Submitted:
25 April 2023
Posted:
26 April 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
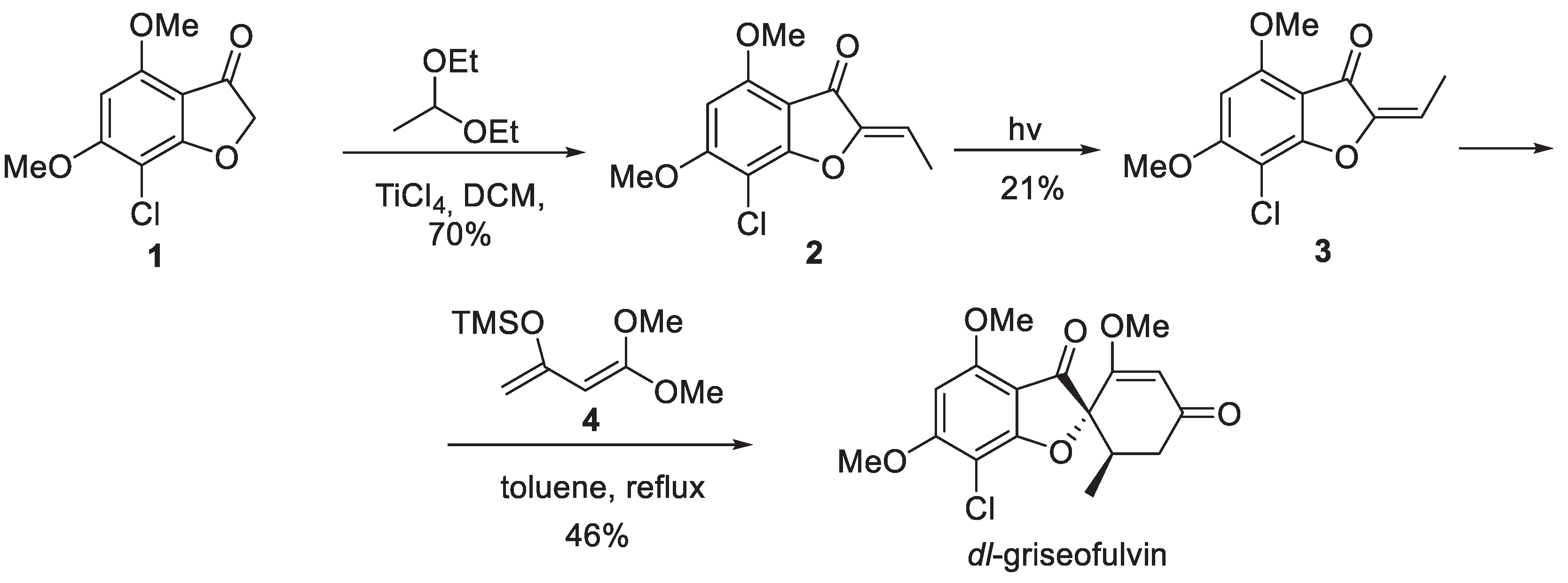
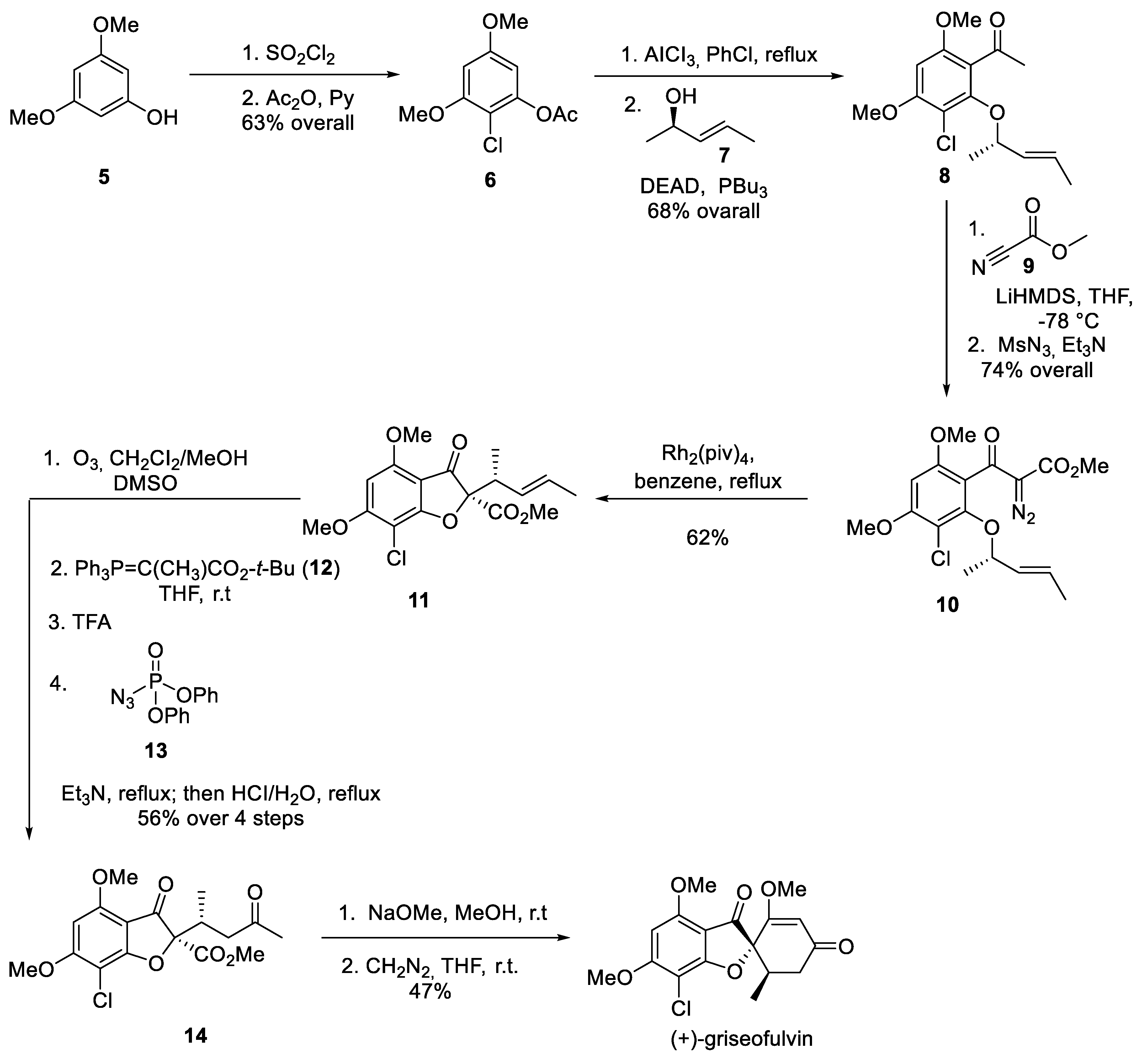
2. Griseofulvin
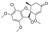
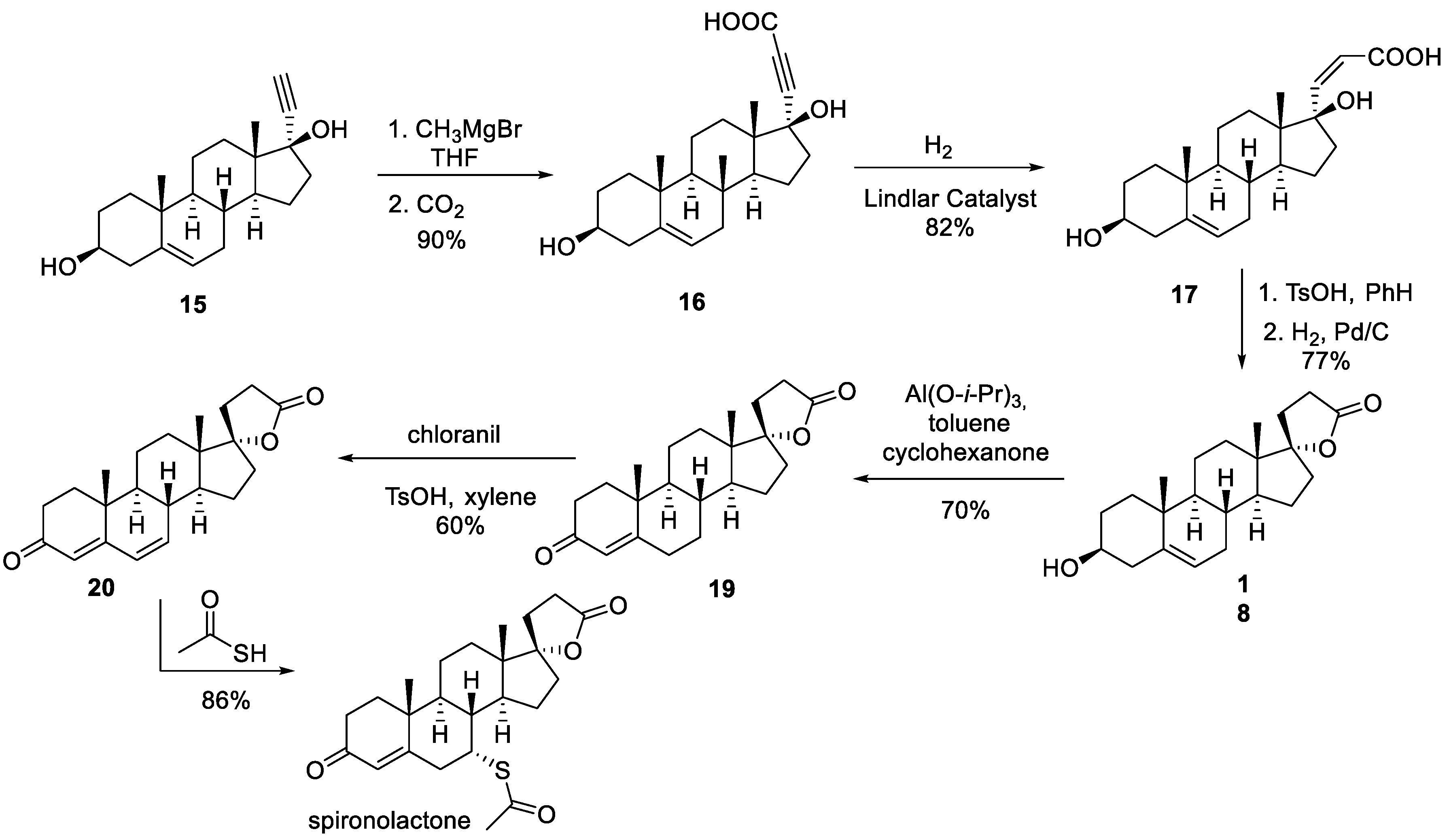
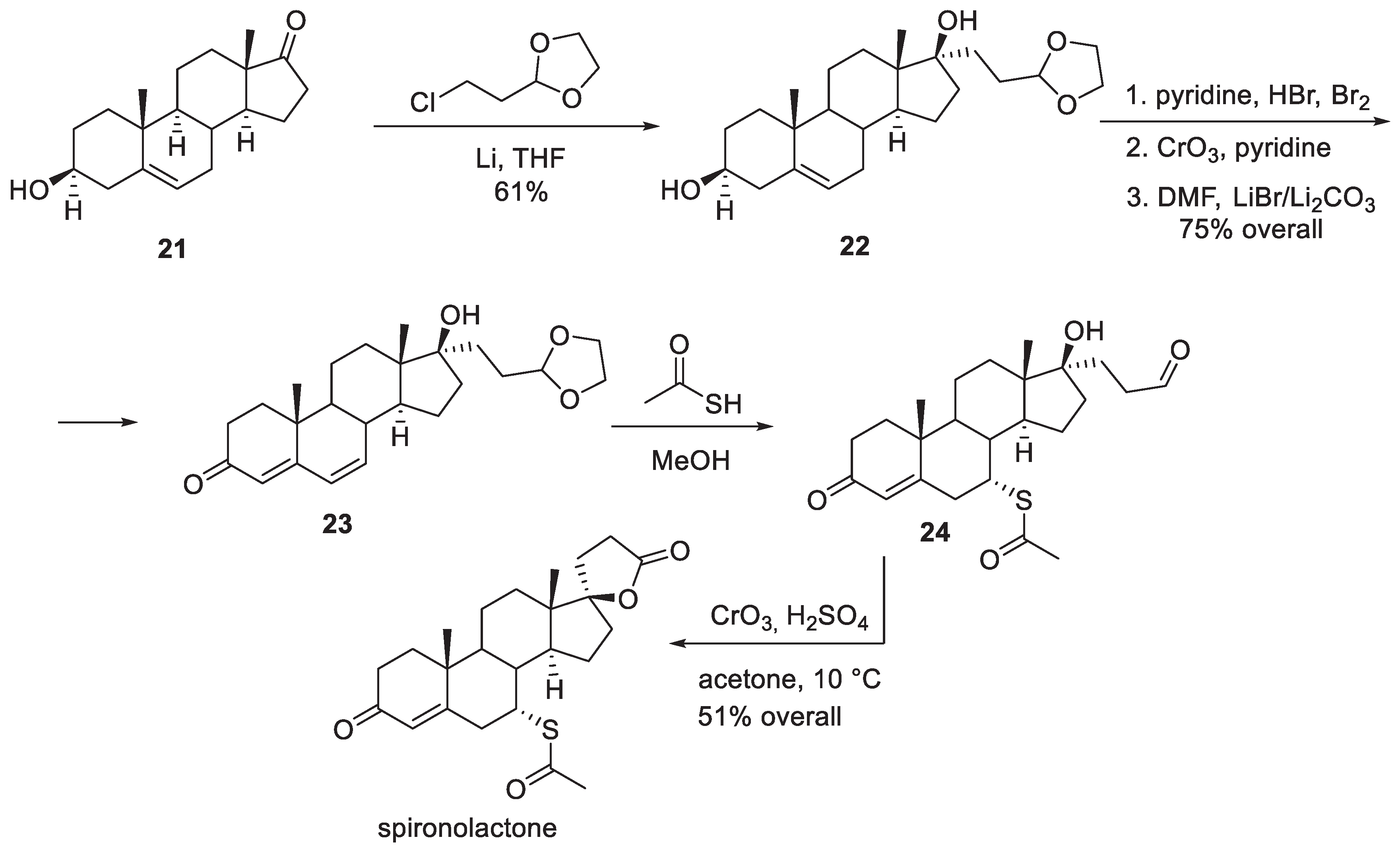
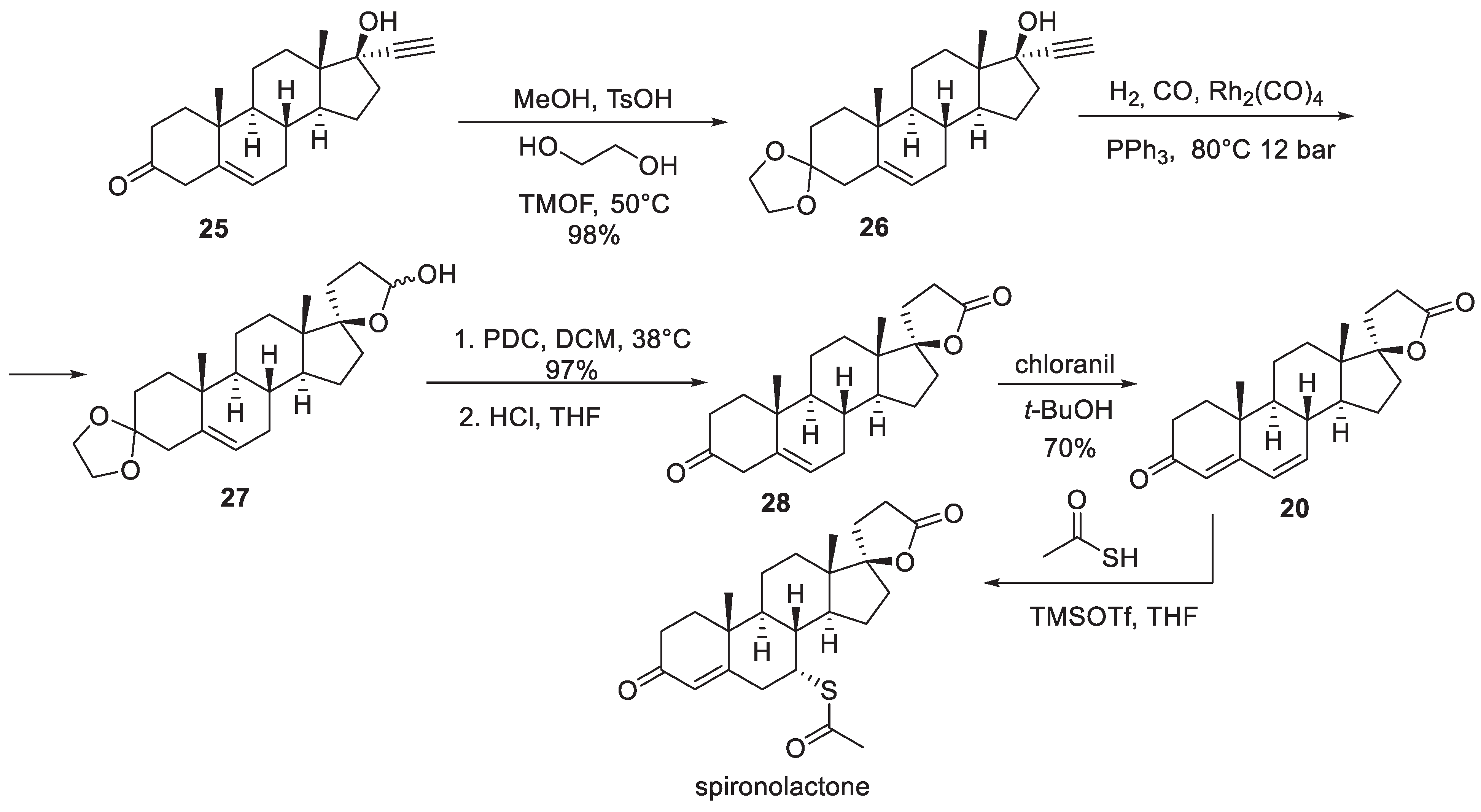
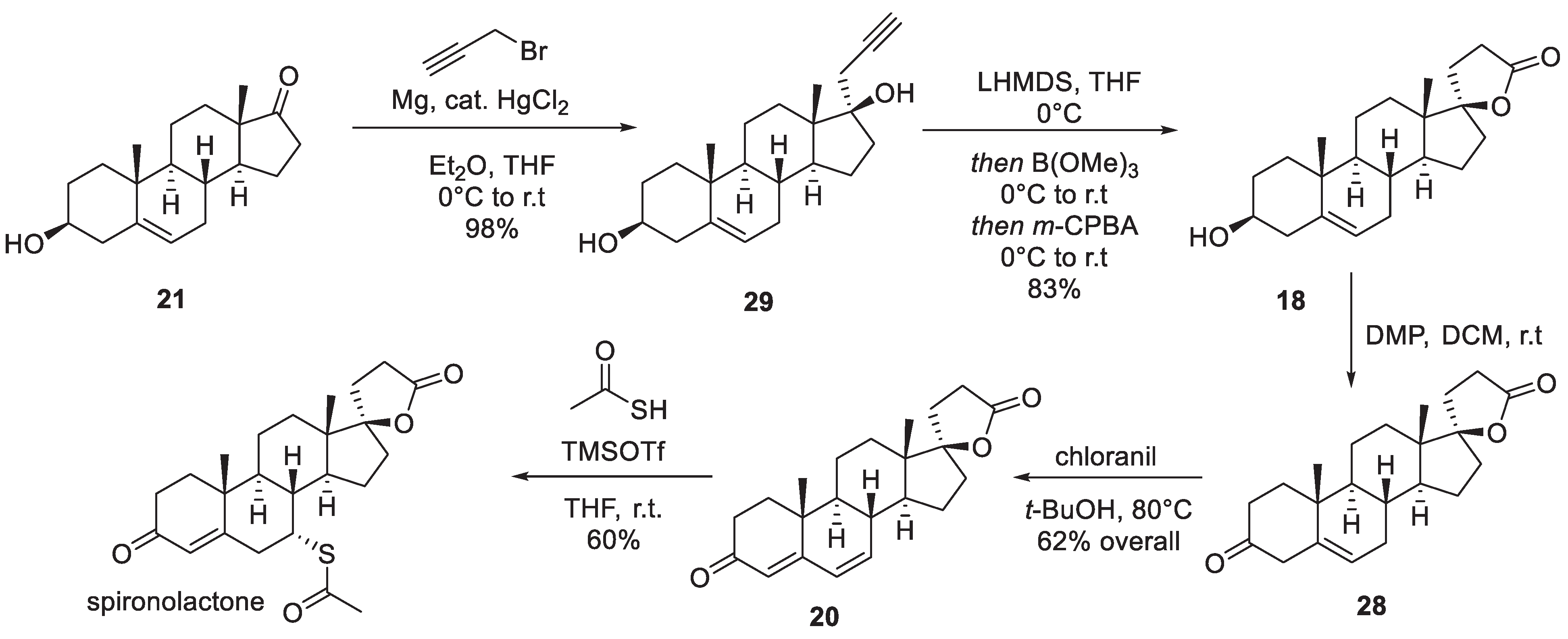
3. Spironolactone
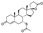
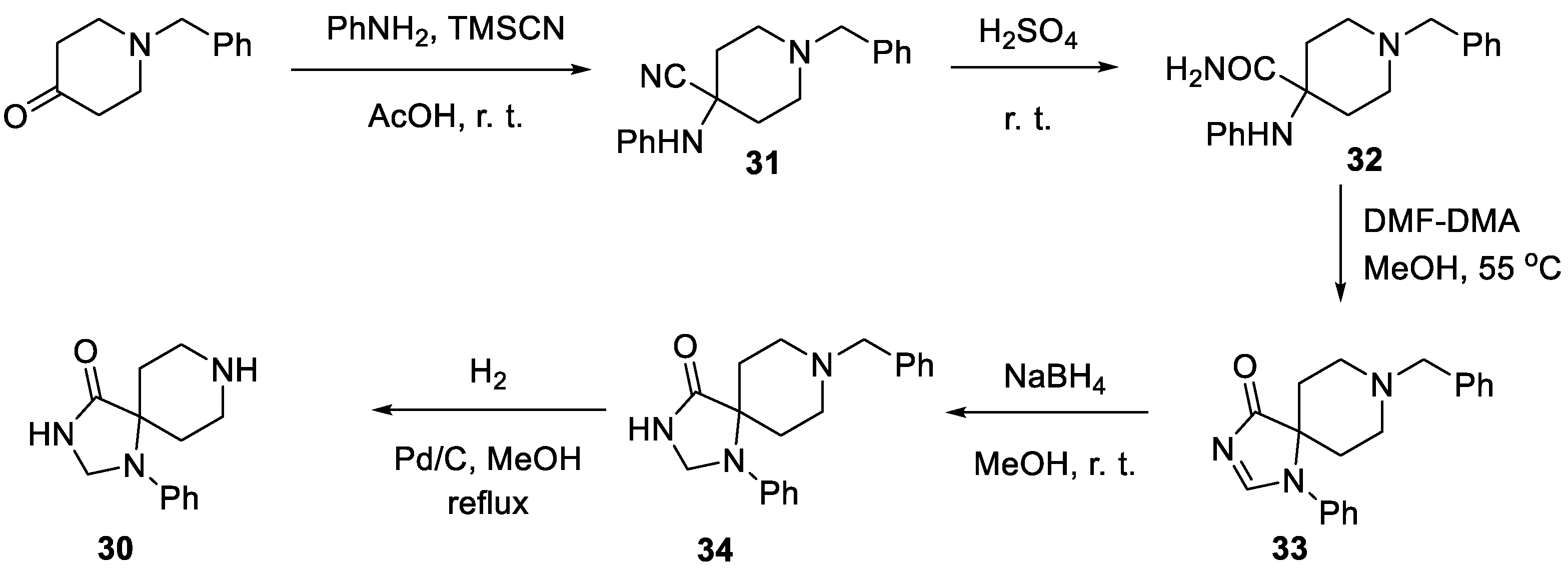
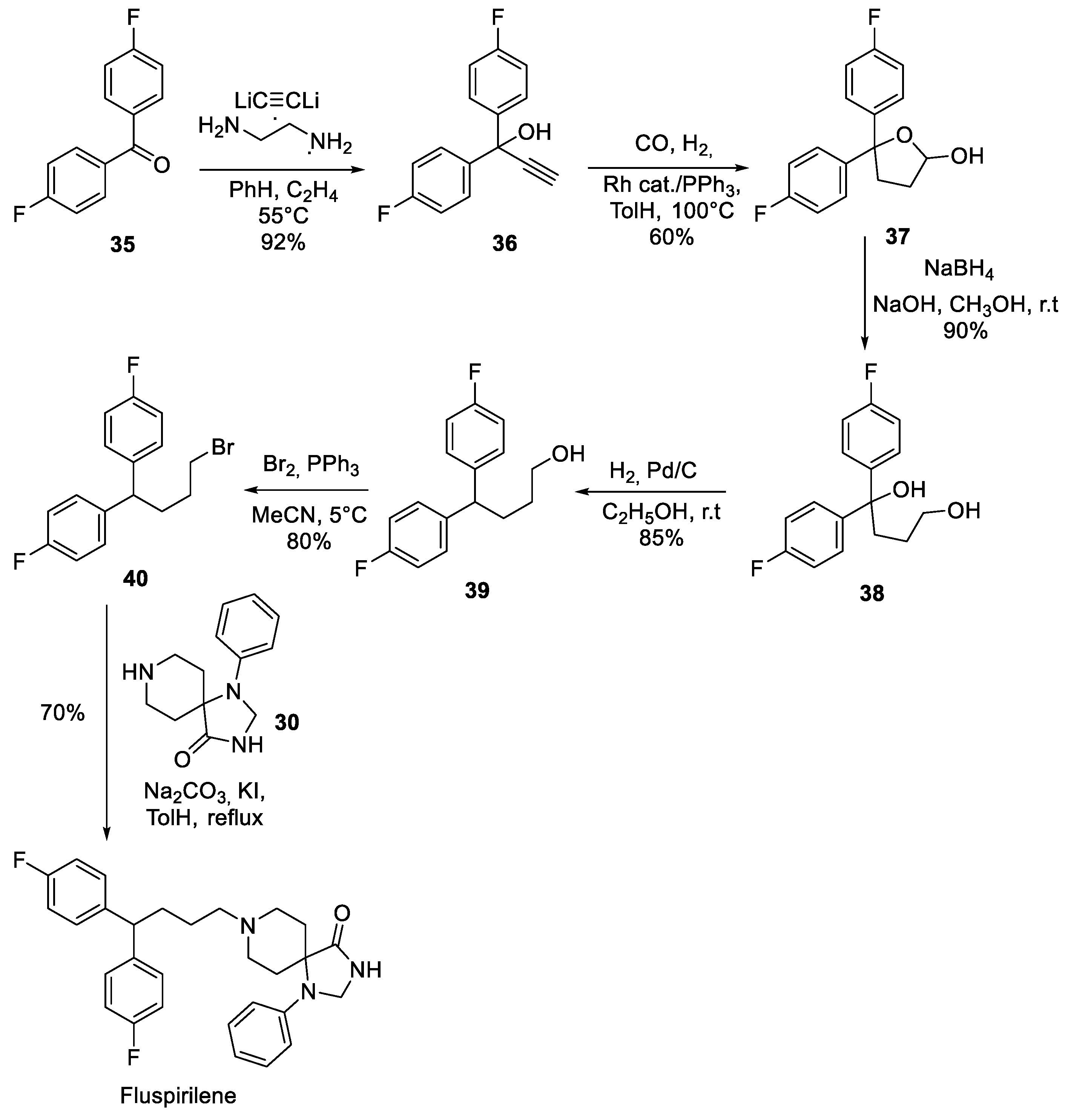
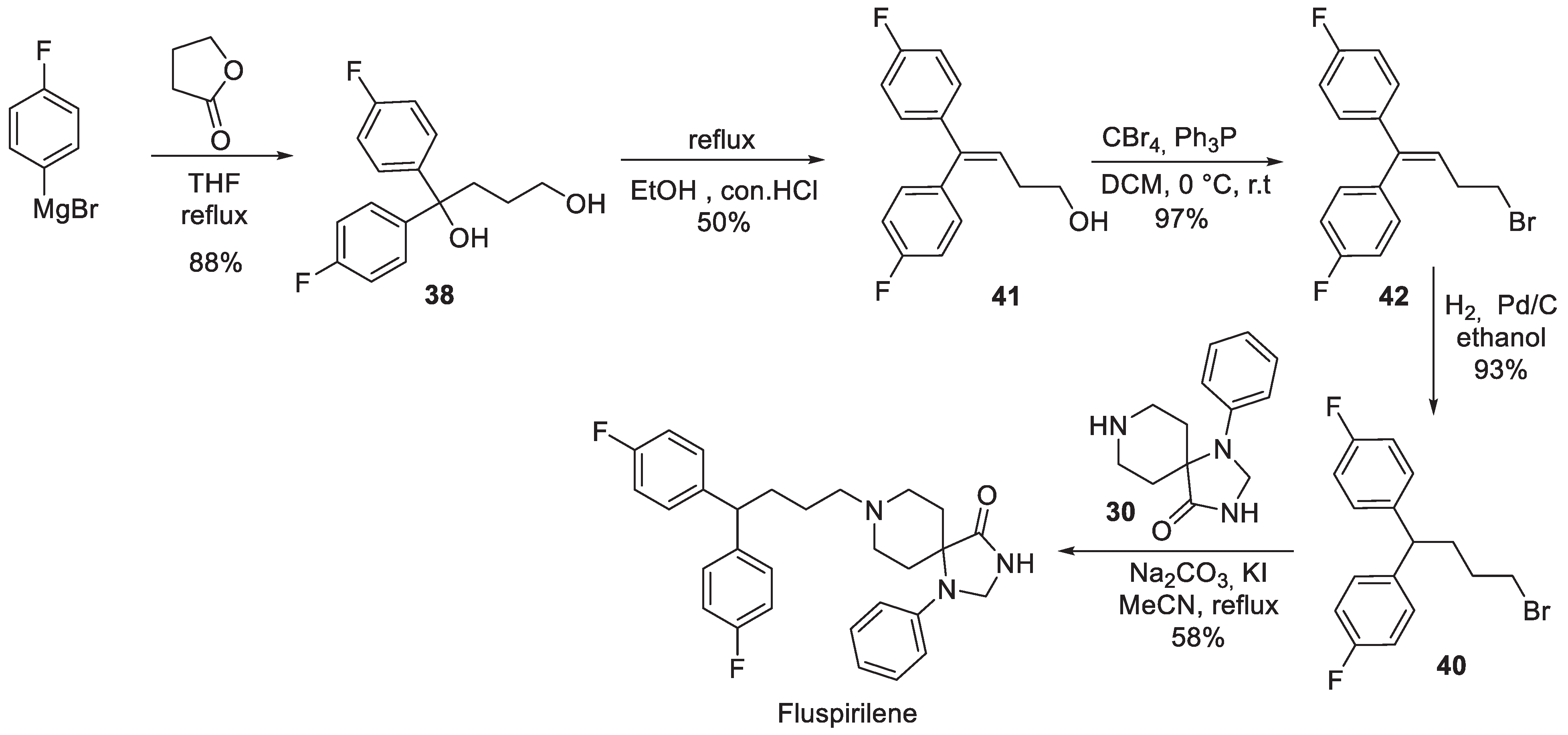
4. Fluspirilene
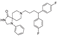
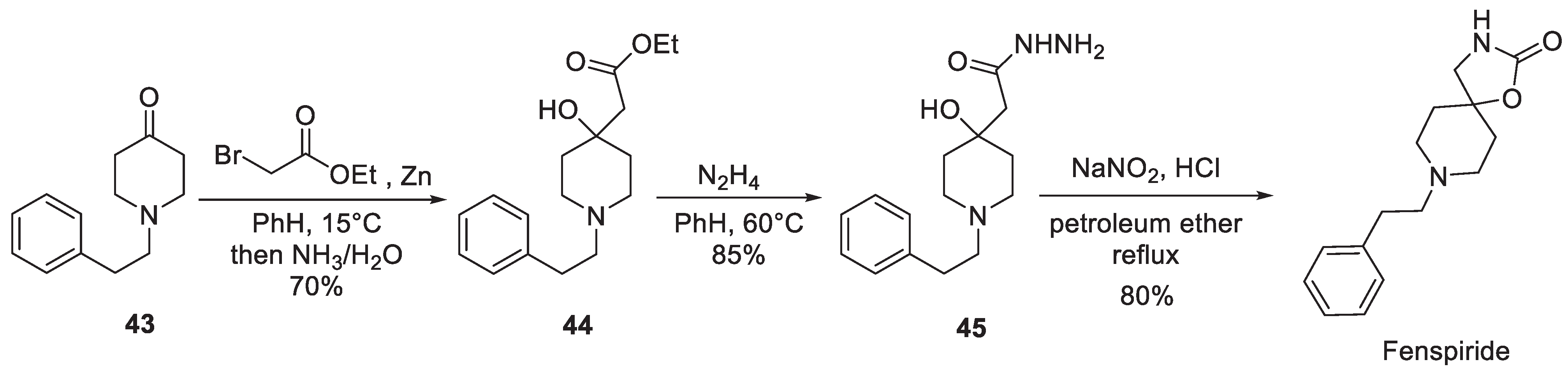
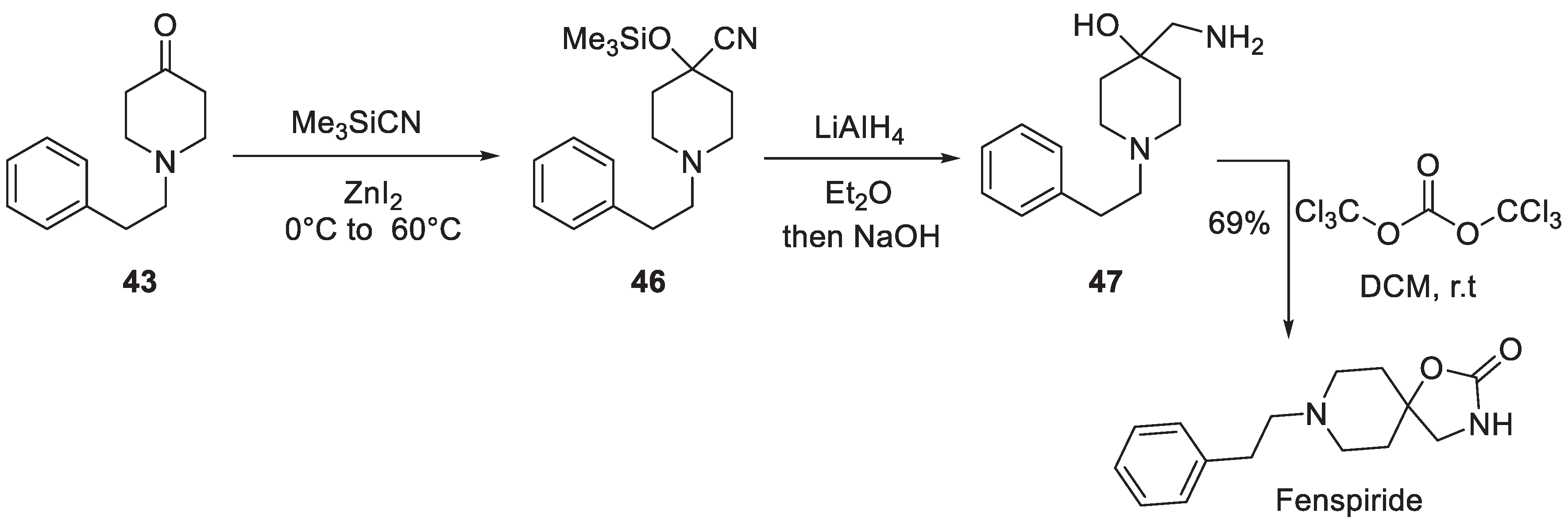
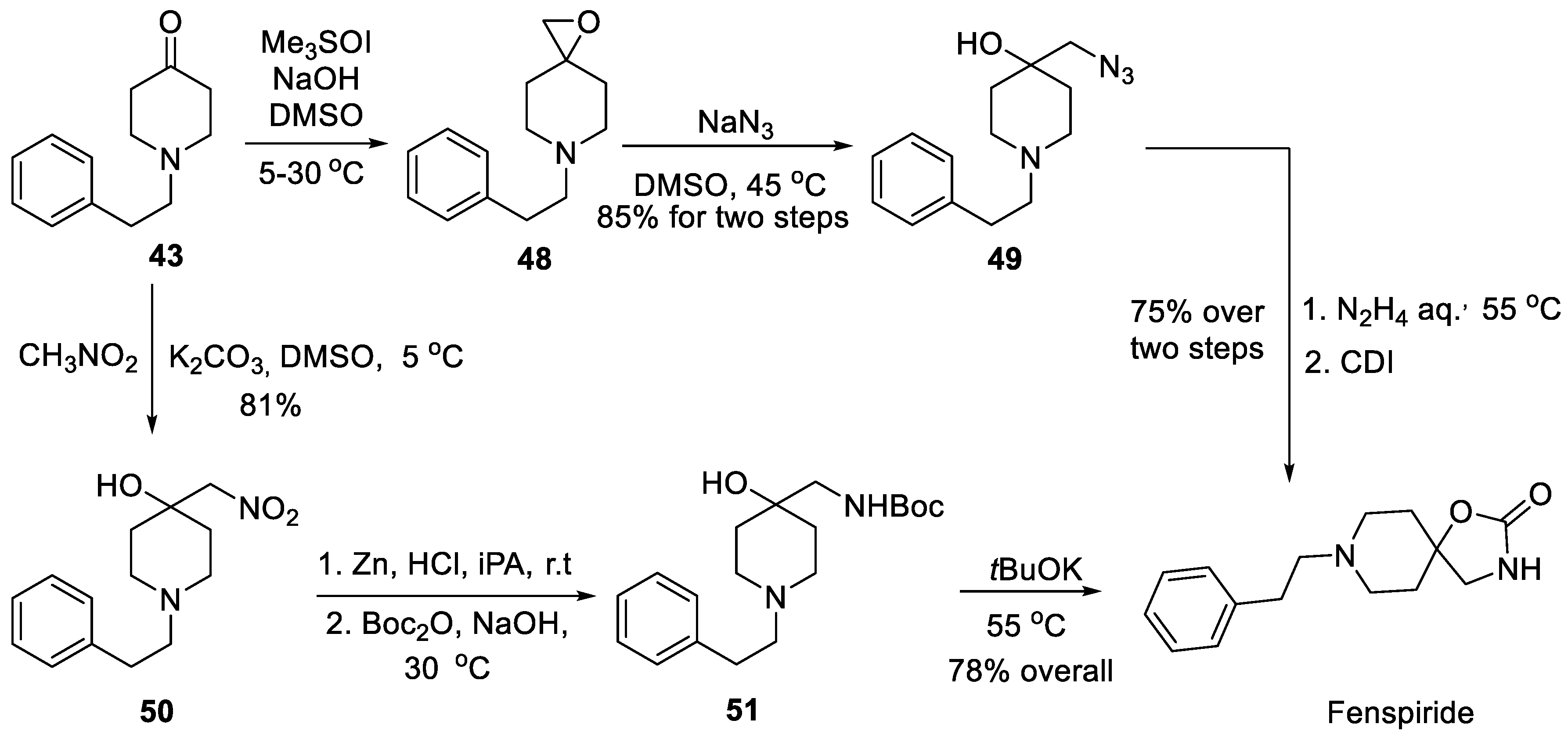
