Submitted:
28 April 2023
Posted:
04 May 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Generation of EBER1 stem-loop mutants, cloning and transfection
2.2. Determination of plasmid copy number
2.3. Cell culture
2.4. Isolation and characterization of exosomes
2.5. Isolation of total and subcellular nucleic acids and qRT-PCR
2.6. Isolation of total and subcellular proteins and Western Blot
2.7. In silico prediction of RNA structure and protein-RNA interactions
2.8. siRNA silencing of RPL22 and La proteins
2.9. Evaluation of the intracellular calcium influx via the SOCE
2.10. Statistical analysis
3. Results
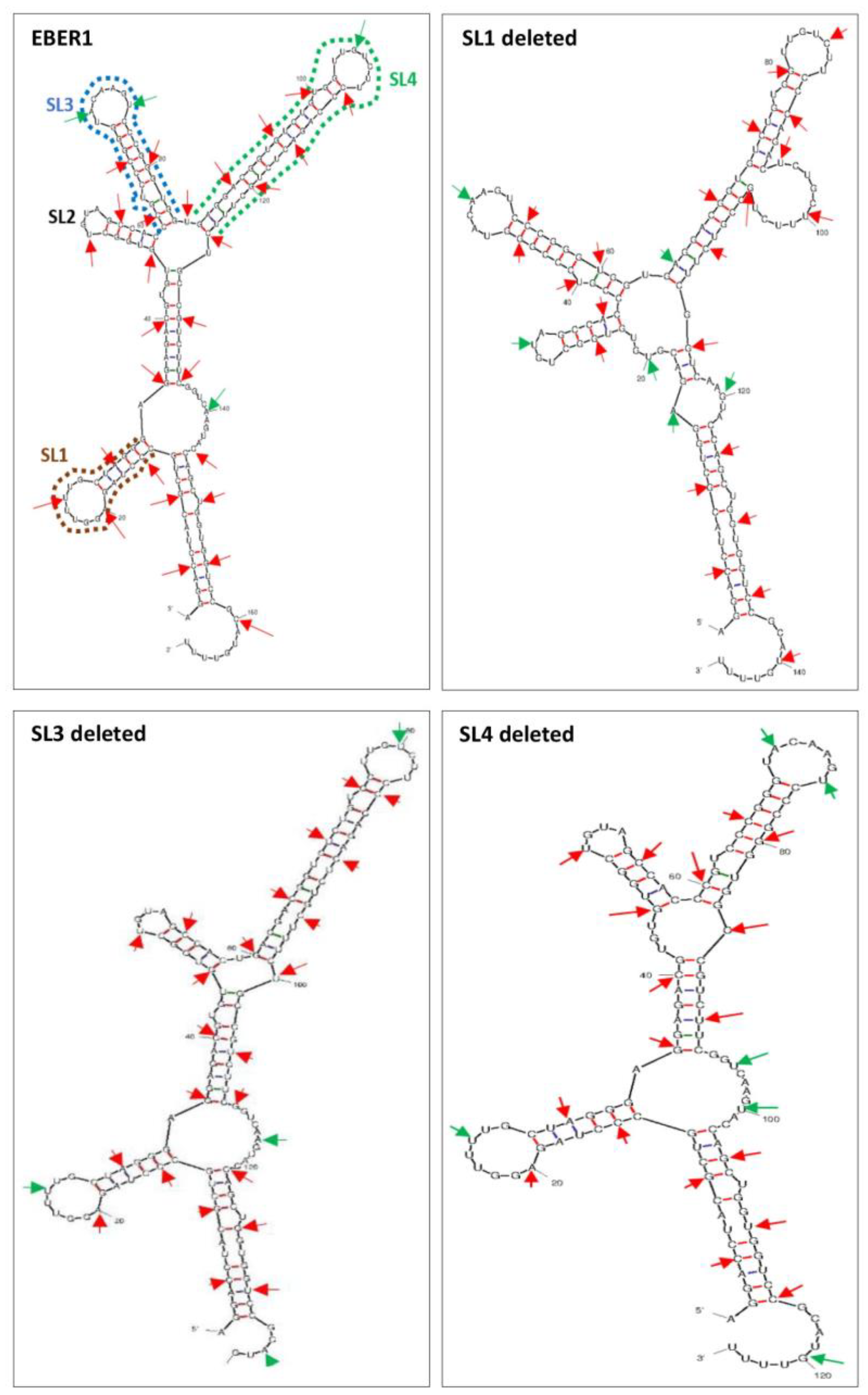
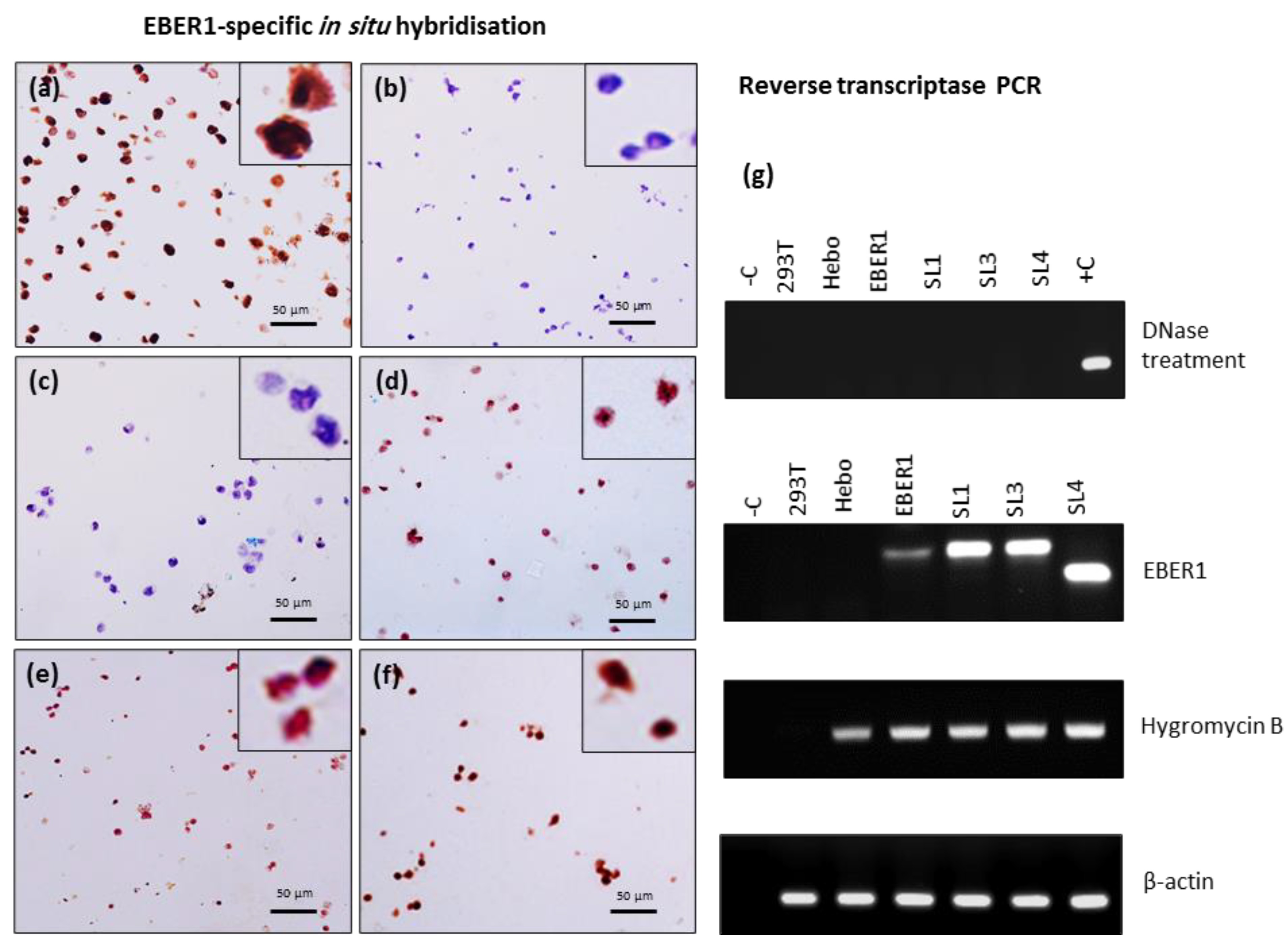
3.1. Prediction of RNA structure, confirmation of mutation and transfection, and determination of plasmid copy number
