Submitted:
21 April 2023
Posted:
21 April 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
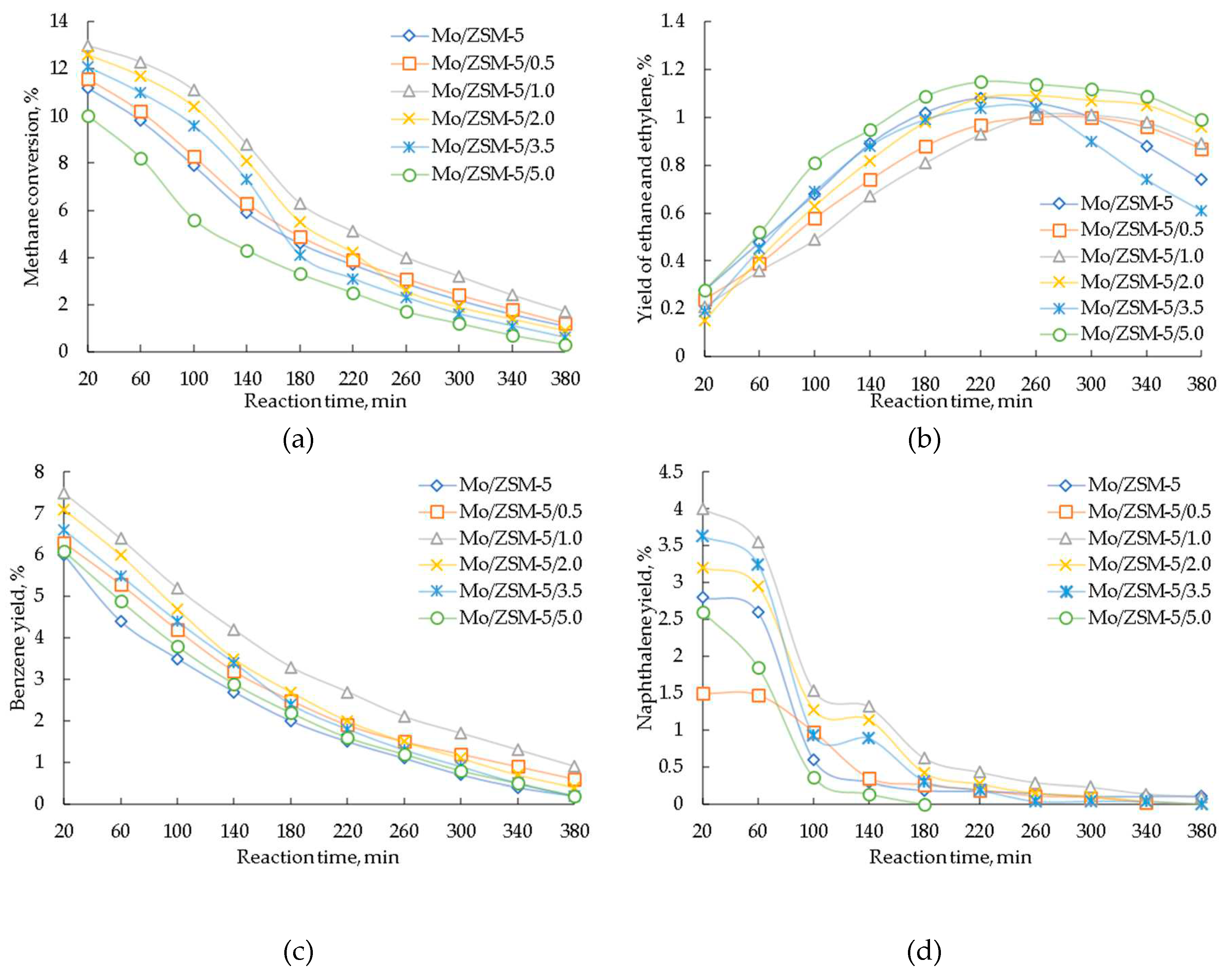
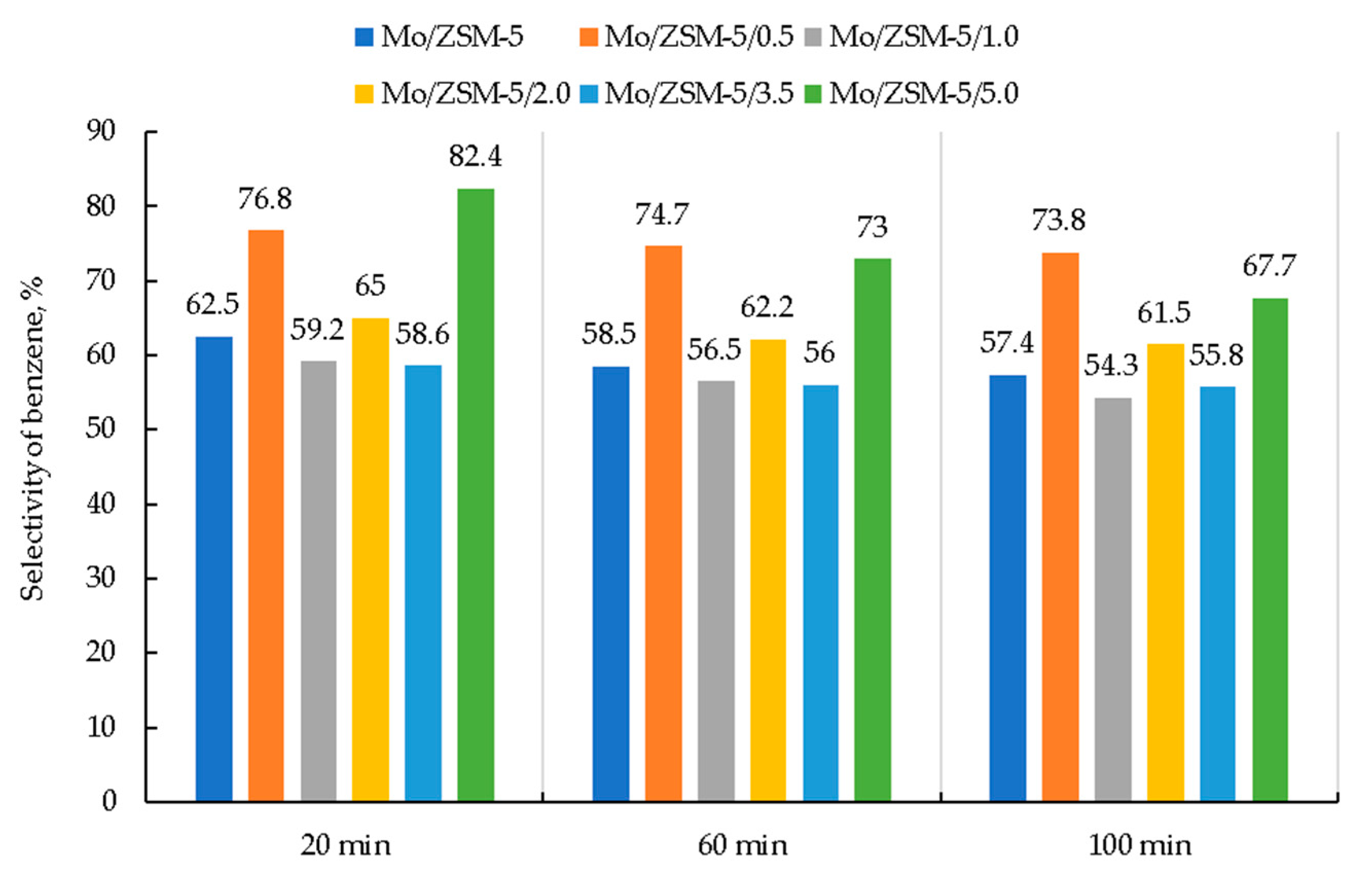
3. Results
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Pinaeva, L.G.; Noskov, A.S.; Parmon, V.N. Prospects for the direct catalytic conversion of methane into useful chemical products. Catalysis in Industry 2017, 9, 283–298. [Google Scholar] [CrossRef]
- Ma, S.; Guo, X.; Zhao, L.; Scott, S.; Bao, X. Recent progress in methane dehydroaromatization: From laboratory curiosities to promising technology. J. En. Chem. 2013, 22, 1–20. [Google Scholar] [CrossRef]
- Taifan, W.; Baltrusaitis, L. CH4 conversion to value added products: Potential, limitations and extensions of a single step heterogeneous catalysis. Appl. Catal. B: Environmental 2016, 198, 525–547. [Google Scholar] [CrossRef]
- Horn, R.; Schlogl, R. Methane activation by heterogeneous catalysis. Catalysis Letters 2015, 145, 23–39. [Google Scholar] [CrossRef]
- Majhi, S.; Mohanty, P.; Wang, H.; Pant, K.K. ; Direct conversion of natural gas to higher hydrocarbons: A review. J. En. Chem 2013, 22, 543–554. [Google Scholar] [CrossRef]
- Wang, B.; Albarracin-Suazo, S.; Pagan-Torres, Y.; Nikolla, E. Advances in methane conversion processes. Catal. Today 2017, 285, 147–158. [Google Scholar] [CrossRef]
- BP Statistical Review of World Energy 2022 71st edition. Available online: https://www.connaissancedesenergies.org/sites/default/files/pdf-actualites/bp-stats-review-2022-full-report.pdf (accessed on 10 April 2023).
