Submitted:
20 April 2023
Posted:
20 April 2023
You are already at the latest version
Abstract
Keywords:
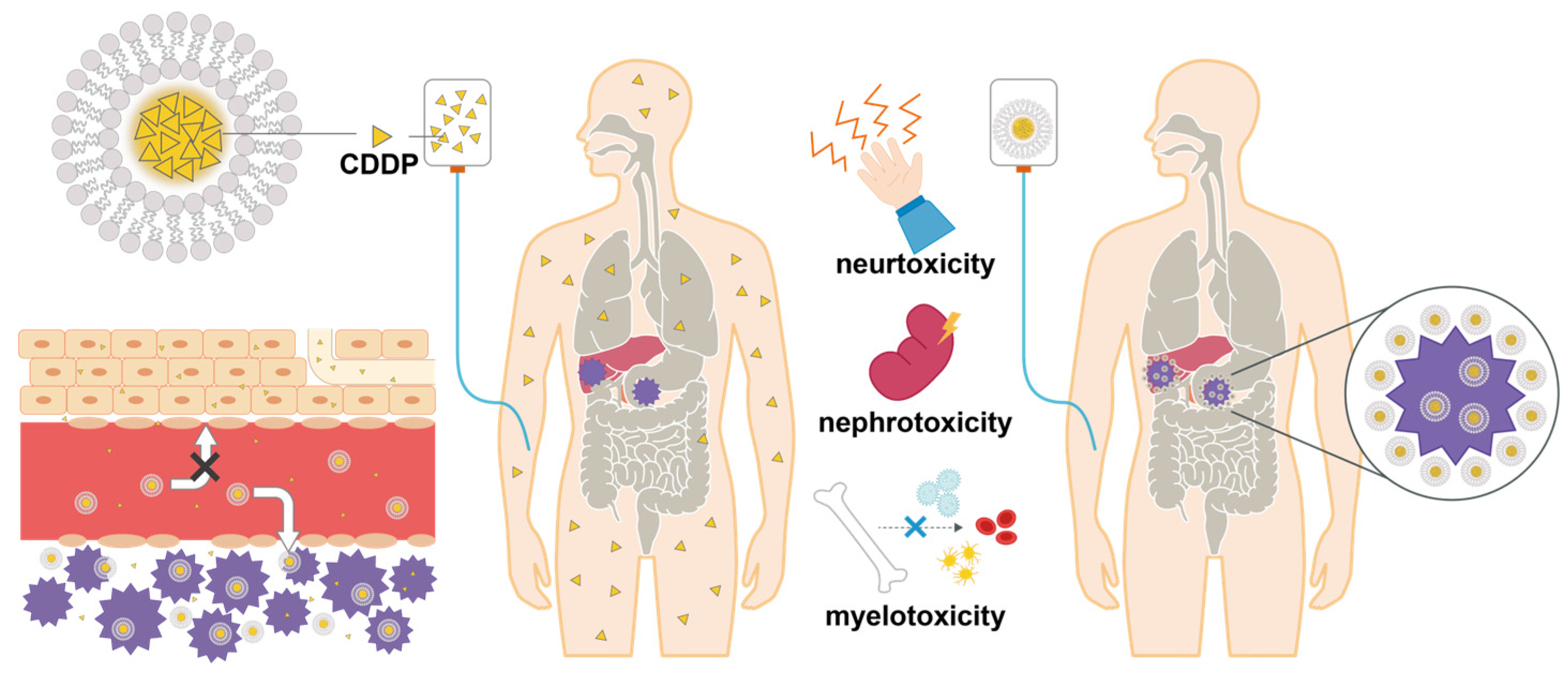
1. Introduction
2. Materials and Methods
2.1. Ethical Review and Informed Consent
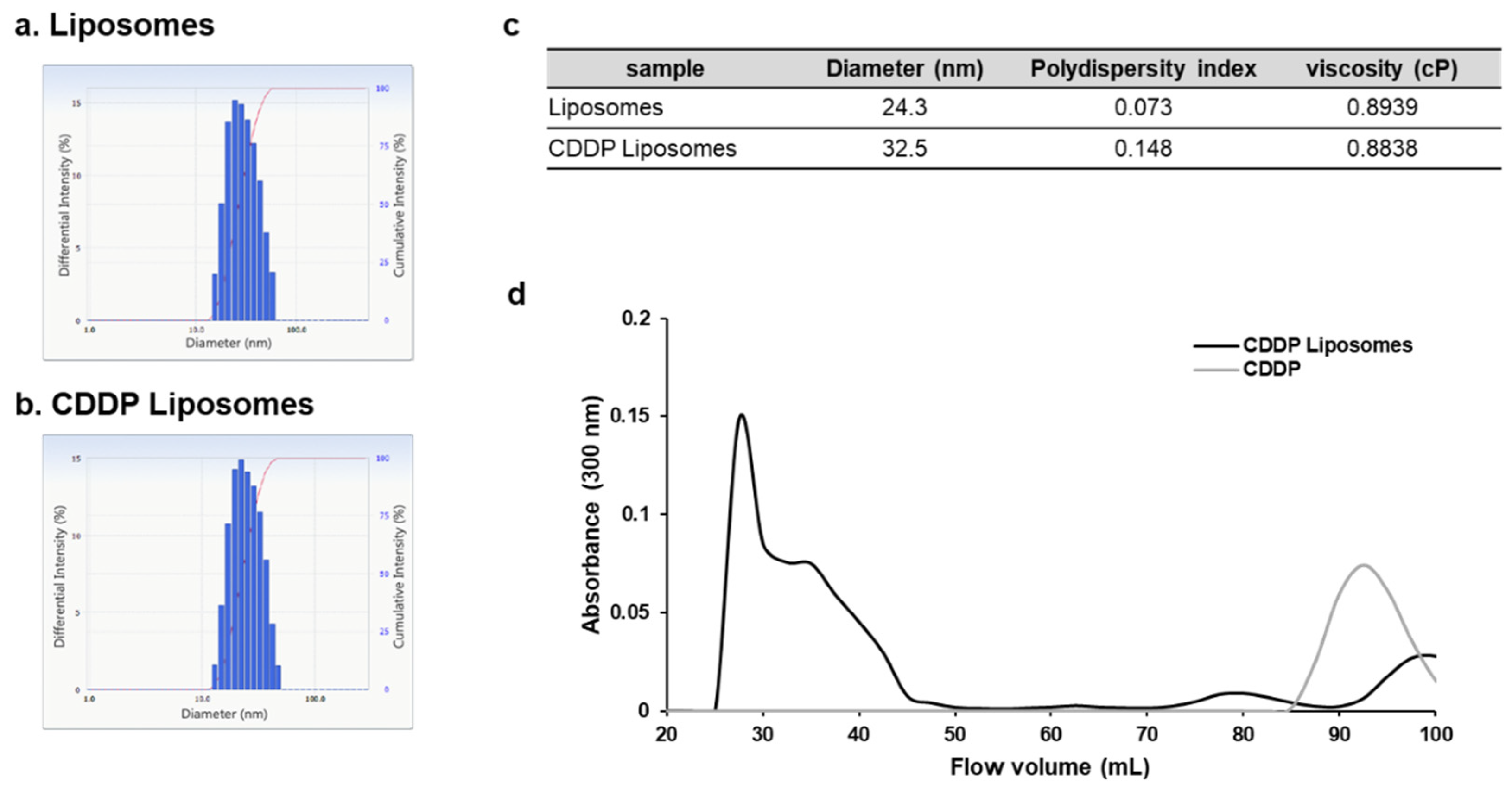
2.2. Preparation of CDDP liposomes
2.3. CDDP liposome therapeutic intervention
2.4. Examination of circulating tumor cells
3. Results