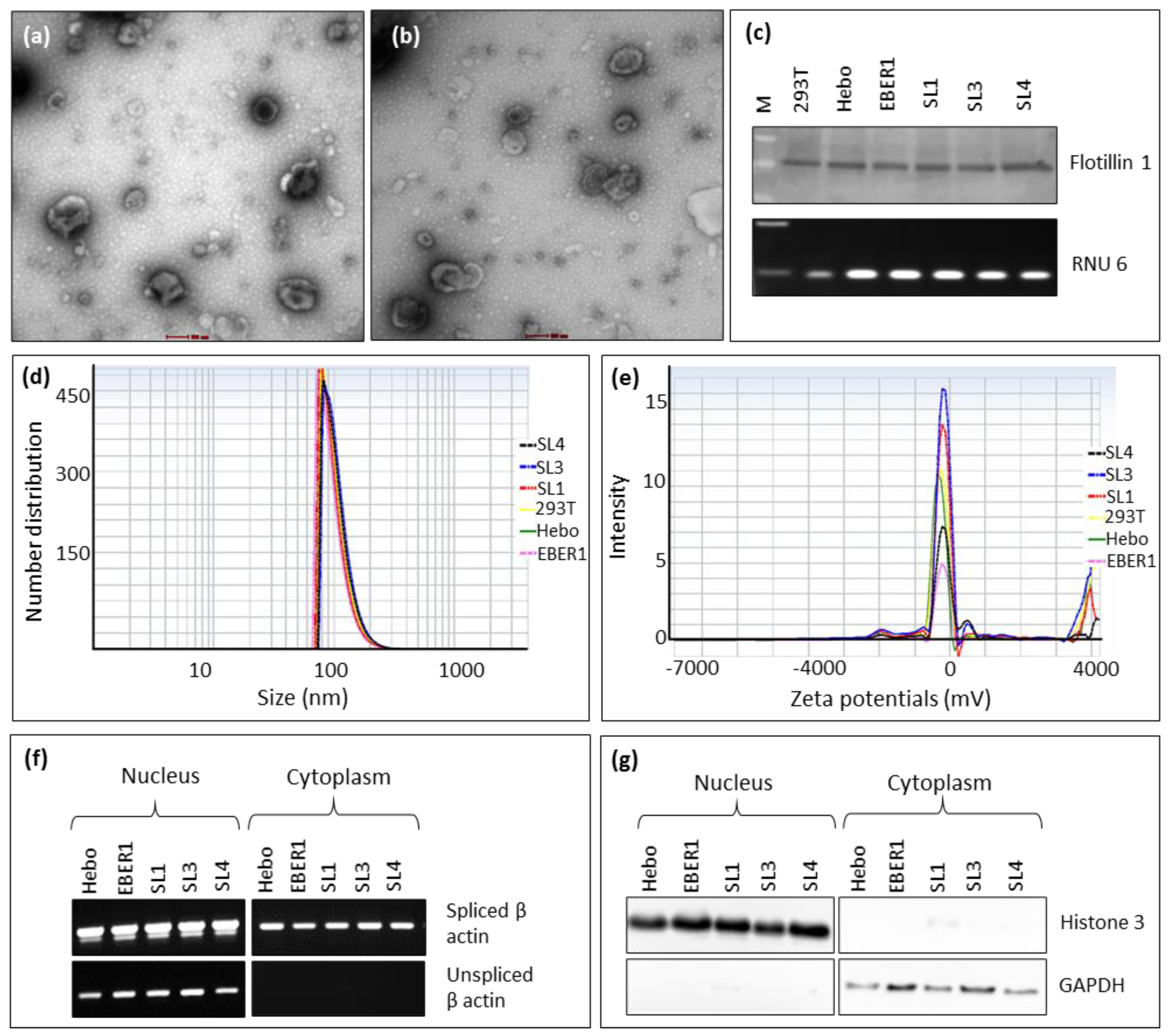
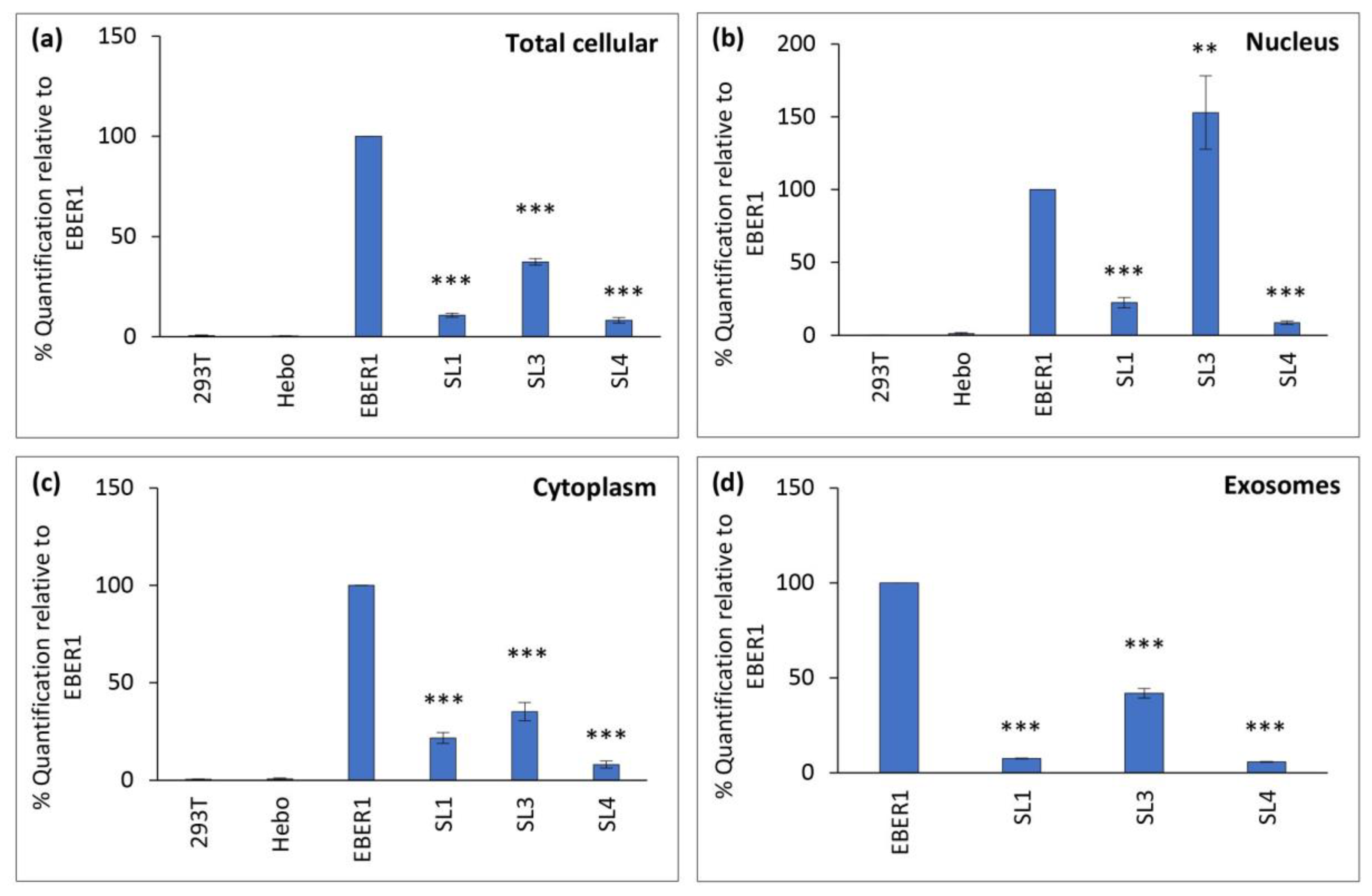
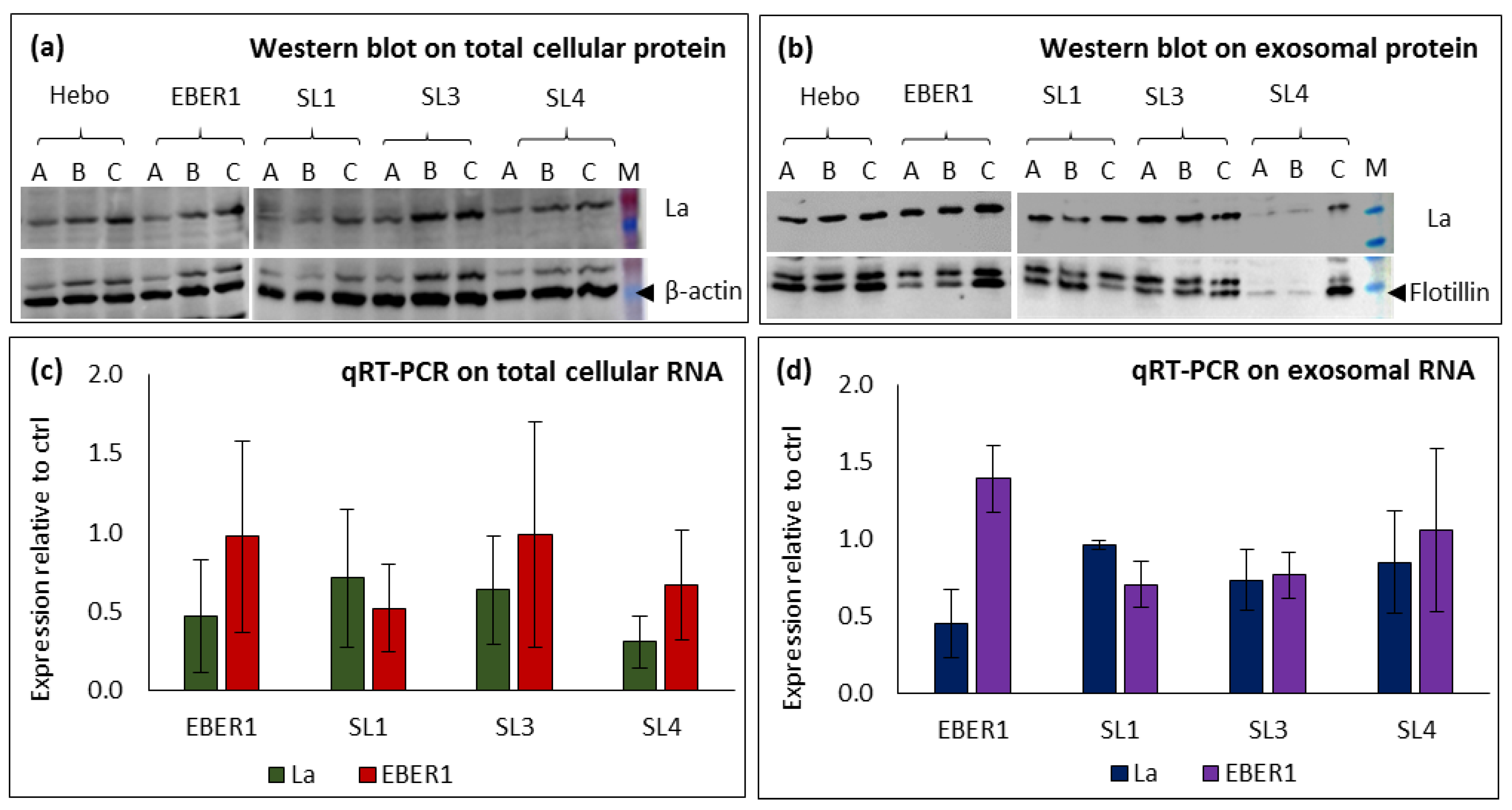
3.2. Characterisation of exosomes and subcellular fractionations
3.3. The conserved structure of EBER1 influences its nuclear-cytoplasmic transport but not its secretion into exosomes
3.4. In silico analysis of EBER1-protein interactions