5. Fenspiride
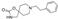
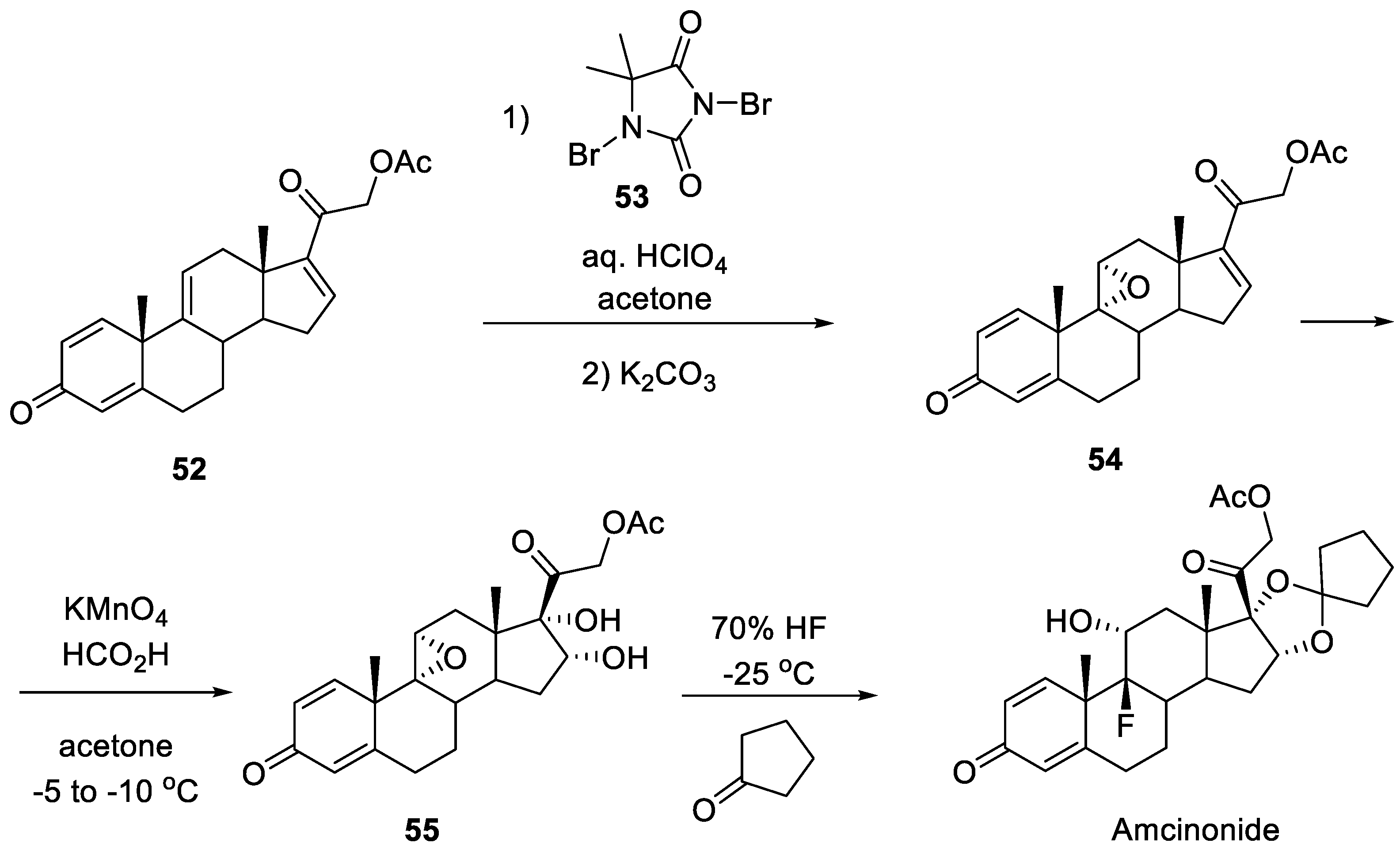
6. Amcinonide
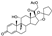
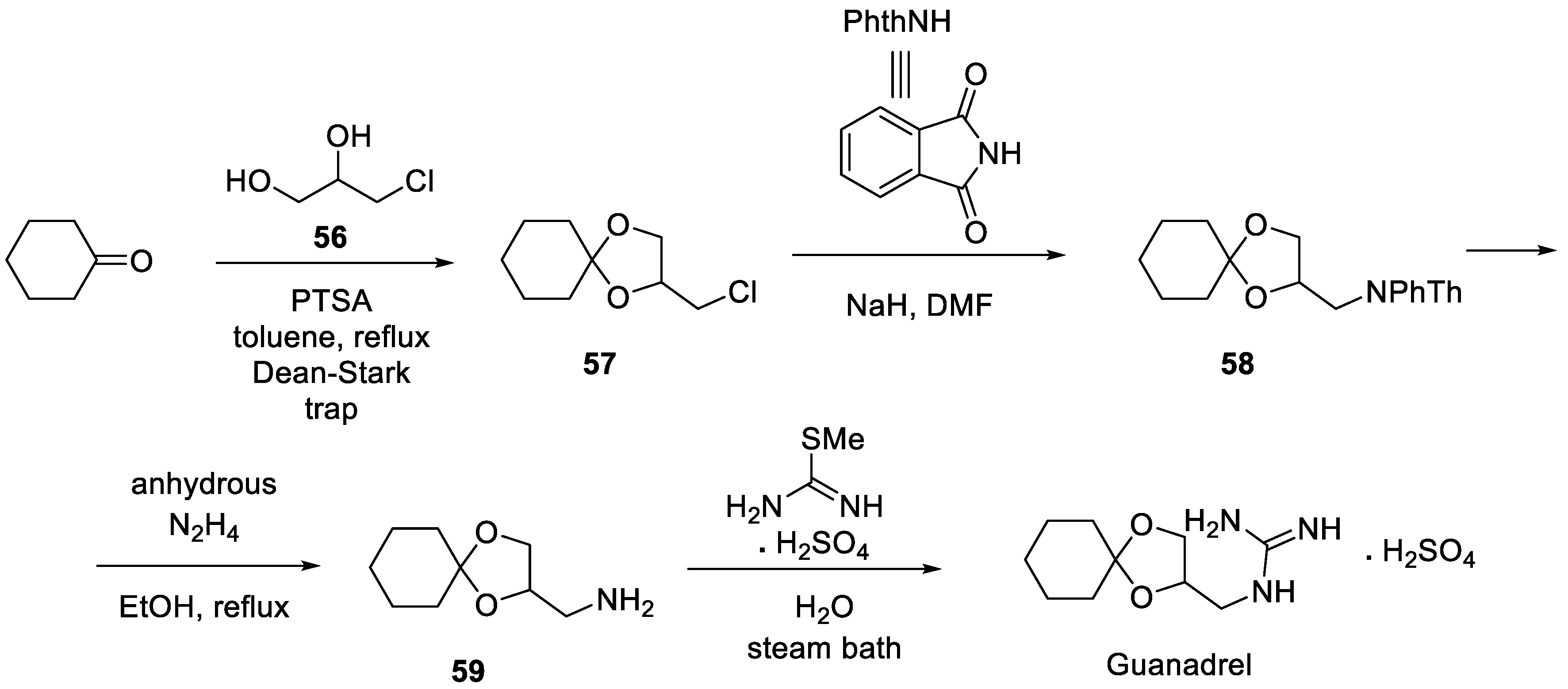
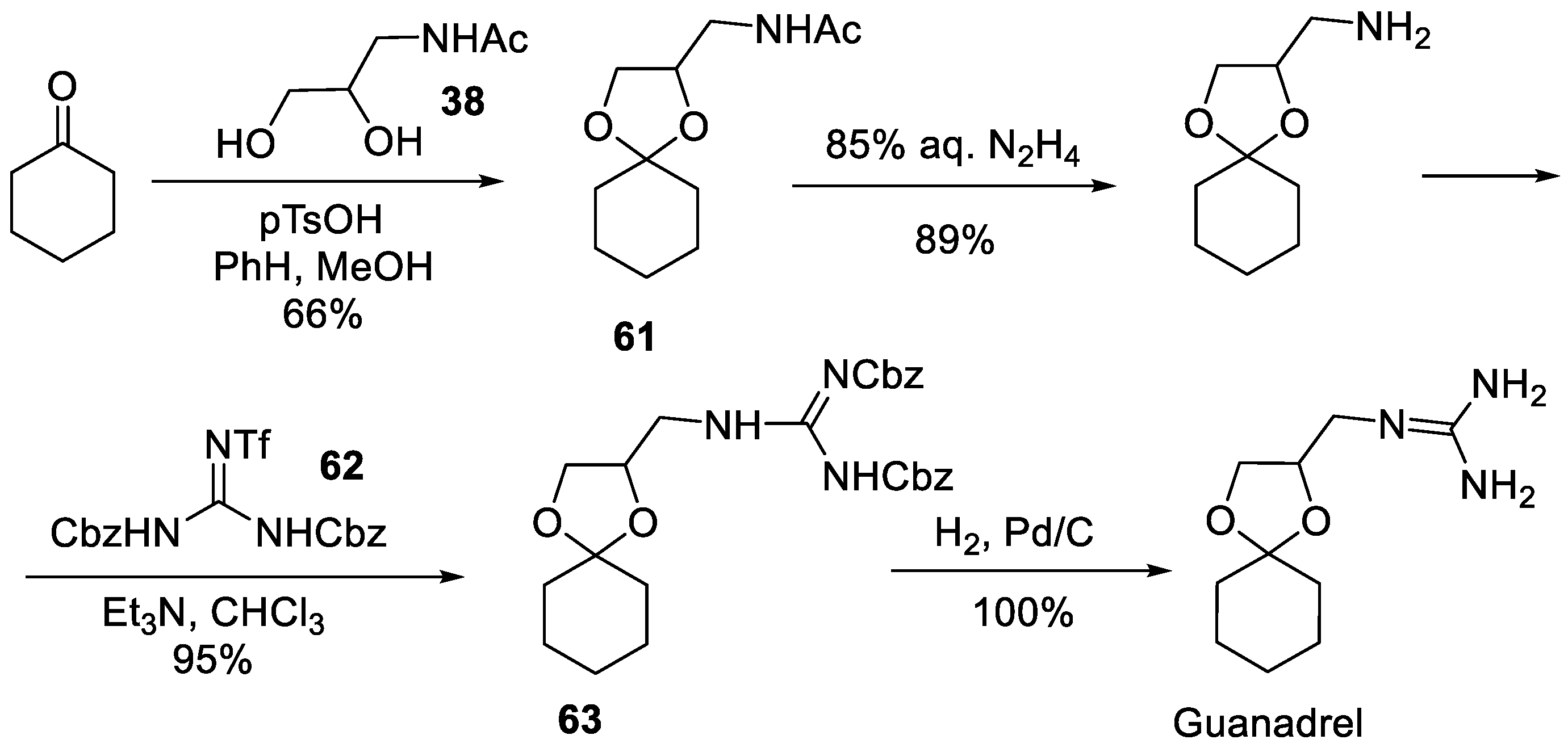
7. Guanadrel
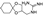
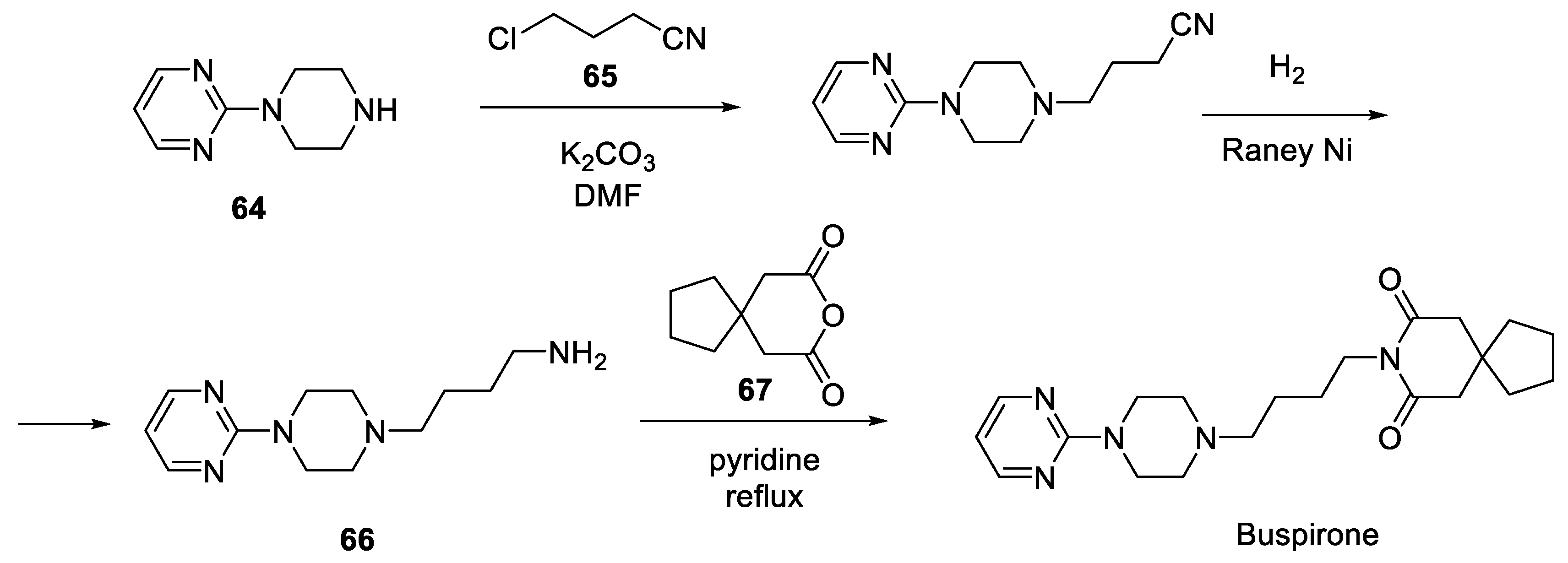
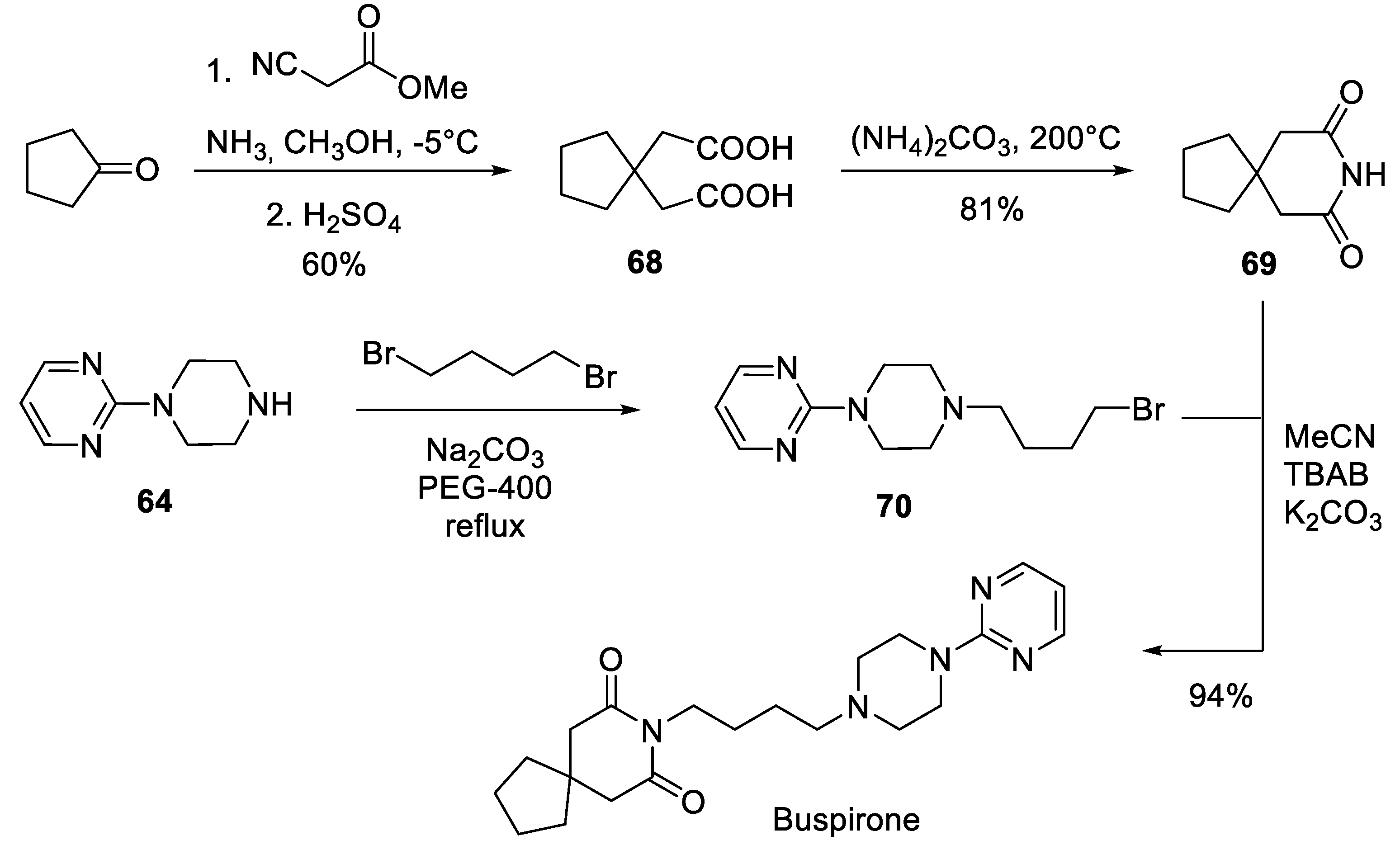
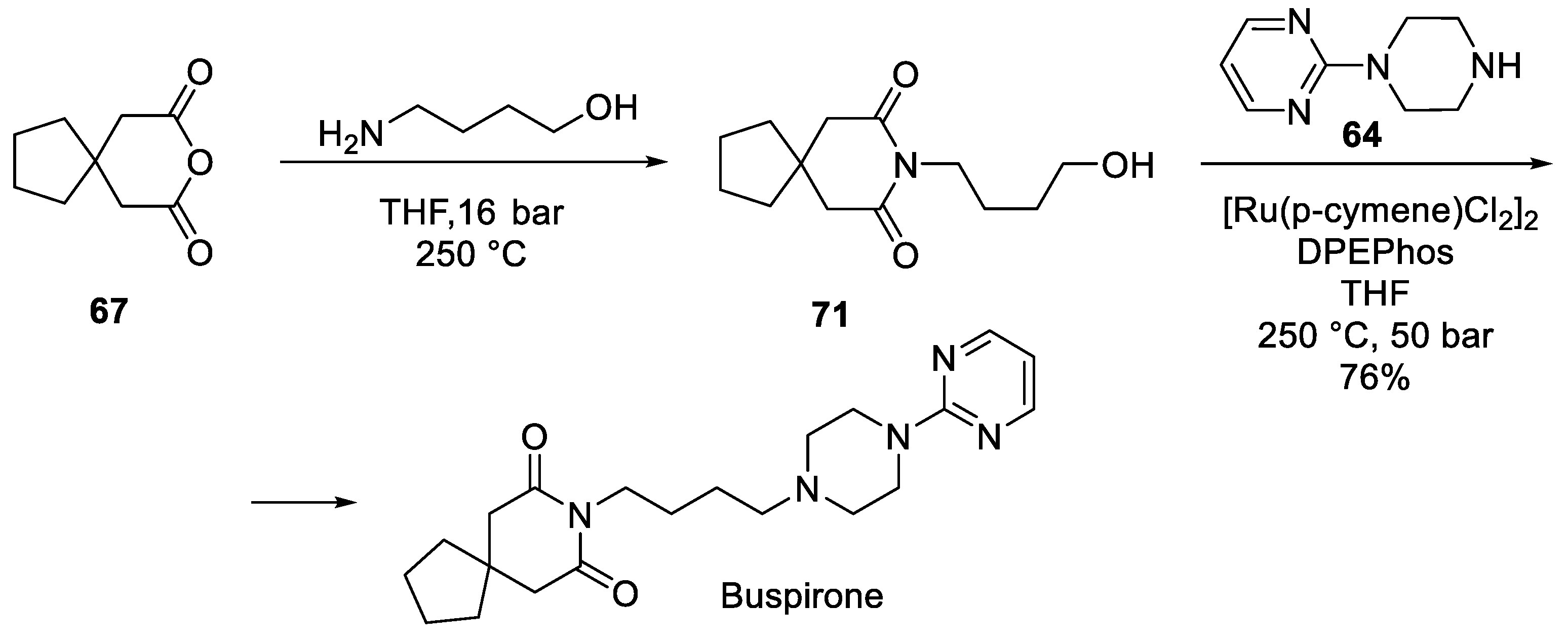
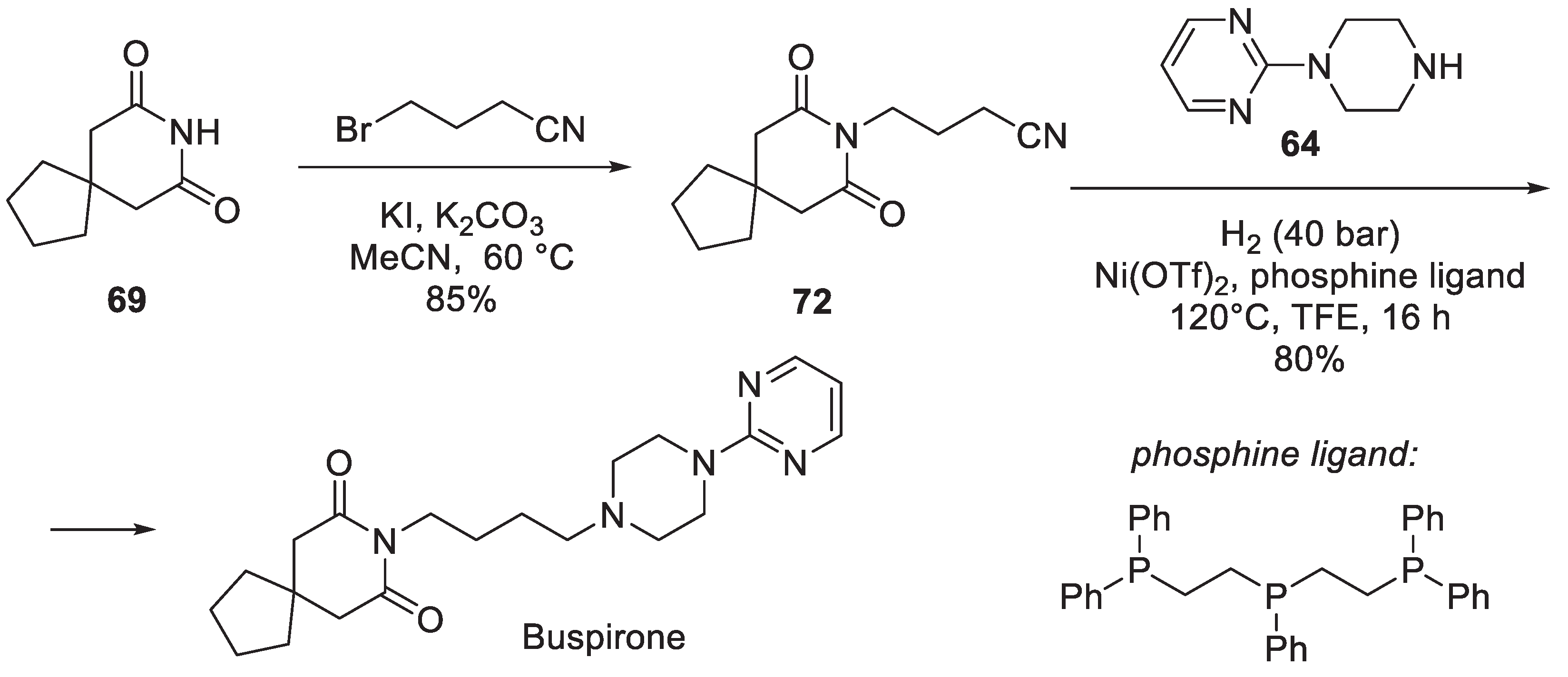
8. Buspirone
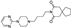
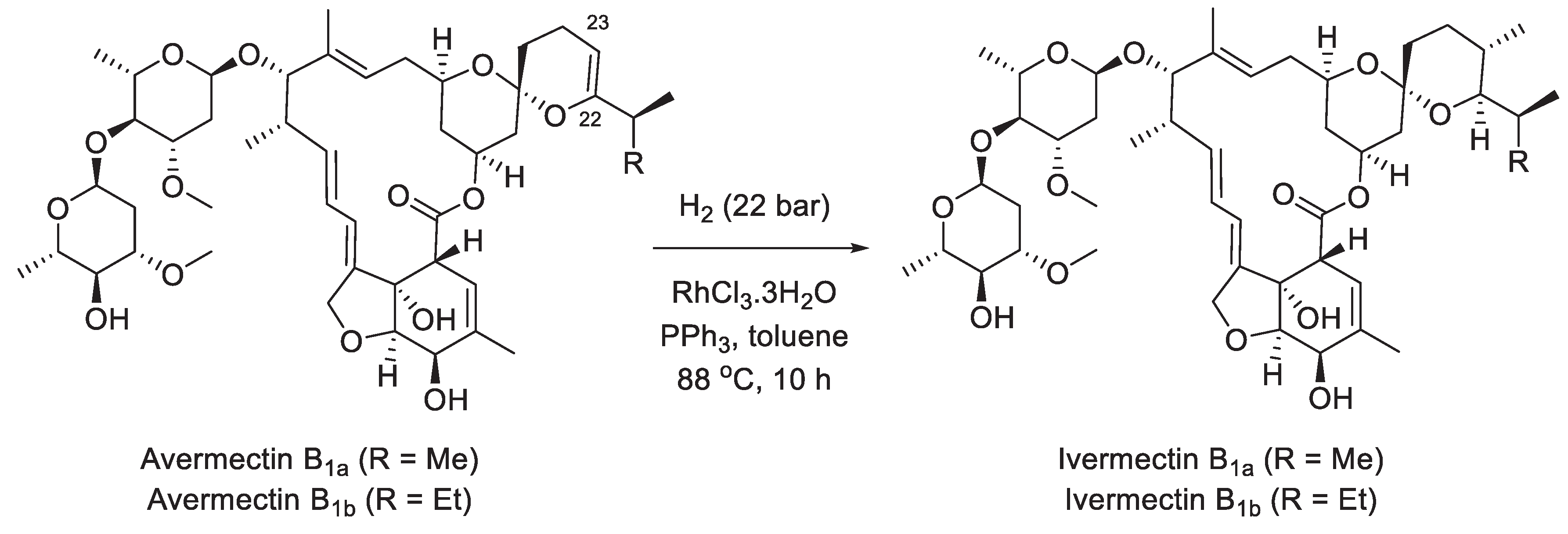
9. Ivermectin
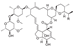
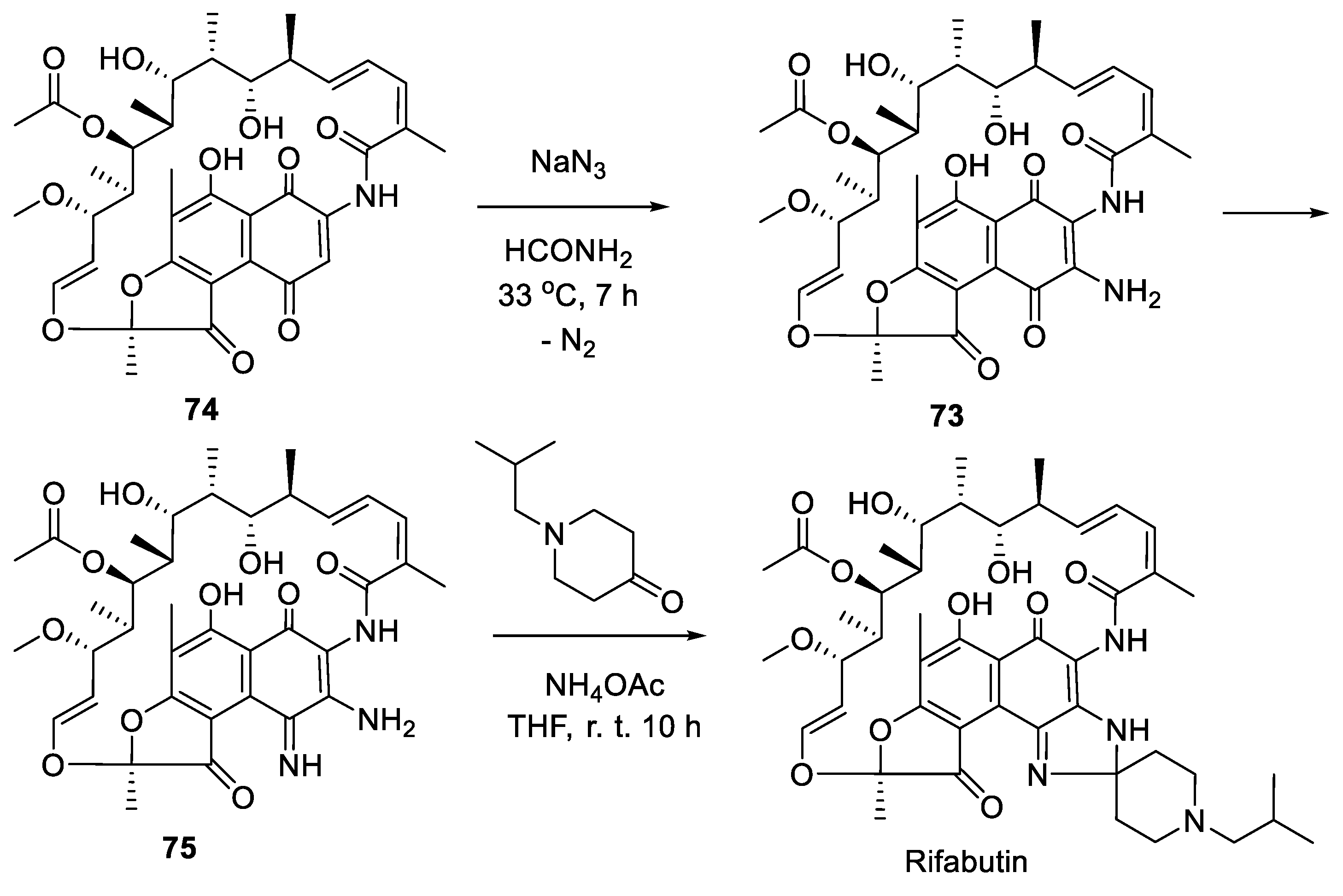
10. Rifabutin
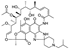
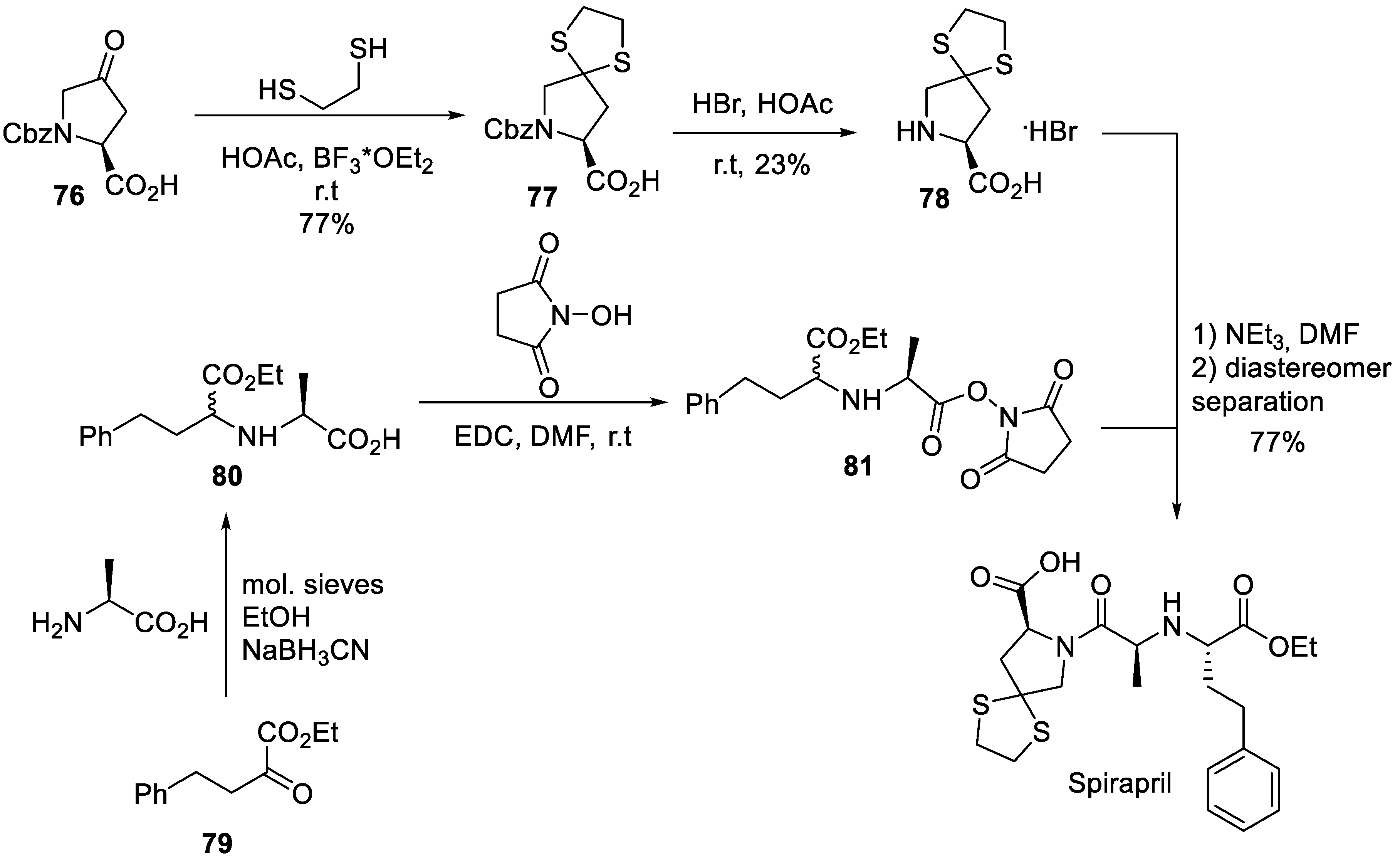
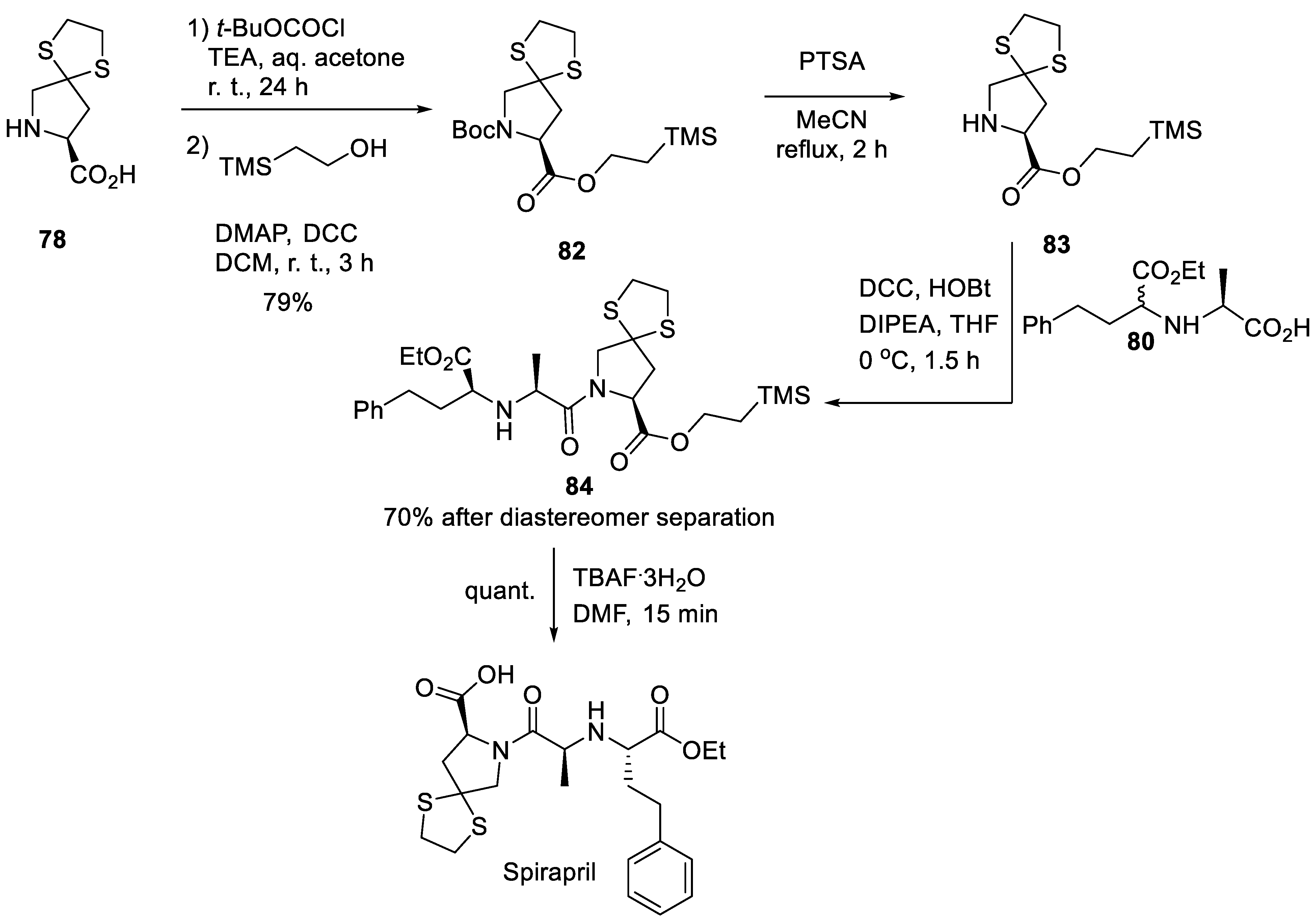