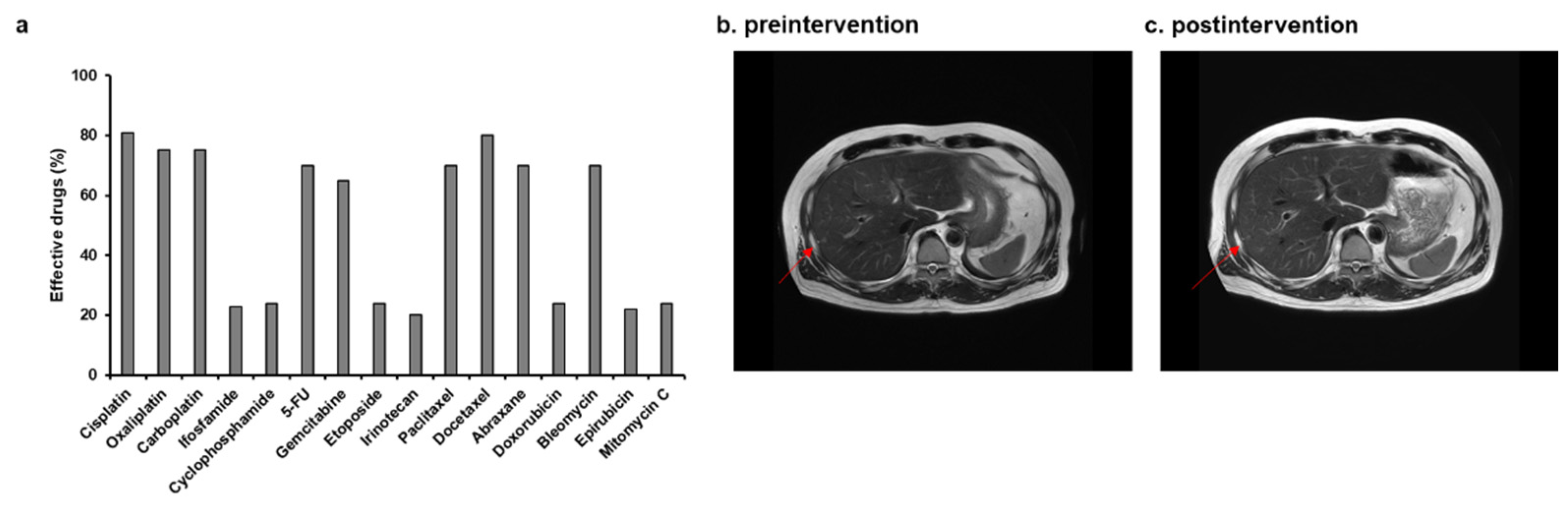
3.1. A case in which CDDP liposomes were administered to a patient with recurrence of multiple liver metastases from metastatic nasal cancer
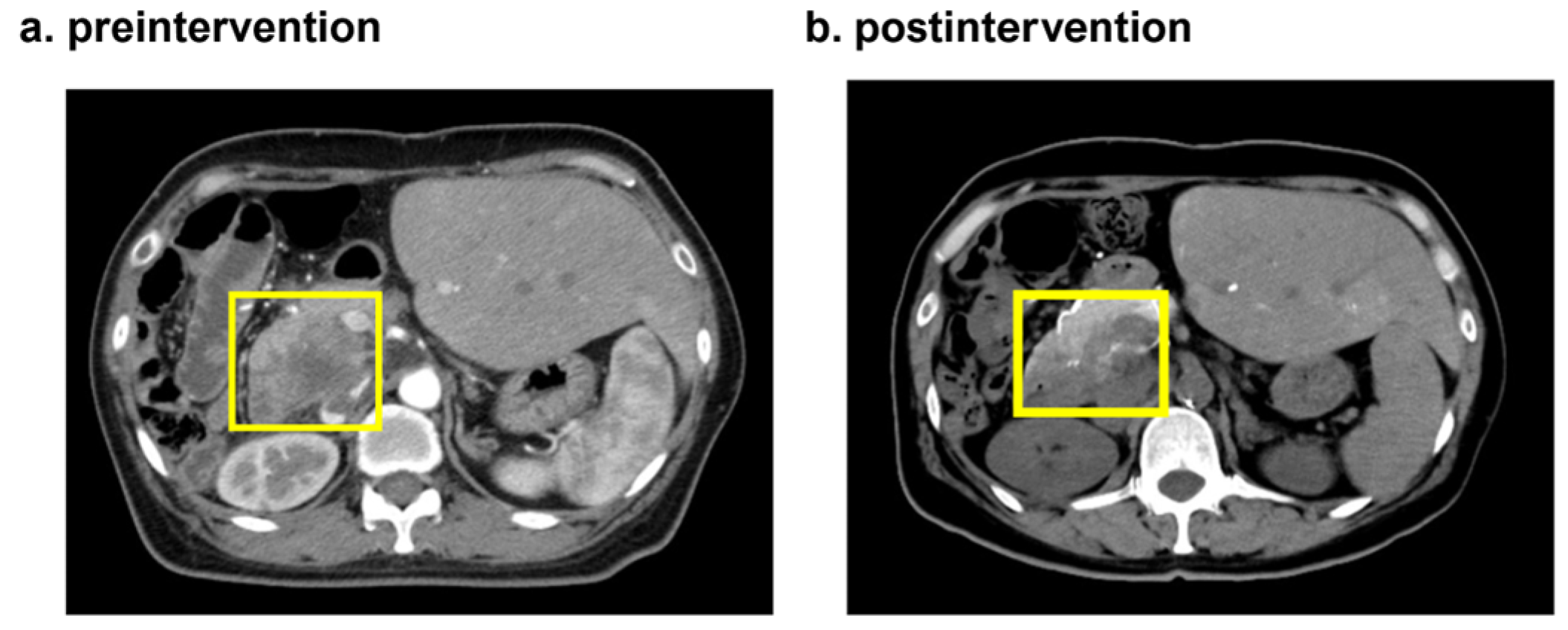
3.2. A case in which CDDP liposomes were administered to a patient undergoing partial pancreatic resection and jejunociliary anastomosis for biliary tract cancer
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rosenberg, B.; Van Camp, L.; Krigas, T. Inhibition of cell division in Escherichia coli by electrolysis products from a platinum electrode. Nature 1965, 205, 698-699. [CrossRef]
- Lippard, S. J. New chemistry of an old molecule: cis-[Pt (NH3) 2Cl2]. Science 1982, 218, 1075-1082. [CrossRef]
- Brabec, V.; Hrabina, O.; Kasparkova, J. Cytotoxic platinum coordination compounds. DNA binding agents. Coord Chem Rev 2017, 351, 2-31. [CrossRef]
- Wagner, A. D.; Grothe, W. Transcatheter Arterial Chemo Embolization Haerting, J.; Kleber, G.; Grothey, A.; Fleig, W. E. Chemotherapy in advanced gastric cancer: a systematic review and meta-analysis based on aggregate data. J Clin Oncol 2006, 24, 2903-2909. [CrossRef]
- Enzinger, P. C.; Ilson, D. H.; Kelsen, D. P. Chemotherapy in esophageal cancer. Semin Oncol 1999, 26, 12-20.