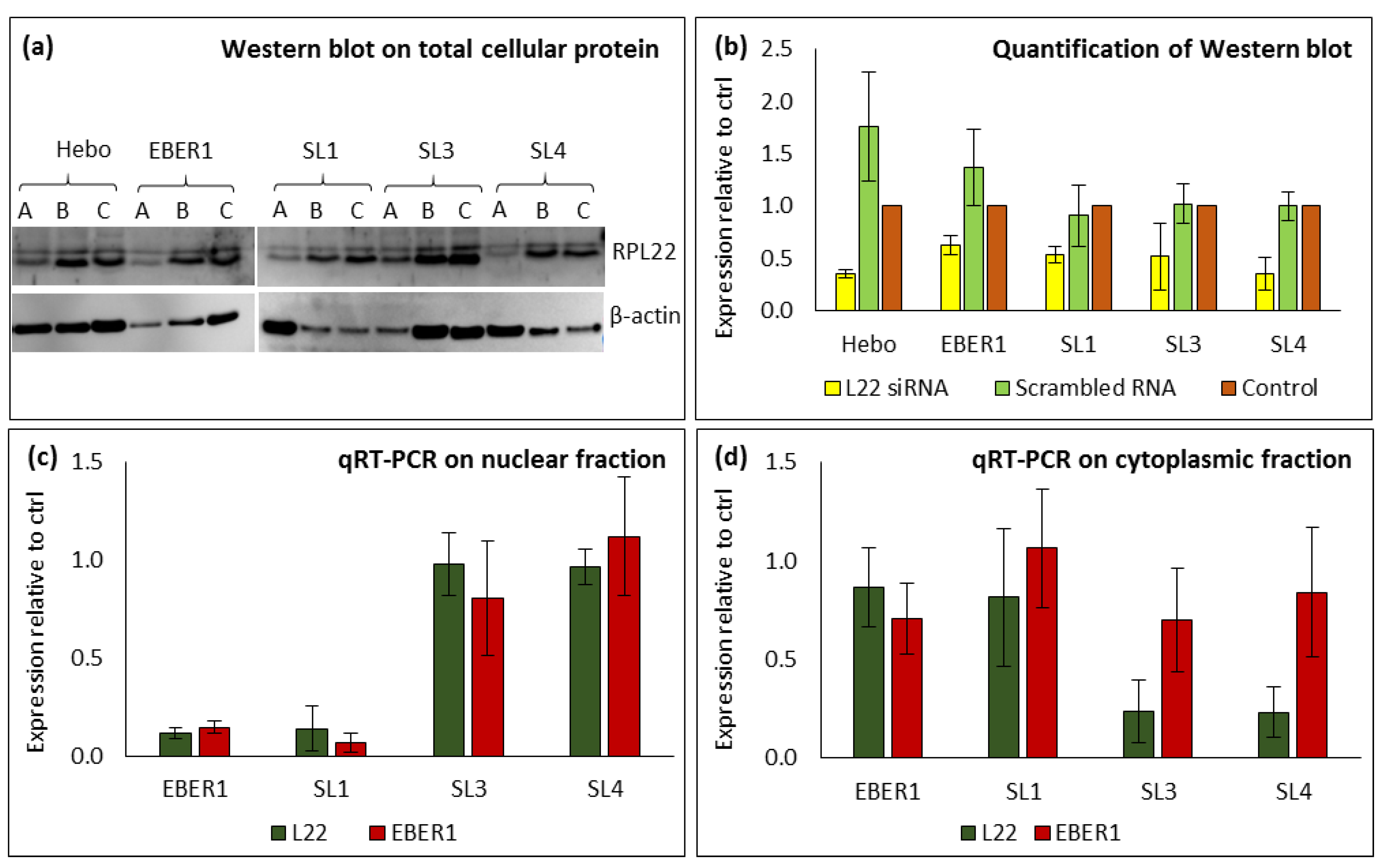
3.5. Interaction of EBER1 with RPL22 affects its nuclear-cytoplasmic transport
3.6. Silencing cellular La protein does not correlate to the expression of EBER1 in the exosomes
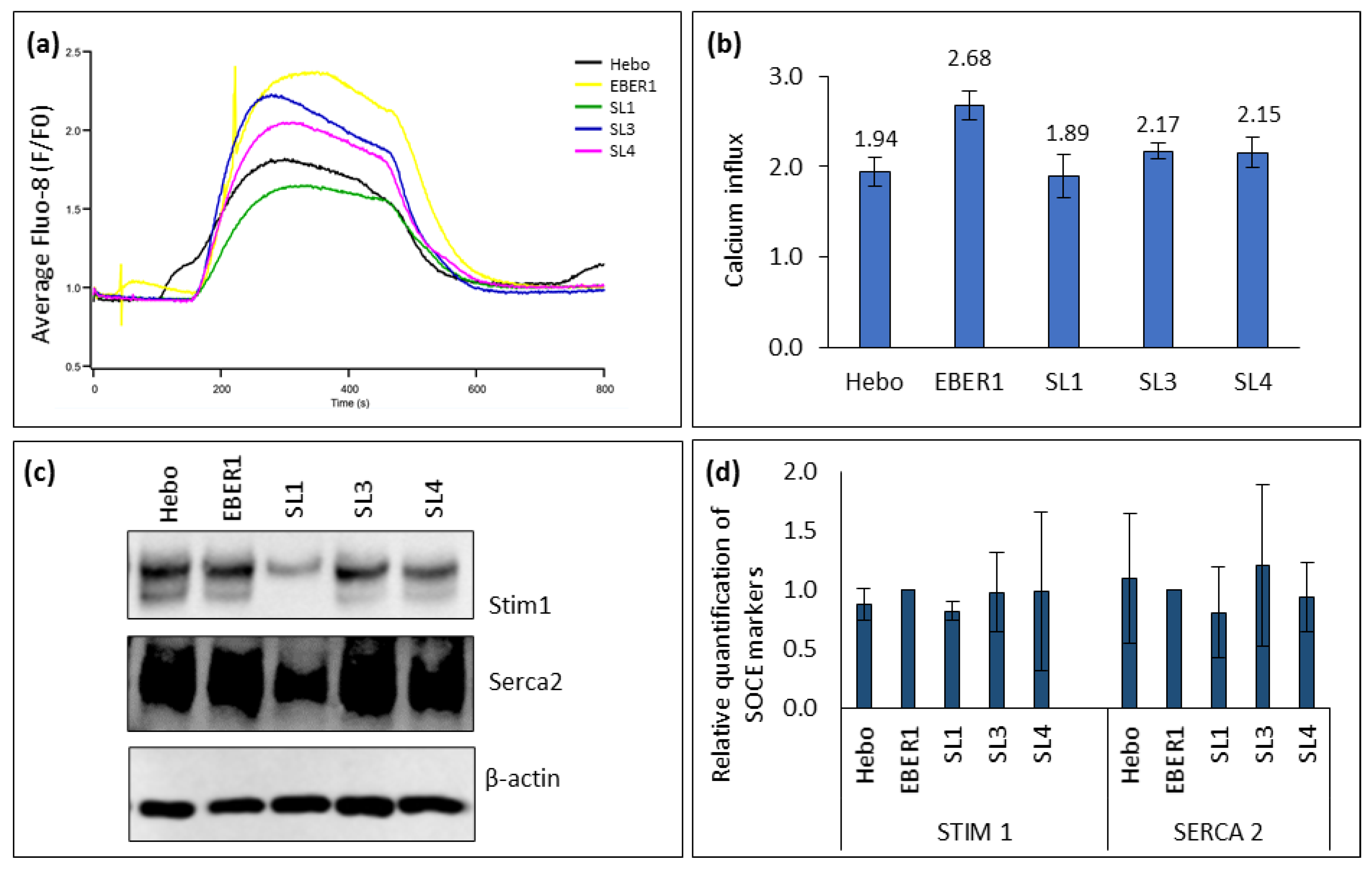
3.7. Intracellular influx of [Ca2+]i could be involved in the secretion of EBER1 into exosomes
4.0. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Thorley-Lawson, D.A.; Allday, M.J. The Curious Case of the Tumour Virus: 50 Years of Burkitt’s Lymphoma. Nature Reviews Microbiology 2008 6:12 2008, 6, 913–924. [Google Scholar] [CrossRef] [PubMed]
- Kieff, E.; Rickinson, A.B. Epstein-Barr Virus and Its Replication. In Fields Virology; Lippincott Williams & Wilkins, Philadelphia, 2007; pp. 2603–2654.
