A.; de Martino, A.; Anel, A.; Monzon, M.; Badiola, J.; Pardo, J.; et al. ESX-1-induced apoptosis is involved in cell-to-cell spread of Mycobacterium tuberculosis. Cell. Microbiol. 2013, 15, 1994–2005. [Google Scholar] [CrossRef]
- Dallenga, T.; Repnik, U.; Corleis, B.; Eich, J.; Reimer, R.; Griffiths, G.W.; Schaible, U.E. M. tuberculosis-Induced Necrosis of Infected Neutrophils Promotes Bacterial Growth Following Phagocytosis by Macrophages. Cell Host & Microbe 2017, 22, 519–530.e513. [Google Scholar] [CrossRef]
- Derrick, S.C.; Morris, S.L. The ESAT6 protein of Mycobacterium tuberculosis induces apoptosis of macrophages by activating caspase expression. Cell. Microbiol. 2007, 9, 1547–1555. [Google Scholar] [CrossRef]
- Anes, E.; Azevedo-Pereira, J.M.; Pires, D. Cathepsins and Their Endogenous Inhibitors in Host Defense During Mycobacterium tuberculosis and HIV Infection. Front. Immunol. 2021, 12, 726984. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.M.; Ramakrishnan, L. The Role of the Granuloma in Expansion and Dissemination of Early Tuberculous Infection. Cell 2009, 136, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Pagán, A.J.; Ramakrishnan, L. Immunity and Immunopathology in the Tuberculous Granuloma. Cold Spring Harb. Perspect. Med. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Wolf, A.J.; Linas, B.; Trevejo-Nuñez, G.J.; Kincaid, E.; Tamura, T.; Takatsu, K.; Ernst, J.D. Mycobacterium tuberculosis Infects Dendritic Cells with High Frequency and Impairs Their Function In Vivo1. The Journal of Immunology 2007, 179, 2509–2519. [Google Scholar] [CrossRef]
- Mishra, B.B.; Rathinam, V.A.K.; Martens, G.W.; Martinot, A.J.; Kornfeld, H.; Fitzgerald, K.A.; Sassetti, C.M. Nitric oxide controls the immunopathology of tuberculosis by inhibiting NLRP3 inflammasome–dependent processing of IL-1β. Nat. Immunol. 2013, 14, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.-W.; Jacobs Jr, W.R. Critical role for NLRP3 in necrotic death triggered by Mycobacterium tuberculosis. Cell. Microbiol. 2011, 13, 1371–1384. [Google Scholar] [CrossRef] [PubMed]
- Brosch, R.; Gordon, S.V.; Marmiesse, M.; Brodin, P.; Buchrieser, C.; Eiglmeier, K.; Garnier, T.; Gutierrez, C.; Hewinson, G.; Kremer, K.; et al. A new evolutionary scenario for the Mycobacterium tuberculosis complex. Proceedings of the National Academy of Sciences 2002, 99, 3684–3689. [Google Scholar] [CrossRef]
- Brosch, R.; Gordon, S.V.; Garnier, T.; Eiglmeier, K.; Frigui, W.; Valenti, P.; Dos Santos, S.; Duthoy, S.; Lacroix, C.; Garcia-Pelayo, C.; et al. Genome plasticity of BCG and impact on vaccine efficacy. Proceedings of the National Academy of Sciences 2007, 104, 5596–5601. [Google Scholar] [CrossRef]
- Cole, S.T.; Brosch, R.; Parkhill, J.; Garnier, T.; Churcher, C.; Harris, D.; Gordon, S.V.; Eiglmeier, K.; Gas, S.; Barry, C.E.; et al. Deciphering the biology of Mycobacterium tuberculosis from the complete genome sequence. Nature 1998, 393, 537–544. [Google Scholar] [CrossRef]
- Marinova, D.; Gonzalo-Asensio, J.; Aguilo, N.; Martin, C. MTBVAC from discovery to clinical trials in tuberculosis-endemic countries. Expert Review of Vaccines 2017, 16, 565–576. [Google Scholar] [CrossRef]