11. Spirapril
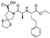
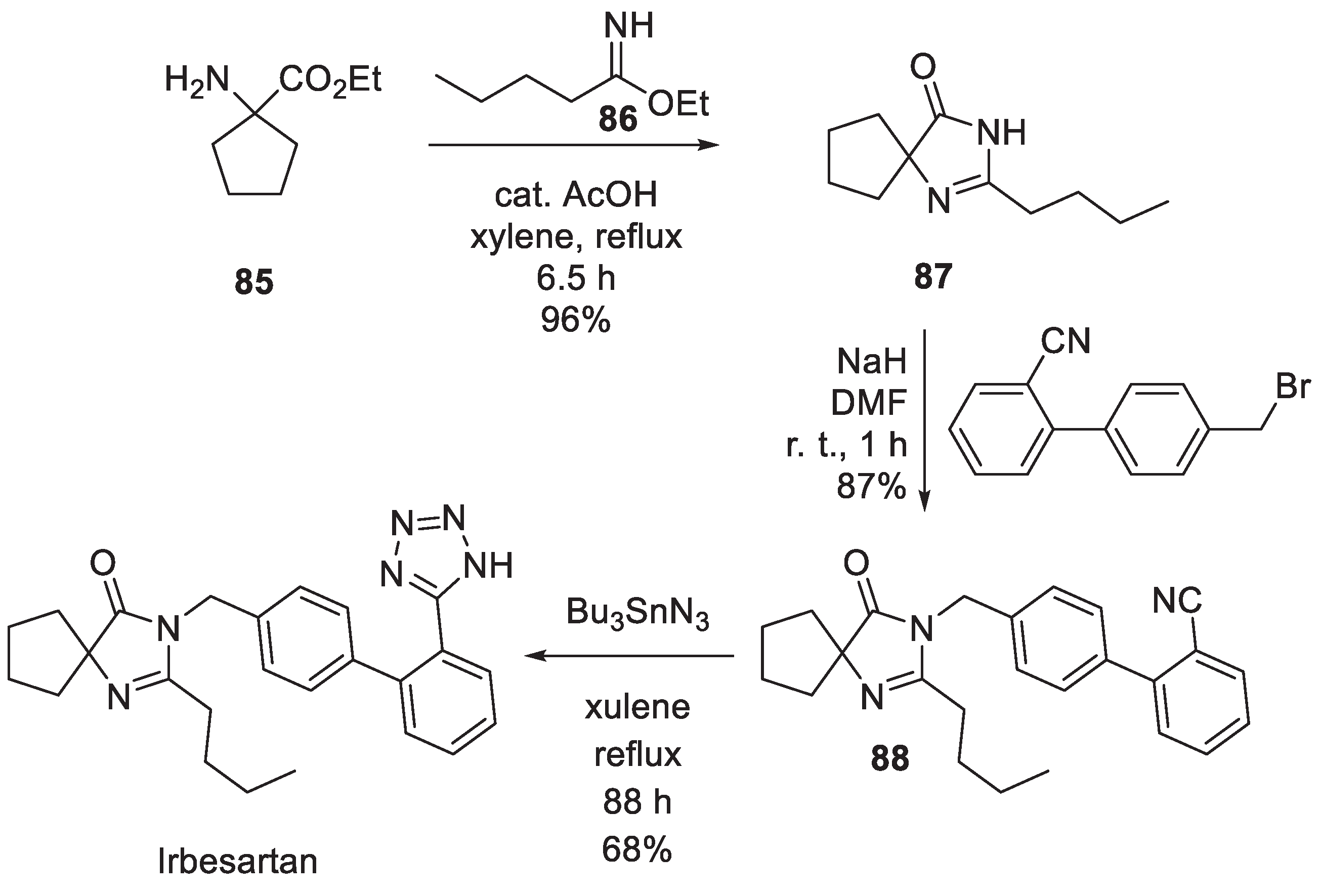
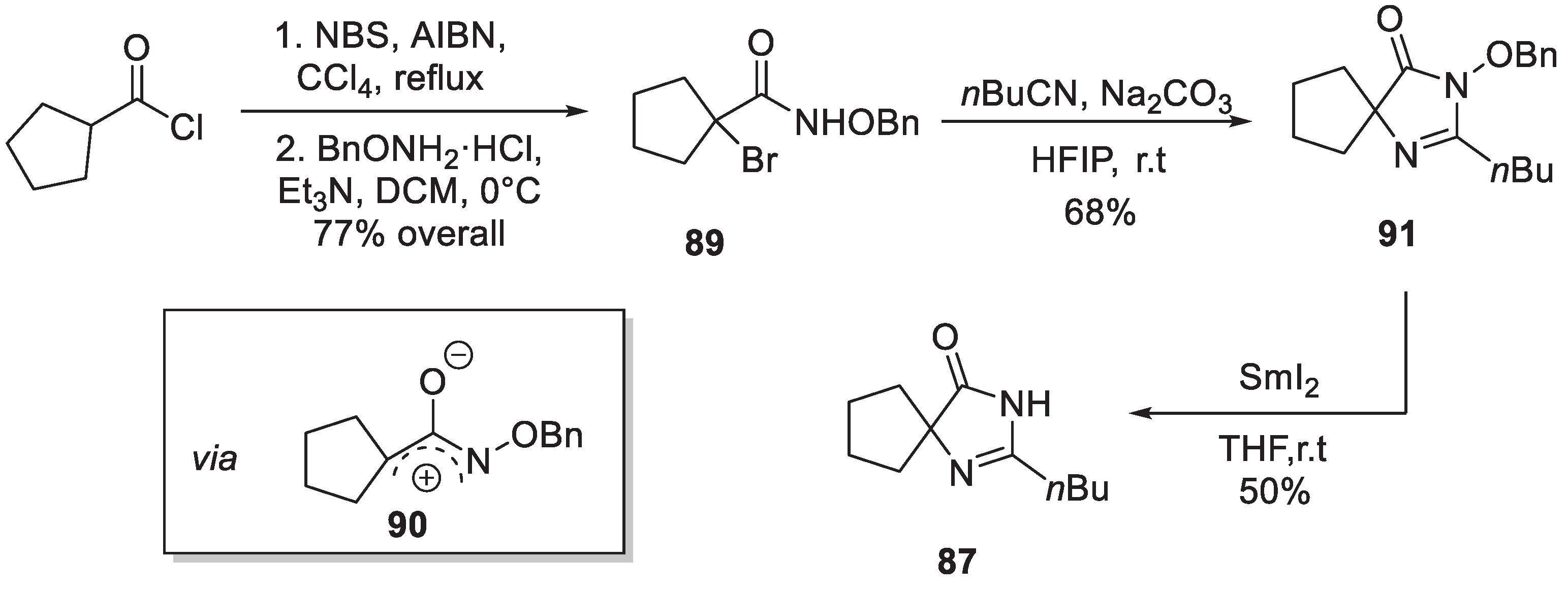
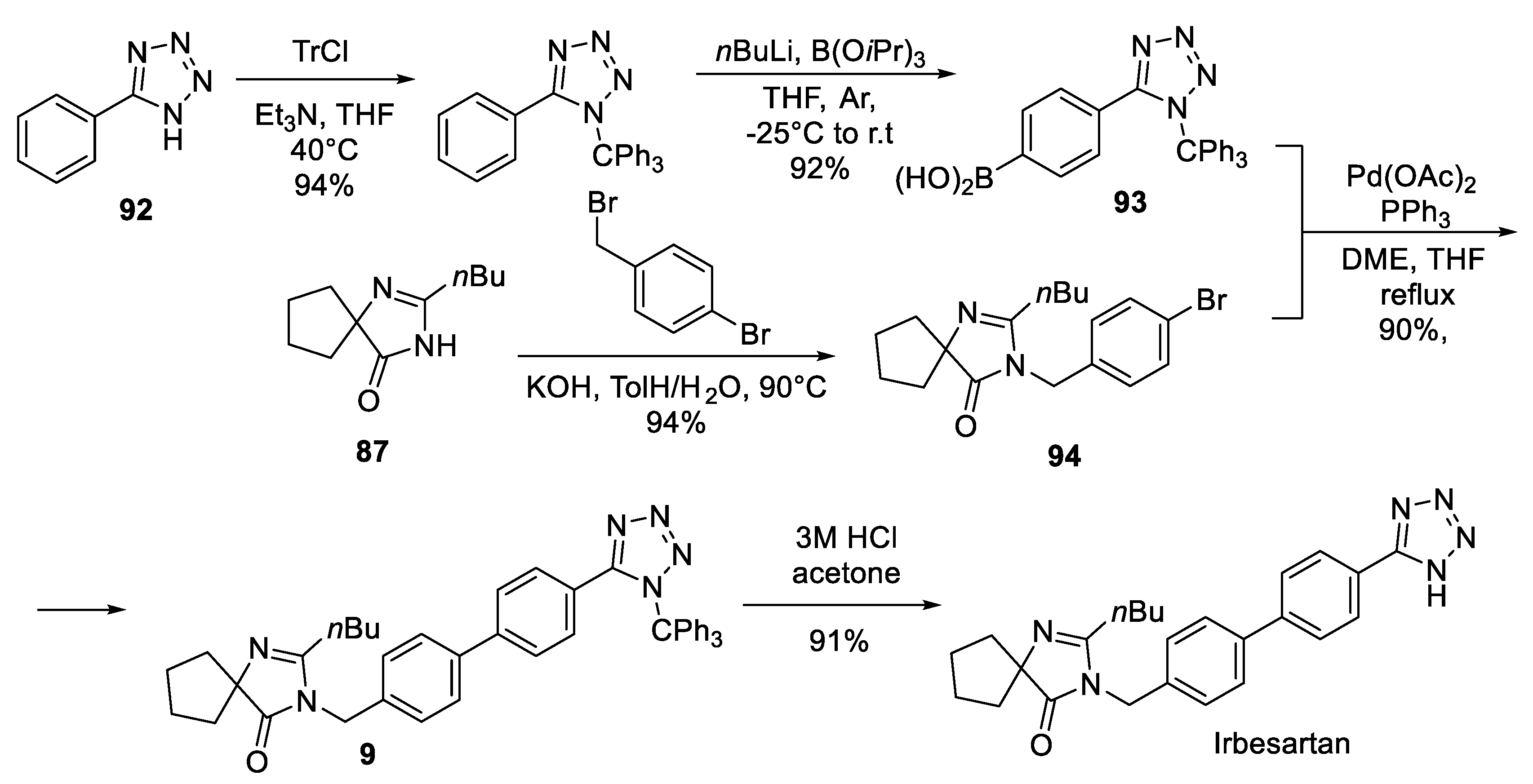
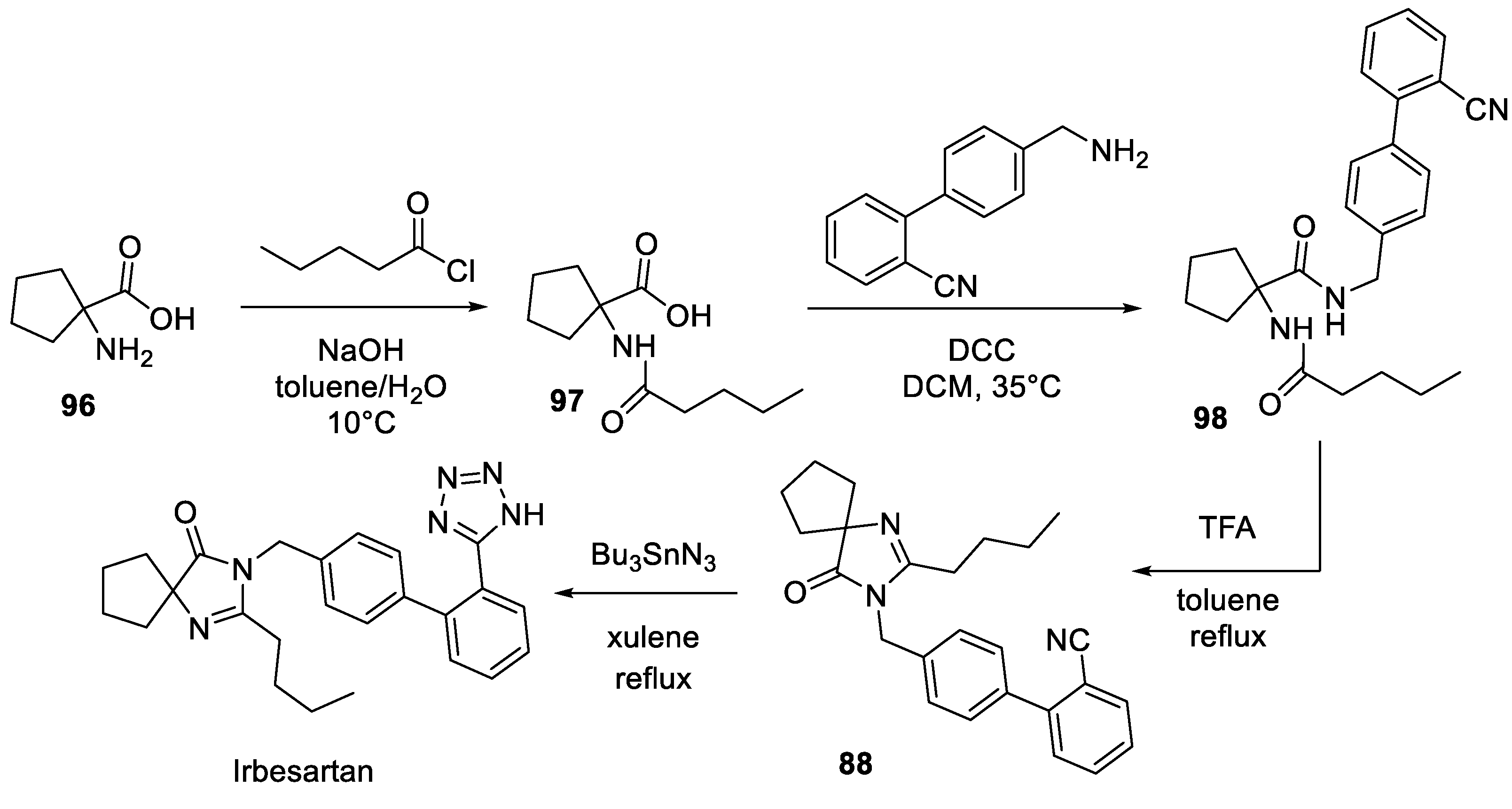
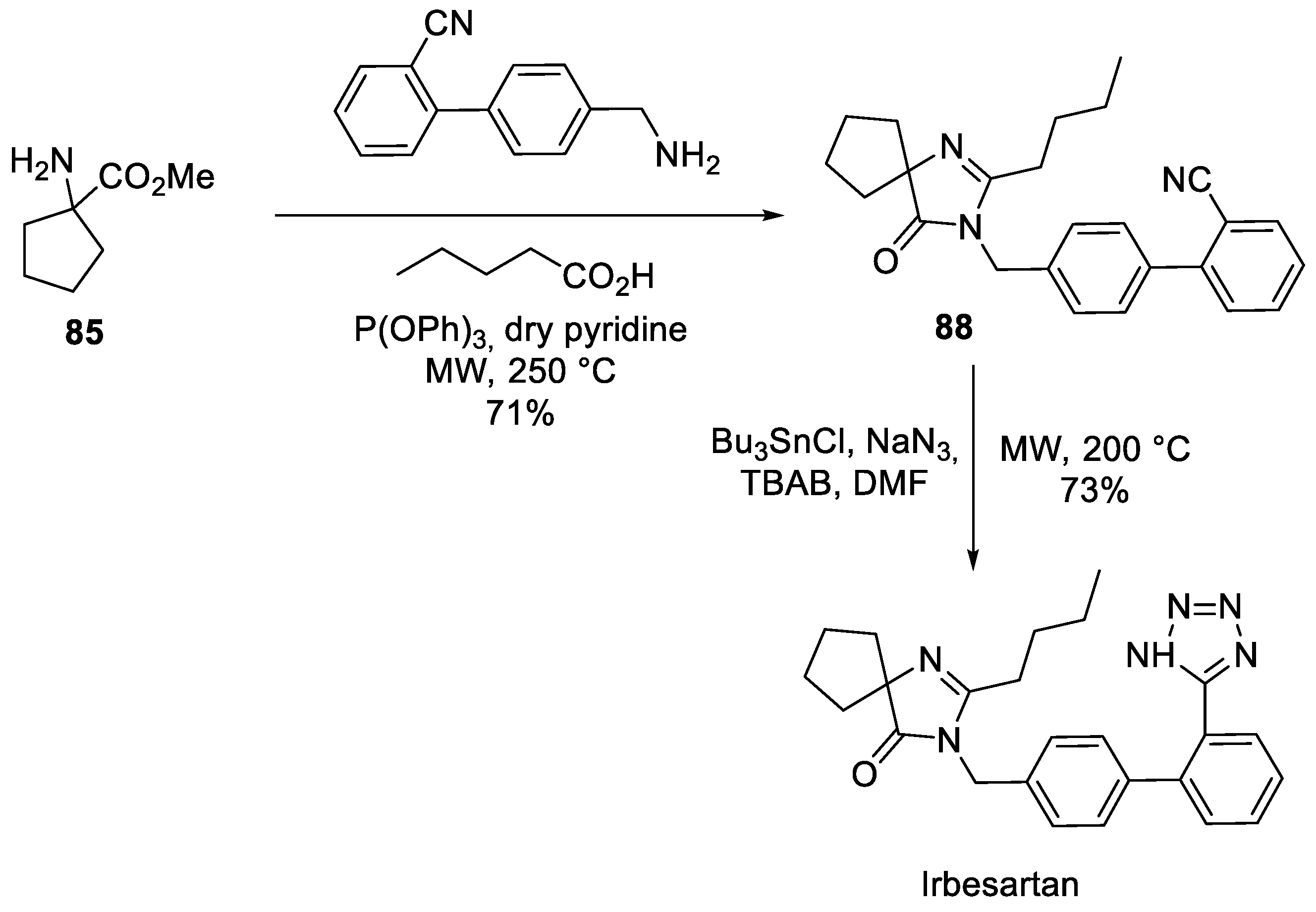
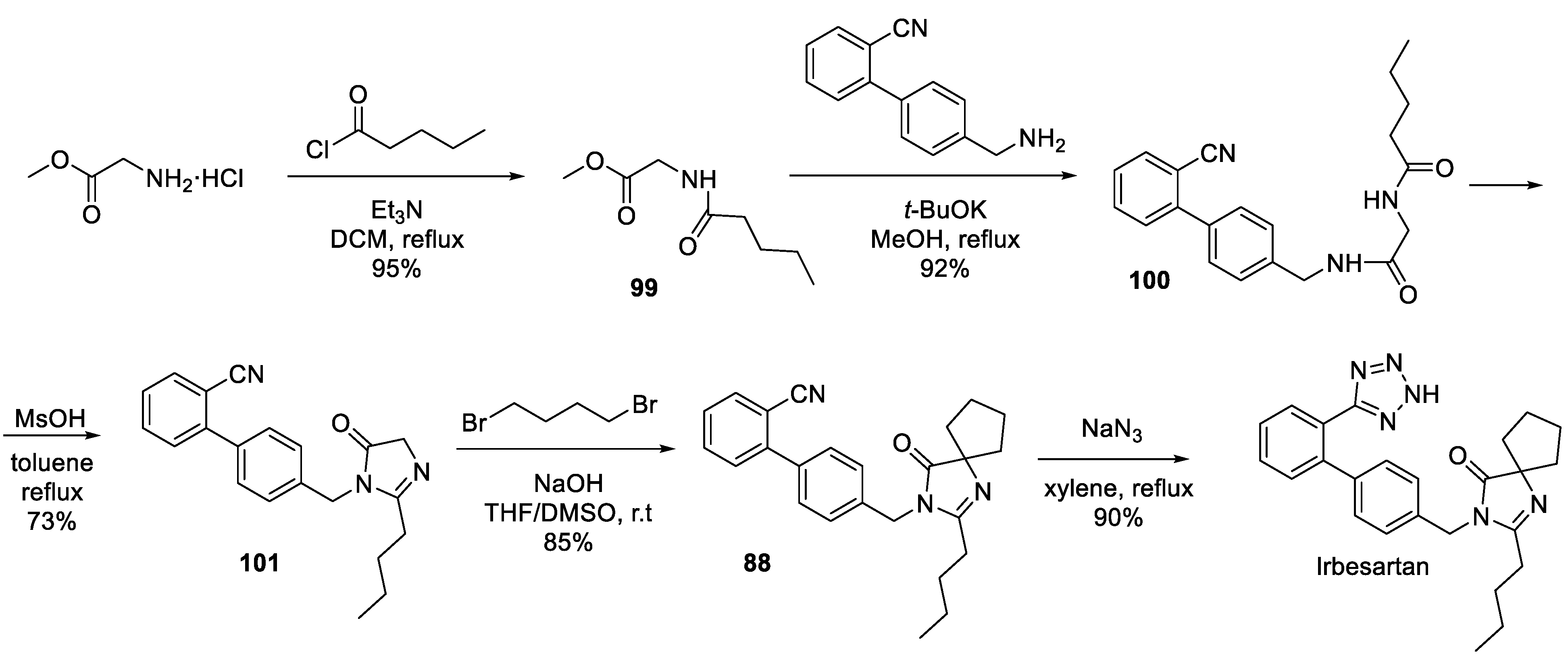
12. Irbesartan
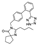
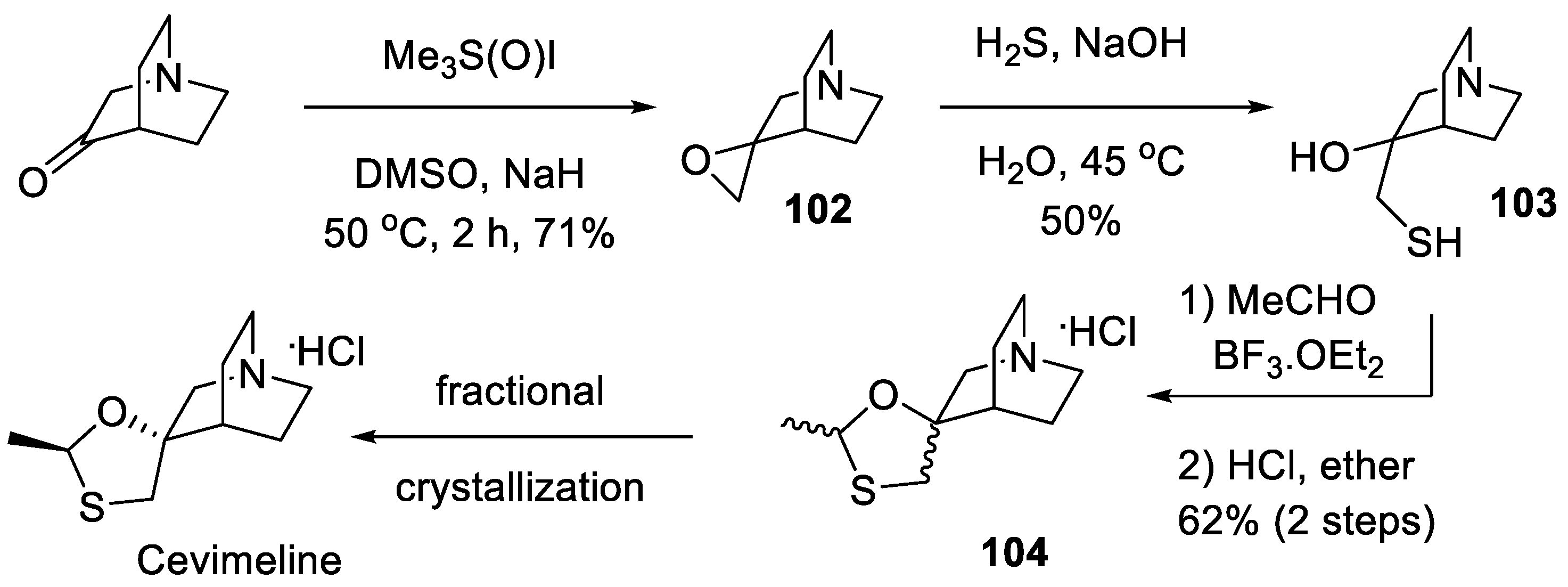
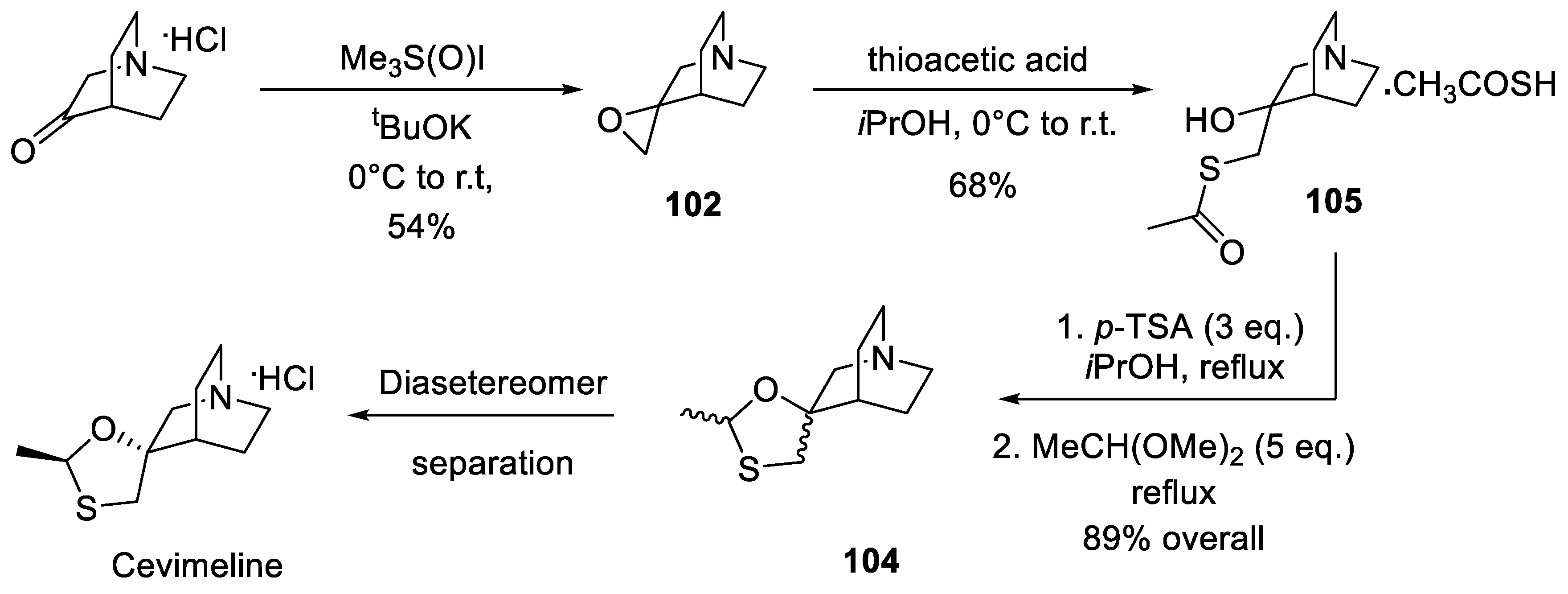
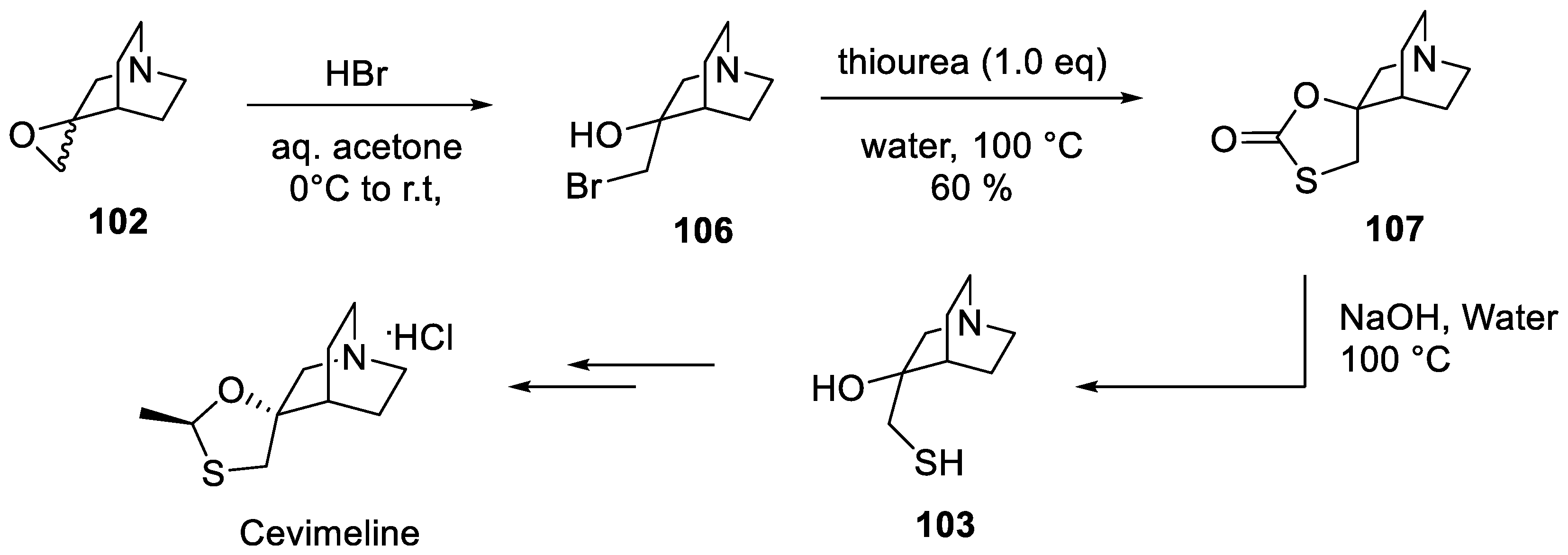
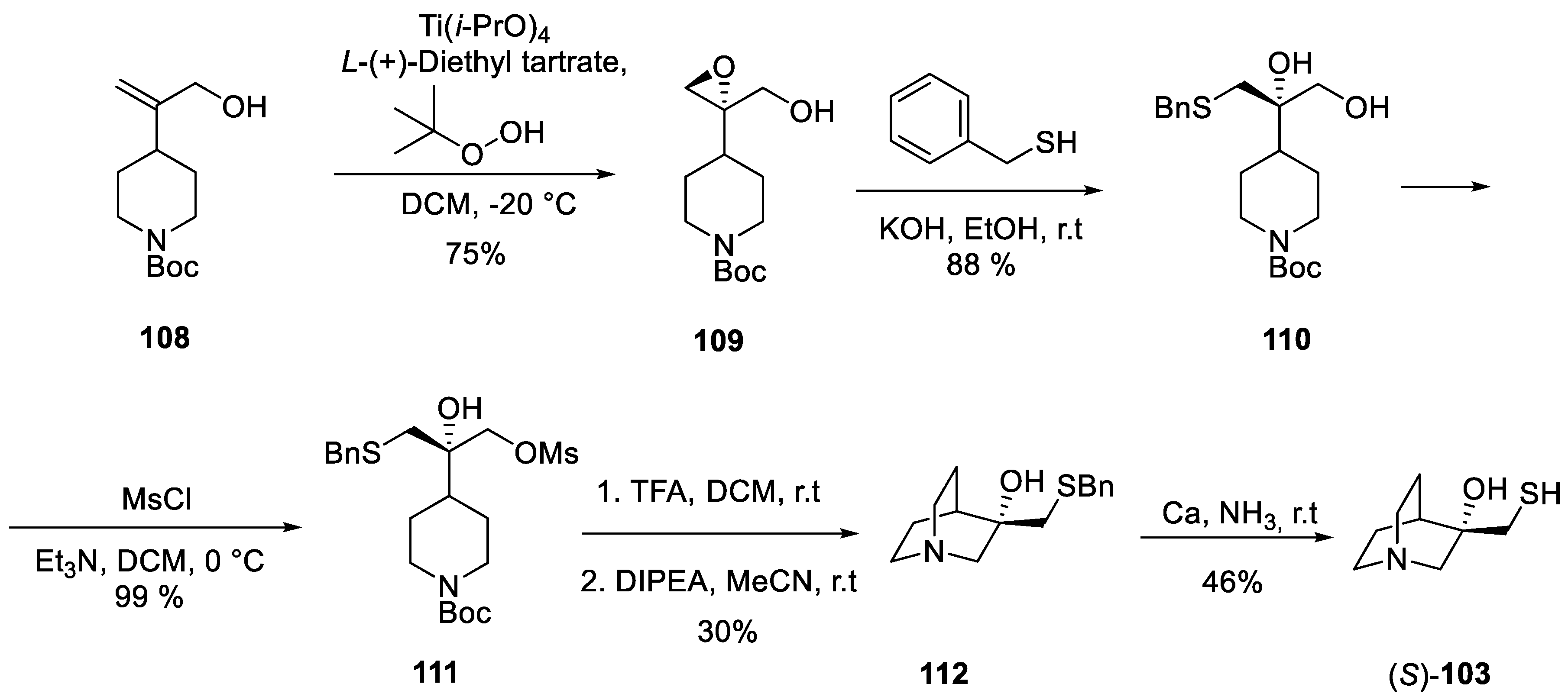
13. Cevimeline
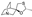
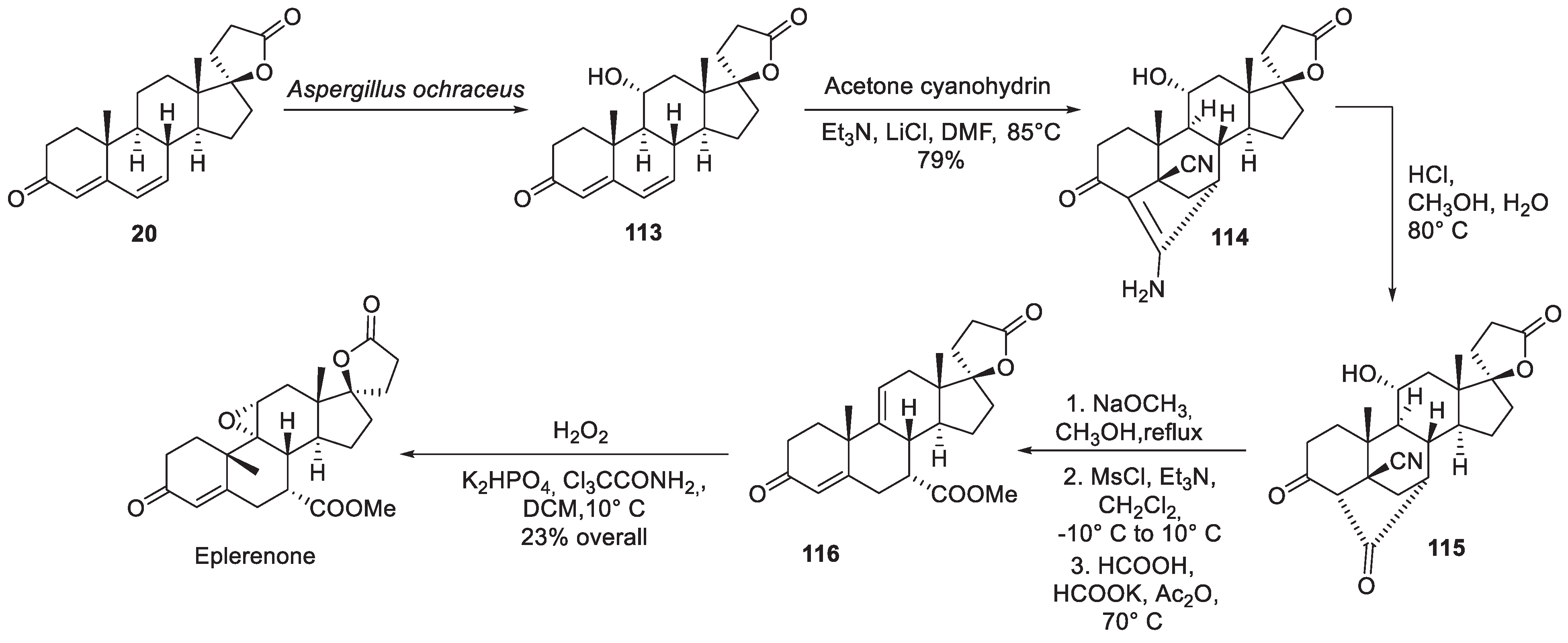
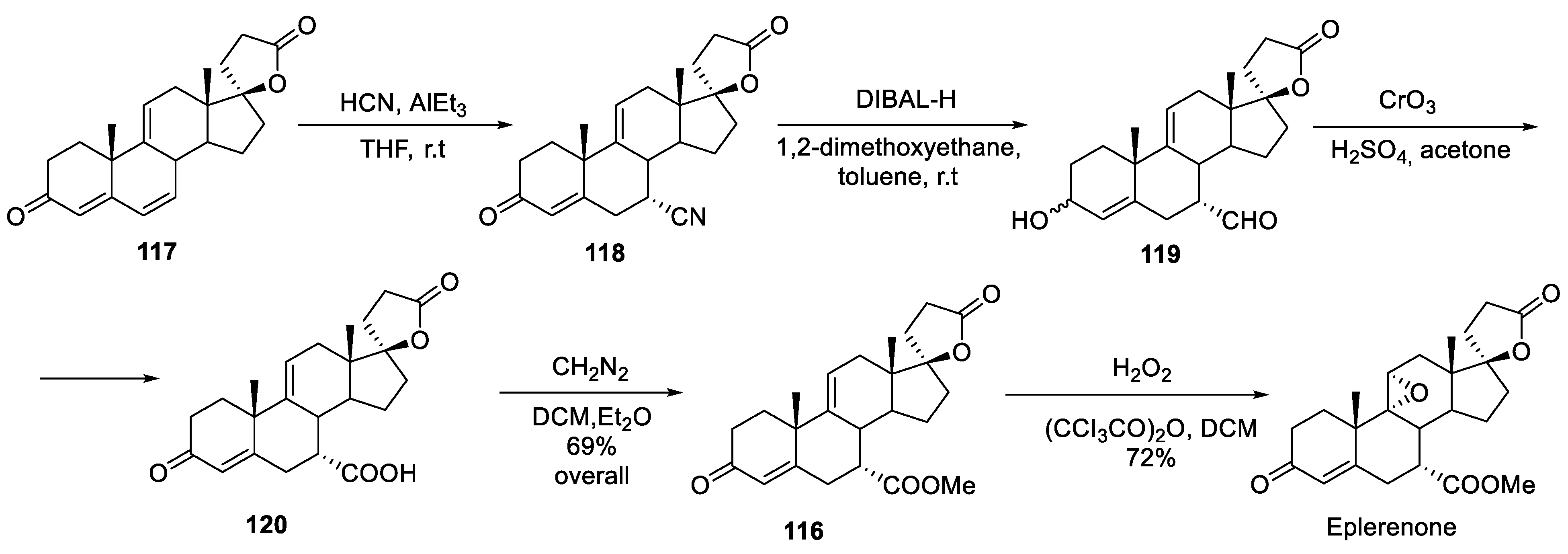
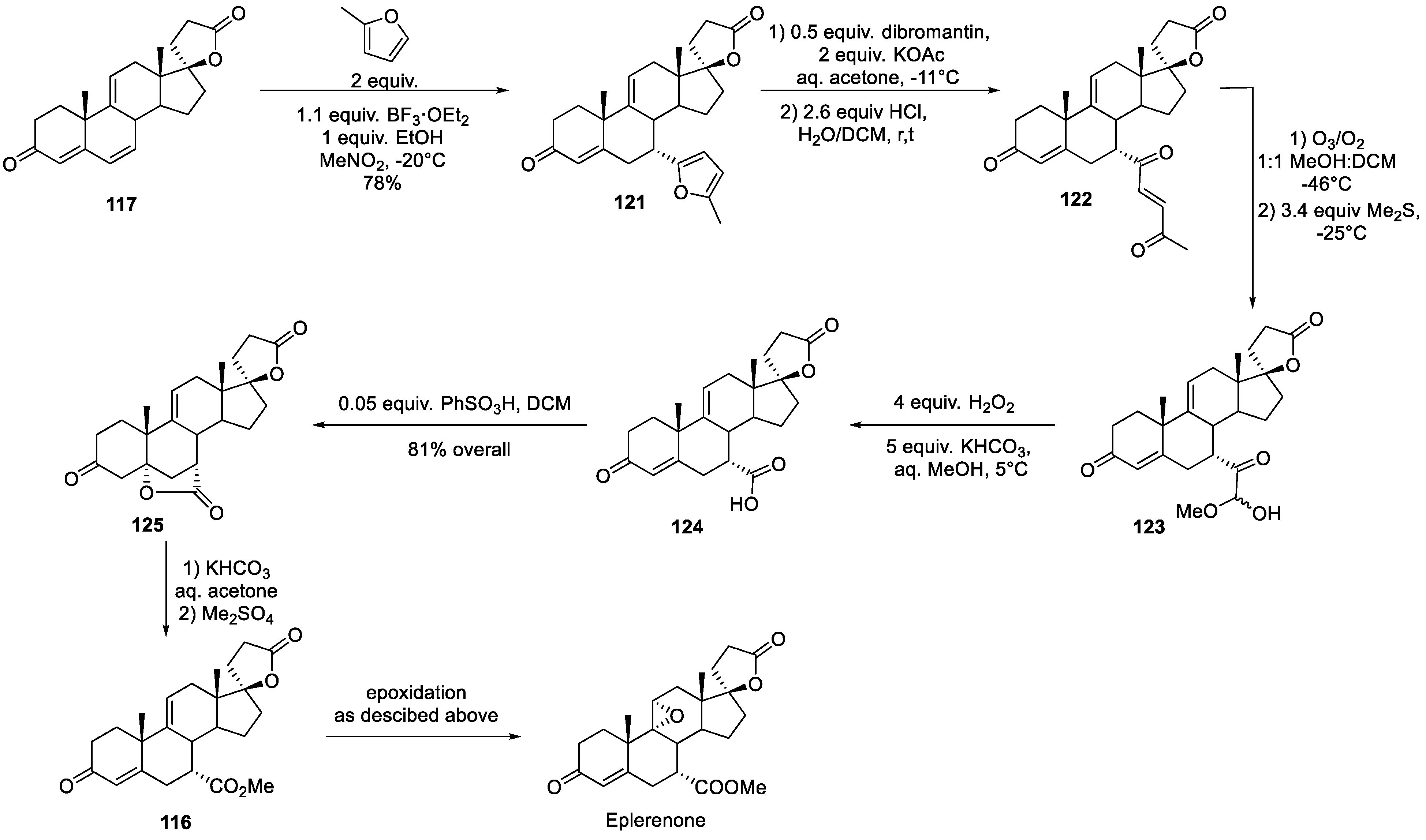
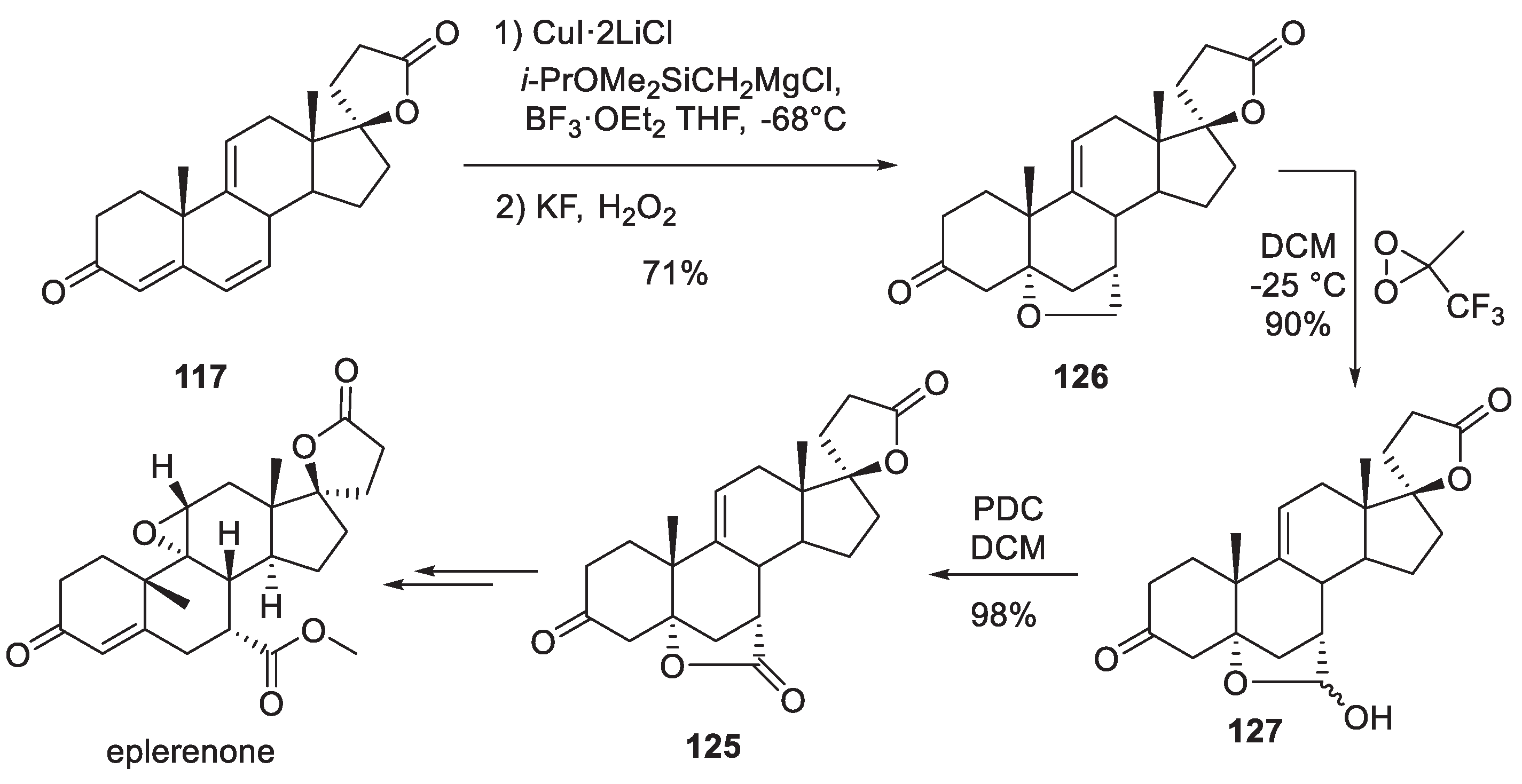
14. Eplerenone
