Submitted:
04 July 2025
Posted:
08 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Model Settings
2.1. General Settings
2.2. The Medical Test and the Accuracy
2.3. Expected NMB with the Medical Test
3. Decision Making
3.1. Decision Making without the Test
3.2. Decision Making with the Test
4. Cost-Effectiveness Analysis For The Medical Test
5. The Case Of Sensitivity And Specificity
-
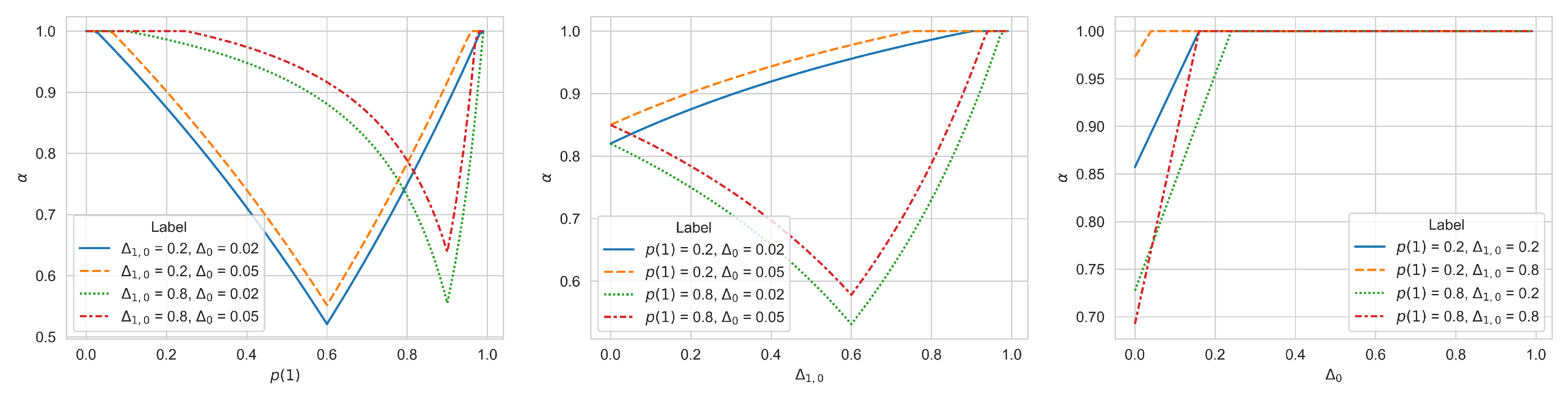
The influence of the percentage of patients in the population():The influence of on the lower bound shows an obvious V-shaped curve. When approaches 0 or 1, the lower bound increases significantly, indicating that detection is difficult. Cost-effectiveness. This can be understood by comparing it with the "no detection" situation: when is close to 0, the simple "no treatment" strategy can achieve better results; when is close to 1, the "treat all" strategy can achieve better results. This also reminds us that the value of testing lies in its ability to significantly change medical decisions, and it should be considered together with the corresponding cost of treatment. This finding has important implications for testing strategies in different disease areas: For rare diseases, multiple factors may need to be considered to consider the value of testing; for common diseases, the cost-effectiveness of a universal prevention strategy versus targeted testing may need to be weighed. In addition, this relationship also explains why the development of diagnostic technologies for certain rare diseases is challenging: not only because the market is small, but also because the test itself is difficult to be cost effective at low .
-
The impact of the relative price of the treatment():When is small, the lower bound is positively correlated with . This is because, in low prevalence settings, expensive treatments must be used more cautiously to avoid wasting resources on many healthy people who do not need treatment. As the cost of treatment increases, medical testing requires greater precision to identify patients who truly need treatment accurately. However, when is large, within certain intervals, expensive treatments require a lower detection accuracy. The following mechanism can explain this seemingly contradictory phenomenon: a) In the case of high , if there is no detection, the optimal decision is to treat everyone universally. b) As the cost of treatment increases, the greater the cost savings through testing to distinguish the few negative cases to avoid unnecessary treatment. c) Therefore, in cases where is not high enough but treatment costs are high, even a test that is not very accurate may be cost-effective because it can help reduce some unnecessary expensive treatments. This complex relationship reveals an essential trade-off in medical decision-making. In the context of high treatment costs and high morbidity, a less precise test that roughly distinguishes patients may be better than no test or a costly test with high accuracy. Testing is more cost-effective. However, it should be noted that when continues to increase beyond a certain threshold, the lower bound will rise again. This is because the incorrect use of extremely expensive treatments is too costly, requiring greater detection accuracy.