- Huang, K.; Miller, J.B.; Huber, G.W.; Dumesic, J.A.; Maravelias, C.T. A general framework for the evaluation of direct nonoxidative methane conversion strategies. Joule 2018, 2, 349–365. [Google Scholar] [CrossRef]
- Ramasubramanian, V.; Ramsurn, H.; Price, G.L. Methane dehydroaromatization – A study on hydrogen use for catalyst reduction, role of molybdenum, the nature of catalyst support and significance of Bronsted acid sites. J. En. Chem. 2019, 34, 20–32. [Google Scholar] [CrossRef]
- Karakaya, C.; Morejudo, S.H.; Zhu, H.; Kee, R.J. Catalytic chemistry for methane dehydroaromatization (MDA) on a bifunctional Mo/HZSM-5 catalyst in a packed bed. Ind. Eng. Chem. Res. 2016, 55, 9895–9906. [Google Scholar] [CrossRef]
- Kosinov, N.; Coumans, F.J.A.G.; Uslamin, E.A.; Wijpkema, A.S.G.; Mezari, B.; Hensen, E.J.M. Methane dehydroaromatization by Mo/HZSM-5: Mono- or Bifunctional catalysis? ACS Catal. 2017, 7, 520–529. [Google Scholar] [CrossRef]
- Menon, U.; Rahman, M.; Khatib, S.J. A critical literature review of the advances in methane dehydroaromatization over multifunctional metal-promoted zeolite catalysts. Appl. Catal. A: Gen. 2020, 608, 117870. [Google Scholar] [CrossRef]