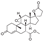
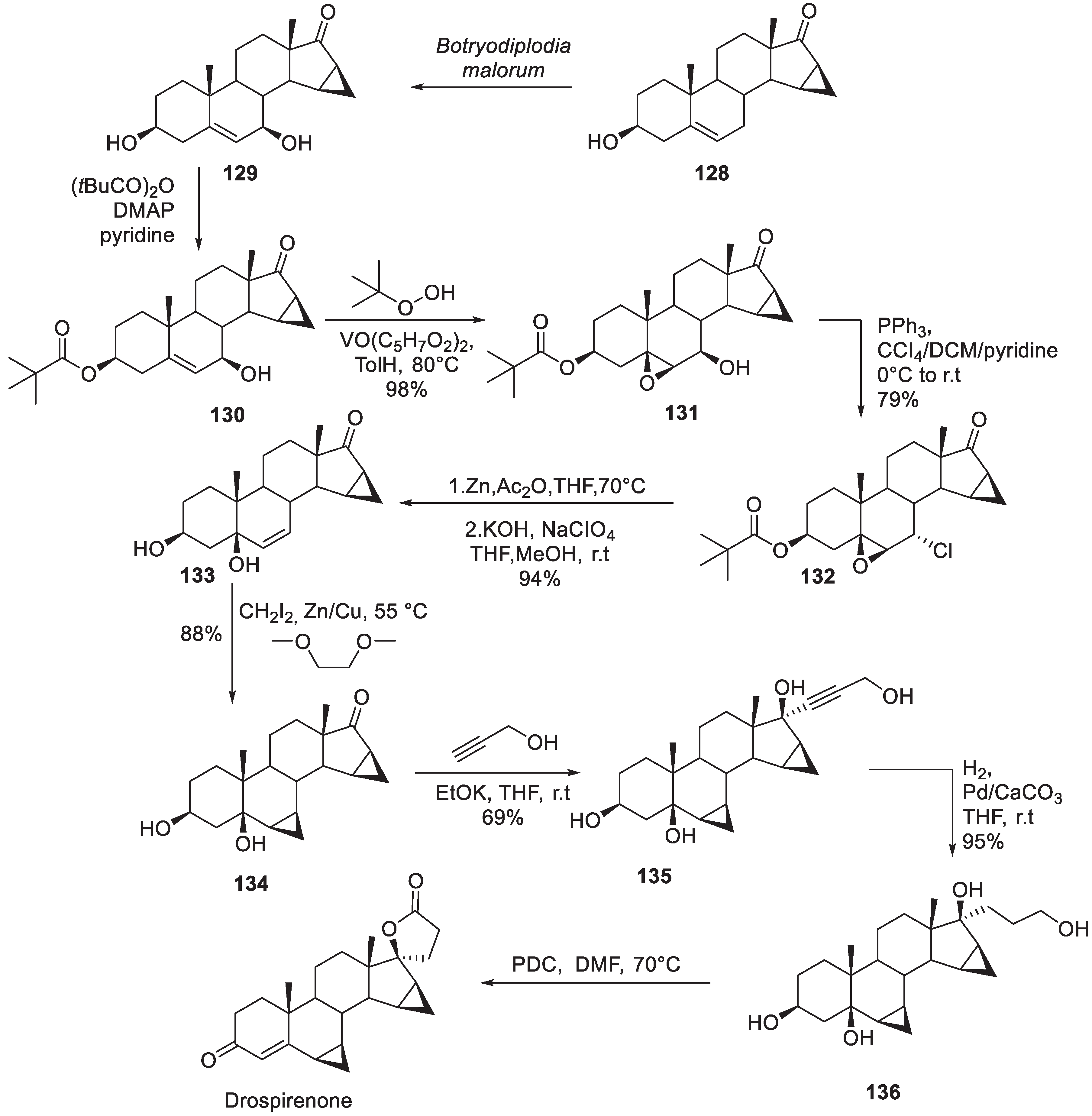
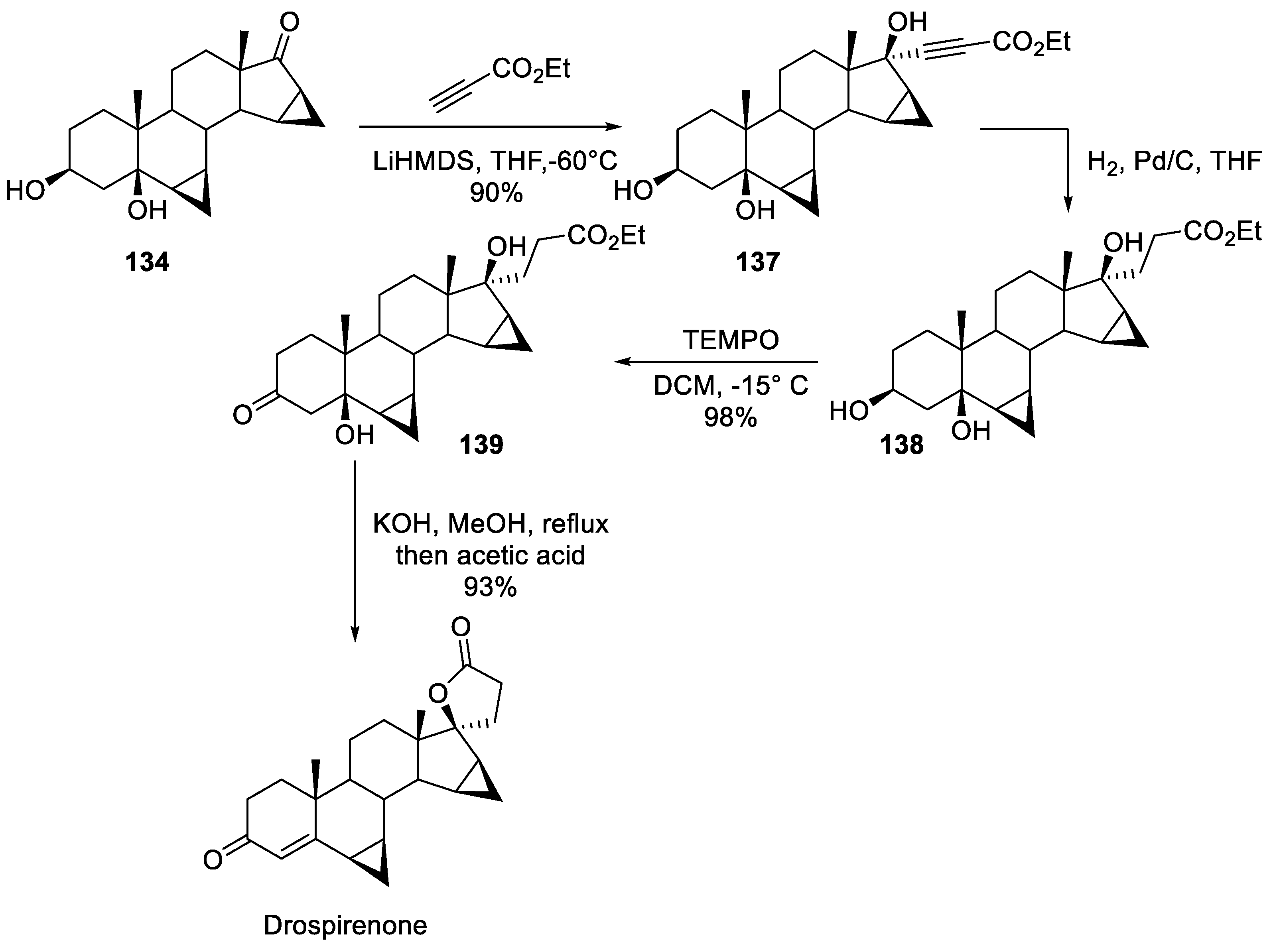
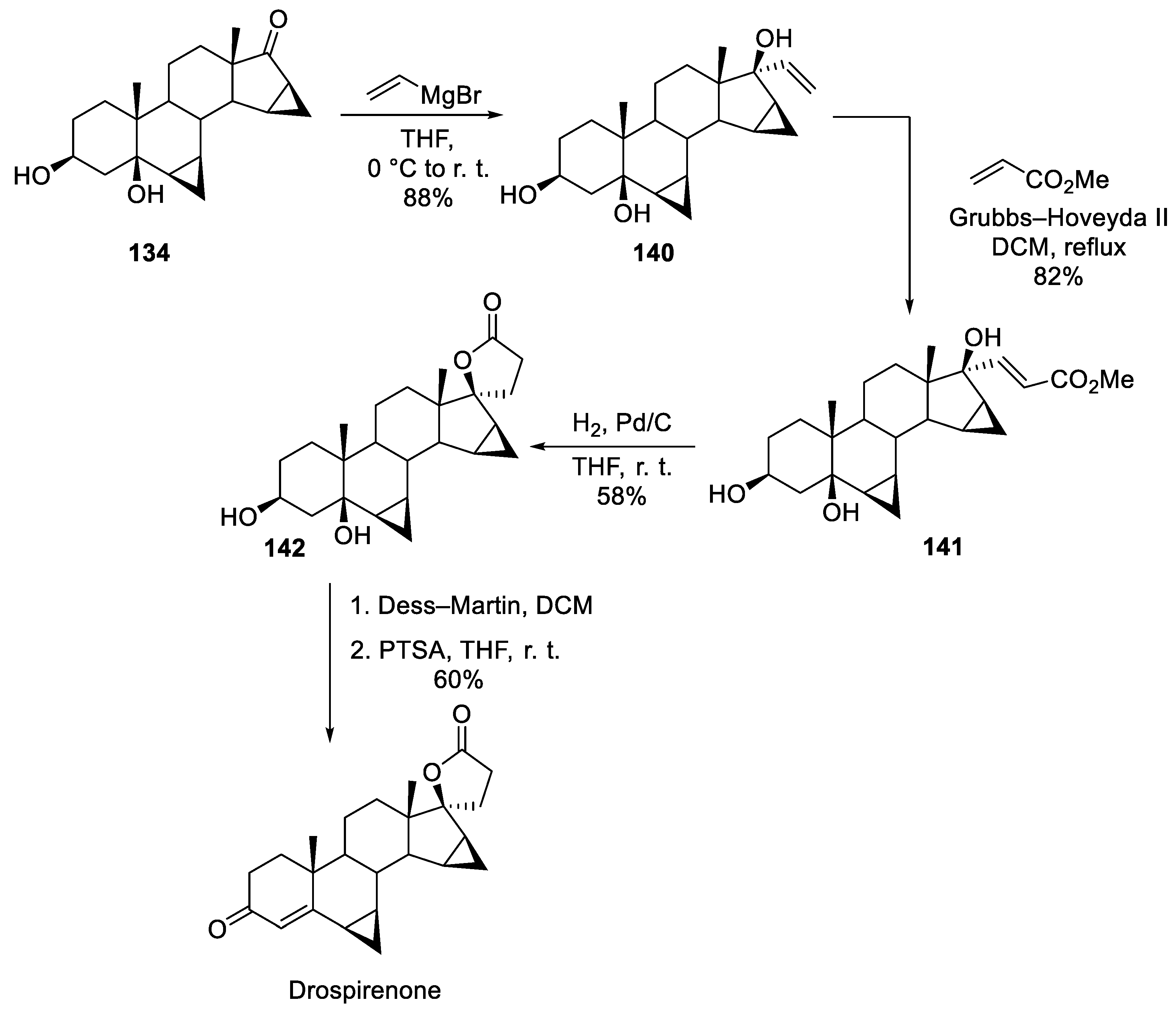
15. Drospirenone
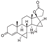
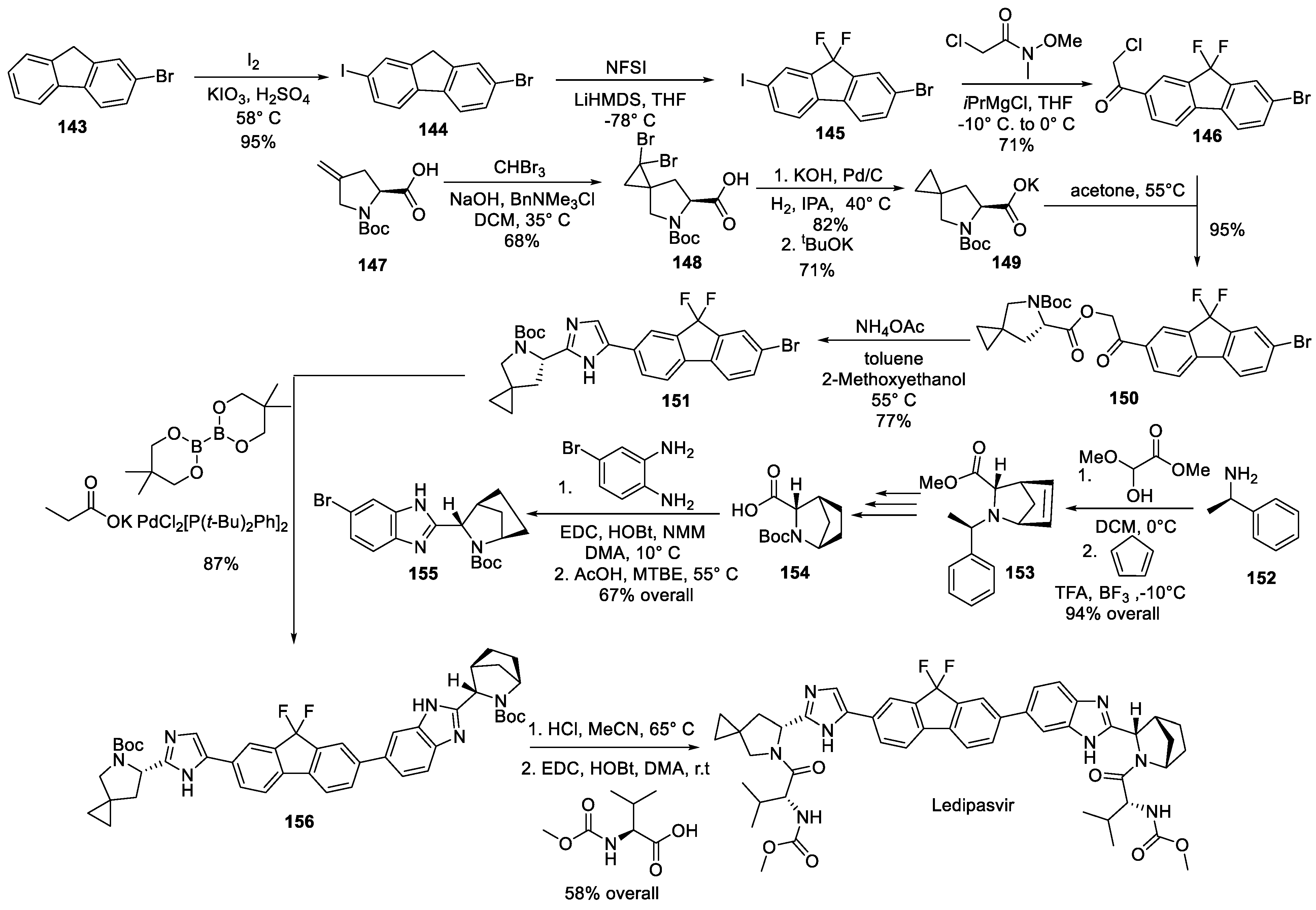
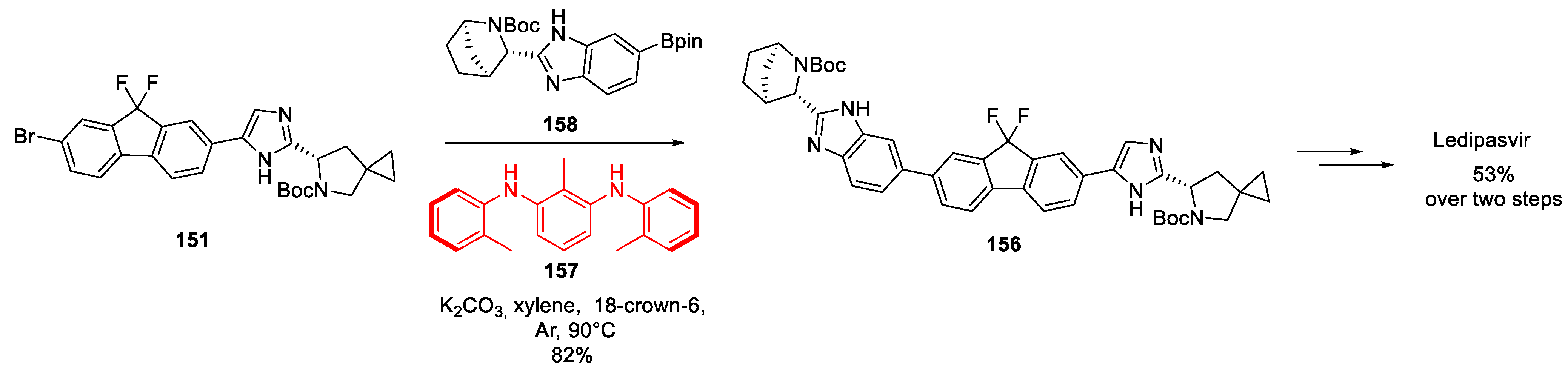
16. Ledipasvir
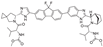
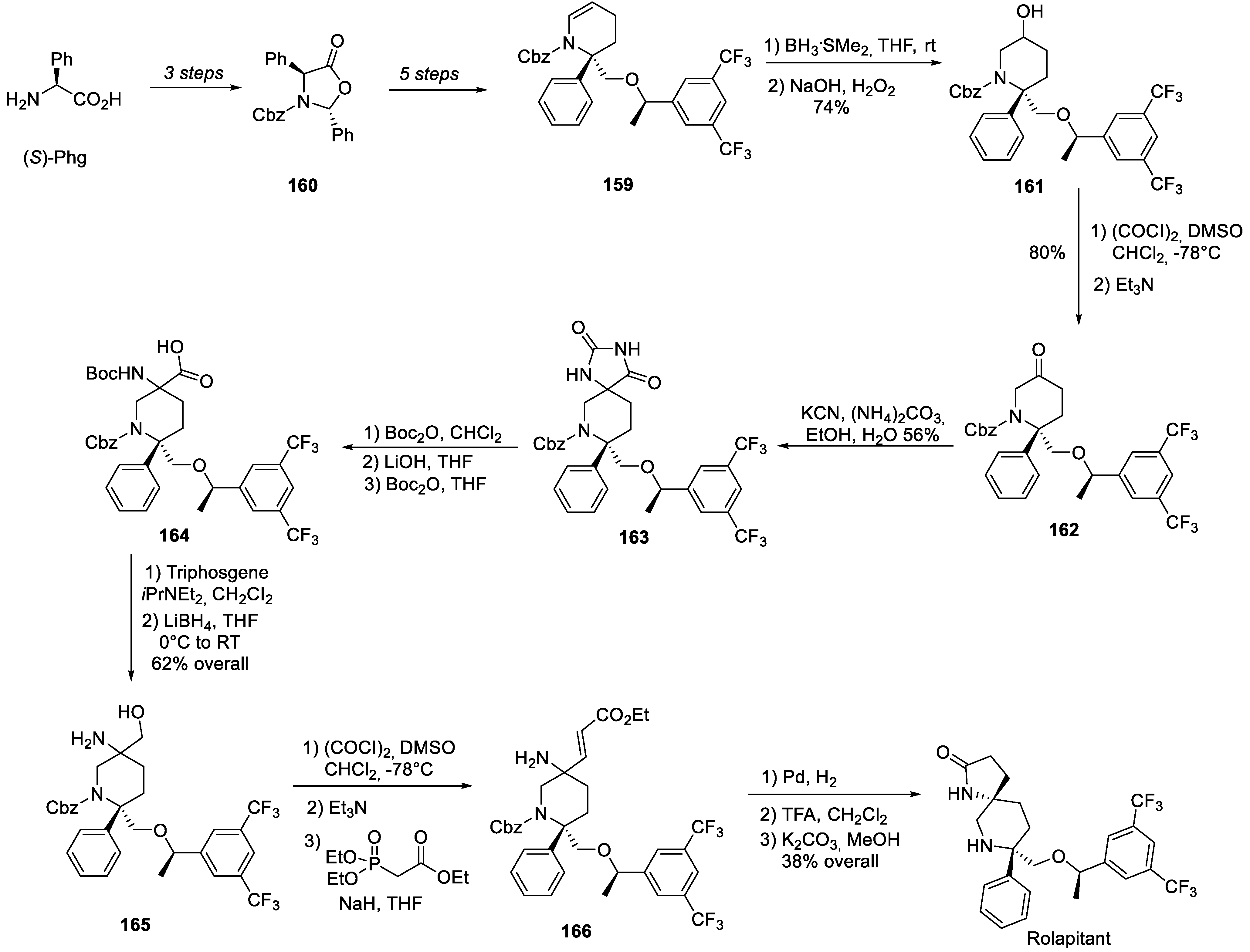
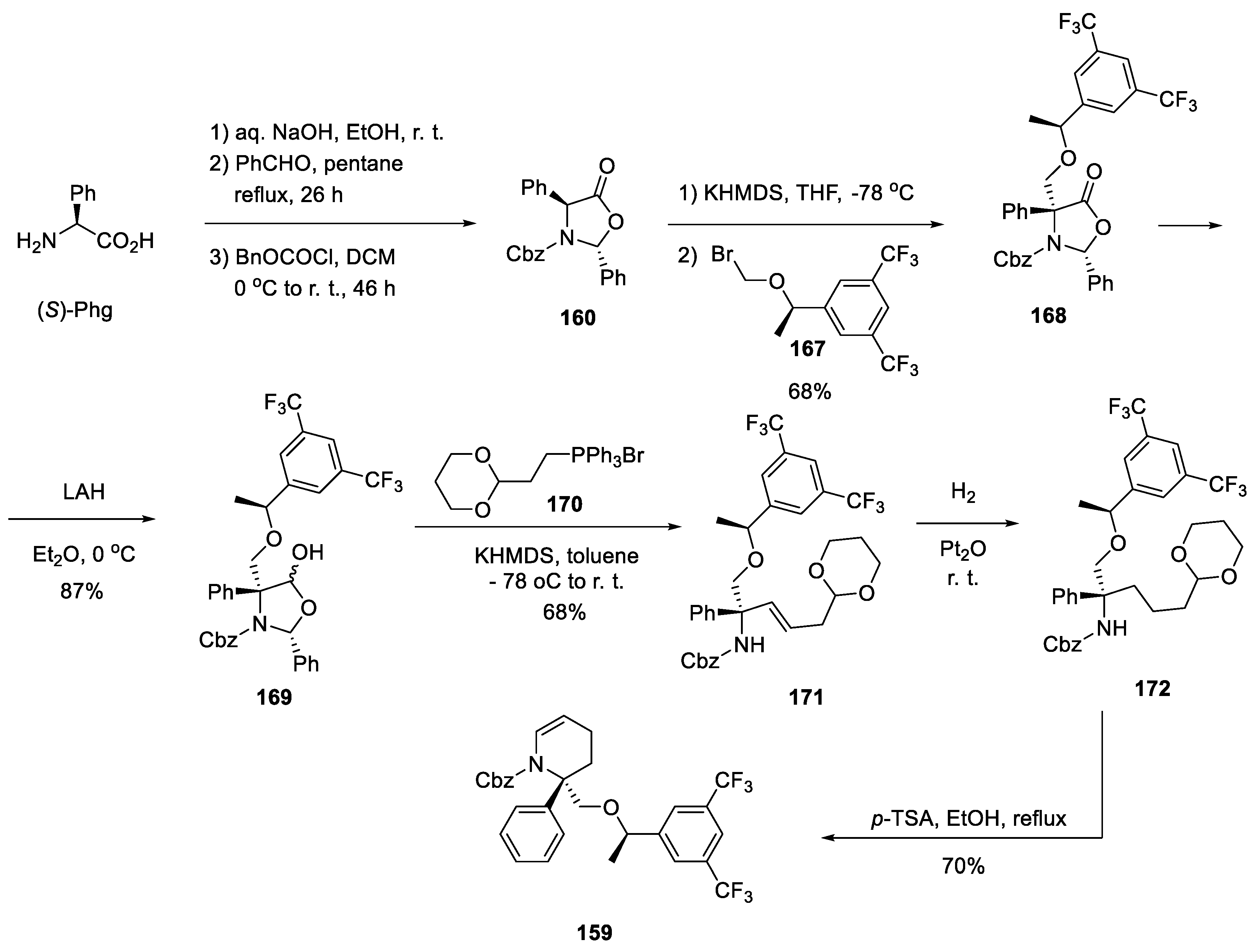
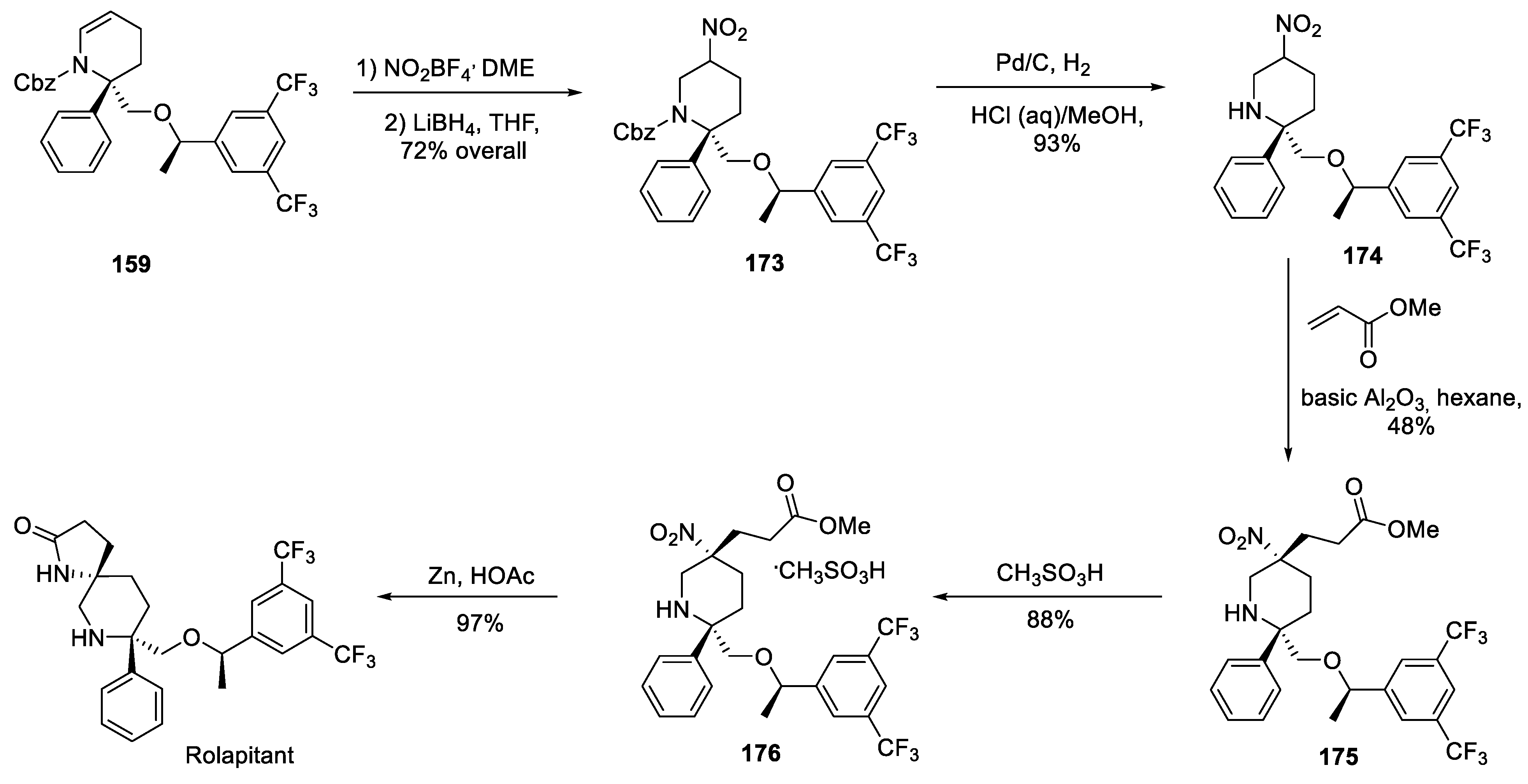
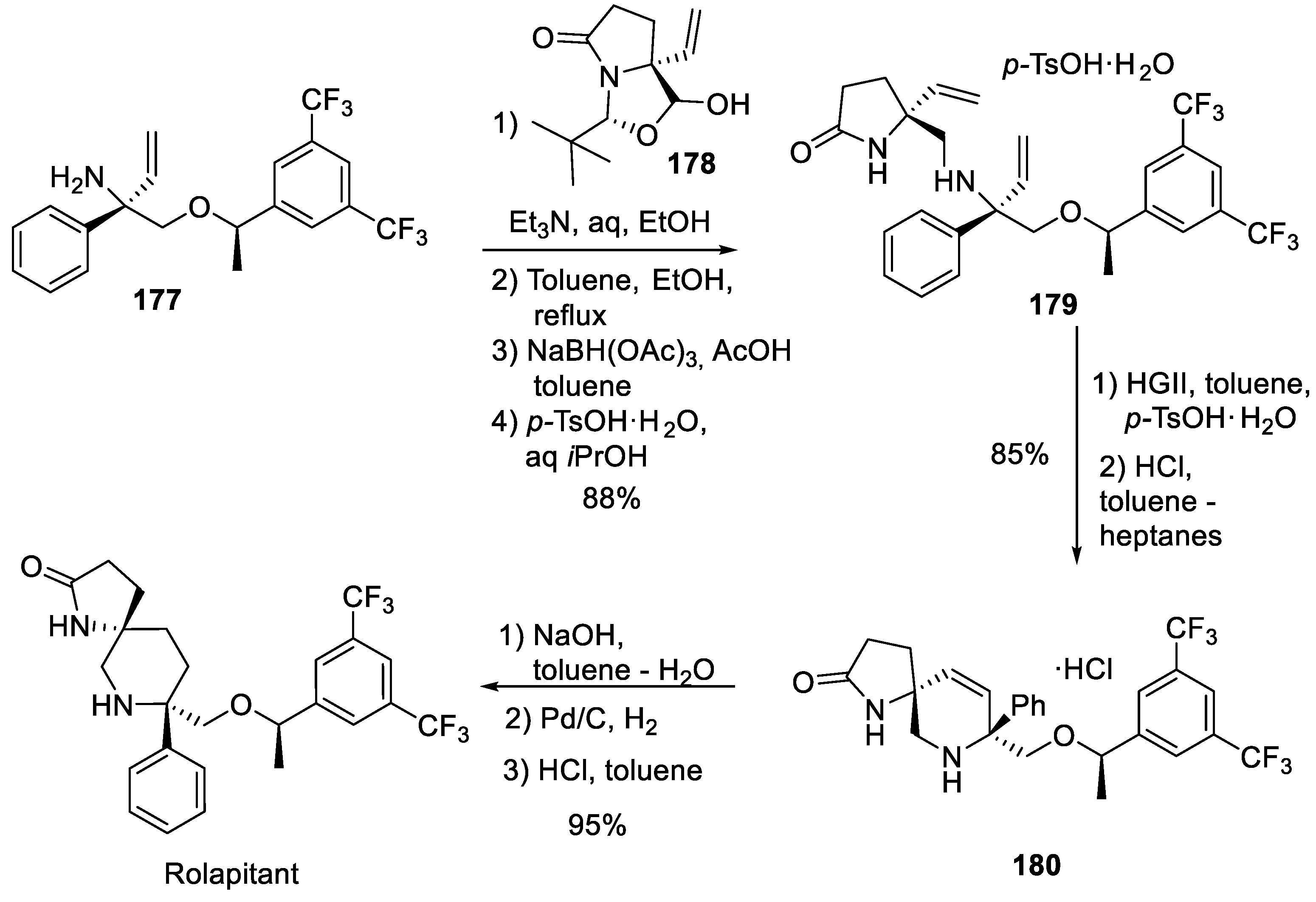
17. Rolapitant
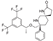
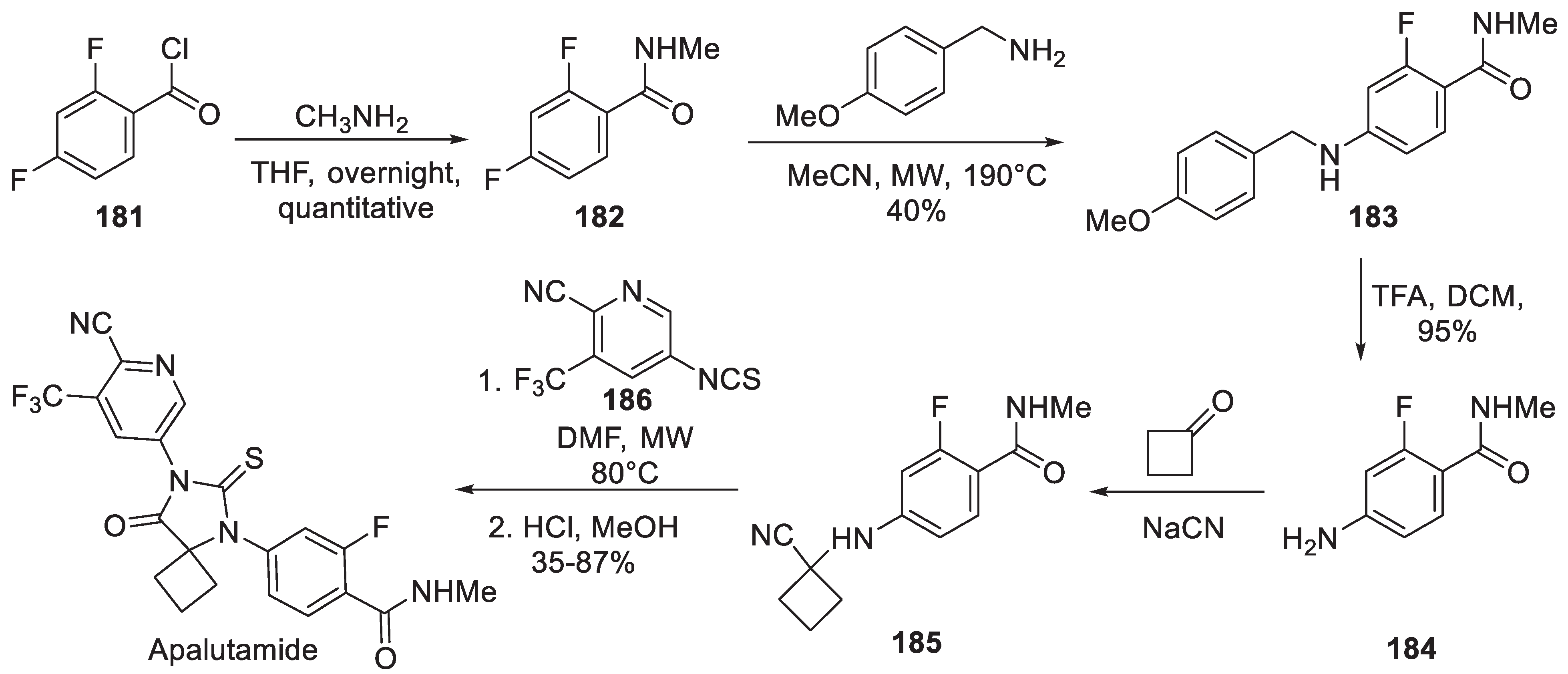
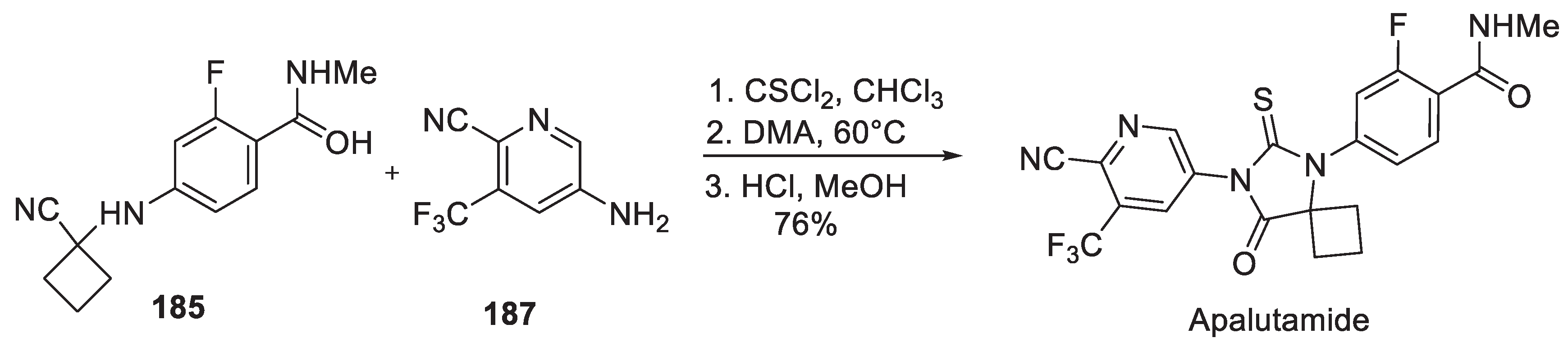
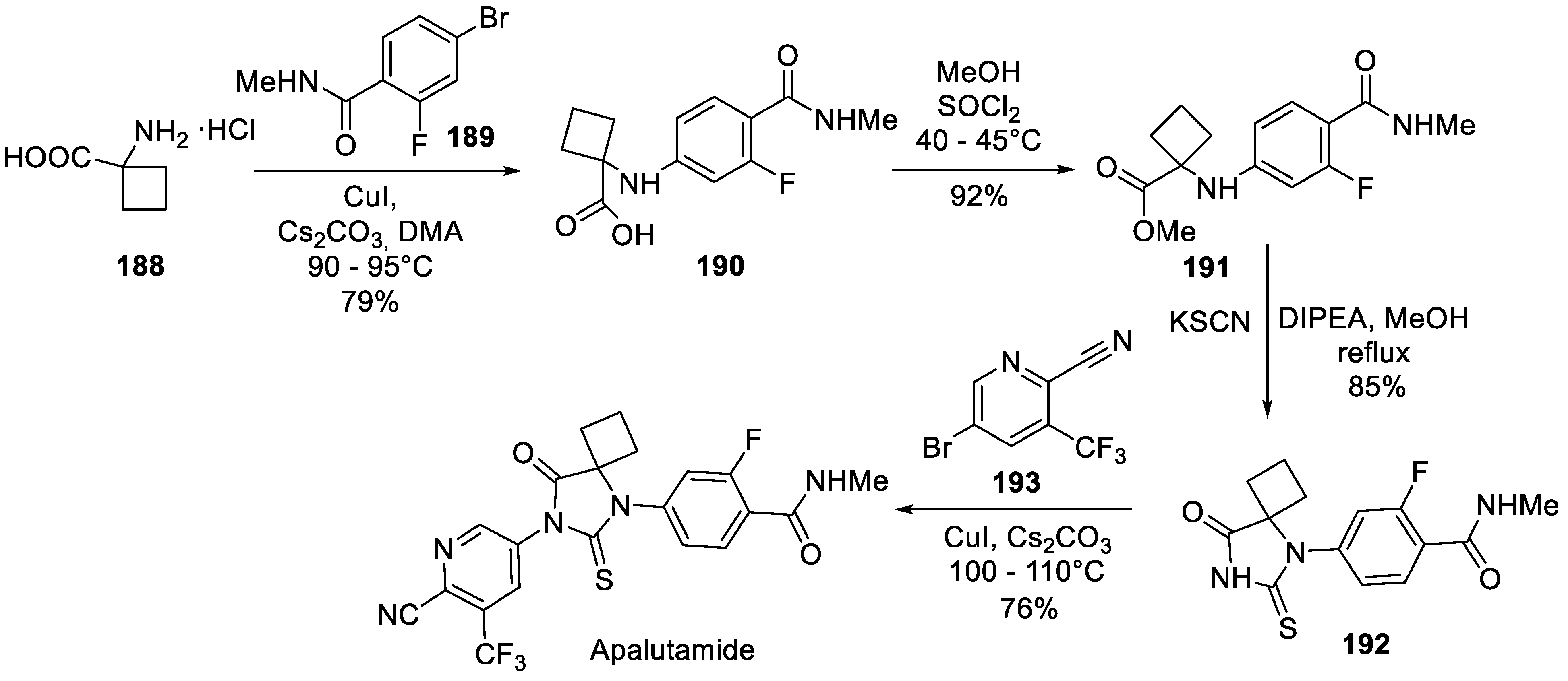
18. Apalutamide
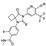