-
The impact of the relative price of the medical test ():When is below a specific critical value, the lower bound increases as increases; however, once exceeds a specific critical value, the lower bound suddenly jumps to greater than 1, which means that no matter how the detection accuracy is improved, cost-effectiveness cannot be achieved. This threshold effect reveals a crucial economic reality: there is an upper limit on the cost of testing, beyond which testing becomes economically unfeasible. It is worth noting that this threshold is not fixed but will be affected by other factors, such as disease prevalence and treatment costs.
6. A Numerical Simulation Example
7. Conclusions and Discussion
8. Declarations
- Funding - No funding has been received for the conduct of this study.
- Conflicts of Interest - Authors Xinyue Zeng, Ermo Chen, and Huajie Jin declare that he has no conflict of interest.
- Availability of data and material - Not applicable.
- Ethics approval - Not applicable.
- Consent to participate - Not applicable.
- Consent for publication - Not applicable.
- Code availability - The code in this study is available on request from the corresponding author.
- Author contributions - Not applicable.
References
- R. Thomas, W. J. M. Probert, R. Sauter, L. Mwenge, S. Singh, S. Kanema, N. Vanqa, A. Harper, R. Burger, A. Cori, M. Pickles, N. Bell-Mandla, B. Yang, J. Bwalya, M. Phiri, K. Shanaube, S. Floyd, D. Donnell, P. Bock, H. Ayles, S. Fidler, R. J. Hayes, C. Fraser, K. Hauck. Cost and cost-effectiveness of a universal hiv testing and treatment intervention in zambia and south africa: evidence and projections from the hptn 071 (popart) trial. The Lancet Global Health 2021, 9, e668–e680. [Google Scholar] [CrossRef] [PubMed]
- E. Jacobsen, S. Sawhney, M. Brazzelli, L. Aucott, G. Scotland, M. Aceves-Martins, C. Robertson, M. Imamura, A. Poobalan, P. Manson, C. Kaye, D. Boyers. Cost-effectiveness and value of information analysis of nephrocheck and ngal tests compared to standard care for the diagnosis of acute kidney injury. BMC nephrology 2021, 22, 399. [Google Scholar] [CrossRef] [PubMed]