- Lorenzetti, M.A.; de Matteo, E.; Gass, H.; Vazquez, P.M.; Lara, J.; Gonzalez, P.; Preciado, M.V.; Chabay, P.A. Characterization of Epstein Barr Virus Latency Pattern in Argentine Breast Carcinoma. PLoS ONE 2010, 5, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, W.; Philip, P.S.; Tariq, S.; Khan, G. Epstein-Barr Virus-Encoded Small RNAs ( EBERs ) Are Present in Fractions Related to Exosomes Released by EBV-Transformed Cells. PloS one 2014, 9, e99163. [Google Scholar] [CrossRef]
- Iwakiri, D.; Takada, K. Role of EBERs in the Pathogenesis of EBV Infection. In Advances in Cancer Research; Elsevier: Amsterdam, The Netherlands, 2010; pp. 119–136. [Google Scholar]
- Herbert, K.M.; Pimienta, G. Consideration of Epstein-Barr Virus-Encoded Noncoding RNAs EBER1 and EBER2 as a Functional Backup of Viral Oncoprotein Latent Membrane Protein 1. MBio 2016, 7, e01926–15. [Google Scholar] [CrossRef] [PubMed]
- Lerner, M.R.; Andrews, N.C.; Miller, G.; Steitz, J.A. Two Small RNAs Encoded by Epstein-Barr Virus and Complexed with Protein Are Precipitated by Antibodies from Patients with Systemic Lupus Erythematosus. Proceedings of the National Academy of Sciences of the United States of America 1981, 78, 805–809. [Google Scholar] [CrossRef]
- Chavez-Calvillo, G.; Martin, S.; Hamm, C.; Sztuba-solinska, J. The Structure-To-Function Relationships of Gammaherpesvirus-Encoded Long RNAs and Their Contributions to Viral Pathogenesis. Non-coding 2018, 4. [Google Scholar] [CrossRef]
- Schwemmle, M.; Clemens, M.J.; Hilse, K.; Pfeifer, K.; Tröster, H.; Müller, W.E.; Bachmann, M. Localization of Epstein-Barr Virus-Encoded RNAs EBER-1 and EBER-2 in Interphase and Mitotic Burkitt Lymphoma Cells. Proc. Natl. Acad. Sci. USA 1992, 89, 10292–10296. [Google Scholar] [CrossRef]
- Ahmed, W.; Tariq, S.; Khan, G. Tracking EBV-Encoded RNAs (EBERs) from the Nucleus to the Excreted Exosomes of B-Lymphocytes. Scientific Reports 2018, 8, 1–11. [Google Scholar] [CrossRef]
- Weeks, K.M. Advances in RNA Secondary and Tertiary Structure Analysis by Chemical Probing. Current Opinion in Structural Biology 2010, 20, 295–304. [Google Scholar] [CrossRef]
- Swadling, J.B.; Ishii, K.; Tahara, T.; Kitao, A. Origins of Biological Function in DNA and RNA Hairpin Loop Motifs from Replica Exchange Molecular Dynamics Simulation. Physical Chemistry Chemical Physics 2018, 20, 2990–3001. [Google Scholar] [CrossRef]
- Wan, Y.; Qu, K.; Zhang, Q.C.; Flynn, R.A.; Manor, O.; Ouyang, Z.; Zhang, J.; Spitale, R.C.; Snyder, M.P.; Segal, E.; et al. Landscape and Variation of RNA Secondary Structure across the Human Transcriptome. Nature 2014, 505, 706–709. [Google Scholar] [CrossRef] [PubMed]
- Houmani, J.L.; Davis, C.I.; Ruf, I.K. Growth-Promoting Properties of Epstein-Barr Virus EBER-1 RNA Correlate with Ribosomal Protein L22 Binding. J Virol. 2009, 83, 9844–9853. [Google Scholar] [CrossRef] [PubMed]
- Eilebrecht, S.; Pellay, F.X.; Odenwälder, P.; Brysbaert, G.; Benecke, B.J.; Benecke, A. EBER2 RNA-Induced Transcriptome Changes Identify Cellular Processes Likely Targeted during Epstein Barr Virus Infection. BMC Research Notes 2008, 1, 11–14. [Google Scholar] [CrossRef] [PubMed]
- Wang; Li, X. ; Zhang, X.; Wang, Q.; Liu, W.; Lu, X.; Gao, S.; Liu, Z.; Liu, M.; Gao, L.; et al. RNA Motifs and Modification Involve in RNA Long-Distance Transport in Plants. Frontiers in Cell and Developmental Biology 2021, 9, 1–10. [Google Scholar] [CrossRef]
- Glimcher, L.H. Structural Mutation Affecting Intracellular Transport and Cell Surface Expression of Murine Class II Molecules. J. Exp. Med. 1988, 167, 541–555. [Google Scholar]
- Yajima, M.; Kanda, T.; Takada, K. Critical Role of Epstein-Barr Virus (EBV)-Encoded RNA in Efficient EBV-Induced B-Lymphocyte Growth Transformation. Journal of Virology 2005, 79, 4298–4307. [Google Scholar] [CrossRef]
- Iwakiri, D. Epstein-Barr Virus-Encoded RNAs: Key Molecules in Viral Pathogenesis. Cancers 2014, 6, 1615–1630. [Google Scholar] [CrossRef]
- Gregorovic, G.; Bosshard, R.; Karstegl, C.E.; White, R.E.; Pattle, S.; Chiang, A.K.S.; Dittrich-breiholz, O.; Kracht, M.; Russ, R.; Farrell, P.J. Cellular Gene Expression That Correlates with EBER Expression in Epstein-Barr Virus-Infected Lymphoblastoid Cell Lines. Journal 2011, 85, 3535–3545. [Google Scholar] [CrossRef]
- Parameswaran, P.; Sklan, E.; Wilkins, C.; Burgon, T.; Samuel, M.A.; Fire, A.Z. Six RNA Viruses and Forty-One Hosts : Viral Small RNAs and Modulation of Small RNA Repertoires in Vertebrate and Invertebrate Systems. PLoS pathogens 2010, 6, e1000764. [Google Scholar] [CrossRef]
