Submitted:
19 April 2023
Posted:
20 April 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
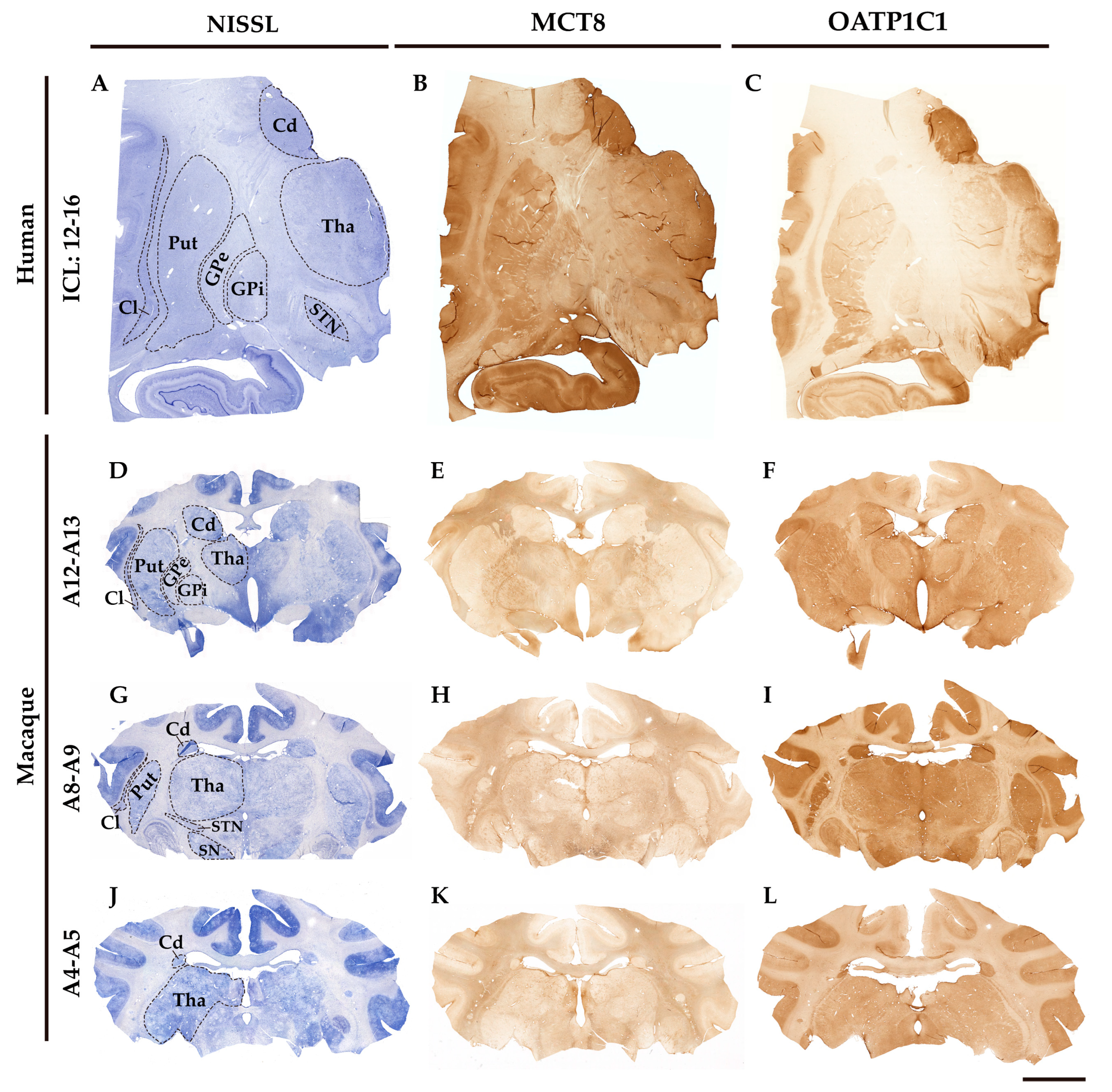
2.1. General pattern of MCT8 and OATP1C1 immunostaining in the adult human and macaque basal ganglia nuclei.
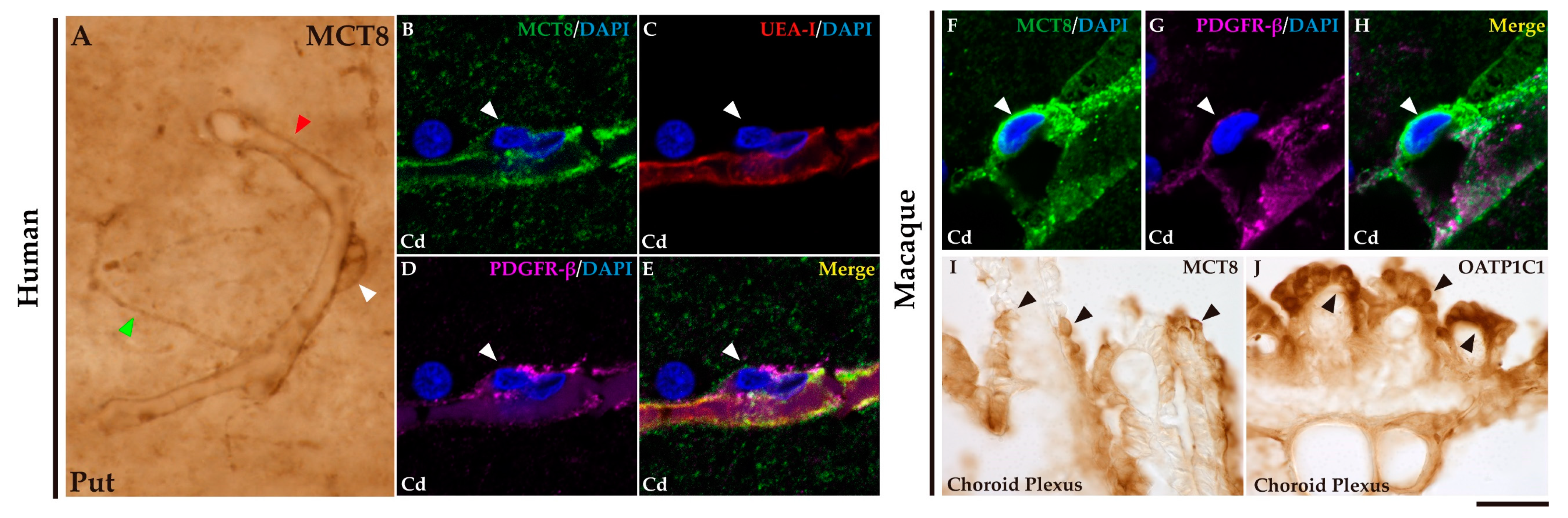
2.2. MCT8 and OATP1C1 in blood vessels and barriers in macaque and human basal ganglia.