- Smith, N.H.; Hewinson, R.G.; Kremer, K.; Brosch, R.; Gordon, S.V. Myths and misconceptions: the origin and evolution of Mycobacterium tuberculosis. Nature Reviews Microbiology 2009, 7, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Broset, E.; Martín, C.; Gonzalo-Asensio, J. Evolutionary Landscape of the Mycobacterium tuberculosis Complex from the Viewpoint of PhoPR: Implications for Virulence Regulation and Application to Vaccine Development. mBio 2015, 6, e01289-01215. [Google Scholar] [CrossRef] [PubMed]
- Frigui, W.; Bottai, D.; Majlessi, L.; Monot, M.; Josselin, E.; Brodin, P.; Garnier, T.; Gicquel, B.; Martin, C.; Leclerc, C.; et al. Control of M. tuberculosis ESAT-6 Secretion and Specific T Cell Recognition by PhoP. PLOS Pathogens 2008, 4, e33. [Google Scholar] [CrossRef] [PubMed]
- Pérez, E.; Samper, S.; Bordas, Y.; Guilhot, C.; Gicquel, B.; Martín, C. An essential role for phoP in Mycobacterium tuberculosis virulence. Mol. Microbiol. 2001, 41, 179–187. [Google Scholar] [CrossRef]
- Mahairas, G.G.; Sabo, P.J.; Hickey, M.J.; Singh, D.C.; Stover, C.K. Molecular analysis of genetic differences between Mycobacterium bovis BCG and virulent M. bovis. J. Bacteriol. 1996, 178, 1274–1282. [Google Scholar] [CrossRef]
- Pym, A.S.; Brodin, P.; Brosch, R.; Huerre, M.; Cole, S.T. Loss of RD1 contributed to the attenuation of the live tuberculosis vaccines Mycobacterium bovis BCG and Mycobacterium microti. Mol. Microbiol. 2002, 46, 709–717. [Google Scholar] [CrossRef]
- de Jong, B.C.; Hill, P.C.; Brookes, R.H.; Gagneux, S.; Jeffries, D.J.; Otu, J.K.; Donkor, S.A.; Fox, A.; McAdam, K.P.W.J.; Small, P.M.; et al. Mycobacterium africanum Elicits an Attenuated T Cell Response to Early Secreted Antigenic Target, 6 kDa, in Patients with Tuberculosis and Their Household Contacts. The Journal of Infectious Diseases 2006, 193, 1279–1286. [Google Scholar] [CrossRef]
- Gonzalo-Asensio, J.; Pérez, I.; Aguiló, N.; Uranga, S.; Picó, A.; Lampreave, C.; Cebollada, A.; Otal, I.; Samper, S.; Martín, C. New insights into the transposition mechanisms of IS6110 and its dynamic distribution between Mycobacterium tuberculosis Complex lineages. PLoS Genet. 2018, 14, e1007282. [Google Scholar] [CrossRef]
- Soto, C.Y.; Menendez, M.C.; Perez, E.; Samper, S.; Gomez, A.B.; Garcia, M.J.; Martin, C. IS6110 Mediates Increased Transcription of the phoP Virulence Gene in a Multidrug-Resistant Clinical Isolate Responsible for Tuberculosis Outbreaks. J. Clin. Microbiol. 2004, 42, 212–219. [Google Scholar] [CrossRef]
- Abdallah, A.M.; Gey van Pittius, N.C.; DiGiuseppe Champion, P.A.; Cox, J.; Luirink, J.; Vandenbroucke-Grauls, C.M.J.E.; Appelmelk, B.J.; Bitter, W. Type VII secretion — mycobacteria show the way. Nature Reviews Microbiology 2007, 5, 883–891. [Google Scholar] [CrossRef]
- Gröschel, M.I.; Sayes, F.; Simeone, R.; Majlessi, L.; Brosch, R. ESX secretion systems: mycobacterial evolution to counter host immunity. Nature Reviews Microbiology 2016, 14, 677–691. [Google Scholar] [CrossRef] [PubMed]