- Beuque, A.; Hao, H.; Berrier, E.; Batalha, N.; Sachse, A.; Paul, J.-F.; Pinard, L. How do the products in methane dehydroaromatization impact the distinct stages of the reaction? Appl. Catal. B: Environmental 2022, 309, 121274. [Google Scholar] [CrossRef]
- Karakaya, C.; Zhu, H.; Kee, R.J. Kinetic modeling of methane dehydroaromatization chemistry on Mo/Zeolite catalysts in packed-bed reactors. Chem. En. Sci. 2015, 123, 474–486. [Google Scholar] [CrossRef]
- Spivey, J.J.; Hutchings, G. Catalytic aromatization of methane. Chem. Soc. Rev. 2014, 43, 792–803. [Google Scholar] [CrossRef]
- Kiani, D.; Sourav, S.; Tang, Y.; Baltrusaitis, J.; Wachs, I.E. Methane activation by ZSM-5-supported transition metal centers. Chem. Soc. Rev. 2021, 50, 1251. [Google Scholar] [CrossRef] [PubMed]
- Pang, T.; Yang, X.; Yuan, C.; Elzatahry, A.A.; Alghamdi, A.; He, X.; Cheng, X.; Deng Y. Recent advance in synthesis and application of heteroatom zeolites Author links open overlay panel. Chinese Chemical Letters 32, 328-338. [CrossRef]
- Sun, K.; Ginosar, D.M.; He, T.; Zhang, Y.; Fan, Y.; Chen, R. Progress in Nonoxidative Dehydroaromatization of Methane in the Last 6 Years. Ind. Eng. Chem. Res. 2018, 57, 1768–1789. [Google Scholar] [CrossRef]
- Hartmann, M.; Machoke, A.G.; Schwieger, W. Catalytic test reactions for the evaluation of hierarchical zeolites. Chem. Soc. Rev. 2016, 45, 3313–3330. [Google Scholar] [CrossRef]