- Fennell, D. A.; Summers, Y.; Cadranel, J.; Benepal, T.; Christoph, D. C.; Lal, R.; Das, M.; Maxwell, F.; Visseren-Grul, C.; Ferry, D. Cisplatin in the modern era: The backbone of first-line chemotherapy for non-small cell lung cancer. Cancer Treat Rev 2016, 44, 42-50. [CrossRef]
- Du Bois, A.; Lück, H. J.; Meier, W.; Adams, H. P.; Möbus, V.; Costa, S.; Bauknecht, T.; Richter, B.; Warm, M.; Schröder, W.; Olbricht, S.; Nitz, U.; Jackisch, C.; Emons, G.; Wagner, U.; Kuhn, W.; Pfisterer, J.; Arbeitsgemeinschaft Gynäkologische Onkologie Ovarian Cancer Study Group. A randomized clinical trial of cisplatin/paclitaxel versus carboplatin/paclitaxel as first-line treatment of ovarian cancer. J Natl Cancer Inst 2003, 95, 1320-1329. [CrossRef]
- de Vries, G.; Rosas-Plaza, X.; van Vugt, M. A. T. M.; Gietema, J. A.; de Jong, S. Testicular cancer: Determinants of cisplatin sensitivity and novel therapeutic opportunities. Cancer Treat Rev 2020, 88, 102054. [CrossRef]
- Lamont, E. B.; Vokes, E. E. Chemotherapy in the management of squamous-cell carcinoma of the head and neck. The Lancet Oncol 2001, 2, 261-269. [CrossRef]
- Santabarbara, G.; Maione, P.; Rossi, A.; Gridelli, C. Pharmacotherapeutic options for treating adverse effects of Cisplatin chemotherapy. Expert Opin Pharmacother 2016, 17, 561-570. [CrossRef]
- Ranganath, P.; Einhorn, L.; Albany, C. Management of chemotherapy induced nausea and vomiting in patients on multiday cisplatin based combination chemotherapy. Biomed Res Int 2015, 2015, 943618. [CrossRef]
- DeConti, R. C.; Toftness, B. R.; Lange, R. C.; Creasey W. A. Clinical and pharmacological studies with cis-diamminedichloroplatinum (II). Cancer Res 1973, 33, 1310-1315.
- Nishiyama, N.; Okazaki, S.; Cabral, H.; Miyamoto, M.; Kato, Y.; Sugiyama, Y.; Nishio, K.; Matsumura, Y.; Kataoka, K. Novel cisplatin-incorporated polymeric micelles can eradicate solid tumors in mice. Cancer Res 2003, 63, 8977-8983.
- Uchino, H.; Matsumura, Y.; Negishi, T.; Koizumi, F.; Hayashi, T.; Honda, T.; Nishiyama, N.; Kataoka, K.; Naito, S.; Kakizoe, T. Cisplatin-incorporating polymeric micelles (NC-6004) can reduce nephrotoxicity and neurotoxicity of cisplatin in rats. Br J Cancer 2005, 93, 678-687. [CrossRef]
- Saisyo, A.; Nakamura, H.; Fang, J.; Tsukigawa, K.; Greish, K.; Furukawa, H.; Maeda, H. pH-sensitive polymeric cisplatin-ion complex with styrene-maleic acid copolymer exhibits tumor-selective drug delivery and antitumor activity as a result of the enhanced permeability and retention effect. Colloids Surf B Biointerfaces 2016, 138, 128-137. [CrossRef]
- Islam, W.; Kimura, S.; Islam, R.; Harada, A.; Ono, K.; Fang, J.; Niidome, T.; Sawa, T.; Maeda, H. EPR-effect enhancers strongly potentiate tumor-targeted delivery of nanomedicines to advanced cancers: Further extension to enhancement of the therapeutic effect. J Pers Med 2021, 11, 487. [CrossRef]