- Andersen, P.; Andersen, A.B.; Sørensen, A.L.; Nagai, S. Recall of long-lived immunity to Mycobacterium tuberculosis infection in mice. The Journal of Immunology 1995, 154, 3359–3372. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, A.L.; Nagai, S.; Houen, G.; Andersen, P.; Andersen, A.B. Purification and characterization of a low-molecular-mass T-cell antigen secreted by Mycobacterium tuberculosis. Infection and Immunity 1995, 63, 1710–1717. [Google Scholar] [CrossRef] [PubMed]
- Brandt, L.; Elhay, M.; Rosenkrands, I.; Lindblad Erik, B.; Andersen, P. ESAT-6 Subunit Vaccination against Mycobacterium tuberculosis. Infection and Immunity 2000, 68, 791–795. [Google Scholar] [CrossRef] [PubMed]
- Ruhwald, M.; de Thurah, L.; Kuchaka, D.; Zaher, M.R.; Salman, A.M.; Abdel-Ghaffar, A.-R.; Shoukry, F.A.; Michelsen, S.W.; Soborg, B.; Blauenfeldt, T.; et al. Introducing the ESAT-6 free IGRA, a companion diagnostic for TB vaccines based on ESAT-6. Sci. Rep. 2017, 7, 45969. [Google Scholar] [CrossRef]
- Brodin, P.; Rosenkrands, I.; Andersen, P.; Cole, S.T.; Brosch, R. ESAT-6 proteins: protective antigens and virulence factors? Trends Microbiol. 2004, 12, 500–508. [Google Scholar] [CrossRef]
- Wang, J.; McIntosh, F.; Radomski, N.; Dewar, K.; Simeone, R.; Enninga, J.; Brosch, R.; Rocha, E.P.; Veyrier, F.J.; Behr, M.A. Insights on the Emergence of Mycobacterium tuberculosis from the Analysis of Mycobacterium kansasii. Genome Biol. Evol. 2015, 7, 856–870. [Google Scholar] [CrossRef]
- Wirth, S.E.; Krywy, J.A.; Aldridge, B.B.; Fortune, S.M.; Fernandez-Suarez, M.; Gray, T.A.; Derbyshire, K.M. Polar assembly and scaffolding proteins of the virulence-associated ESX-1 secretory apparatus in mycobacteria. Mol. Microbiol. 2012, 83, 654–664. [Google Scholar] [CrossRef]
- Anes, E.; Peyron, P.; Staali, L.; Jordao, L.; Gutierrez, M.G.; Kress, H.; Hagedorn, M.; Maridonneau-Parini, I.; Skinner, M.A.; Wildeman, A.G.; et al. Dynamic life and death interactions between Mycobacterium smegmatis and J774 macrophages. Cell. Microbiol. 2006, 8, 939–960. [Google Scholar] [CrossRef]
- Coros, A.; Callahan, B.; Battaglioli, E.; Derbyshire, K.M. The specialized secretory apparatus ESX-1 is essential for DNA transfer in Mycobacterium smegmatis. Mol. Microbiol. 2008, 69, 794–808. [Google Scholar] [CrossRef]
- Flint, J.L.; Kowalski, J.C.; Karnati, P.K.; Derbyshire, K.M. The RD1 virulence locus of Mycobacterium tuberculosis regulates DNA transfer in Mycobacterium smegmatis. Proceedings of the National Academy of Sciences 2004, 101, 12598–12603. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.; Becq, J.; Gicquel, B.; Deschavanne, P.; Neyrolles, O. Horizontally acquired genomic islands in the tubercle bacilli. Trends Microbiol. 2008, 16, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Ates, L.S.; Brosch, R. Discovery of the type VII ESX-1 secretion needle? Mol. Microbiol. 2017, 103, 7–12. [Google Scholar] [CrossRef]
- Boritsch, E.C.; Supply, P.; Honoré, N.; Seeman, T.; Stinear, T.P.; Brosch, R. A glimpse into the past and predictions for the future: the molecular evolution of the tuberculosis agent. Mol. Microbiol. 2014, 93, 835–852. [Google Scholar] [CrossRef] [PubMed]