- N. Choi, S. Jang, K. H. Yoo, C. K. Rhee, Y. Kim. The effectiveness and harms of screening for chronic obstructive pulmonary disease: An updated systematic review and meta-analysis. Journal of Korean medical science 2022, 37, e117. [Google Scholar] [CrossRef]
- C. Fang, H. J. Otero, D. Greenberg, P. J. Neumann. Cost-utility analyses of diagnostic laboratory tests: A systematic review, Value in health. Value Health 2011, 14, 1010–1018. [Google Scholar] [CrossRef]
- Y. Yang, L. Abel, J. Buchanan, T. Fanshawe, B. Shinkin. Shinkins, Use of decision modelling in economic evaluations of diagnostic tests: An appraisal and review of health technology assessments in the uk. PharmacoEconomics 2019, 3, 281–291. [Google Scholar] [CrossRef] [PubMed]
- T. Snowsill. Modelling the cost-effectiveness of diagnostic tests. PharmacoEconomics 2023, 41, 339–351. [Google Scholar] [CrossRef]
- D. I. Ling, L. D. Lynd, M. Harrison, A. H. Anis, N. Bansback. Early cost–effectiveness modeling for better decisions in public research investment of personalized medicine technologies. Journal of Comparative Effectiveness Research 2019, 8, 7–19. [Google Scholar] [CrossRef]
- M. Kasztura, A. Richard, N.-E. Bempong, D. Loncar, A. Flahault. Cost-effectiveness of precision medicine: a scoping review. International journal of public health 2019, 64, 1261–1271. [Google Scholar] [CrossRef]
- A. I. Mushlin, H. S. Ruchlin, M. A. Callahan. Costeffectiveness of diagnostic tests. The Lancet 2001, 358, 1353–1355. [Google Scholar] [CrossRef]
- H. Koffijberg, B. van Zaane, K. G. Moons. From accuracy to patient outcome and cost-effectiveness evaluations of diagnostic tests and biomarkers: an exemplary modelling study. BMC medical research methodology 2013, 13, 1–12. [Google Scholar] [CrossRef]
- A. F. Smith, A. Sutton, B. Shinkins. Early cost-effectiveness analysis of new medical tests: Response. International journal of technology assessment in health care 2016, 32, 324–325. [Google Scholar] [CrossRef] [PubMed]
- S. P. Gavan, A. J. Thompson, K. Payne. The economic case for precision medicine. Expert review of precision medicine and drug development 2018, 3, 1–9. [Google Scholar] [CrossRef] [PubMed]
- L. D. Mattos-Arruda, G. Siravegna. How to use liquid biopsies to treat patients with cancer. ESMO Open 2021, 6. [Google Scholar] [CrossRef]