- Feng, R.; Wang, X.; Lin, J.; Li, Z.; Hou, K.; Yan, X.; Hu, X.; Yan, Z.; Rood, M.J. Two-stage glucose-assisted crystallization of ZSM-5 to improve methanol to propylene (MTP). Micropor. Mesopor. Mater. 2018, 270, 57–66. [Google Scholar] [CrossRef]
- Parkhomchuk, E.V.; Sashkina, K.A.; Parmon, V.N. New heterogeneous catalysts based on zeolites with hierarchical pore system. Petroleum Chemistry 2016, 56, 197–204. [Google Scholar] [CrossRef]
- Liu, H.; Yang, S.; Hu, J.; Shang, F.; Li, Z.; Xu, C.; Guan, J.; Kan, Q. A Comparison study of mesoporous Mo/H-ZSM-5 and conventional Mo/H-ZSM-5 catalysts in methane non-oxidative aromatization. Furl Processing Technology 2012, 96, 195–202. [Google Scholar] [CrossRef]
- Na, K.; Choi, M.; Ryoo, R. Recent advances in the synthesis of hierarchically nanoporous zeolites. Micropor. Mesopor. Mater. 2013, 166, 3–19. [Google Scholar] [CrossRef]
- Yang, K.; Zhou, F.; Ma, H. , Yu, L., Wu, G. Glucose-Assisted Synthesis of Hierarchical HZSM-5 for Catalytic Fast Pyrolysis of Cellulose to Aromatics. Chemistry Select 2021, 6, 11591–11598. [Google Scholar] [CrossRef]
- Liu, H.; Kan, Q. Improved performance of hierarchical porous Mo/H-IM-5 catalyst in methane non-oxidative aromatization. Appl. Petrochem. Res. 2017, 7, 2–4. [Google Scholar] [CrossRef]