- MacGurn, J.A.; Raghavan, S.; Stanley, S.A.; Cox, J.S. A non-RD1 gene cluster is required for Snm secretion in Mycobacterium tuberculosis. Mol. Microbiol. 2005, 57, 1653–1663. [Google Scholar] [CrossRef]
- Vaziri, F.; Brosch, R. ESX/Type VII Secretion Systems—An Important Way Out for Mycobacterial Proteins. Microbiology Spectrum 2019, 7, 7.4.10. [Google Scholar] [CrossRef]
- Gao, L.-Y.; Guo, S.; McLaughlin, B.; Morisaki, H.; Engel, J.N.; Brown, E.J. A mycobacterial virulence gene cluster extending RD1 is required for cytolysis, bacterial spreading and ESAT-6 secretion. Mol. Microbiol. 2004, 53, 1677–1693. [Google Scholar] [CrossRef]
- Guinn, K.M.; Hickey, M.J.; Mathur, S.K.; Zakel, K.L.; Grotzke, J.E.; Lewinsohn, D.M.; Smith, S.; Sherman, D.R. Individual RD1-region genes are required for export of ESAT-6/CFP-10 and for virulence of Mycobacterium tuberculosis. Mol. Microbiol. 2004, 51, 359–370. [Google Scholar] [CrossRef]
- Hsu, T.; Hingley-Wilson, S.M.; Chen, B.; Chen, M.; Dai, A.Z.; Morin, P.M.; Marks, C.B.; Padiyar, J.; Goulding, C.; Gingery, M.; et al. The primary mechanism of attenuation of bacillus Calmette–Guérin is a loss of secreted lytic function required for invasion of lung interstitial tissue. Proceedings of the National Academy of Sciences 2003, 100, 12420–12425. [Google Scholar] [CrossRef]
- Lewis, K.N.; Liao, R.; Guinn, K.M.; Hickey, M.J.; Smith, S.; Behr, M.A.; Sherman, D.R. Deletion of RD1 from Mycobacterium tuberculosis Mimics Bacille Calmette-Guérin Attenuation. The Journal of Infectious Diseases 2003, 187, 117–123. [Google Scholar] [CrossRef]
- Sassetti, C.M.; Rubin, E.J. Genetic requirements for mycobacterial survival during infection. Proceedings of the National Academy of Sciences 2003, 100, 12989–12994. [Google Scholar] [CrossRef] [PubMed]
- Stanley, S.A.; Raghavan, S.; Hwang, W.W.; Cox, J.S. Acute infection and macrophage subversion by Mycobacterium tuberculosis require a specialized secretion system. Proceedings of the National Academy of Sciences 2003, 100, 13001–13006. [Google Scholar] [CrossRef] [PubMed]
- Simeone, R.; Bottai, D.; Frigui, W.; Majlessi, L.; Brosch, R. ESX/type VII secretion systems of mycobacteria: Insights into evolution, pathogenicity and protection. Tuberculosis 2015, 95, S150–S154. [Google Scholar] [CrossRef] [PubMed]
- Steenken, W., Jr.; Oatway, W.H., Jr.; Petroff, S.A. Biological Studies of the Tubercle Bacillus : Iii. Dissociation and Pathogenicity of the R and S Variants of the Human Tubercle Bacillus (H(37)). J. Exp. Med. 1934, 60, 515–540. [Google Scholar] [CrossRef] [PubMed]
- Solans, L.; Aguiló, N.; Samper, S.; Pawlik, A.; Frigui, W.; Martín, C.; Brosch, R.; Gonzalo-Asensio, J. A Specific Polymorphism in Mycobacterium tuberculosis H37Rv Causes Differential ESAT-6 Expression and Identifies WhiB6 as a Novel ESX-1 Component. Infection and Immunity 2014, 82, 3446–3456. [Google Scholar] [CrossRef] [PubMed]