- Yorozu, K.; Kaibori, M.; Kimura, S.; Ichikawa, M.; Matsui, K.; Kaneshige, S.; Kobayashi, M.; Jimbo, D.; Torikai, Y.; Fukuzawa, Y.; Okamoto, Y. Experience with Photodynamic Therapy Using Indocyanine Green Liposomes for Refractory Cancer. J Pers Med 2022, 12, 1039. [CrossRef]
- DasGupta, R.; Yap, A.; Yaqing, E. Y.; Chia, S. Evolution of precision oncology-guided treatment paradigms. WIREs Mech Dis 2023, 15, e1585. [CrossRef]
- Borgeaud, M.; Kim, F.; Friedlaender, A.; Lococo, F.; Addeo, A.; Minervini, F. The Evolving Role of Immune-Checkpoint Inhibitors in Malignant Pleural Mesothelioma. J Clin Med 2023, 12, 1757. [CrossRef]
- Boulikas, T.; Vougiouka, M. Recent clinical trials using cisplatin, carboplatin and their combination chemotherapy drugs. Oncol Rep 2004, 11, 559-595. [CrossRef]
- Dewit, L. Combined treatment of radiation and cisdiamminedichloroplatinum (II): a review of experimental and clinical data. Int J Radiat Oncol Biol Phys 1987, 13, 403-426. [CrossRef]
- Ichikawa, M.; Matsunaga, W.; Ishikawa, T.; Gotoh, A. Lentiviral vector-mediated gene transfer combined with cisplatin enhances tumor suppression in human bladder cancer cell lines. Personalized Medicine Universe 2019, 8, 15-19. [CrossRef]
- Aapro, M. S.; Walko, C. M. Aprepitant: drug–drug interactions in perspective. Ann Oncol 2010, 21, 2316-2323. [CrossRef]
- Grunberg, S. M. Antiemetic activity of corticosteroids in patients receiving cancer chemotherapy: dosing, efficacy, and tolerability analysis. Ann Oncol 2007, 18, 233-240. [CrossRef]
- Brock, P. R.; Maibach, R.; Childs, M.; Rajput, K.; Roebuck, D.; Sullivan, M. J.; Laithier, V.; Ronghe, M.; Dall'Igna, P.; Hiyama, E.; Brichard, B.; Skeen, J.; Mateos, M. E.; Capra, M.; Rangaswami, A. A.; Ansari, M.; Rechnitzer, C.; Veal, G. J.; Covezzoli, A.; Brugières, L.; Perilongo, G.; Czauderna, P.; Morland, B.; Neuwelt, E. A. Sodium thiosulfate for protection from cisplatin-induced hearing loss. N Engl J Med 2018, 378, 2376-2385. [CrossRef]
- Araújo, R. S.; Cristina Oliveira, M.; Cardoso, V. N.; Keefe, D. M.; Stringer, A. M. The effect of free and encapsulated cisplatin into long-circulating and pH-sensitive liposomes on IEC-6 cells during wound healing in the presence of host–microbiota. J Pharm Pharmacol 2022, 74, 711-717. [CrossRef]
- Boulikas, T. Clinical overview on Lipoplatin™: a successful liposomal formulation of cisplatin. Expert Opin Investig Drugs 2009, 18, 1197-1218. [CrossRef]
| Characteristics | preintervention | postintervention | preintervention | postintervention |
|---|---|---|---|---|
| Age (years) | 57 | 52 | ||
| Sex | Male | Female | ||
| Height | 165.0 | 160.0 | ||
| Weight | 60.0 | 57.0 | ||
| PS | 0 | 0 | ||
| Chills | 0 | 0 | 0 | 0 |
| Edema face | 0 | 0 | 0 | 0 |
| Fatigue | 0 | 1 | 0 | 1 |
| Gait disturbance | 0 | 0 | 0 | 0 |
| Infusion site extravasation | 0 | 0 | 0 | 0 |
| Injection site reaction | 0 | 0 | 0 | 0 |