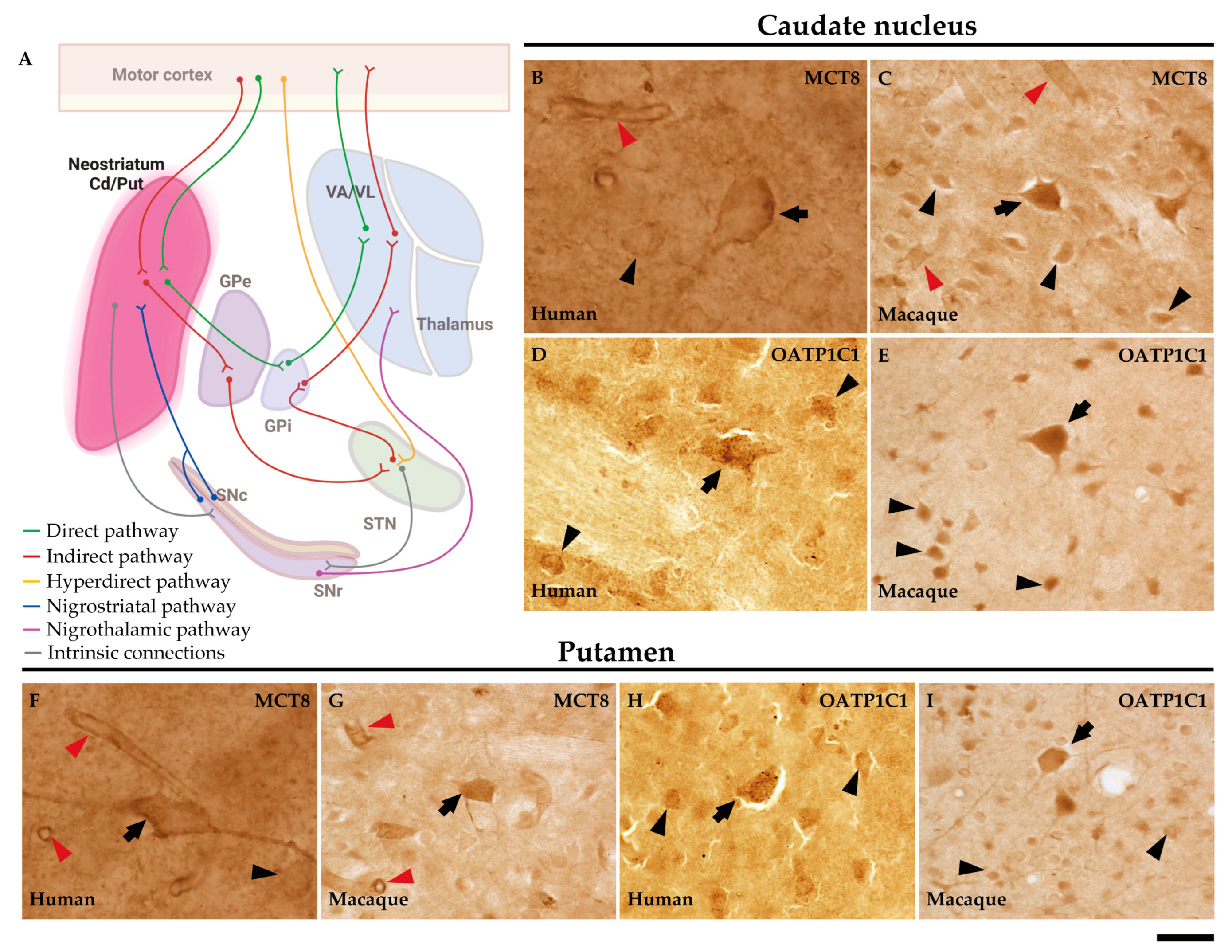
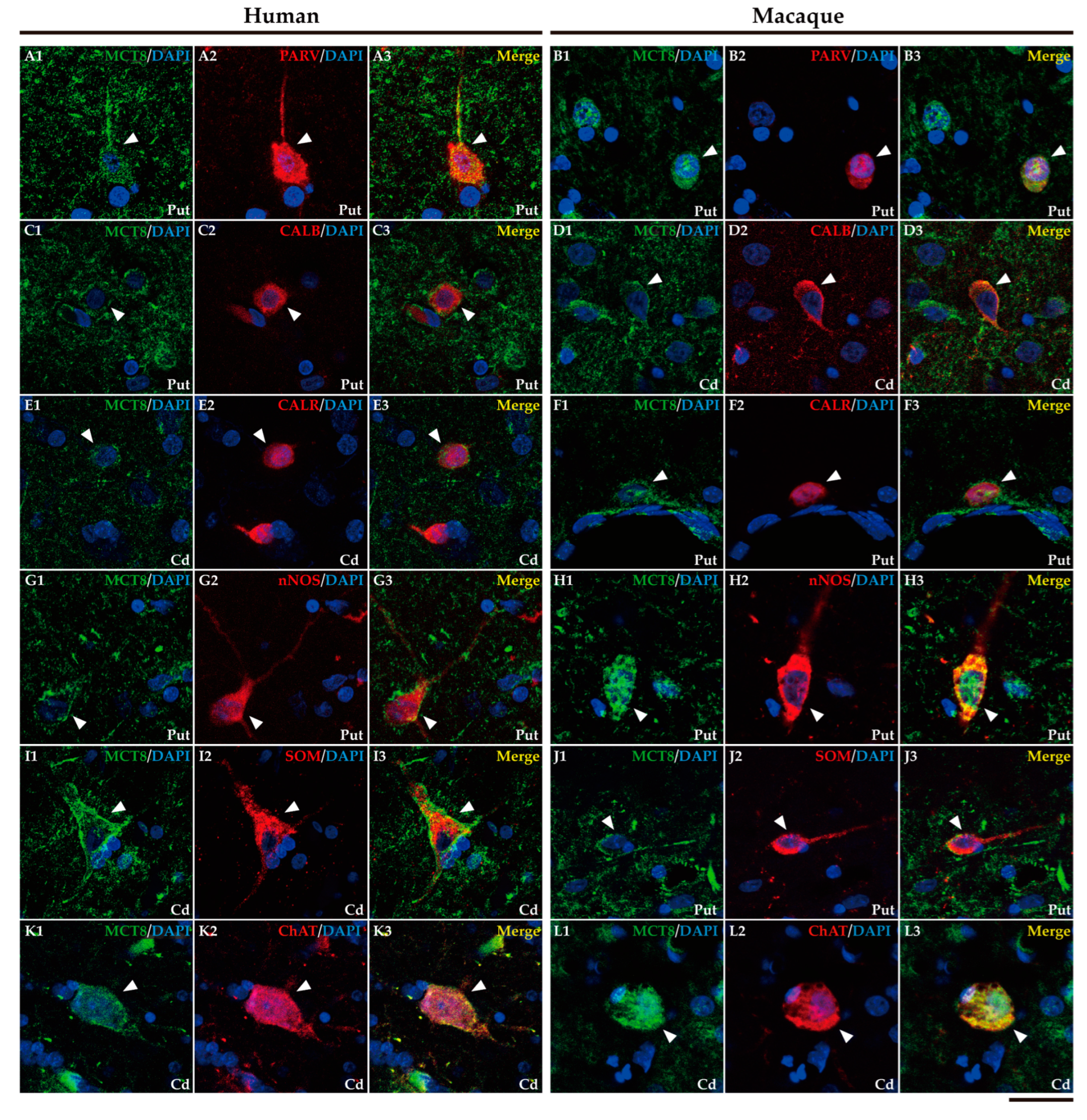
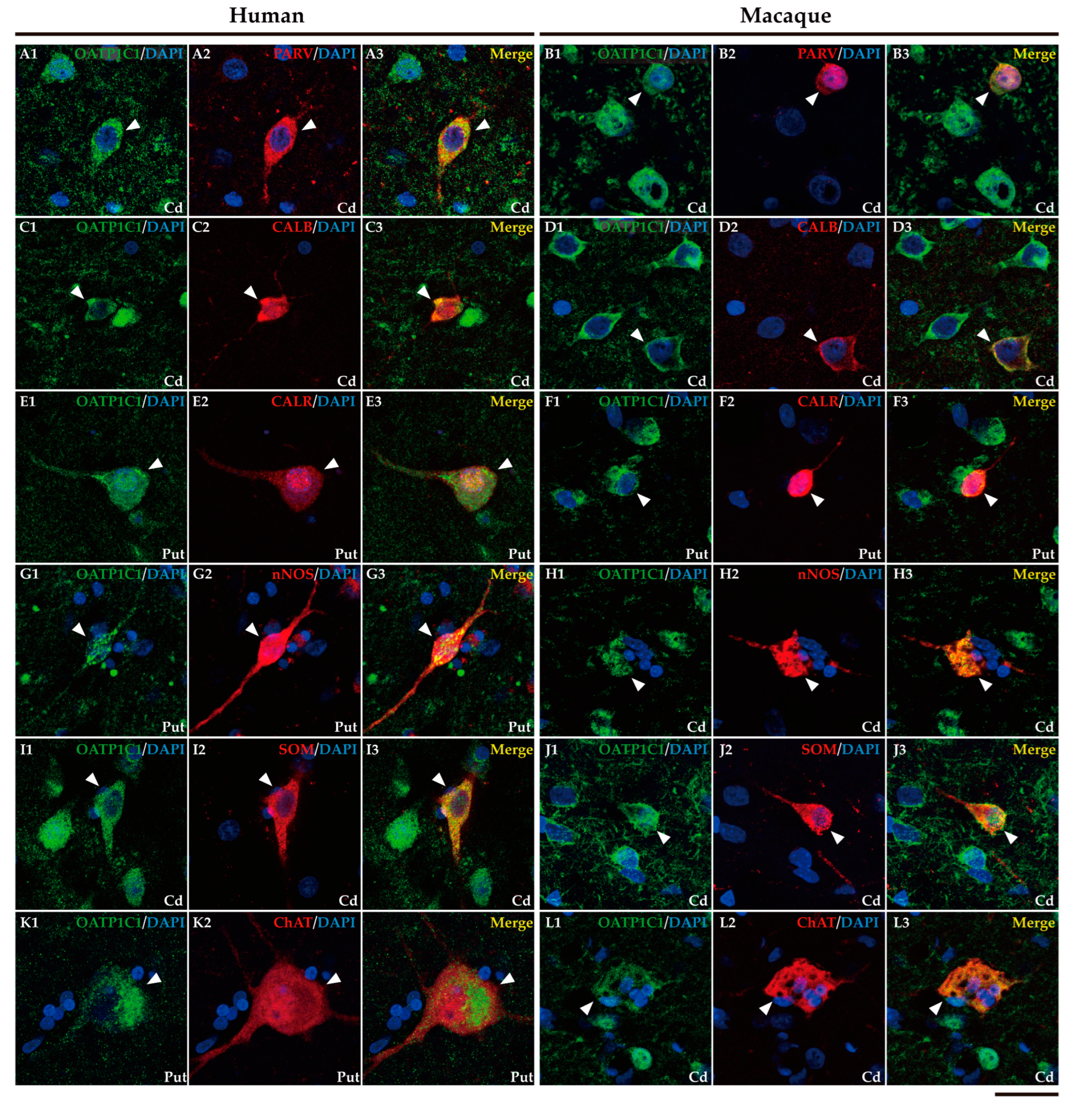
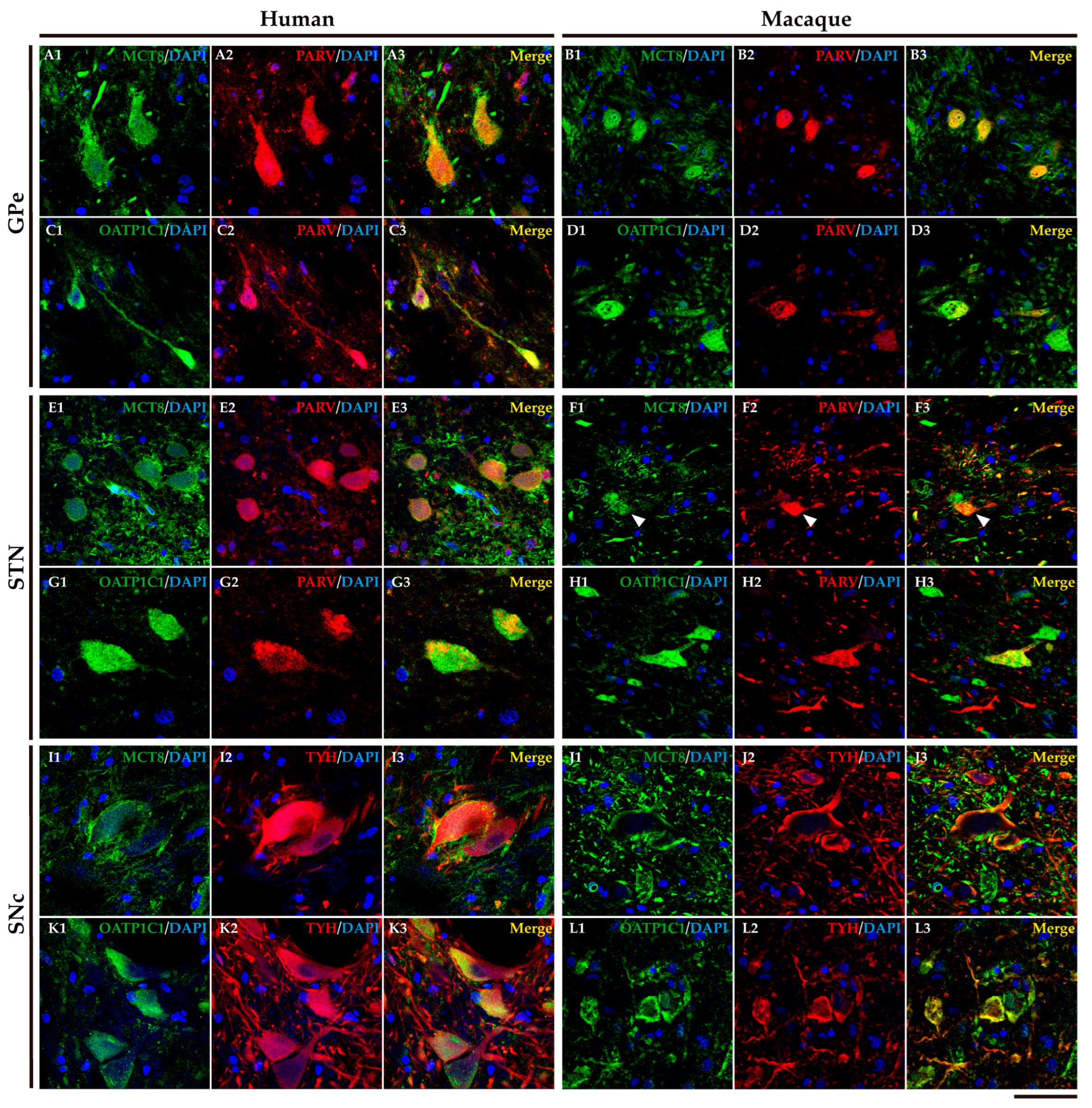
2.3. MCT8 and OATP1C1 are expressed in input nuclei of the human and macaque basal ganglia.
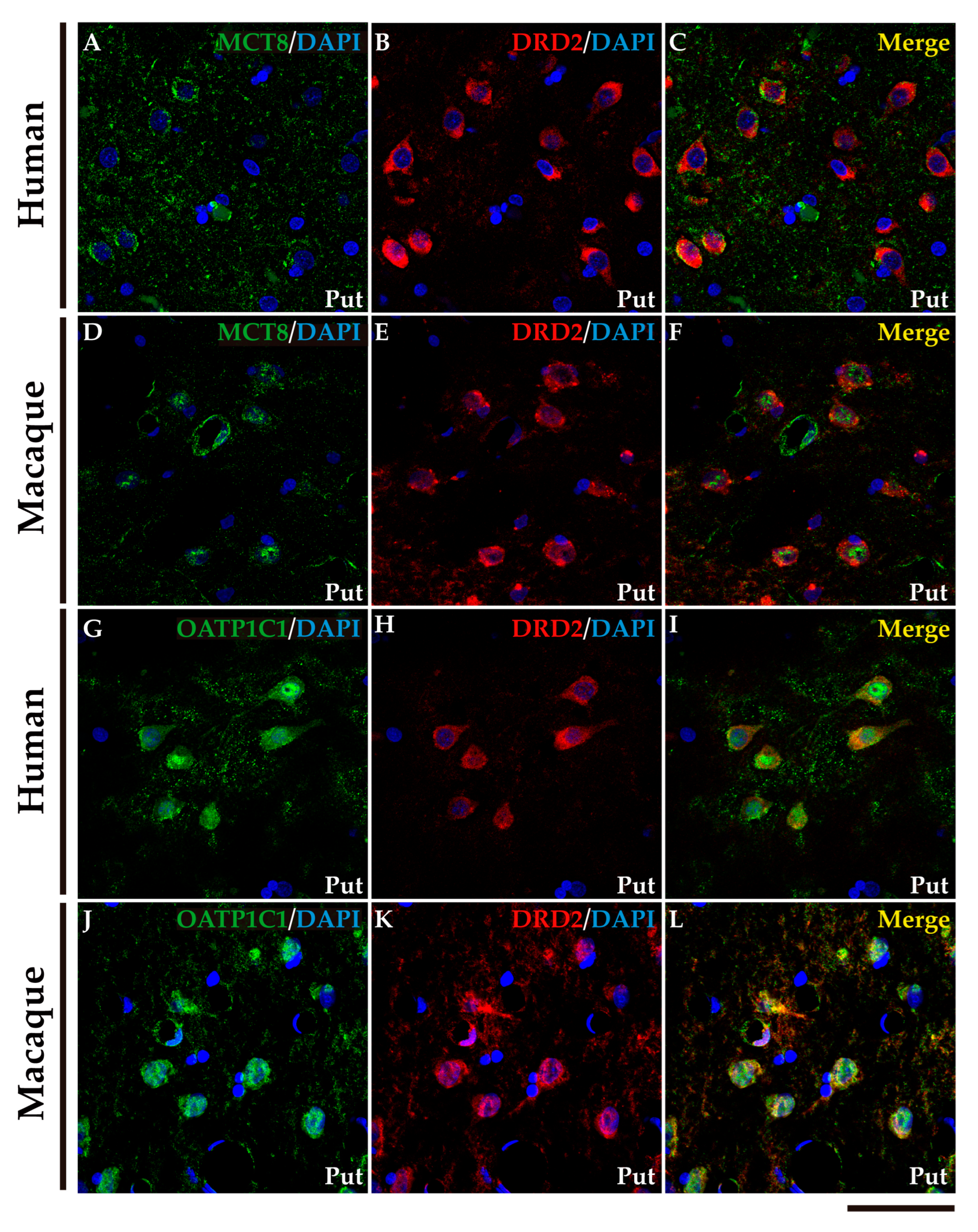