- Wu, Y.; Maruo, S.; Yajima, M.; Kanda, T.; Takada, K. Epstein-Barr Virus (EBV)-Encoded RNA 2 (EBER2) but Not EBER1 Plays a Critical Role in EBV-Induced B-Cell Growth Transformation. J Virol. 2007, 80, 11236–11245. [Google Scholar] [CrossRef]
- Zhang; Jiang1, X. ; Bao, J.; Wang, Y.; Liu, H.; Tang, L. Exosomes in Pathogen Infections: A Bridge to Deliver Molecules and Link Functions. Frontiers in Immunology 2018, 9, 90. [Google Scholar] [CrossRef] [PubMed]
- Low, J.T.; Shukla, A.; Behrendorff, N.; Thorn, P. Exocytosis, Dependent on Ca2+ Release from Ca2+ Stores, Is Regulated by Ca2+ Microdomains. Journal of Cell Science 2010, 123, 3201–3208. [Google Scholar] [CrossRef] [PubMed]
- Hansen, N.J.; Antonin, W.; Edwardson, J.M. Identification of SNAREs Involved in Regulated Exocytosis in the Pancreatic Acinar Cell *. Journal of Biological Chemistry 1999, 274, 22871–22876. [Google Scholar] [CrossRef] [PubMed]
- Gaisano, H.Y.; Ghai, M.; Malkus, P.N.; Sheu, L.; Bouquillon, A.; Bennett, M.K.; Trimble, W.S. Distinct Cellular Locations of the Syntaxin Family of Proteins in Rat Pancreatic Acinar Cells. Molecular biology of the cell 1996, 7, 2019–2027. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, W.; Hassan, Z.; Abdelmowla, Y.A.A.; Philip, P.S.; Shmygol, A.; Khan, G. Epstein-Barr Virus Noncoding Small RNA (EBER1) Induces Cell Proliferation by up-Regulating Cellular Mitochondrial Activity and Calcium Influx. Virus Research 2021, 305, 198550. [Google Scholar] [CrossRef] [PubMed]
- Heckman, K.L.; Pease, L.R. Gene Splicing and Mutagenesis by PCR-Driven Overlap Extension. Nature Protocols 2007, 2, 924–932. [Google Scholar] [CrossRef] [PubMed]
- Akhlaq, S.; Panicker, N.G.; Philip, P.S.; Ali, L.M.; Dudley, J.P.; Rizvi, T.A.; Mustafa, F. A Cis-Acting Element Downstream of the Mouse Mammary Tumor Virus Major Splice Donor Critical for RNA Elongation and Stability. J Mol Biol 2018, 430, 4307–4324. [Google Scholar] [CrossRef]
- Khan, G.; Coates, P.J.; Kangro, H.O.; Slavin, G. Epstein Barr Virus ( EBV ) Encoded Small RNAs : Targets for Detection by in Situ Hybridisation with Oligonucleotide Probes. J clin pathol 1992, 45, 616–620. [Google Scholar] [CrossRef]
- Weiss, L.M.; Chen, Y.-Y.; Liu, X.-F.; Shibata, D. Epstein-Barr Virus and Hodgkin’s Disease A Correlative In Situ Hybridization and Polymerase Chain Reaction Study. American Journal of Pathology 1991, 139. [Google Scholar]
- Ryan, J.L.; Fan, H.; Glaser, S.L.; Schichman, S.A.; Raab-Traub, N.; Gulley, M.L. Epstein-Barr Virus Quantitation by Real-Time PCR Targeting Multiple Gene Segments : A Novel Approach to Screen for the Virus in Paraffin-Embedded Tissue and Plasma. The Journal of molecular diagnostics 2004, 6, 378–385. [Google Scholar] [CrossRef]
- Pauling, L. Elements and Compounds. Atomic and Molecular Masses. In General Chemistry, reprinted by Courier in 2014; 992 pages; 1970; p. 96 ISBN 9780486134659.
- Théry, C.; Amigorena, S.; Raposo, G.; Clayton, A. Isolation and Characterization of Exosomes from Cell Culture Supernatants and Biological Fluids. Current Protocols in Cell Biology 2006, 30, 3–22. [Google Scholar] [CrossRef] [PubMed]
- Stetefeld, J.; McKenna, S.A.; Patel, T.R. Dynamic Light Scattering: A Practical Guide and Applications in Biomedical Sciences. Biophysical Reviews 2016, 8, 409. [Google Scholar] [CrossRef] [PubMed]
- Pecora, R. Doppler Shifts in Light Scattering from Pure Liquids and Polymer Solutions. The Journal of Chemical Physics 2004, 40, 1604. [Google Scholar] [CrossRef]
- Greenberg, M.E.; Bender, T.P. Identification of Newly Transcribed RNA. In Curr Protoc Mol Biol; 2007.
- Liao, J.; Ma, L.; Guo, Y.; Zhang, Y.; Zhou, H.; Shao, P.; Chen, Y. Deep Sequencing of Human Nuclear and Cytoplasmic Small RNAs Reveals an Unexpectedly Complex Subcellular Distribution of MiRNAs and TRNA 3 9 Trailers. PLoS One 2010, 5, e10563. [Google Scholar] [CrossRef] [PubMed]
- Baghirova, S.; Hughes, B.G.; Hendzel, M.J.; Schulz, R. Sequential Fractionation and Isolation of Subcellular Proteins from Tissue or Cultured Cells. MethodsX 2015, 2, e440–e445. [Google Scholar] [CrossRef]
- Towbin, H.; Staehelin, T.; Gordon, J. Electrophoretic Transfer of Proteins from Polyacrylamide Gels to Nitrocellulose Sheets: Procedure and Some Applications. Proceedings of the National Academy of Sciences of the United States of America 1979, 76, 4350–4354. [Google Scholar] [CrossRef]
- Sanchez de Groot, N.; Armaos, A.; Graña-Montes, R.; Alriquet, M.; Calloni, G.; Vabulas, R.M.; Tartaglia, G.G. RNA Structure Drives Interaction with Proteins. Nature Communications 2019, 10, 1–13. [Google Scholar] [CrossRef]