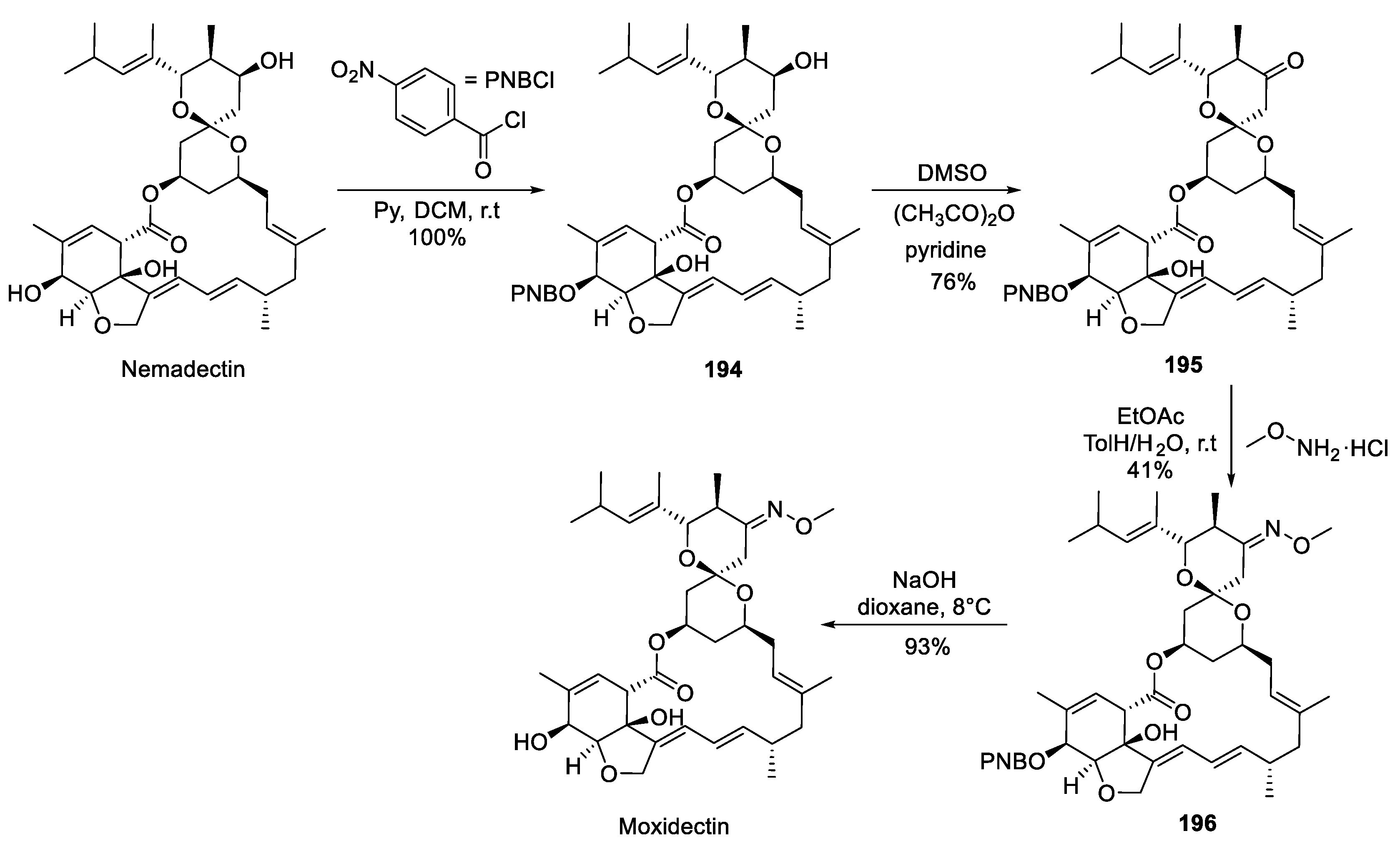
19. Moxidectin
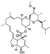
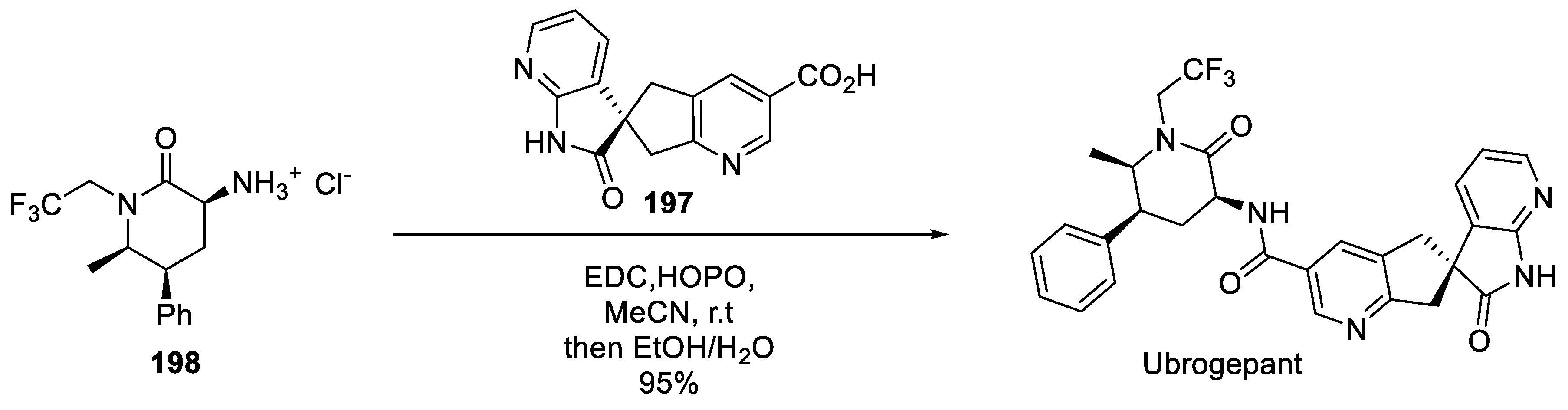
20. Ubrogepant
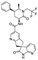
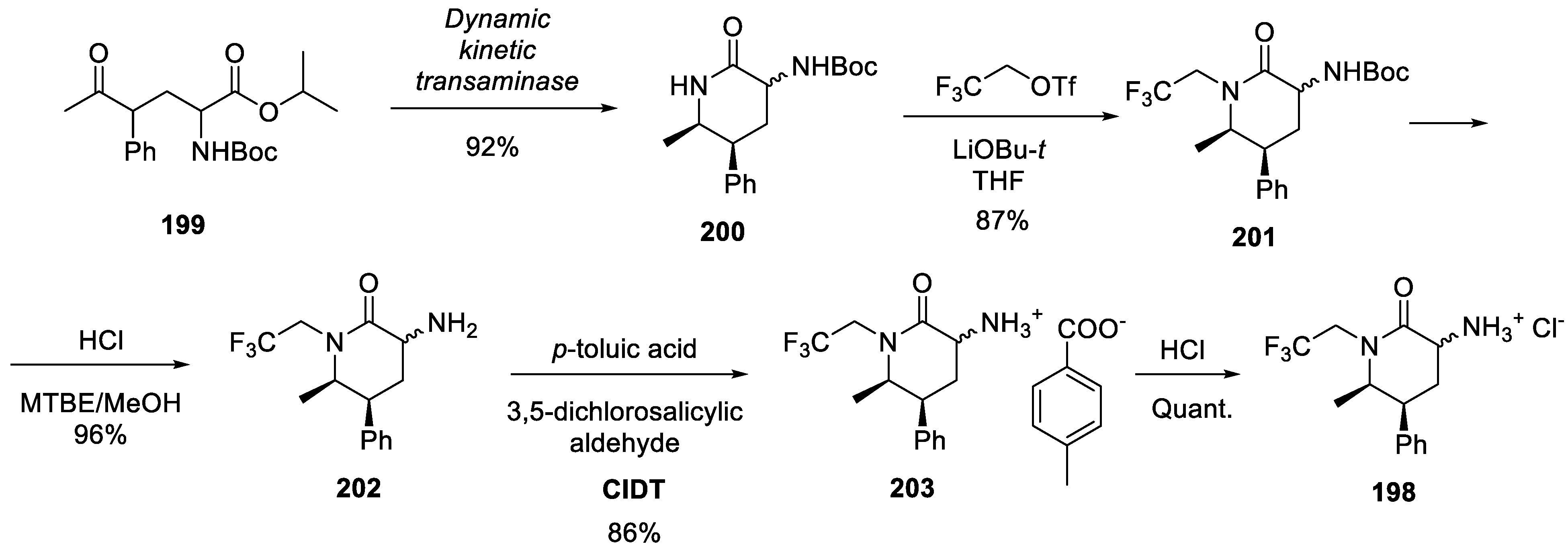
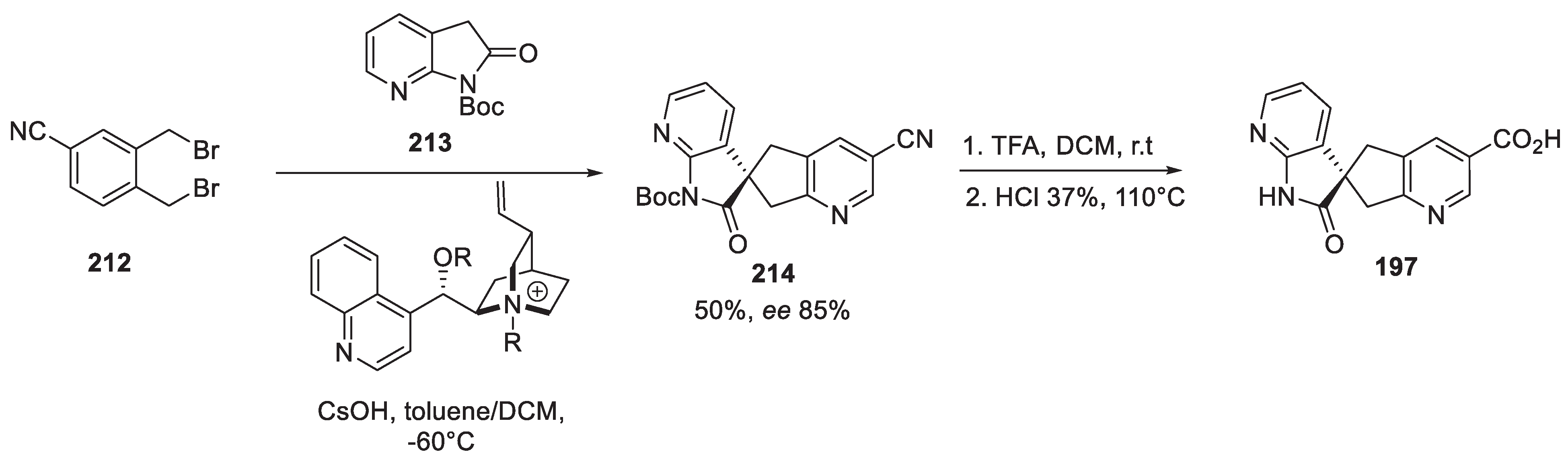
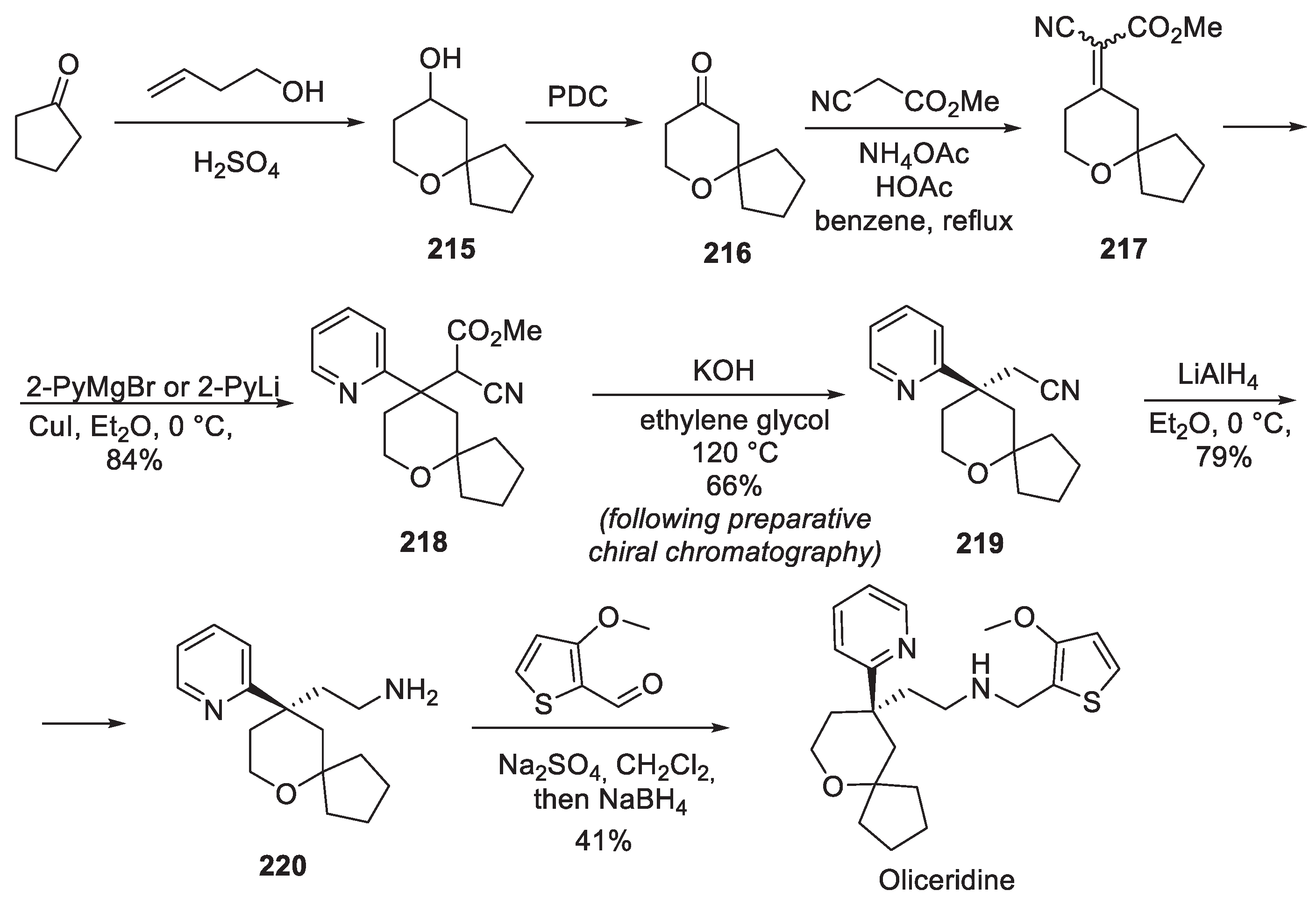
21. Oliceridine
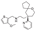
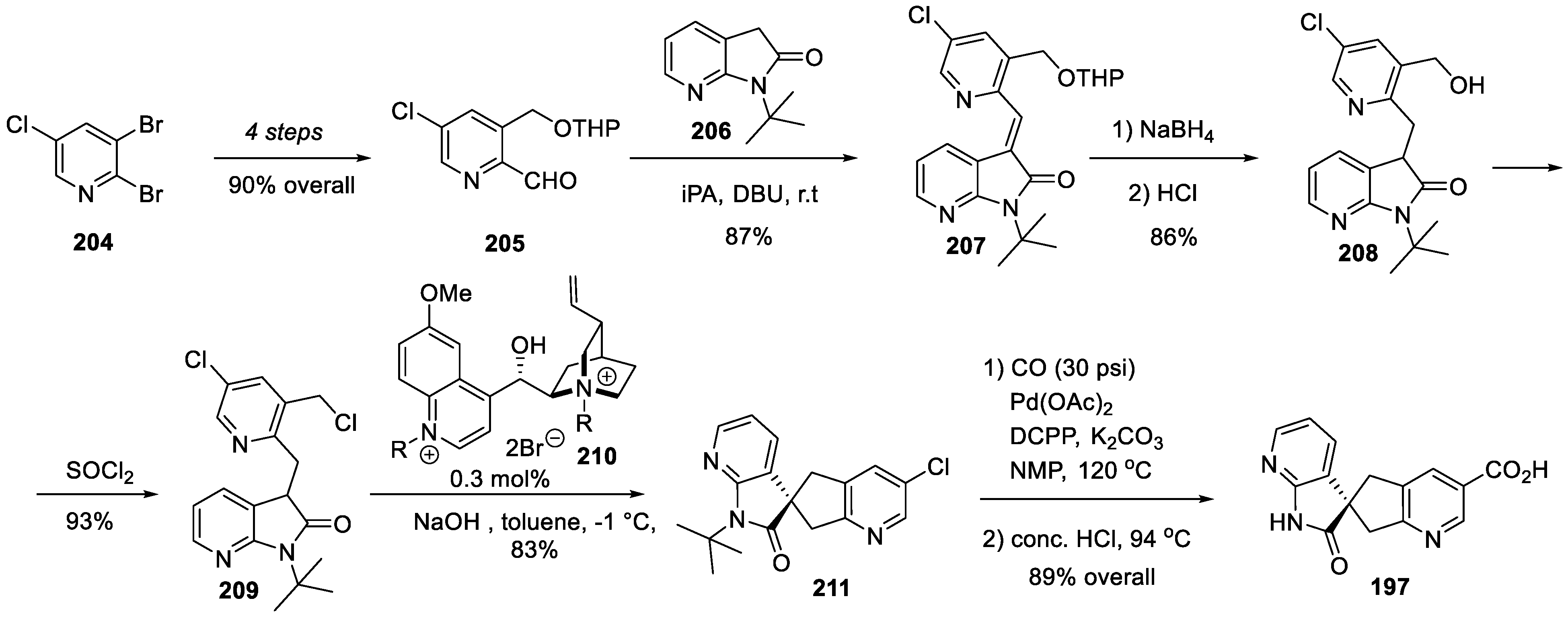
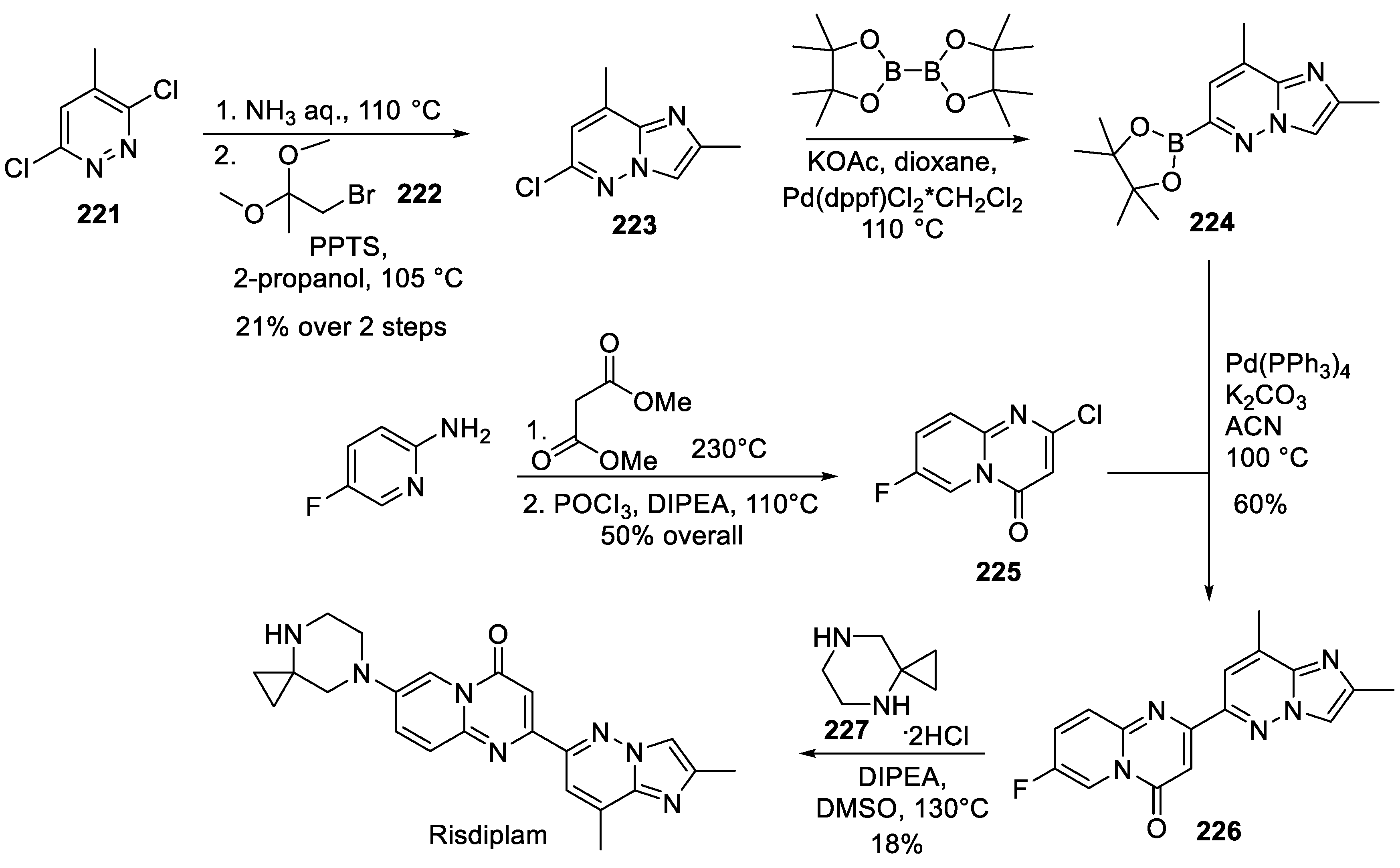
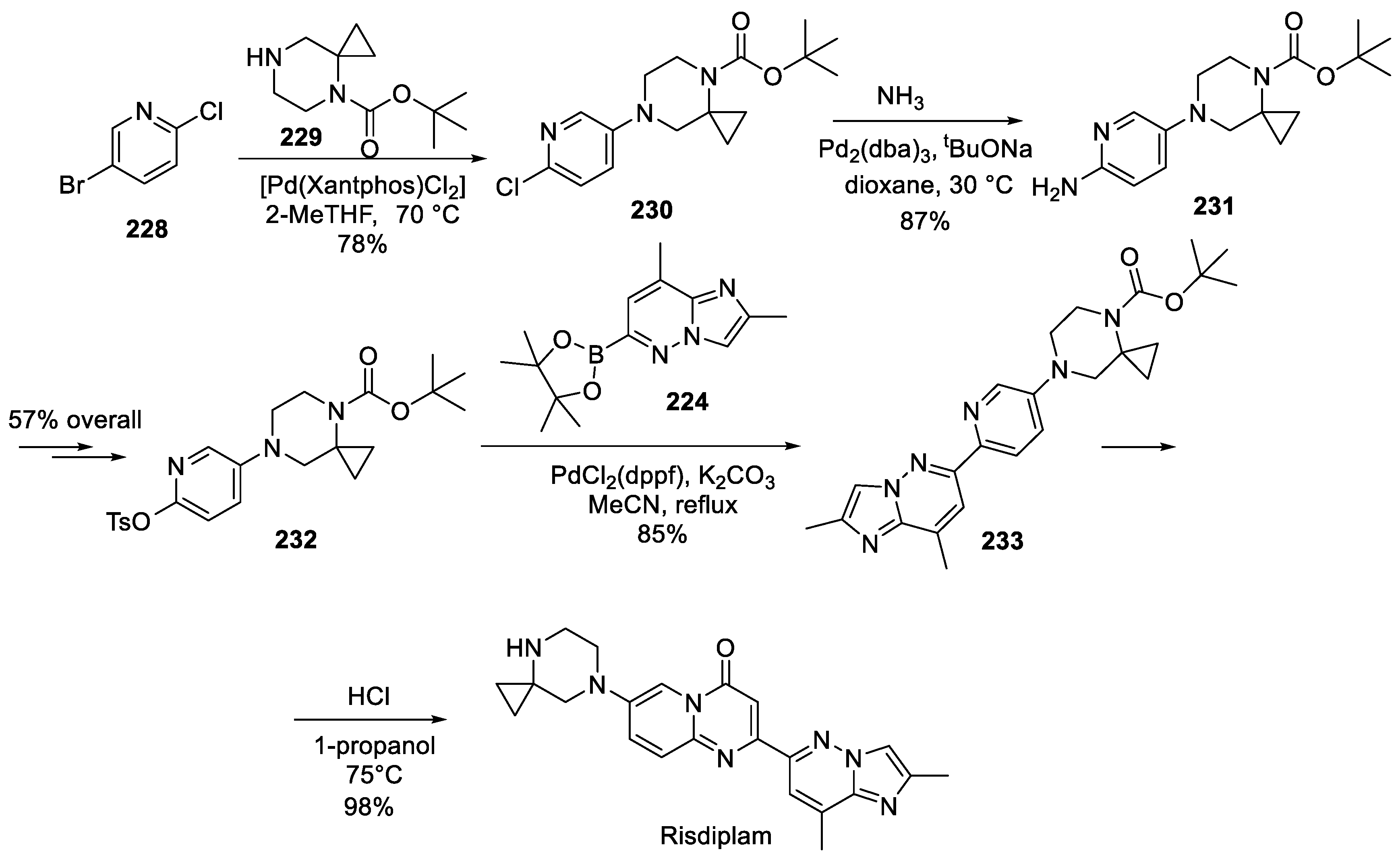
22. Risdiplam
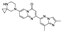
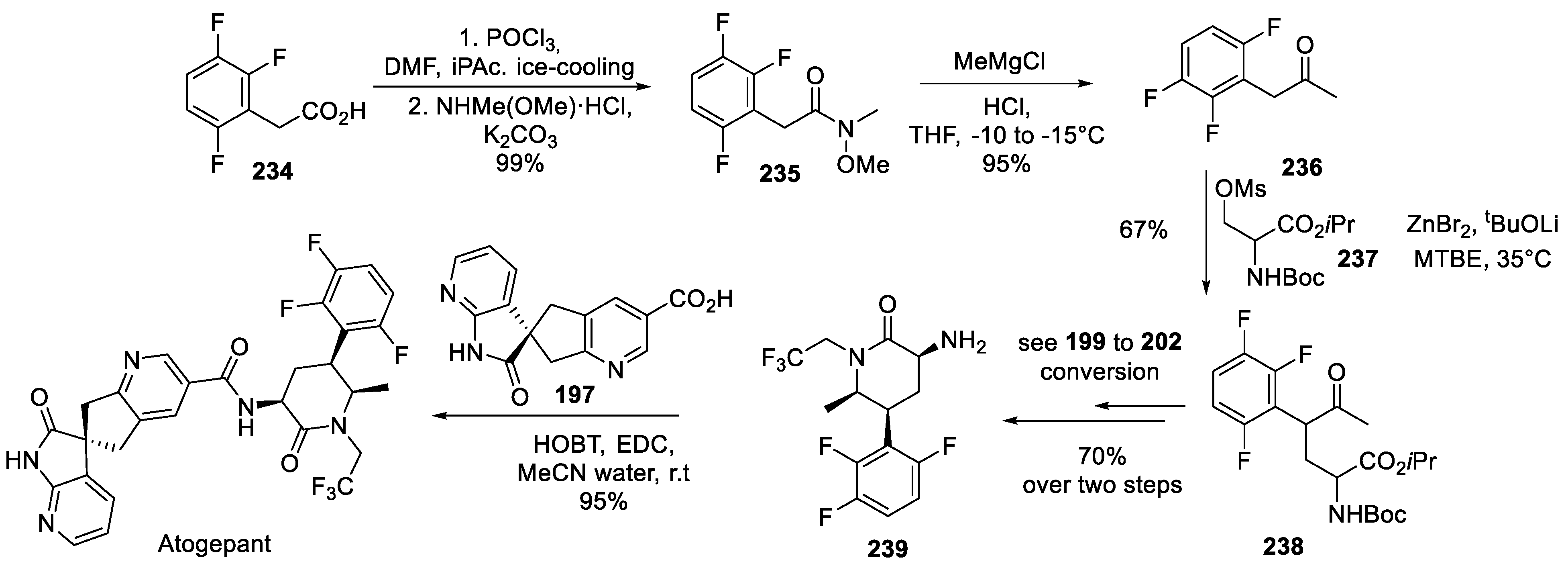
23. Atogepant
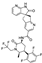
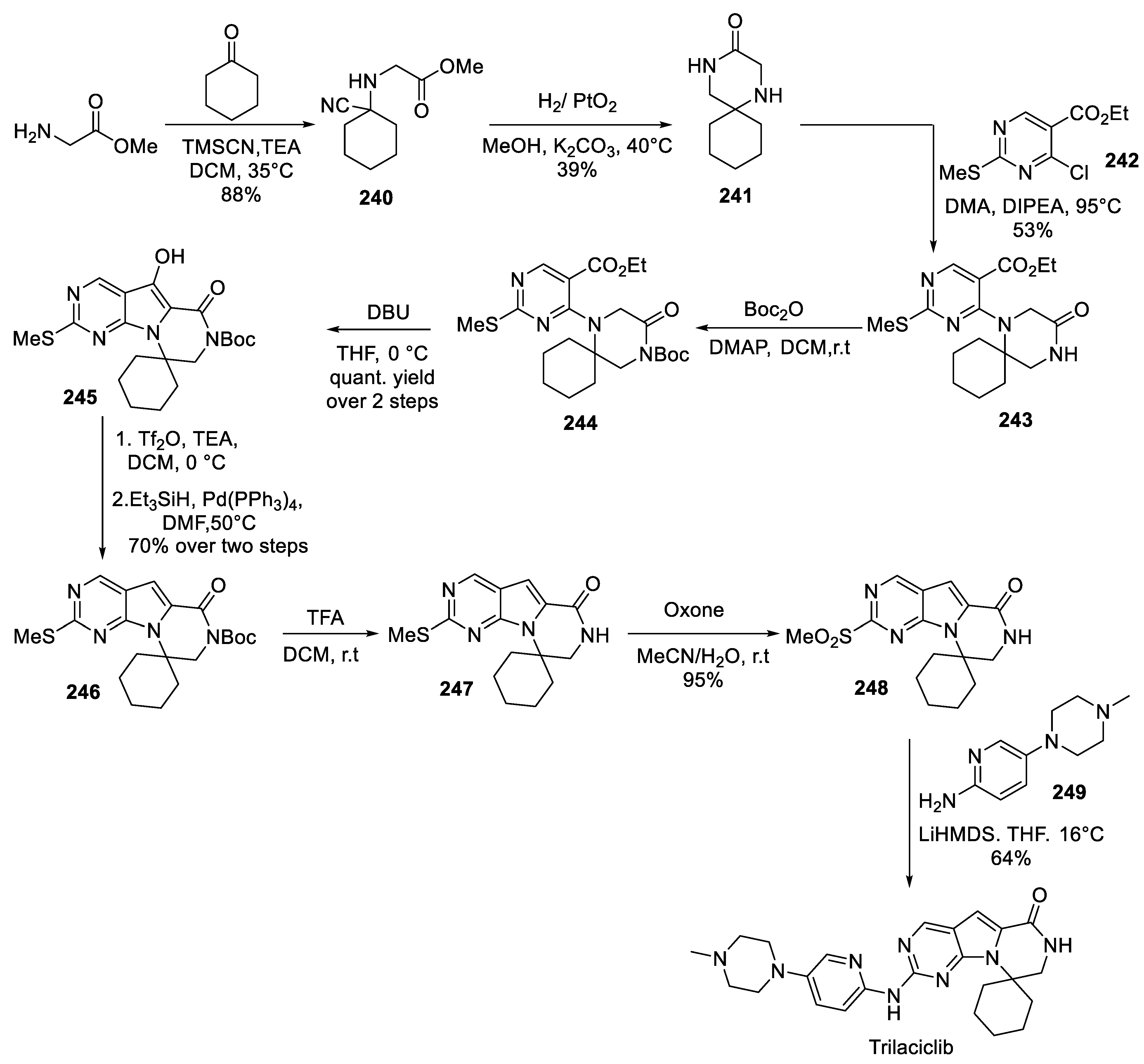
24. Trilaciclib
25. Conclusions and perspectives
Disclosure Statement