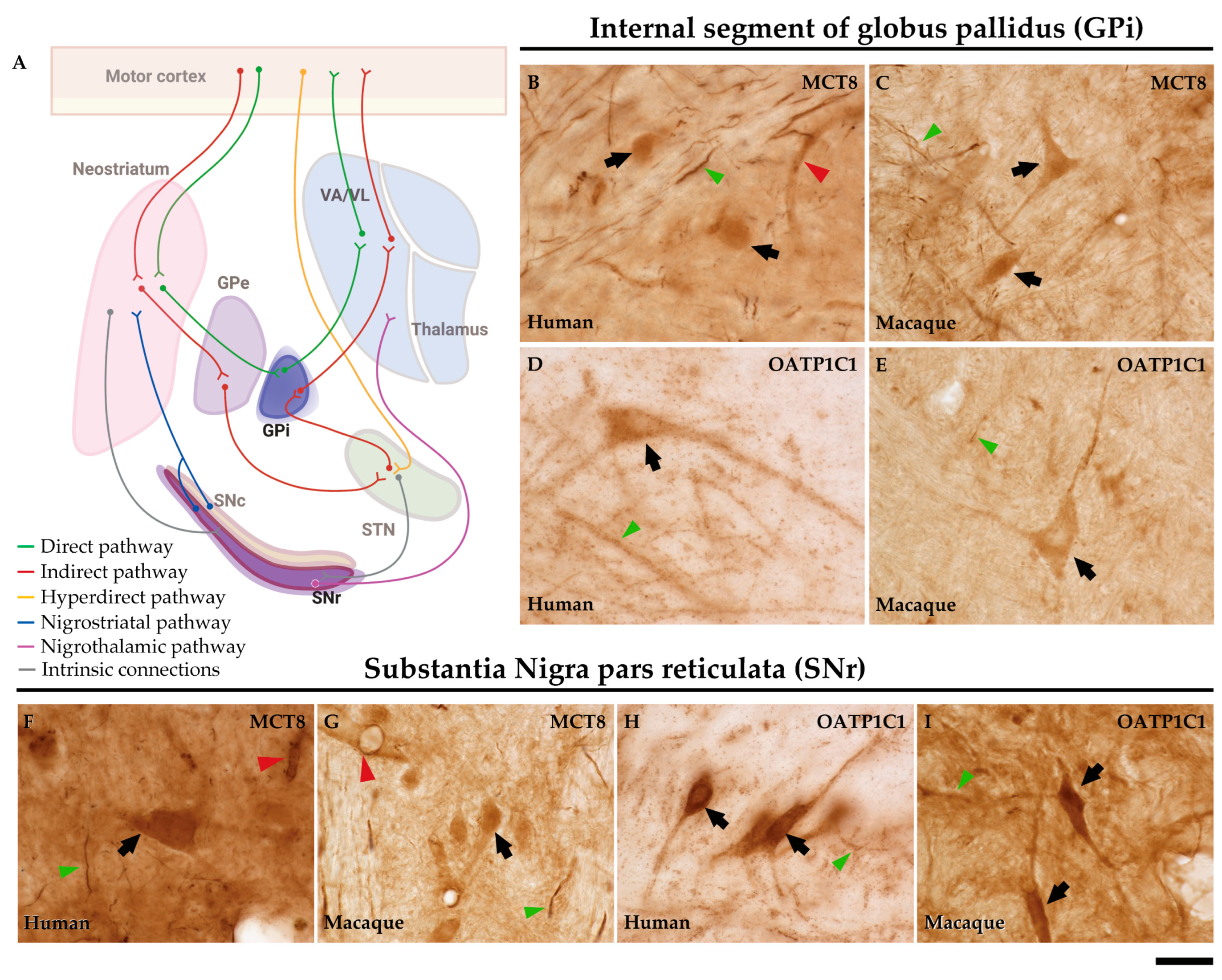
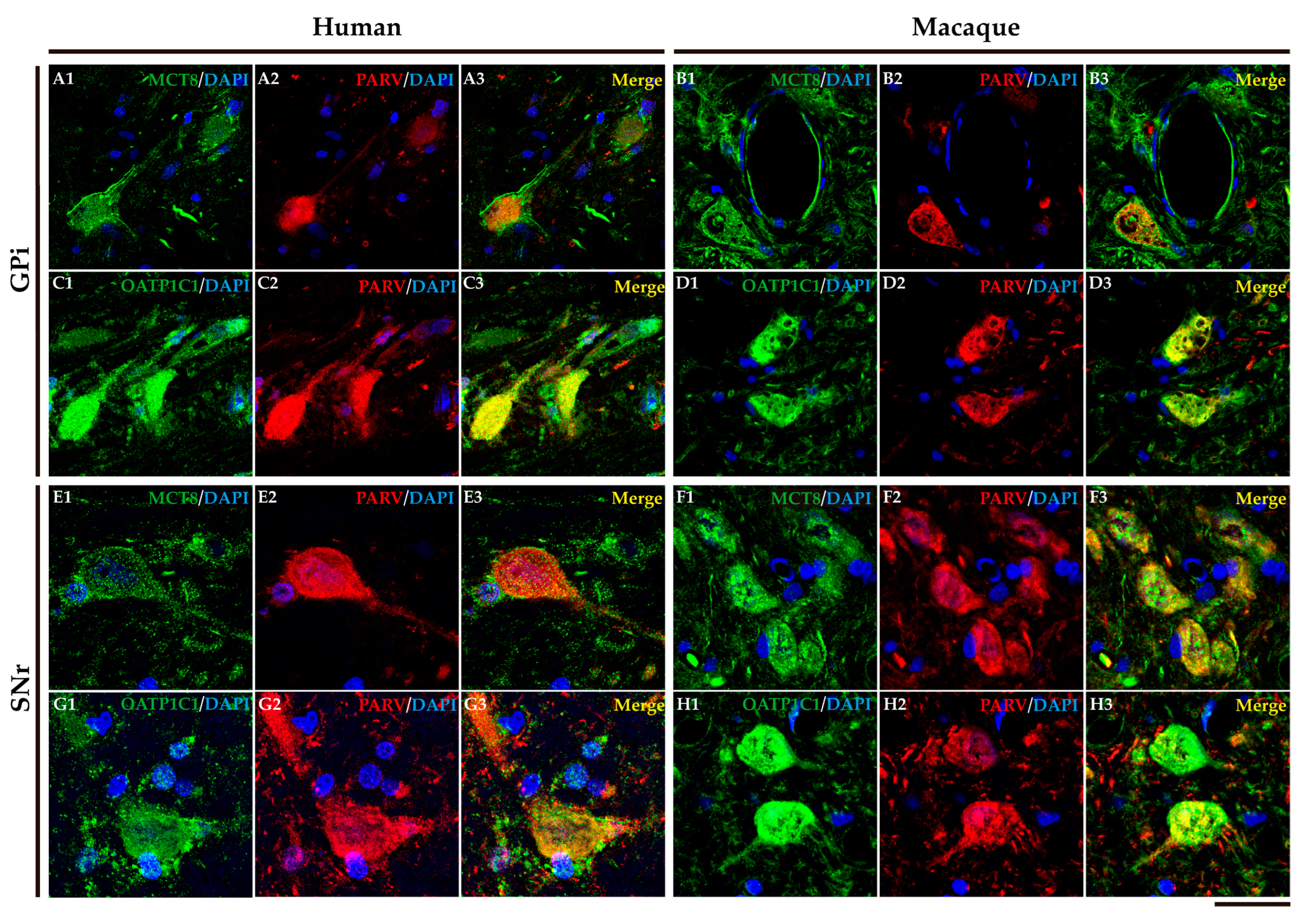
2.4. MCT8 and OATP1C1 are expressed in projection neurons of output nuclei of the human and macaque basal ganglia.
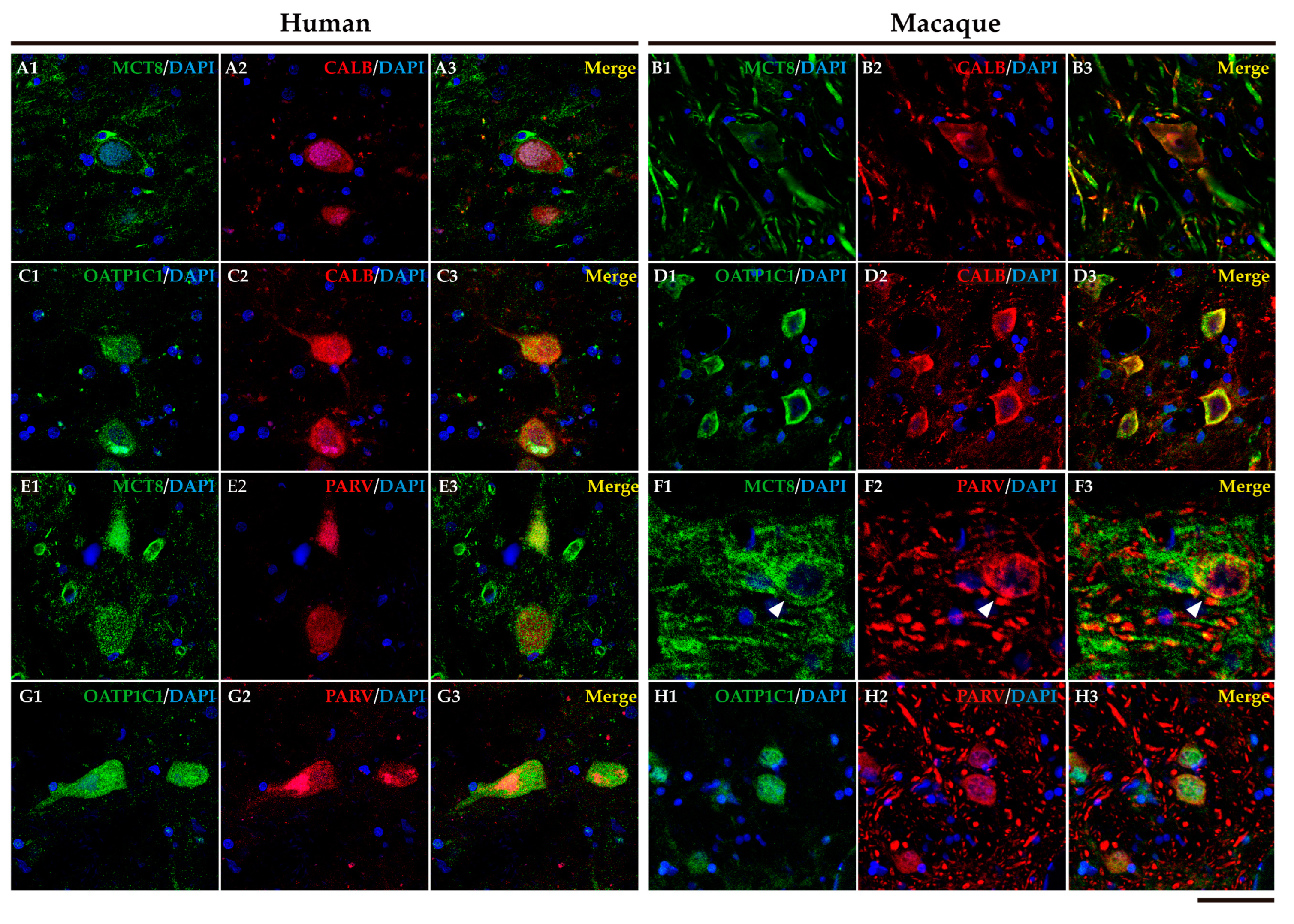
2.5. MCT8 and OAdTP1C1 are expressed in projection neurons of the human and macaque intrinsic nuclei.