- W. Chen, T. Anothaisintawee, D. Butani, Y. Wang, Y. Zemlyanska, C. Boon, N. Wong, S. Virabhak, M. A. Hrishikesh, Y. Teerawattananon. Assessing the cost-effectiveness of precision medicine: protocol for a systematic review and meta-analysis. BMJ Open 2022, 12. [Google Scholar] [CrossRef]
- J. S. Hernandez. Cost-effectiveness of laboratory testing. Archives of pathology and laboratory medicine 2003, 127, 440–445. [Google Scholar] [CrossRef]
- A. Van den Bruel, I. Cleemput, B. Aertgeerts, D. Ramaekers, F. Buntinx. The evaluation of diagnostic tests: evidence on technical and diagnostic accuracy, impact on patient outcome and cost-effectiveness is needed. Journal of clinical epidemiology 2007, 60, 1116–1122. [Google Scholar] [CrossRef]
- M. K. Seo, M. Strong. A practical guide to modeling and conducting a cost-effectiveness analysis of companion biomarker tests for targeted therapies using r: Tutorial paper. PharmacoEconomics 2021, 39, 1373–1381. [Google Scholar] [CrossRef]
- N. Novielli, N. J. Cooper, K. R. Abrams, A. J. Sutton. How is evidence on test performance synthesized for economic decision models of diagnostic tests? a systematic appraisal of health technology assessments in the uk since 1997. Value in Health 2010, 13, 952–957. [Google Scholar] [CrossRef]
- N. Novielli, N. J. Cooper, A. J. Sutton. Evaluating the cost-effectiveness of diagnostic tests in combination: Is it important to allow for performance dependency? Value in health 2013, 16, 536–541. [Google Scholar] [CrossRef]
- V. Garg, N. Y. Gu, M. E. Borrego, D. W. Raisch. A literature review of cost-effectiveness analyses of prostate-specific antigen test in prostate cancer screening. Expert review of pharmacoeconomics and outcomes research 2013, 13, 327–342. [Google Scholar] [CrossRef]
- E. Biltaji, B. Bellows, D. Stenehjem, D. Brixner. Validation of a cost-effectiveness model comparing accuracy of genetic tests for brca mutations. Value in health 2016, 19, A79. [Google Scholar] [CrossRef]
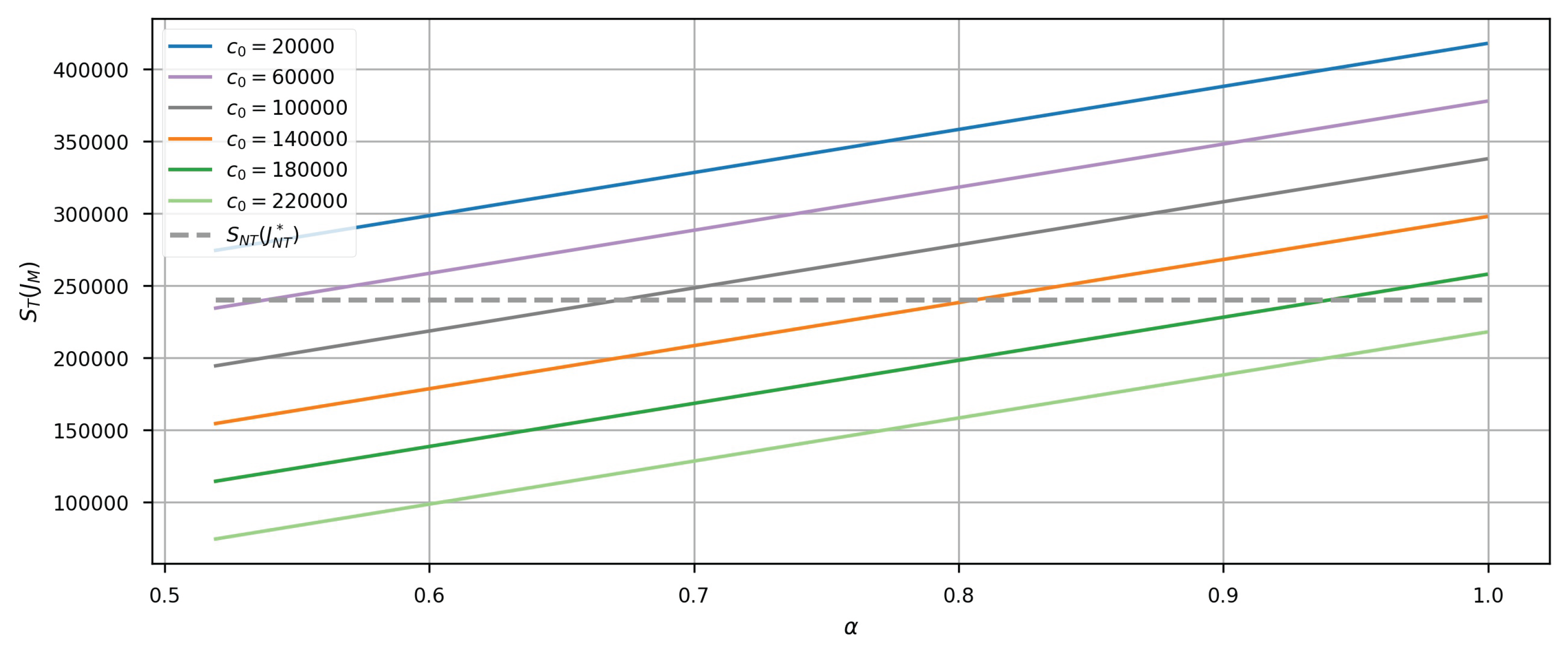
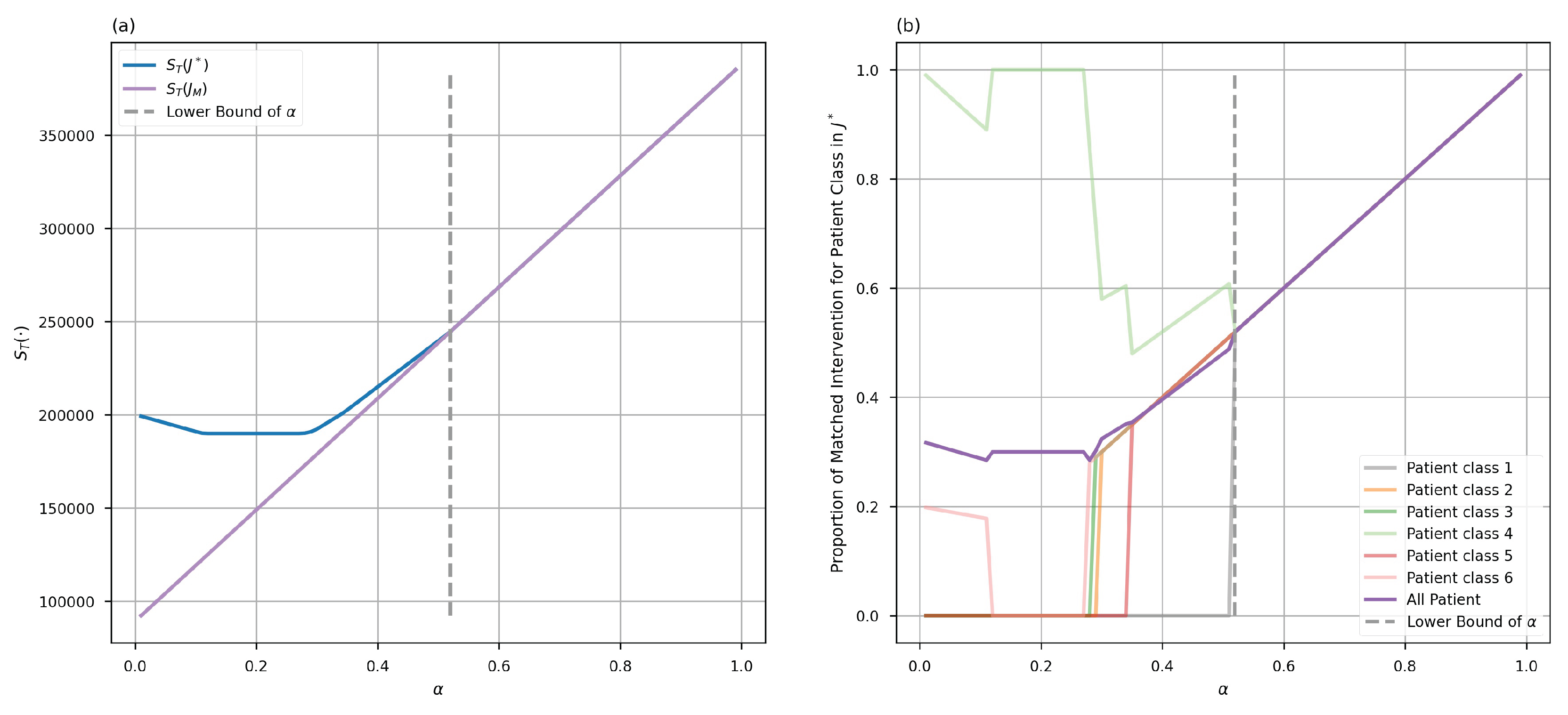
| Patient Class i | 1 | 2 | 3 | 4 | 5 | 6 |
| 200,000 | 180,000 | 175,000 | 150,000 | 140,000 | 120,000 | |
| 0.1 | 0.2 | 0.2 | 0.3 | 0.1 | 0.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