- Tong, J.; McCarthy, T. V.; MacLennan, D.H. Measurement of Resting Cytosolic Ca2+ Concentrations and Ca2+ Store Size in HEK-293 Cells Transfected with Malignant Hyperthermia or Central Core Disease Mutant Ca2+ Release Channels. Journal of Biological Chemistry 1999, 274, 693–702. [Google Scholar] [CrossRef]
- Hassan, Z.; Philip, P.S.; Khan, G. The Impact of Deleting Stem-Loop 1 of Epstein – Barr Virus-Encoded RNA 1 on Cell Proliferation. Viruses 2022, 14. [Google Scholar] [CrossRef]
- Glickman, J.N.; Howe, J.G.; Steitz, J.A. Structural Analyses of EBERI and EBER2 Ribonucleoprotein Particles Present in Epstein-Barr Virus-Infected Cells. J. Virol. 1988, 62, 902–911. [Google Scholar] [CrossRef]
- Ha, D.; Yang, N.; Nadithe, V. Exosomes as Therapeutic Drug Carriers and Delivery Vehicles across Biological Membranes : Current Perspectives and Future Challenges. Acta Pharmaceutica Sinica B 2016, 6, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Urbanelli, L.; Magini, A.; Buratta, S.; Brozzi, A.; Sagini, K.; Polchi, A.; Tancini, B.; Emiliani, C. Signaling Pathways in Exosomes Biogenesis, Secretion and Fate. Genes 2013, 4, 152–170. [Google Scholar] [CrossRef] [PubMed]
- Vlassov, A. V.; Magdaleno, S.; Setterquist, R.; Conrad, R. Exosomes: Current Knowledge of Their Composition, Biological Functions and Diagnostic and Therapeutic Potentials. Biochim Biophys Acta 2012, 1820, 940–948. [Google Scholar] [CrossRef] [PubMed]
- Midekessa, G.; Godakumara, K.; Ord, J.; Viil, J.; Lättekivi, F.; Dissanayake, K.; Kopanchuk, S.; Rinken, A.; Andronowska, A.; Bhattacharjee, S.; et al. Zeta Potential of Extracellular Vesicles: Toward Understanding the Attributes That Determine Colloidal Stability. ACS Omega 2020, 5, 16701–16710. [Google Scholar] [CrossRef] [PubMed]
- Hilleren, P.; McCarthy, T.; Rosbash, M.; Parker, R.; Jensen, T.H. Quality Control of MRNA 3′-End Processing Is Linked to the Nuclear Exosome. Nature 2001, 413, 538–542. [Google Scholar] [CrossRef] [PubMed]
- Ohno, M.; Segref, A.; Kuersten, S.; Mattaj, I.W. Identity Elements Used in Export of MRNAs. Molecular Cell 2002, 9, 659–671. [Google Scholar] [CrossRef] [PubMed]
- Toczyski, D.P.; Matera, A.G.; Ward, D.C.; Steitz, J.A. The Epstein–Barr Virus (EBV) Small RNA EBER1 Binds and Relocalizes Ribosomal Protein L22 in EBV-Infected Human B Lymphocytes. PNAS 1994, 91, 3463–3467. [Google Scholar] [CrossRef] [PubMed]
- Muppirala, U.K.; Honavar, V.G.; Dobbs, D. Predicting RNA-Protein Interactions Using Only Sequence Information. BMC Bioinformatics 2011, 12, 489. [Google Scholar] [CrossRef]
- Baglio, S.R.; Eijndhoven, M.A.J. van; Koppers-Lalic, D.; Berenguer, J.; Lougheed, S.M.; Gibbs, S.; Léveillé, N.; Rinkel, R.N.P.M.; Hopmans, E.S.; Swaminathan, S.; et al. Sensing of Latent EBV Infection through Exosomal Transfer of 5′pppRNA. Proceedings of the National Academy of Sciences 2016, 113, E587–E596. [Google Scholar] [CrossRef]
- Gregorovic, G.; Boulden, E.A.; Bosshard, R.; Elgueta Karstegl, C.; Skalsky, R.; Cullen, B.R.; Gujer, C.; Rämer, P.; Münz, C.; Farrell, P.J.; et al. Epstein-Barr Viruses (EBVs) Deficient in EBV-Encoded RNAs Have Higher Levels of Latent Membrane Protein 2 RNA Expression in Lymphoblastoid Cell Lines and Efficiently Establish Persistent Infections in Humanized Mice. Journal of Virology 2015, 89, 11711–11714. [Google Scholar] [CrossRef]
- Kapsogeorgou, E.K.; Abu-Helu, R.F.; Moutsopoulos, H.M.; Manoussakis, M.N. Salivary Gland Epithelial Cell Exosomes: A Source of Autoantigenic Ribonucleoproteins. Arthritis and Rheumatism 2005, 52, 1517–1521. [Google Scholar] [CrossRef] [PubMed]
| Constructs | Zeta potential |
|---|---|
| Untransfected | -312.44 |
| pHebo | -260.16 |
| Wildtype EBER1 | -214.68 |
| ∆SL1 mutant | -198.52 |
| ∆SL3 mutant | -180.00 |
| ∆SL4 mutant | -190.65 |
| Transport | Wildtype | ∆SL1 | ∆SL3 | ∆SL4 |
|---|---|---|---|---|
| Nuclear-cytoplasmic | 17.12 | 11.78 | 9.32 | 17.85 |
| Cytoplasmic-exosomes | 5.41 | 6.76 | 5.22 | 5.47 |
| RNA ID | Interaction with RPL22 | Interaction with La | ||
|---|---|---|---|---|
| RF Classifier | SVM Classifier | RF Classifier | SVM Classifier | |
| Wildtype EBER1 | 0.55 | 0.66 | 0.40 | 0.45 |
| ∆SL1 mutant | 0.55 | 0.56 | 0.40 | 0.37 |
| ∆SL3 mutant | 0.50 | 0.41 | 0.40 | 0.28 |
| ∆SL4 mutant | 0.50 | 0.74 | 0.30 | 0.50 |
| EBER1 | SL1 | SL3 | SL4 | |||||
|---|---|---|---|---|---|---|---|---|
| RPL22 | EBER1 | RPL22 | EBER1 | RPL22 | EBER1 | RPL22 | EBER1 | |
| Nucleus | 11.93 | 17.38 | 14.90 | 6.17 | 80.50 | 53.56 | 80.46 | 57.13 |
| Cytoplasm | 88.07 | 82.62 | 85.10 | 93.83 | 19.50 | 46.44 | 19.54 | 42.87 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