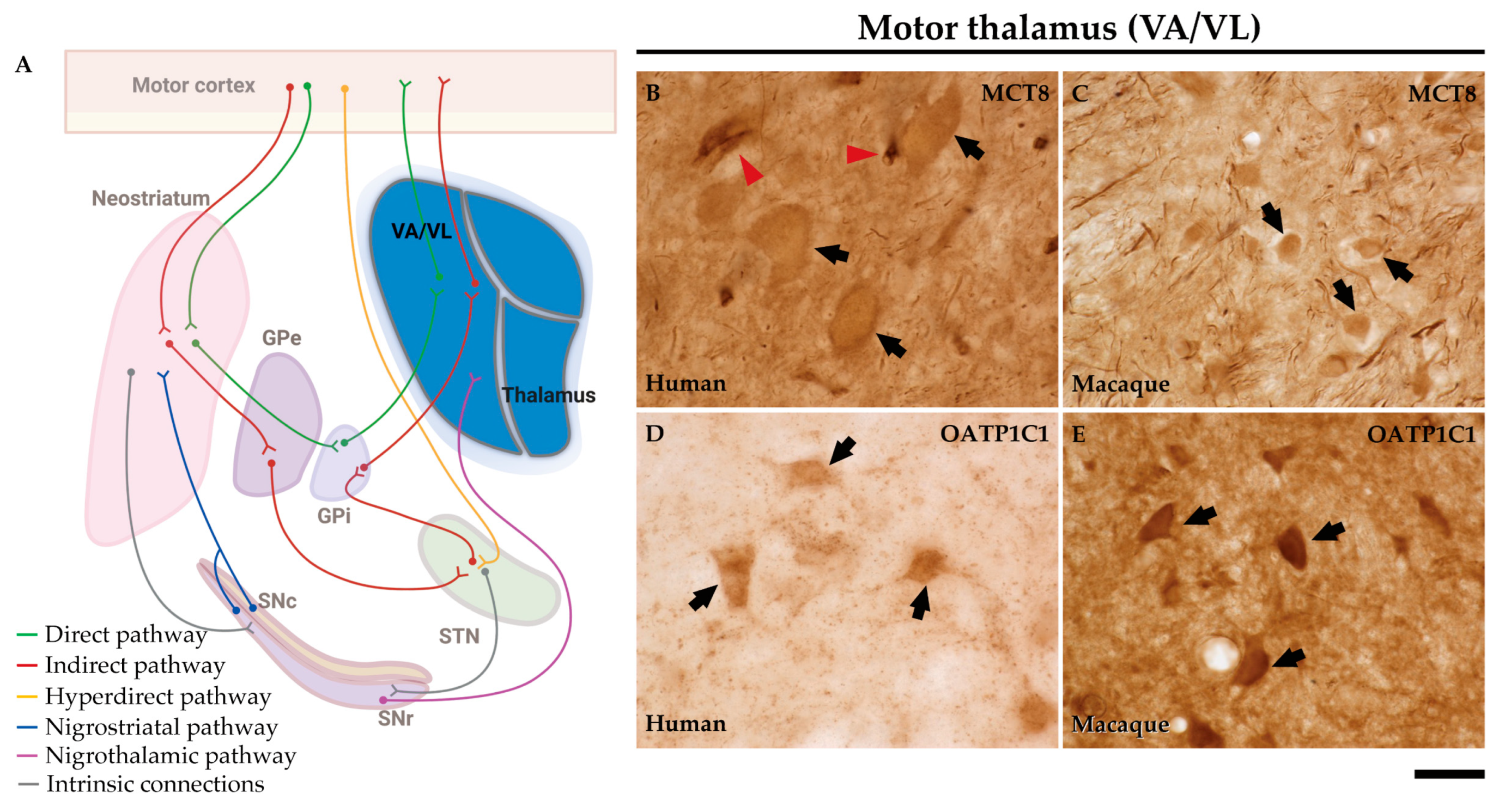
2.6. MCT8 and OATP1C1 are expressed in thalamocortical neurons of the human and macaque motor thalamus.
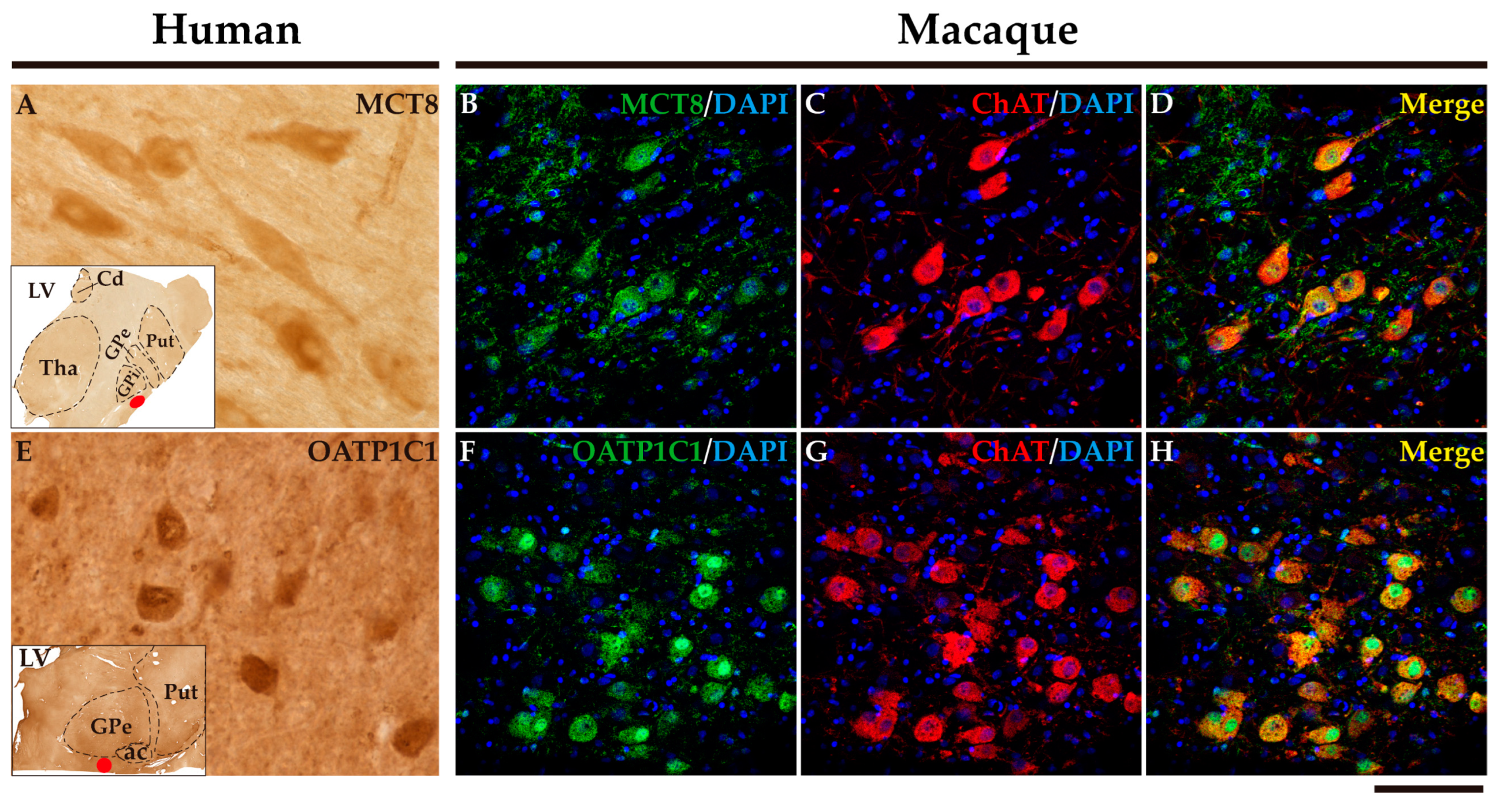
2.7. MCT8 and OATP1C1 are expressed in the cholinergic neurons of the human and macaque nucleus basalis of Meynert.
3. Discussion
3.1. Study limitations.