- Liu, K.; Caglayan, M.; Dikhtiarenko, A.; Zhang, X.; Sayidov, O.; Abou-Hamad, E.; Gascon, J.; Chowdhury, A.D. Are hierarchical zeolites good catalysts for Methane Dehydroaromatization? A critical analysis. Catal. Today 2023, 408, 22–35. [Google Scholar] [CrossRef]
- Dipak, B.S.; Viomesh, P.P. Estimation of crystalline phase in ZSM-5 zeolites by infrared spectroscopy. J. Chem. Tech. Biotechnol. 1989, 44, 147–154. [Google Scholar]
- Bukhtiyarov, V.I.; Zaikovskii, V.I.; Kashin, A.S.; Ananikov, V.P. Modern electron microscopy in the study of chemical systems at the boundary of organic synthesis and catalysis. Russian Chemical Reviews 2016, 85, 1198–1214. [Google Scholar] [CrossRef]
- Vosmerikov, A.V.; Erofeev, V.I. Influence of mechanical treatment on the catalytic properties of zeolite-containing catalysts for aromatization of lower alkanes. Russian Journal of Physical Chemistry A 1995, 69, 787–790. [Google Scholar]
- Vosmerikov, A.V.; Korobitsyna, L.L.; Barabashin, Ya.E.; Arbuzova, N.V.; Echevskii, G.V.; Kodenev, E.G.; Zhuravkov, S.P. Deactivation of Molybdenum-Containing Zeolites in the Course of Nonoxidative Methane Conversion. Kinetics and Catalysis 2005, 46, 724–728. [Google Scholar] [CrossRef]