| Malaise | 0 | 0 | 0 | 0 |
| Pain | 0 | 0 | 0 | 0 |
| Test items | Reference ranges | Unit | 12/6/22 | 12/13/22 | 12/20/22 | 12/27/22 | 1/6/23 | 1/11/23 | 1/16/23 | 1/24/23 | 1/31/23 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| TP | 6.5~8.2 | g/dL | 7.4 | 7.5 | 7.4 | 7.7 | 7.0 | 7.3 | 7.5 | 7.3 | 7.8 |
| ALB | 3.8~5.2 | g/dL | 4.5 | 4.5 | 4.7 | 4.9 | 4.7 | 4.5 | 4.8 | 4.6 | 4.7 |
| T-Bil | 0.2~1.2 | mg/dL | 0.5 | 0.5 | 0.4 | 0.6 | 0.4 | 0.4 | 0.6 | 0.4 | 0.5 |
| ALP | 38~113 | U/L | 39 | 38 | 41 | 36 | 32 | 33 | 35 | 31 | 46 |
| AST | 10~40 | U/L | 17 | 25 | 17 | 18 | 16 | 16 | 19 | 18 | 22 |
| ALT | 6~40 | U/L | 16 | 17 | 14 | 14 | 13 | 13 | 13 | 12 | 17 |
| LD | 124~222 | U/L | 127 | 219 | 139 | 133 | 125 | 114 | 134 | 134 | 154 |
| γ-GT | 80 ≦ | U/L | 38 | 43 | 42 | 40 | 34 | 36 | 40 | 35 | 46 |
| Ch-E | 200~465 | U/L | 286 | 268 | 289 | 301 | 277 | 270 | 291 | 265 | 281 |
| GLU | 70~109 | mg/dL | 116 | 122 | 144 | 114 | 160 | 165 | 136 | 147 | 149 |
| TC | 130~219 | mg/dL | 226 | 205 | 210 | 215 | 206 | 213 | 220 | 192 | 214 |
| TG | 35~149 | mg/dL | 185 | 126 | 145 | 140 | 148 | 191 | 102 | 110 | 177 |
| UN | 8.0~21.0 | mg/dL | 11.9 | 13.9 | 16.7 | 14.3 | 14.9 | 17.3 | 15.8 | 18.4 | 17.4 |
| CRE | 0.60~1.15 | mg/dL | 0.91 | 1.05 | 1.05 | 1.01 | 1.03 | 1.02 | 1.08 | 1.05 | 1.01 |
| eGFR | 1.73 m2 ≧ 60 | mL/min | 67.4 | 57.6 | 57.6 | 60.1 | 59.5 | 59.5 | 55.9 | 57.6 | 59.8 |
| CRP | 0.30 ≦ | mg/dL | 0.17 | 1.07 | 0.37 | 0.13 | 0.27 | 0.86 | 0.76 | 0.71 | 1.75 |
| WBC | 38~98 | 102/μL | 45 | 33 | 48 | 45 | 41 | 56 | 42 | 43 | 47 |
| RBC | 420~570 | 104/μL | 423 | 427 | 422 | 427 | 413 | 411 | 473 | 408 | 421 |
| Hgb | 13.2~17.6 | g/dL | 12.7 | 12.8 | 12.7 | 13.0 | 12.4 | 12.1 | 13.1 | 12.4 | 12.7 |
| Hct | 39.2~51.8 | % | 40.4 | 40.9 | 41.1 | 40.4 | 40.0 | 39.6 | 41 | 38.9 | 41.3 |
| MCV | 83.0~101.5 | fL | 95.5 | 95.8 | 97.4 | 94.6 | 96.9 | 96.4 | 93.8 | 95.3 | 98.1 |
| MCH | 28.0~34.5 | pg | 30.0 | 30.0 | 30.1 | 30.4 | 30.0 | 29.4 | 30.0 | 30.4 | 30.2 |
| MCHC | 31.5~35.5 | % | 31.4 | 31.3 | 30.9 | 32.2 | 31.0 | 30.6 | 32.0 | 31.9 | 30.8 |
| PLT | 14.0~36.0 | 104/μL | 23.7 | 25.0 | 27.4 | 27.2 | 27.3 | 27.1 | 29.9 | 27.5 | 29.4 |
| MYELO | 0.0 | % | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| MET. M | 0.0 | % | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| NEUT | 36.0~69.0 | % | 71.4 | 59.5 | 67.7 | 70.4 | 66.5 | 73.4 | 68.5 | 69.7 | 71.9 |
| EOS | 1.0~5.0 | % | 1.3 | 3.1 | 1.5 | 0.9 | 1.5 | 0.9 | 1.4 | 0.9 | 1.1 |
| BASO | 0.0~2.0 | % | 1.1 | 1.8 | 1.3 | 1.1 | 0.5 | 0.5 | 0.7 | 0.9 | 0.8 |
| LYMP | 27.0~53.0 | % | 20.4 | 27.0 | 22.8 | 22.5 | 24.7 | 19 | 23.4 | 22.2 | 20.7 |
| AT. LY | 0.0 | % | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| MONO | 2.0~10.0 | % | 5.8 | 8.6 | 6.7 | 5.1 | 6.8 | 6.2 | 6.0 | 6.3 | 5.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