- Behura, A.; Das, M.; Kumar, A.; Naik, L.; Mishra, A.; Manna, D.; Patel, S.; Mishra, A.; Singh, R.; Dhiman, R. ESAT-6 impedes IL-18 mediated phagosome lysosome fusion via microRNA-30a upon Calcimycin treatment in mycobacteria infected macrophages. Int. Immunopharmacol. 2021, 101, 108319. [Google Scholar] [CrossRef]
- Tan, T.; Lee, W.L.; Alexander, D.C.; Grinstein, S.; Liu, J. The ESAT-6/CFP-10 secretion system of Mycobacterium marinum modulates phagosome maturation. Cell. Microbiol. 2006, 8, 1417–1429. [Google Scholar] [CrossRef]
- Simeone, R.; Bobard, A.; Lippmann, J.; Bitter, W.; Majlessi, L.; Brosch, R.; Enninga, J. Phagosomal Rupture by Mycobacterium tuberculosis Results in Toxicity and Host Cell Death. PLOS Pathogens 2012, 8, e1002507. [Google Scholar] [CrossRef]
- van der Wel, N.; Hava, D.; Houben, D.; Fluitsma, D.; van Zon, M.; Pierson, J.; Brenner, M.; Peters, P.J. M. tuberculosis and M. leprae Translocate from the Phagolysosome to the Cytosol in Myeloid Cells. Cell 2007, 129, 1287–1298. [Google Scholar] [CrossRef]
- Elkington, P.T.; Green, J.A.; Emerson, J.E.; Lopez-Pascua, L.D.; Boyle, J.J.; O'Kane, C.M.; Friedland, J.S. Synergistic up-regulation of epithelial cell matrix metalloproteinase-9 secretion in tuberculosis. Am. J. Respir. Cell Mol. Biol. 2007, 37, 431–437. [Google Scholar] [CrossRef]
- Volkman, H.E.; Pozos, T.C.; Zheng, J.; Davis, J.M.; Rawls, J.F.; Ramakrishnan, L. Tuberculous Granuloma Induction via Interaction of a Bacterial Secreted Protein with Host Epithelium. Science 2010, 327, 466–469. [Google Scholar] [CrossRef] [PubMed]
- Augenstreich, J.; Arbues, A.; Simeone, R.; Haanappel, E.; Wegener, A.; Sayes, F.; Le Chevalier, F.; Chalut, C.; Malaga, W.; Guilhot, C.; et al. ESX-1 and phthiocerol dimycocerosates of Mycobacterium tuberculosis act in concert to cause phagosomal rupture and host cell apoptosis. Cell. Microbiol. 2017, 19, e12726. [Google Scholar] [CrossRef] [PubMed]
- Watson, R.O.; Bell, S.L.; MacDuff, D.A.; Kimmey, J.M.; Diner, E.J.; Olivas, J.; Vance, R.E.; Stallings, C.L.; Virgin, H.W.; Cox, J.S. The Cytosolic Sensor cGAS Detects Mycobacterium tuberculosis DNA to Induce Type I Interferons and Activate Autophagy. Cell Host & Microbe 2015, 17, 811–819. [Google Scholar] [CrossRef]
- Grover, A.; Izzo, A.A. BAT3 Regulates Mycobacterium tuberculosis Protein ESAT-6-Mediated Apoptosis of Macrophages. PLoS One 2012, 7, e40836. [Google Scholar] [CrossRef] [PubMed]
- Welin, A.; Eklund, D.; Stendahl, O.; Lerm, M. Human Macrophages Infected with a High Burden of ESAT-6-Expressing M. tuberculosis Undergo Caspase-1- and Cathepsin B-Independent Necrosis. PLoS One 2011, 6, e20302. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, M.G.; Master, S.S.; Singh, S.B.; Taylor, G.A.; Colombo, M.I.; Deretic, V. Autophagy is a defense mechanism inhibiting BCG and Mycobacterium tuberculosis survival in infected macrophages. Cell 2004, 119, 753–766. [Google Scholar] [CrossRef] [PubMed]
- Wassermann, R.; Gulen, M.F.; Sala, C.; Perin, S.G.; Lou, Y.; Rybniker, J.; Schmid-Burgk, J.L.; Schmidt, T.; Hornung, V.; Cole, S.T.; et al. Mycobacterium tuberculosis Differentially Activates cGAS- and Inflammasome-Dependent Intracellular Immune Responses through ESX-1. Cell Host & Microbe 2015, 17, 799–810. [Google Scholar] [CrossRef]