- Break, D.W. Zeolite molecular sieves: Structure, chemistry, and use; Publisher: Wiley, USA, 1973; 771. [Google Scholar]
- Mamonov, N.A.; Fadeeva, E.V.; Grigoriev, D.A.; Mikhailov, M.N.; Kustov, L.M.; Alkhimov, S.A. Metal/zeolite catalysts of methane dehydroaromatization. Russian Chemical Reviews 2013, 82, 567–585. [Google Scholar] [CrossRef]
- Xiong, Z.-T.; Zhang, H.-B.; Lin, G.-D.; Zeng, J.-L. Study of W/HZSM-5 based catalysts for dehydroaromatization of CH4 in absence of O2. II. Action of promoters Zn and Li. Catal. Lett. 2001, 74, 233–243. [Google Scholar] [CrossRef]
- Ma, D.; Lu, Y.; Su, L.; Xu, Z.; Tian, Z.; Xu, Y.; Lin, L.; Bao, X. Remarkable improvement on the methane aromatization reaction: A highly selective and coking-resistant catalyst. J. Phys. Chem. B 2002, 106, 8524–8530. [Google Scholar] [CrossRef]
- Zhao, K.; Jia, L.; Wang, J.; Hou, B.; Li. D. The influence of the Si/Al ratio of Mo/HZSM-5 on methane non-oxidative dehydroaromatization. New J. Chem. 2019, 43, 4130–4136. [Google Scholar] [CrossRef]
- Baerlocher, C.; McCusker, L.B.; Olson, D.H. Atlas of zeolite framework types, 6th ed.; Publisher: Elsevier, Netherlands, 2007; 398. [Google Scholar]