Funding
References
- Lovering, F.; Bikker, J.; Humblet, C. Escape from Flatland: Increasing Saturation as an Approach to Improving Clinical Success. J. Med. Chem. 2009, 52, 6752–6756. [Google Scholar] [CrossRef] [PubMed]
- Hiesinger, K.; Dar'in, D.; Proschak, E.; Krasavin, M. Spirocyclic Scaffolds in Medicinal Chemistry. J. Med. Chem. 2021, 64, 150–183. [Google Scholar] [CrossRef] [PubMed]
- Tomozane, H.; Takeuchi, Y.; Choshi, T.; Kishida, S.; Yamato, M. Syntheses and Antifungal Activities of dl-Griseofulvin and Its Congeners. I. Chem. Pharm. Bull. 1990, 38, 925–929. [Google Scholar] [CrossRef] [PubMed]
- Pirrung, M.C.; Brown, W.L.; Rege, S.; Laughton, P. Total Synthesis of (+)-Griseofulvin. J. Am. Chem. Soc. 1991, 113, 8561–8562. [Google Scholar] [CrossRef]
- Larik, F.A.; Saeed, A.; Shahzad, D.; Faisal, M.; El-Seedi, H.; Mehfooz, H.; Channar, P.A. Synthetic approaches towards the multi target drug spironolactone and its potent analogues/derivatives. Steroids 2017, 118, 76–92. [Google Scholar] [CrossRef]
- Cella, J.A.; Kagawa, C.M. Steroidal Lactones. J. Am. Chem. Soc. 1957, 79, 4808–4809. [Google Scholar] [CrossRef]
- Cella, J.A.; Brown, E.A.; Burtner, R.R. Steroidal Aldosterone Blockers. I. J. Org. Chem. 1959, 24, 743–748. [Google Scholar] [CrossRef]
- Cella, J.A.; Tweit, R.C. Steroidal Aldosterone Blockers. II. J. Org. Chem. 1959, 24, 1109–1110. [Google Scholar] [CrossRef]
- Anner, G.; Wehrli, H. Process for the preparation of steroid carbolactones. Swiss Patent CH617443A5 (publication 30.05.1980).