- Majlessi, L.; Brosch, R. Mycobacterium tuberculosis Meets the Cytosol: The Role of cGAS in Anti-mycobacterial Immunity. Cell Host & Microbe 2015, 17, 733–735. [Google Scholar] [CrossRef]
- Shafiani, S.; Dinh, C.; Ertelt, J.M.; Moguche, A.O.; Siddiqui, I.; Smigiel, K.S.; Sharma, P.; Campbell, D.J.; Way, S.S.; Urdahl, K.B. Pathogen-Specific Treg Cells Expand Early during Mycobacterium tuberculosis Infection but Are Later Eliminated in Response to Interleukin-12. Immunity 2013, 38, 1261–1270. [Google Scholar] [CrossRef]
- Banaiee, N.; Kincaid, E.Z.; Buchwald, U.; Jacobs, W.R., Jr.; Ernst, J.D. Potent Inhibition of Macrophage Responses to IFN-γ by Live Virulent Mycobacterium tuberculosis Is Independent of Mature Mycobacterial Lipoproteins but Dependent on TLR21. The Journal of Immunology 2006, 176, 3019–3027. [Google Scholar] [CrossRef]
- Sreejit, G.; Ahmed, A.; Parveen, N.; Jha, V.; Valluri, V.L.; Ghosh, S.; Mukhopadhyay, S. The ESAT-6 Protein of Mycobacterium tuberculosis Interacts with Beta-2-Microglobulin (β2M) Affecting Antigen Presentation Function of Macrophage. PLOS Pathogens 2014, 10, e1004446. [Google Scholar] [CrossRef] [PubMed]
- Bretl, D.J.; Demetriadou, C.; Zahrt, T.C. Adaptation to Environmental Stimuli within the Host: Two-Component Signal Transduction Systems of Mycobacterium tuberculosis. Microbiology and Molecular Biology Reviews 2011, 75, 566–582. [Google Scholar] [CrossRef] [PubMed]
- Polena, H.; Boudou, F.; Tilleul, S.; Dubois-Colas, N.; Lecointe, C.; Rakotosamimanana, N.; Pelizzola, M.; Andriamandimby, S.F.; Raharimanga, V.; Charles, P.; et al. Mycobacterium tuberculosis exploits the formation of new blood vessels for its dissemination. Sci. Rep. 2016, 6, 33162. [Google Scholar] [CrossRef] [PubMed]
- Lerner, T.R.; Borel, S.; Greenwood, D.J.; Repnik, U.; Russell, M.R.G.; Herbst, S.; Jones, M.L.; Collinson, L.M.; Griffiths, G.; Gutierrez, M.G. Mycobacterium tuberculosis replicates within necrotic human macrophages. J. Cell Biol. 2017, 216, 583–594. [Google Scholar] [CrossRef] [PubMed]
- Mayer-Barber, K.D.; Andrade, B.B.; Oland, S.D.; Amaral, E.P.; Barber, D.L.; Gonzales, J.; Derrick, S.C.; Shi, R.; Kumar, N.P.; Wei, W.; et al. Host-directed therapy of tuberculosis based on interleukin-1 and type I interferon crosstalk. Nature 2014, 511, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Mourik, B.C.; Lubberts, E.; de Steenwinkel, J.E.M.; Ottenhoff, T.H.M.; Leenen, P.J.M. Interactions between Type 1 Interferons and the Th17 Response in Tuberculosis: Lessons Learned from Autoimmune Diseases. Front. Immunol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- McNab, F.; Mayer-Barber, K.; Sher, A.; Wack, A.; O'Garra, A. Type I interferons in infectious disease. Nature Reviews Immunology 2015, 15, 87–103. [Google Scholar] [CrossRef]
- Behar, S.M.; Martin, C.J.; Booty, M.G.; Nishimura, T.; Zhao, X.; Gan, H.X.; Divangahi, M.; Remold, H.G. Apoptosis is an innate defense function of macrophages against Mycobacterium tuberculosis. Mucosal Immunol. 2011, 4, 279–287. [Google Scholar] [CrossRef]