- Hu, J.; Wu, S.; Ma, Y.; Yang, X.; Li, Z.; Liu, H.; Huo, Q.; Guan, J.; Kan, Q. Effect of the particle size of MoO3 on the catalytic activity of Mo/ZSM-5 in methane non-oxidative aromatization. New J. Chem. 2015, 39, 5459–5469. [Google Scholar] [CrossRef]
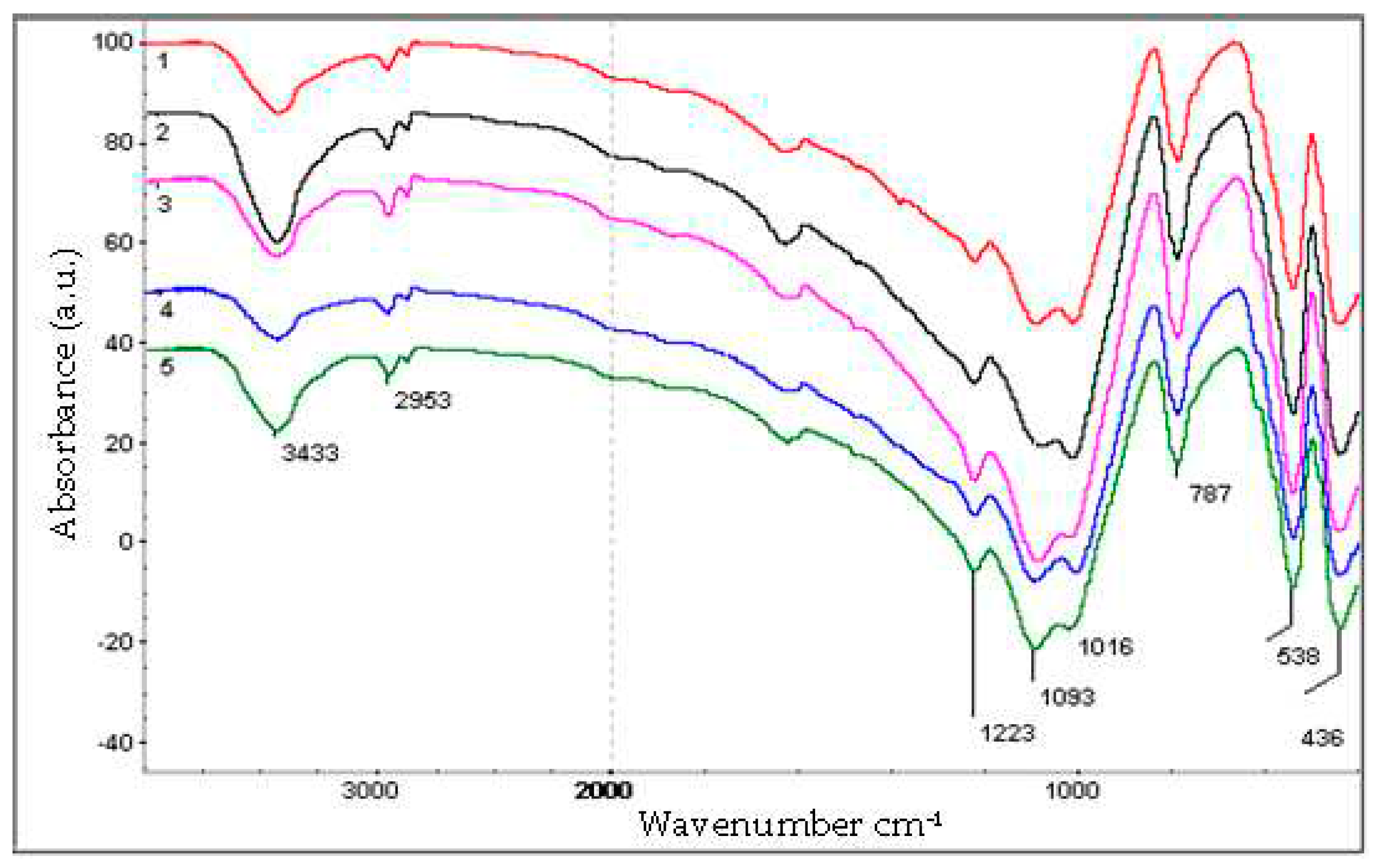
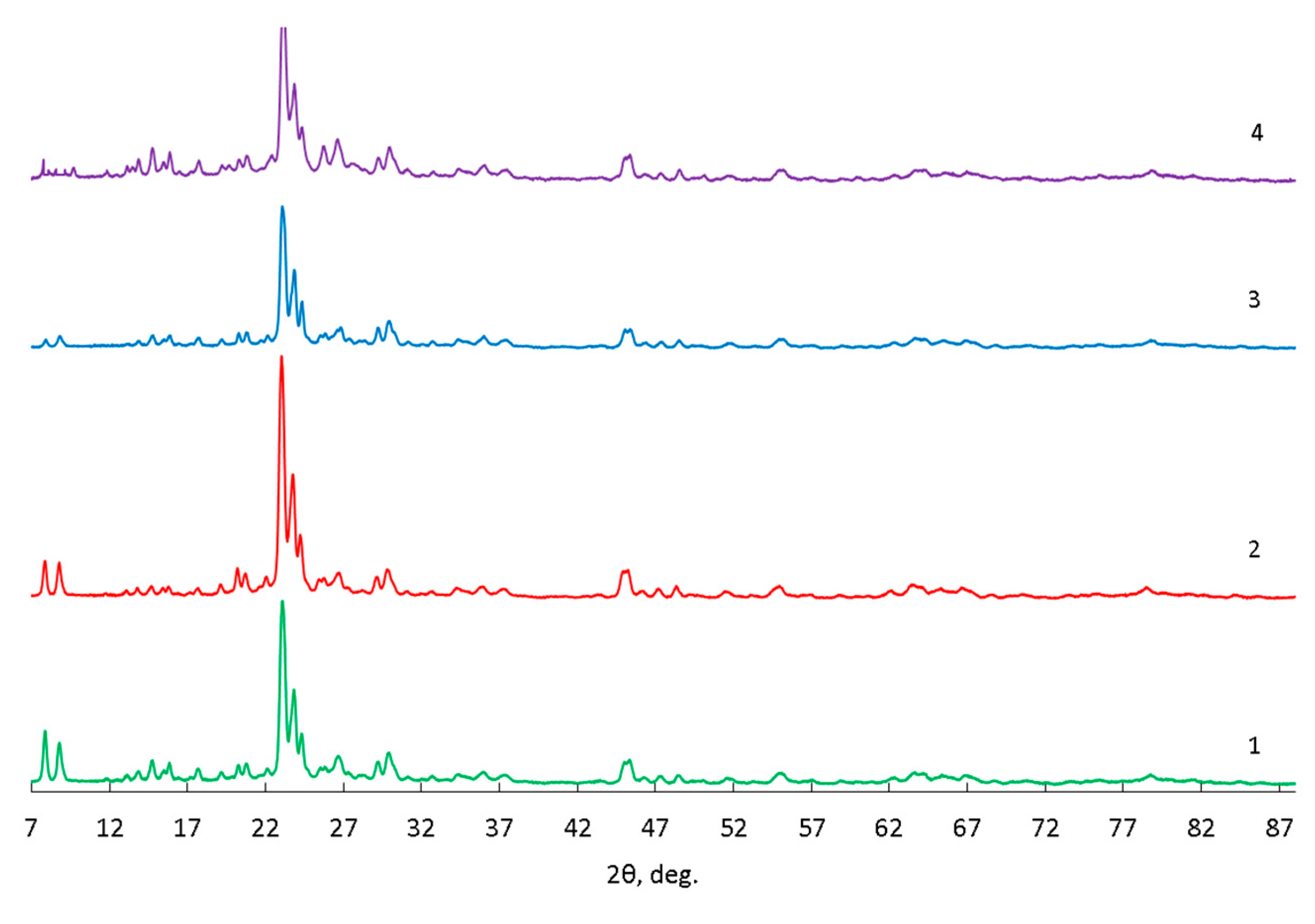


| Zeolite | Element content, mass % | |||
|---|---|---|---|---|
| Si | O | Al | *M | |
| ZSM-5 | 49.09 | 45.90 | 2.46 | 2.55 |
| ZSM-5/1.0 | 54.65 | 42.40 | 2.77 | 0.18 |
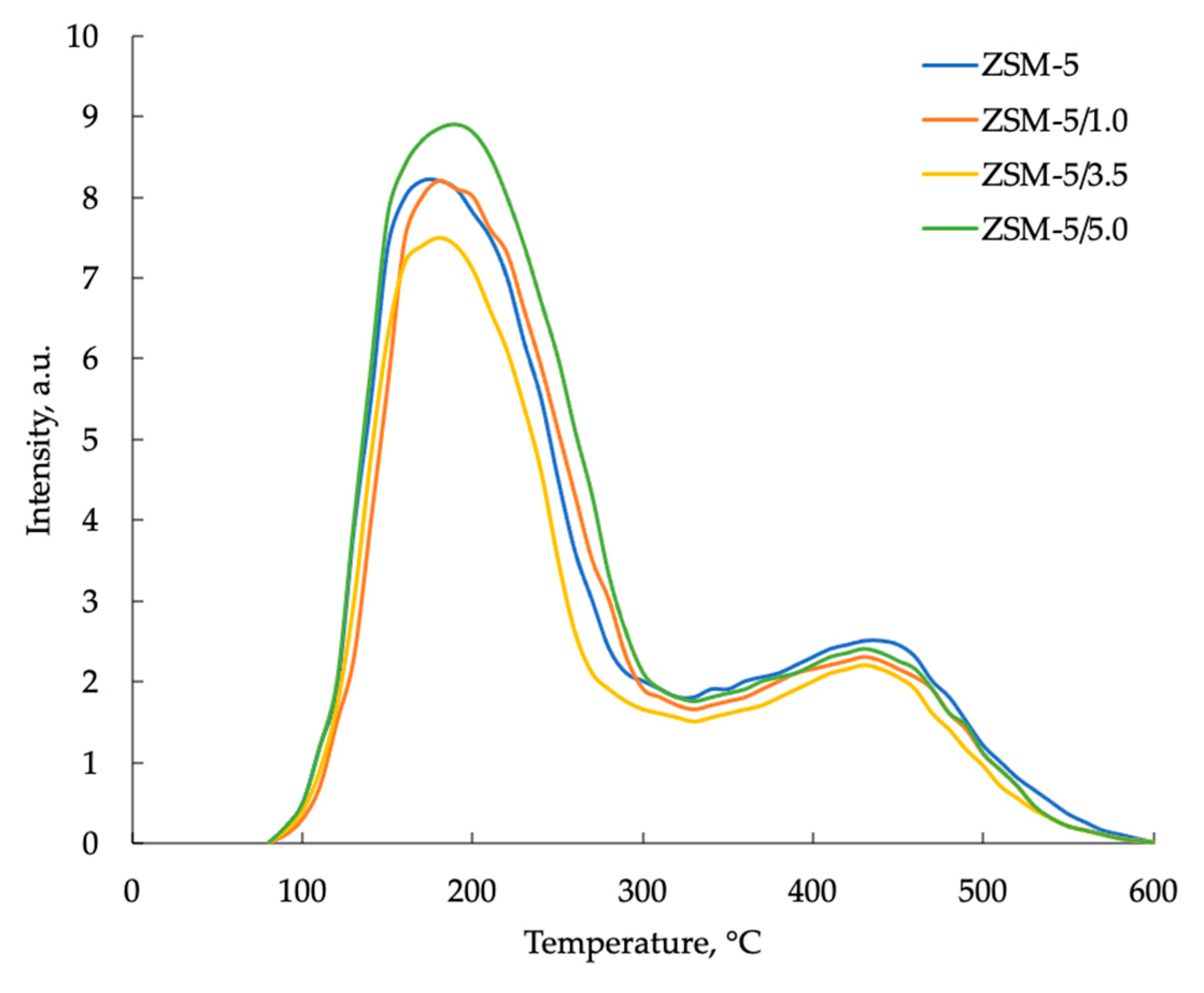
| Sample | Т, °С | Concentration, μmol/g | |||
|---|---|---|---|---|---|
| TI | TII | CI | CII | C∑ | |
| ZSM-5 | 185 | 440 | 753 | 388 | 1141 |
| 4%Mo/ZSM-5 | 180 | 430 | 807 | 355 | 1162 |
| ZSM-5/1.0 | 185 | 435 | 767 | 389 | 1156 |
| ZSM-5/2.0 | 180 | 430 | 787 | 364 | 1151 |
| ZSM-5/3.5 | 185 | 430 | 793 | 348 | 1141 |
| ZSM-5/5.0 | 195 | 435 | 824 | 322 | 1146 |
| 4%Mo/ZSM-5/1.0 | 160 | 430 | 810 | 315 | 1125 |
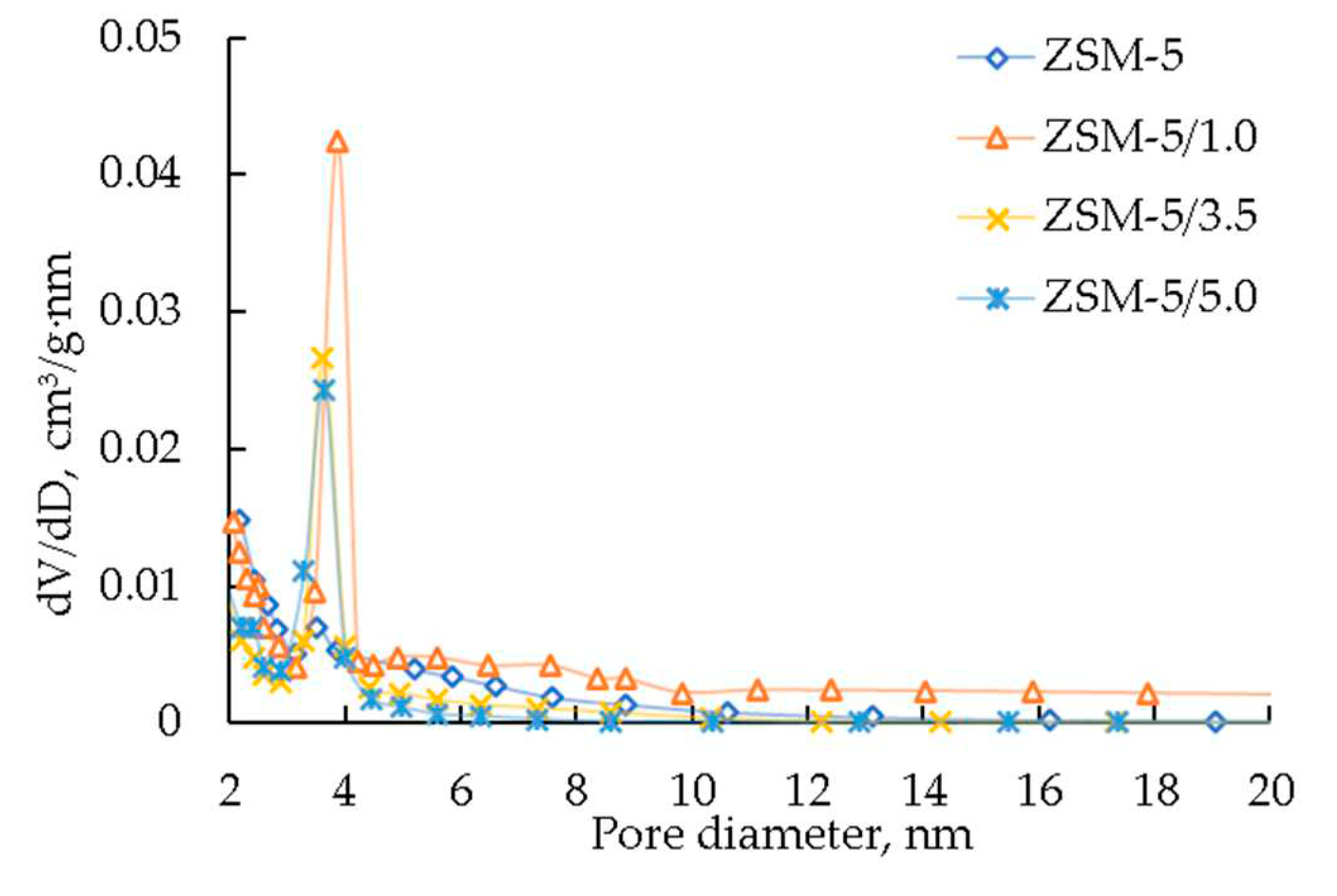
| Sample | SBET, m2/g | Vmicro, cm3/g | Vmeso, cm3/g | Vtotal, cm3/g |
|---|---|---|---|---|
| ZSM-5 | 285 | 0,13 | 0,02 | 0,15 |
| ZSM-5/1.0 | 379 | 0,12 | 0,08 | 0,20 |
| ZSM-5/2.0 | 378 | 0,12 | 0,10 | 0,22 |
| ZSM-5/3.5 | 375 | 0,11 | 0,11 | 0,22 |
| ZSM-5/5.0 | 373 | 0,11 | 0,13 | 0,24 |
| 4%Mo/ZSM-5/1.0 | 343 | 0,10 | 0,07 | 0,17 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