- Wuts, P.G.M.; Ritter, A.R. A Novel Synthesis of Spironolactone. An Application of the Hydroformylation Reaction. J. Org. Chem. 1989, 54, 5180–5182. [Google Scholar] [CrossRef]
- Yamane, D.; Tanaka, H.; Hirata, A.; Tamura, Y.; Takahashi, D.; Takahashi, Y.; Nagamitsu, T.; Ohtawa, M. One-Pot γ-Lactonization of Homopropargyl Alcohols via Intramolecular Ketene Trapping. Org. Lett. 2021, 23, 2831–2835. [Google Scholar] [CrossRef] [PubMed]
- Stepnicki, P.; Kondej, M.; Kaczor, A.A. Current Concepts and Treatments of Schizophrenia. Molecules 2018, 23, 2087. [Google Scholar] [CrossRef]
- Janssen, P.A.; Niemegeers, C.J.; Schellekens, K.H.; Lenaerts, F.M.; Verbruggen, F.J.; van Nueten, J.M.; Marsboom, R.H.; Hérin, V.V.; Schaper, W. K. The pharmacology of fluspirilene (R 6218), a potent, long-acting and injectable neuroleptic drug. Arzneimittelroschung 1970, 20, 1689–1698. [Google Scholar]
- Morciano, G.; Preti, D.; Pedriali, G.; Aquila, G.; Missiroli, S.; Fantinati, A.; Caroccia, N.; Pacifico, S.; Bonora, M.; Talarico, A.; et al. Discovery of Novel 1,3,8-Triazaspiro[4.5]decane Derivatives That Target the c Subunit of F1/FO-Adenosine Triphosphate (ATP) Synthase for the Treatment of Reperfusion Damage in Myocardial Infarction. J. Med. Chem. 2018, 61, 7131–7143. [Google Scholar] [CrossRef]
- Botteghi, C.; Marchetti, M.; Paganelli, S.; Persi-Paoli, F. Rhodium catalyzed hydroformylation of 1,1-bis(p-fluorophenyl)allyl or propargyl alcohol: A key step in the synthesis of Fluspirilen and Penfluridol. Tetrahedron 2001, 57, 1631–1637. [Google Scholar] [CrossRef]
- Chen, G.; Xia, H.; Cai, Y.; Ma, D.; Yuan, J.; Yuan, C. Synthesis and SAR study of diphenylbutylpiperidines as cell autophagy inducers. Bioorg. Med. Chem. Lett. 2011, 21, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Romanet, P.; Stewart, A.G. Value of Fenspiride in the Treatment of Inflammation Diseases in Otorhinolaryngology. Eur. Respir. Rev. 1991, 1, 105–110. [Google Scholar]
- Vandenberg, J.I.; Perozo, E.; Allen, T.W. Towards a structural view of drug binding to hERG K+ channels. Trends Pharmacol. Sci. 2017, 38, 899–907. [Google Scholar] [CrossRef]
- Wiśniowska, B.; Holbrook, M.; Pollard, C.; Polak, S. Utilization of mechanistic modelling and simulation to analyse fenspiride proarrhythmic potency - Role of physiological and other non-drug related parameters. J. Clin. Pharm. Ther. 2022, 47, 2152–2161. [Google Scholar] [CrossRef]
- Taccone, I. Method for the preparation of derivatives of spiro[4,5]decane and derivatives thus obtained. US Patent 4,028, 351.
- Somanathan, R.; Rivero, I.A.; Nunez, G.I.; Hellberg, L.H. Convenient synthesis of 1-oxa-3,8-diazaspiro[4,5]decan-2-ones. Synth. Commun. 1994, 24, 1483–1387. [Google Scholar] [CrossRef]
- Pramanik, C.; Bapat, K.; Patil, P.; Kotharkar, S.; More, Y.; Gotrane, D.; Chaskar, Mahajan, U. ; Tripathy, N.K. Nitro-Aldol Approach for Commercial Manufacturing of Fenspiride Hydrochloride. Org. Proc. Res. Dev. 2019, 23, 1252–1256. [Google Scholar] [CrossRef]
- Woodford, R.; Barry, B.W.; Frosch, P.J. Activity and Bioavailability of Amcinonide and Triamcinolone Acetonide in Experimental and Proprietary Topical Preparations. Curr. Ther. Res. 1977, 21, 877–886. [Google Scholar]
- Shultz, W.; Sieger, G.M.; Krieger, C. Novel triamcinolone derivative. Brittish patent GB1442925 (published 14.07.1976).
- Trimathi, V.; Kumar, R.; Bhuwania, R.; Bhuwania, B.K. Novel process for preparation of corticosteroids. PCT Int. Appl. WO2018037423 (published 01.03.2018).
- Finnerty, F.A.; Brogden, R.N. Guanadrel. A review of its pharmacodynamic and pharmacokinetic properties and therapeutic use in hypertension. Drugs 1985, 30, 22–31. [Google Scholar] [CrossRef]
- Vardanyan, R.S.; Hruby, V.J. 12 - Adrenoblocking Drugs, In Synthesis of Essential Drugs; R.S. Vardanyan, V.J. Hruby, Eds.; Elsevier, 2006, pp. 161–177.
- Hardie, W.R.; Aaron, J.E. 1,3-Dioxolan-4-yl-alkyl guanidines. US Patent 3,547, 951.
- Baker, T.J.; Goodman, M. Synthesis of Biologically Important Guanidine-Containing Molecules Using Triflyl-Diurethane Protected Guanidines. Synthesis 1999, 1423–1426. [Google Scholar] [CrossRef]
- Taylor, D.P.; Moon, S. L. Buspirone and related compounds as alternative anxiolytics. Neuropeptides 1991, 19, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Rayburn, J. N-(Heteroarcyclic)piperazinylalkylazaspiroalkanediones. US Patent 3,717, 634.
- Wu, Y.H.; Rayburn, J.W. N-[(4-Pyridyl-piperazino)alkyl]azaspiroalkanediones. US Patent 3,907, 801.
- Wu, Y.H.; Rayburn, J.W. Tranquilizer process employing N-(heteroarcyclic)piperazinylalkylazaspiroalkanediones. US Patent 3,976, 776.
- Mou, J.; Zong, Z.-M.; Wei, X.-Y. Facile synthesis of anxyolytic buspirone. Org. Prep. Proc. Int. 2008, 40, 391–394. [Google Scholar] [CrossRef]
- Labes, R.; Mateos, C.; Battilocchio, C.; Chen, Y.; Dingwall, P.; Cumming, G.R.; Rincon, J.A.; Nieves-Remacha, M.J.; Ley, S.V. Fast continous alcohol amination employing a hydrogen borrowing protocol. Green Chem. 2019, 21, 59–63. [Google Scholar] [CrossRef]
- Chandrashekhar, V.G.; Baumann, W.; Beller, M.; Jagadeesh, R.V. Nickel-catalyzed hydrogenative coupling of nitriles and amines for general amine synthesis. Science 2022, 376, 1433–1441. [Google Scholar] [CrossRef] [PubMed]
- Campbell, W.C. Ivermectin as an antiparasitic agent for use in humans. Ann. Rev. Microbiol. 1991, 45, 445–474. [Google Scholar] [CrossRef] [PubMed]
- Crump, A. Ivermectin: Enigmatic multifaceted 'wonder' drug continues to surprise and exceed expectations. J. Antibiotics 2017, 70, 495–505. [Google Scholar] [CrossRef]
- Martin, R.J.; Robertson, A.P.; Choudhary, S. Ivermectin: An Anthelmintic, an Insecticide, and Much More. Trends Parasitol. 2021, 37, 48–64. [Google Scholar] [CrossRef]
- Molyneux, D.H.; Ward, S.A. Reflections on the Nobel Prize for Medicine 2015 - The Public Health Legacy and Impact of Avermectin and Artemisinin. Trends Parasitol. 2015, 31, 605–607. [Google Scholar] [CrossRef] [PubMed]
- Arlt, D.; Bonse, G.; Reisewitz, F. Method for the preparation of Ivermectin. Eur. Patent EP072997 (published 04.09.1996).
- Yamashita, S.; Hayashi, D.; Nakano, A.; Hayashi, Y.; Hirama, M. Total synthesis of avermectin B1a revisited. J. Antibiotics 2016, 69, 31–50. [Google Scholar] [CrossRef] [PubMed]
- Rothstein, D.M. Rifamycins, Alone and in Combination. Cold Spring Harb. Perpect. Med. 2016, 6, a027011. [Google Scholar] [CrossRef] [PubMed]
- Nasiri, M.J.; Ebrahimi, G.; Arefzadeh, S.; Zamani, S.; Nikpor, Z.; Mirsaeidi, M. Antibiotic therapy success rate in pulmonary Mycobacterium avium complex: A systematic review and meta-analysis. Expert Rev. Anti. Infect. Ther. 2020, 18, 263–273. [Google Scholar] [CrossRef]
- Pinheiro, M.; Nunes, C.; Caio, J.M.; Lucio, M.; Brezesinski, Reis, S. The Influence of Rifabutin on Human and Bacterial Membrane Models: Implications for Its Mechanism of Action. J. Phys. Chem. B 2013, 117, 6187–6193. [Google Scholar] [CrossRef]
- Marsili, L.; Pasqualucci, C.; Vigevani, A.; Gioia, B.; Schioppacassi, G.; Oronzo, G. New rifamycins modified at positions 3 and 4. Synthesis, structure and biological evaluation. J. Antiobiot. 1981, 34, 1033–1038. [Google Scholar] [CrossRef] [PubMed]
- Marsili, L.; Rosetti, V.; Pasqualucci, C. Method of preparing derivatives of rifamycin S. US Patent 4,007, 169.
- Noble, S.; Sorkin, E.M. Spirapril: A Preliminary Review of its Pharmacology and Therapeutic Efficacy in the Treatment of Hypertension. Drugs 1995, 49, 750–766. [Google Scholar] [CrossRef]
- Hossein-Nia, M.; Surve, A.H.; Weglein, R.; Gerbeau, C.; Holt, D.W. Radioimmunoassays for spirapril and its active metabolite spiraprilate: Performance and application. Ther. Drug Monit. 1992, 14, 234–242. [Google Scholar] [CrossRef]
- Gold, E.H.; Neustadt, B.R.; Smith, E.M. 7-Carboxyalkylaminoacyl-1,4-dithia-7-azaspiro[4.4]-nonane-8-carboxylic acids. US Patent 4,470, 972.
- Smith, E.M.; Swiss, G.F.; Neustadt, B.R.; McNamara, P.; Gold, E.H.; Sybertz, E.J.; Baum, T. Angiotensin Converting Enzyme Inhibitors: Spirapril and Related Compounds. J. Med. Chem. 1989, 32, 1600–1606. [Google Scholar] [CrossRef]
- Krapcho, J.; Turk, C.; Cushman, D.W.; Powell, J.R.; DeForrest, J.M.; Spitzmiller, E.R.; Karanewsky, D.S.; Duggan, M.; Rovnyak, G.; Schwartz, J.; et al. Angiotensin-Converting Enzyme Inhibitors. Mercaptan, Carboxyalkyl Dipeptide, and Phosphinic Acid Inhibitors Incorporating 4-Substituted Prolines. J. Med. Chem. 1988, 31, 1148–1160. [Google Scholar] [CrossRef]
- Muszalska, I.; Sobczak, Dolhan, A. ; Jelinska, A. Analysis of Sartans: A review. J. Pharm. Sci. 2014, 103, 2–28. [Google Scholar] [CrossRef] [PubMed]
- Siragy, H. Angiotensin II receptor blockers: Review of the binding characteristics. Am. J. Cardiol. 1999, 84, 3S–8S. [Google Scholar] [CrossRef] [PubMed]
- Bernhart, C.A.; Perreaut, P.M.; Ferrari, B.P.; Muneaux, Y.A.; Assens, J.-L. A.; Clement, J.; Haudricourt, F.; Muneaux, C.F.; Taillades, J.E.; Vignal, M.-A.; et al. A New Series of Imidazolones: Highly Specific and Potent Nonpeptide AT1Angiotensin II Receptor Antagonists. J. Med. Chem. 1993, 36, 3371–3380. [Google Scholar] [CrossRef] [PubMed]
- DiPoto, M.C.; Wu, J. Synthesis of 2-Aminoimidazolones and Imidazolones by (3 + 2) Annulation of Azaoxyallyl Cations. Org. Lett. 2018, 20, 499–501. [Google Scholar] [CrossRef] [PubMed]
- Nisnevich, G.; Rukhman, I.; Pertsikov, B.; Kaftanov, J.; Dolitzky, B.-Z. Novel synthesis of irbesartan. US Patent Appl. US2004192713 (published 30.09.2004).
- Reddy, R.B.; Suhakar, S.; Sridhar, C.; Rao, S.N.; Venkataraman, S.; Reddy, P.P. Satyanarayana, B. Process for preparing irbesartan. PCT Int. Appl. WO2005113518 (published 01.12.2005).
- Ye, P.; Sargent, K.; Stewart, E.; Liu, J.-F.; Yohannes, D.; Yu, L. Novel and Expeditious Microwave-Assisted Three-Component Reactions for the Synthesis of Spiroimidazolin-4-ones. J. Org. Chem. 2006, 71, 3137–3140. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Guo, Y.; Niu, H.; Wang, J.; Li, J.; Li, C.; Qiao, R. Development of a New Synthetic Route of the Key Intermediate of Irbesartan. Org. Proc. Res. Dev. 2022, 26, 2438–2446. [Google Scholar] [CrossRef]
- Yasuda, H.; Niki, H. Review of the Pharmacological Properties and Clinical Usefulness of Muscarinic Agonists for Xerostomia in Patients with Sjögren's Syndrome. Clin. Drug Investig. 2002, 22, 67–73. [Google Scholar] [CrossRef]
- Ono, M.; Takamura, E.; Shinozaki, K.; Tsumura, T.; Hamano, T.; Yagi, Y.; Tsubota, K. Therapeutic effect of cevimeline on dry eye in patients with Sjögren's syndrome: A randomized, double-blind clinical study. Am. J. Ophthalmol. 2004, 138, 6–17. [Google Scholar] [CrossRef]
- Oleksak, P.; Novotny, M.; Patocka, J.; Nepovimova, E.; Hort, J.; Pavlik, J.; Klimova, B.; Valis, M.; Kuca, K. Neuropharmacology of Cevimeline and Muscarinic Drugs—Focus on Cognition and Neurodegeneration. Int. J. Mol. Sci. 2021, 22, 8908. [Google Scholar] [CrossRef]
- Fisher, A.; Karton, I.; Heldman, E.; Levy, A.; Grunfeld, Y. Derivatives of quinuclidine. US Patent 4,855, 290.
- Bratovanov, S.; Bejan, E.; Wang, Z.-X.; Horne, S. Process for the preparation of 2-methylspiro(1,3-oxathiolane-5,3')quiniclidine. US Patent Appl. US2008249312 (published 09.10.2008).
- Pramanik, C.; Patil, P.; Kotharkar, S.; Tripathy, N.K.; Gurjar, M.K. A new route to cevimeline. Tetrahedron Lett. 2013, 54, 3043–3045. [Google Scholar] [CrossRef]
- Bös, M.; Canesso, R. EPC-synthesis of (S)-3-hydroxy-3-mercaptomethylquinucudine, a chiral building block for the synthesis of the muscarinic agonist AF1028. Heterocycles 1994, 38, 1889–1896. [Google Scholar] [CrossRef]
- Pitt, B.; Remme, W.; Zannad, F.; Neaton, J.; Martinez, F.; Roniker, B.; Bittman, R.; Hurley, S.; Kleiman, J.; Gatlin, M. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. N. Engl. J. Med. 2003, 348, 1309–1321. [Google Scholar] [CrossRef] [PubMed]
- Menard, J. The 45-year story of the development of an anti-aldosterone more specific than spironolactone. Mol. Cell. Endocrinol. 2004, 217, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Ng, J.S.; Liu, C.; Anderson, D.K.; Lawson, J.P.; Wieczorek, J.; Kunda, S.A.; Letendre, L.J.; Pozzo, M.J.; Sing, Y.-L. L.; Wang, P.T.; et al. Processes for preparation of 9,11-epoxy steroids and intermediates useful therein. PCT Int. Appl. WO9825948 (published 18.06.1998).
- Grob, J.; Boillaz, M.; Schmidlin, J.; Wehrli, H.; Wieland, P.; Fuhrer, H.; Rihs, G.; Joss, U.; de Gasparo, M.; Haenni, H.; et al. Steroidal, Aldosterone Antagonists: Increased Selectivity of 9,11-Epoxy Derivatives. Helv. Chim. Acta 1997, 80, 566–585. [Google Scholar] [CrossRef]
- Pearlman, B.A.; Padilla, A.G.; Hach, J.T.; Havens, J.L.; Pillai, M.D. A New Approach to the Furan Degradation Problem Involving Ozonolysis of the trans-Enedione and Its Use in a Cost-Effective Synthesis of Eplerenone. Org. Lett. 2006, 8, 2111–2113. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Chen, H.; Feng, H.; Li, Y. Stereoselective construction of a steroid 5a,7a-oxymethylene derivative and its use in the synthesis of eplerenone. Steroids 2011, 76, 56–59. [Google Scholar] [CrossRef]
- Krattenmacher R: Drospirenone: Pharmacology and pharmacokinetics of a unique progestogen. Contraception 2000, 62, 29–38. [CrossRef] [PubMed]
- Muhn P, Fuhrmann U, Fritzemeier KH, Krattenmacher R, Schillinger E: Drospirenone: A novel progestogen with antimineralocorticoid and antiandrogenic activity. Ann. N. Y. Acad. Sci. 1995, 761, 311–335. [CrossRef]
- Bittler, D.; Hofmeister, H.; Laurent, H.; Nickisch, K.; Nickolson, R.; Petzoldt, K.; Wiechert, R. Synthesis of Spirorenone- A Novel Highly Active Aldosterone Antagonist. Angew. Chem. Int. Ed. Engl. 1982, 21, 696–697. [Google Scholar] [CrossRef]
- Nickisch, K.; Acosta, K.; Santhamma, B. Methods for the preparation of drospirenone. US Patent Appl. US2010261896 (published 14.10.2010).
- Bandini, M.; Contento, M.; Garelli, A.; Monari, M.; Tolomelli, A.; Umani-Ronchi, A.; Andriolo, E.; Montorsi, M. A Nonclassical Stereoselective Semi-Synthesis of Drospirenone via Cross-Metathesis Reaction. Synthesis 2008, 3801–3904. [Google Scholar] [CrossRef]
- Scott, L.J. Ledipasvir/Sofosbuvir: A Review in Chronic Hepatitis C. Drugs 2018, 78, 245–256. [Google Scholar] [CrossRef] [PubMed]
- Scott, R.W.; Vitale, J.P.; Matthews, K.S.; Teresk, M.G.; Formella, A.; Evans, J.W. Synthesis of antiviral compound. US Patent Appl. US20130324740 (published 05.12.2013).
- Xu, L.; Liu, F.-Y.; Zhang, Q.; Chang, W.-J.; Liu, Z.-L.; Lv, Y.; Yu, H.-Z.; Xu, J.; Dai, J.-J.; Xu, H.-J. The amine-catalysed Suzuki–Miyaura-type coupling of aryl halides and arylboronic acids. Nat. Catal. 2021, 4, 71–78. [Google Scholar] [CrossRef]
- Heo, Y.-A.; Deeks, E.D. Rolapitant: A Review in Chemotherapy-Induced Nausea and Vomiting. Drugs 2017, 77, 1687–1694. [Google Scholar] [CrossRef] [PubMed]
- Paliwal, S.; Reichard, G.A.; Wang, C.; Xiao, D.; Tsui, H.-C.; Shih, N.-Y.; Arredondo, J.D.; Wrobleski, M.L.; Palani, A. US Patent 7,049,320 (published 23. 05. 2006.
- O'Donnell, M.; Fang, Z.; Ma, X.; Huffman, J.C. New methodology for the synthesis of -dialkylamino acids using the "self-regeneration of stereocenters" method: -ethyl--phenylglycine. Heterocycles 1997, 46, 617–630. [Google Scholar] [CrossRef]
- Mergelsberg, I.; Scherer, D.H.; Huttenloch, M.E.; Tsui, H.-C.; Paliwal, S.; Shih, N.-Y. Process and intermediates for the synthesis of 8-[{1-(3,5-bis-(thrifluoromethyl)phenyl)ethoxy}methyl]-8-phenyl-1,7-diazadpiro[4,5]decan-2-one compounds. US Patent 8,552, 191.
- Wu, G.G.; Werne, G.; Fu, X.; Orr, R.K.; Chen, F.X.; Cui, J.; Sprague, V.M.; Zhang, F.; Xie, J.; Zeng, L.; et al. Process and intermediates for the synthesis of 8-[{1-(3,5-bis-(trifluoromethyl)phenyl)-ethoxy}-methyl]-8-phenyl-1,7-diaza-spiro[4.5]decan-2-one compounds. US Patent 9,249, 144.
- Hughes, D.L. Review of Synthetic Routes and Crystalline Forms of the Antiandrogen Oncology Drugs Enzalutamide, Apalutamide, and Darolutamide. Org. Proc. Res. Dev. 2020, 24, 347–362. [Google Scholar] [CrossRef]
- Jung, M.E.; Sawyers, C.L.; Ouk, S.; Tran, C.; Wongvipat, J. Androgen Receptor Modulator for the Treatment of Prostate Cancer and Androgen Receptor-Associated Diseases. PCT Int. Patent Appl. WO 2007126765 (published 08.11.2007).
- Ouerfelli, O.; Dilhas, A.; Yang, G.; Zhao, H. Synthesis of Thiohydantoins. PCT Int. Patent Appl. WO 2008119015 (published 02.10.2008).
- Zheng, X.; Zhang, Y. Synthesis method of apalutamide and its intermediates. Chinese Patent CN108383749 (published 10.08.2018).
- Takiguchi, Y.; Mishima, H.; Okuda, M.; Terao, M. Milbemycins, a new family of macrolide antibiotics: Fermentation, isolation and physico-chemical properties. J. Antibiot. 1980, 23, 1120–1127. [Google Scholar] [CrossRef]
- Prichard, R.K.; Geary, T.G. Perspectives on the utility of moxidectin for the control of parasitic nematodes in the face of developing anthelmintic resistance. Int. J. Parasitol. Drugs Drug Resist. 2019, 10, 69–83. [Google Scholar] [CrossRef]
- Milton, P.; Hamley, J.I.D.; Walker, M.; Basáñez, M.G. Moxidectin: An oral treatment for human onchocerciasis. Exp. Rev. Anti-infect. Ther. 2020, 18, 1067–1081. [Google Scholar] [CrossRef]
- Takiguchi, Y.; Ono, M.; Muramatsu, S.; Ide, J.; Mishima, H.; Terao, M. Milbemycins, a new family of macrolide antibiotics. Fermentation, isolation and physico-chemical properties of milbemycins D, E, F, G, and H. J. Antibiot. 1983, 36, 502–508. [Google Scholar] [CrossRef]
- Maulding, D.R.; Kumar, A. Process for the preparation of 23-(C1-C6 alkyloxime)-LL-F28249 compounds. US Patent 4,988, 824.
- Russell, F.A.; King, R.; Smillie, S.J.; Kodji, X.; Brain, S. D. Calcitonin gene-related peptide: Physiology and pathophysiology". Physiol. Rev. 2014, 94, 1099–1142. [Google Scholar] [CrossRef]
- Moore, E.; Fraley, M.E.; Bell, I.M.; Burgey, C.S.; White, R.B.; Li, C.C.; Rega, C.P.; Danziger, A.; Michener, M.S.; Hostetler, E.; et al. Characterization of Ubrogepant: A Potent and Selective Antagonist of the Human Calcitonin Gene‒Related Peptide Receptor. J. Pharmacol. Exp. Ther. 2020, 373, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, N.; Cleator, E.; Kosjek, B.; Yin, J.; Xiang, B.; Chen, F.; Kuo, S.-C.; Belyk, K.; Mullens, P.R.; Goodyear, A.; et al. Practical Asymmetric Synthesis of a Calcitonin Gene-Related Peptide (CGRP) Receptor Antagonist Ubrogepant. Org. Proc. Res. Dev. 2017, 21, 1851–1858. [Google Scholar] [CrossRef]
- Gao, M.; Li, Y.; Li, X.; Hu, L. Phase-Transfer-Catalyzed Asymmetric Annulations of Alkyl Dihalides with Oxindoles: Unified Access to Chiral Spirocarbocyclic Oxindoles. Org. Lett. 2022, 24, 875–880. [Google Scholar] [CrossRef]
- Smith, J.S.; Lefkowitz, R.J.; Rajagopal, S. Biased signalling: From simple switches to allosteric microprocessors. Nat. Rev. Drug Discov. 2018, 17, 243–260. [Google Scholar] [CrossRef] [PubMed]
- Markham, A. Oliceridine: First Approval. Drugs 2020, 80, 1739–1744. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, D.; Gotchev, D.; Pitis, P.; Chen, X.-T.; Liu, G.; Yuan, C.C.K. Opioid receptor ligands and methods of using and making same. PCT Int. Appl. WO2012129495 (published 27.09.2012).
- Hanschke, E. Zur Kenntnis der Prinsschen Reaktion, III. Mitteil.1): Über die Reaktion von Allylcarbinol mit Aldehyden und Ketonen. Chem. Ber. 1955, 88, 1053–1061. [Google Scholar] [CrossRef]
- D'Amico, A.; Mercuri, E.; Tiziano, F.D.; Bertini, E. Spinal muscular atrophy. Orphanet J. Rare Dis. 2011, 6, 71. [Google Scholar] [CrossRef]
- Dhillon, S. Risdiplam: First Approval. Drugs 2020, 80, 1853–1858. [Google Scholar] [CrossRef]
- Ratni, H.; Scalco, R.S.; Stephan, A.H. Risdiplam, the First Approved Small Molecule Splicing Modifier Drug as a Blueprint for Future Transformative Medicines. ACS Med. Chem. Lett. 2021, 12, 874–877. [Google Scholar] [CrossRef]
- Ratni, H.; Green, L.; Naryshkin, N.A.; Weetall, M.L. Compounds for treating spinal muscular atrophy. PCT Int. Appl. WO2015173181 (published 19.11.2015).
- Adam, J.-M.; Fantasia, S.M.; Fishlock, D.V.; Hoffmann-Emery, F.; Moine, G.; Pfleger, C.; Moessner, C. Process for the prepration of 7-(4,7-diazaspiro[2.5]octan-7-yl)-2-(2,8-dimethylimidazo[1,2-b]pyridazin-6-yl)pyrido[1,2-a]pyrimidin-4-one derivatives. PCT Int. Appl. 2019057740 (published 28.03.2019).
- Ailani, J.; Lipton, R.B.; Goadsby, P.J.; Guo, H.; Miceli, R.; Severt, L.; Finnegan, M.; Trugman, J.M.; ADVANCE Study Group. Atogepant for the Preventive Treatment of Migraine. N. Engl. J. Med. 2021, 385, 695–706. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Molinaro, C.; Wuelfing, W.P.; Yasuda, N.; Yin, J.; Zhong, Y.-L.; Lynch, J.; Andreani, T. Process for Making CGRP Receptor Antagonists. US Patent Appl. US2016130273 (published 12.05.2016).
- Dhillon, S. Trilaciclib: First Approval. Drugs 2021, 81, 867–874. [Google Scholar] [CrossRef] [PubMed]
- Sharpless, N.E.; Strum, J.C.; Bisi, J.E.; Roberts, P.J.; Tavares, F.X. Highly Active Anti-Neoplastic and Anti-Proliferative Agents. WO2014144740A2 (published 18.09.2014).
- Smith, A.; White, H.S.; Tavares, F.X.; Krasutsky, S.; Chen, J.-X.; Dorrow, R.L.; Zhong, H. Synthesis of N-(Heteroaryl)Pyrrolo[3,2-d]Pyrimidin-2-Amines. WO201800 5865A1, 2018.
| Entry | Drug name | Year of approval | Origin of spirocycle |
|---|---|---|---|
| 1 | Griseofulvin | 1959 | Cycloaddition, intramolecular Claisen condensation |
| 2 | Spironolactone | 1960 | Lactonization |
| 3 | Fluspirilene | 1970 | Cyclocondensation |
| 4 | Fenspiride | 1970 | Lactamization |
| 5 | Amcinonide | 1979 | Ketalization |
| 6 | Guanadrel | 1979 | Ketalization |
| 7 | Buspirone | 1986 | Commercially available spiro building block |
| 8 | Ivermectin | 1986 | Biosynthesis |
| 9 | Rifabutin | 1992 | Cyclocondensation |
| 10 | Spirapril | 1995 | Thioketalization |
| 11 | Irbesartan | 1997 | Cyclocondensation |
| 12 | Cevimeline | 2000 | Acetalization |
| 13 | Eplerenone | 2002 | Biosynthesis |
| 14 | Drospirenone | 2000 | Lactonization |
| 15 | Ledipasvir | 2014 | Cyclopropanation |
| 16 | Rolapitant | 2015 | Lactamization, ring-closing metathesis |
| 17 | Apalutamide | 2018 | Lactonization |
| 18 | Moxidectin | 2018 | Biosynthesis |
| 19 | Ubrogepant | 2020 | Intramolecular C-alkylation |
| 20 | Oliceridine | 2020 | Prins reaction |
| 21 | Risdiplam | 2020 | Commercially available spiro building block |
| 22 | Atogepant | 2021 | Intramolecular C-alkylation |
| 23 | Trilaciclib | 2021 | Intramolecular Claisen condensation |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