- Roca, F.J.; Ramakrishnan, L. TNF Dually Mediates Resistance and Susceptibility to Mycobacteria via Mitochondrial Reactive Oxygen Species. Cell 2013, 153, 521–534. [Google Scholar] [CrossRef]
- Russell, D.G.; Cardona, P.-J.; Kim, M.-J.; Allain, S.; Altare, F. Foamy macrophages and the progression of the human tuberculosis granuloma. Nat. Immunol. 2009, 10, 943–948. [Google Scholar] [CrossRef]
- Griffin, G.K.; Newton, G.; Tarrio, M.L.; Bu, D.-x.; Maganto-Garcia, E.; Azcutia, V.; Alcaide, P.; Grabie, N.; Luscinskas, F.W.; Croce, K.J.; et al. IL-17 and TNF-α Sustain Neutrophil Recruitment during Inflammation through Synergistic Effects on Endothelial Activation. The Journal of Immunology 2012, 188, 6287–6299. [Google Scholar] [CrossRef] [PubMed]
- Poh, X.Y.; Loh, F.K.; Friedland, J.S.; Ong, C.W.M. Neutrophil-Mediated Immunopathology and Matrix Metalloproteinases in Central Nervous System – Tuberculosis. Front. Immunol. 2022, 12. [Google Scholar] [CrossRef] [PubMed]
- Dallenga, T.; Schaible, U.E. Neutrophils in tuberculosis--first line of defence or booster of disease and targets for host-directed therapy? Pathogens and Disease 2016, 74, ftw012. [Google Scholar] [CrossRef] [PubMed]
- Martín, C.; Marinova, D.; Aguiló, N.; Gonzalo-Asensio, J. MTBVAC, a live TB vaccine poised to initiate efficacy trials 100 years after BCG. Vaccine 2021, 39, 7277–7285. [Google Scholar] [CrossRef] [PubMed]
- Camacho, L.R.; Constant, P.; Raynaud, C.; Lanéelle, M.-A.; Triccas, J.A.; Gicquel, B.; Daffé, M.; Guilhot, C. Analysis of the Phthiocerol Dimycocerosate Locus of Mycobacterium tuberculosis: EVIDENCE THAT THIS LIPID IS INVOLVED IN THE CELL WALL PERMEABILITY BARRIER *. J. Biol. Chem. 2001, 276, 19845–19854. [Google Scholar] [CrossRef] [PubMed]
- Quigley, J.; Hughitt, V.K.; Velikovsky Carlos, A.; Mariuzza Roy, A.; El-Sayed Najib, M.; Briken, V. The Cell Wall Lipid PDIM Contributes to Phagosomal Escape and Host Cell Exit of Mycobacterium tuberculosis. mBio 2017, 8, e00148-00117. [Google Scholar] [CrossRef] [PubMed]
- Sable, S.B.; Posey, J.E.; Scriba, T.J. Tuberculosis Vaccine Development: Progress in Clinical Evaluation. Clin. Microbiol. Rev. 2019, 33, e00100–00119. [Google Scholar] [CrossRef] [PubMed]
- Tkachuk, A.P.; Gushchin, V.A.; Potapov, V.D.; Demidenko, A.V.; Lunin, V.G.; Gintsburg, A.L. Multi-subunit BCG booster vaccine GamTBvac: Assessment of immunogenicity and protective efficacy in murine and guinea pig TB models. PLoS One 2017, 12, e0176784. [Google Scholar] [CrossRef]
- Moguche, A.O.; Musvosvi, M.; Penn-Nicholson, A.; Plumlee, C.R.; Mearns, H.; Geldenhuys, H.; Smit, E.; Abrahams, D.; Rozot, V.; Dintwe, O.; et al. Antigen Availability Shapes T Cell Differentiation and Function during Tuberculosis. Cell Host & Microbe 2017, 21, 695–706.e695. [Google Scholar] [CrossRef]
- Pai, M.; Denkinger Claudia, M.; Kik Sandra, V.; Rangaka Molebogeng, X.; Zwerling, A.; Oxlade, O.; Metcalfe John, Z.; Cattamanchi, A.; Dowdy David, W.; Dheda, K.; et al. Gamma Interferon Release Assays for Detection of Mycobacterium tuberculosis Infection. Clin. Microbiol. Rev. 2014, 27, 3–20. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