3.2. Expression pattern of TH transporters in the human and non-human primate basal ganglia and thalamus.
3.3. TH transporters in the nucleus basalis of Meynert.
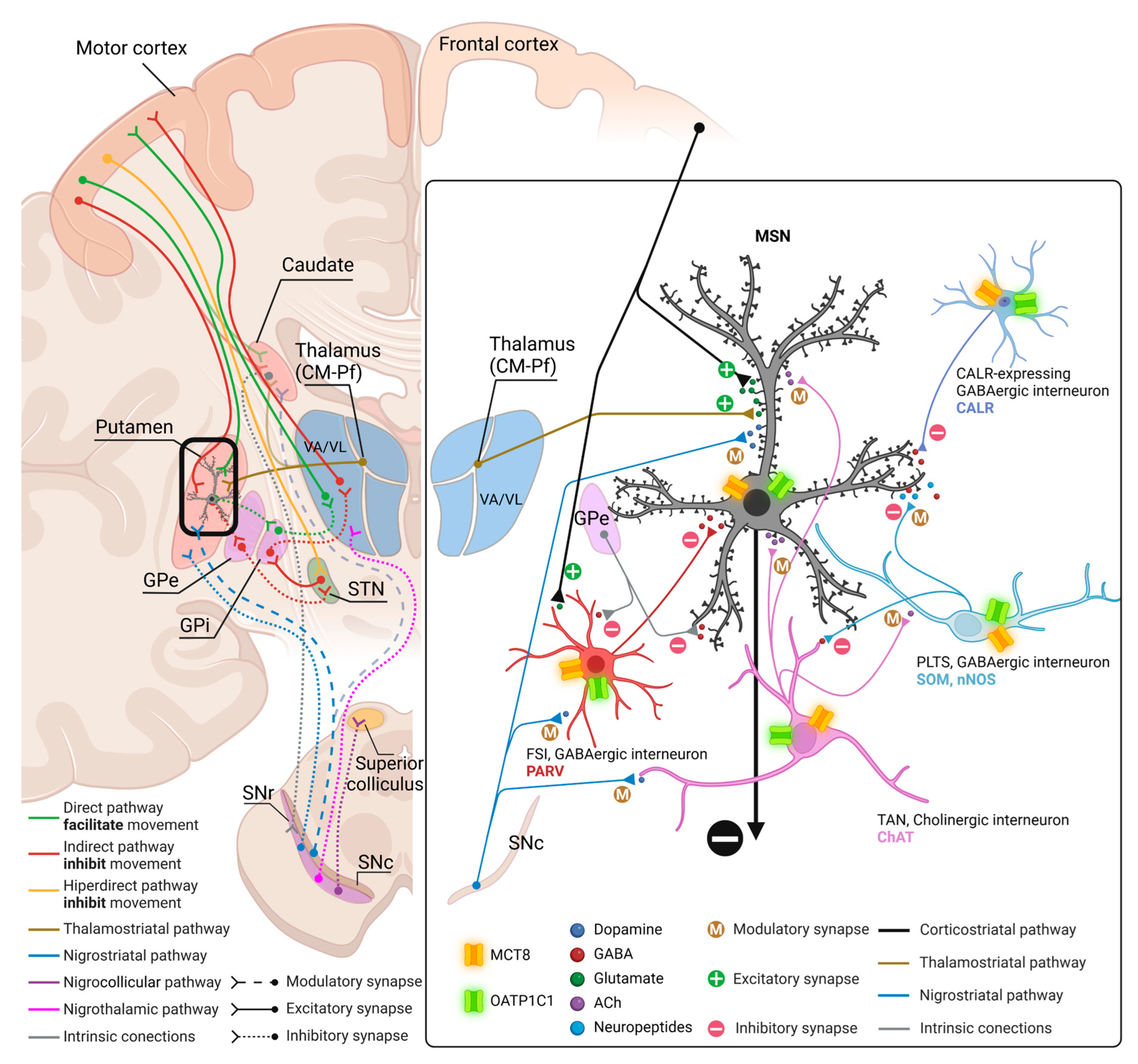
3.4. Model of TH transport in human and non-human primate neostriatum microcircuitry.
4. Materials and Methods
4.1. Human samples and tissue preparation.
4.2. Monkey samples and tissue preparation.
4.3. Histological analysis.
4.4. Immunohistochemistry (IHC).
4.5. Double/triple immunofluorescence (IF).
4.6. Image Acquisition and Processing.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bernal, J.; Guadaño-Ferraz, A.; Morte, B. Perspectives in the study of thyroid hormone action on brain development and function. Thyroid 2003, 13, 1005–1012. [Google Scholar] [CrossRef] [PubMed]
- Tedeschi, L.; Vassalle, C.; Iervasi, G.; Sabatino, L. Main Factors Involved in Thyroid Hormone Action. Molecules 2021, 26, 7337. [Google Scholar] [CrossRef] [PubMed]
- Giammanco, M.; Di Liegro, C.M.; Schiera, G.; Di Liegro, I. Genomic and Non-Genomic Mechanisms of Action of Thyroid Hormones and Their Catabolite 3,5-Diiodo-L-Thyronine in Mammals. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef] [PubMed]
- Bianco, A.C.; Kim, B.W. Deiodinases: implications of the local control of thyroid hormone action. J Clin Invest 2006, 116, 2571–2579. [Google Scholar] [CrossRef] [PubMed]
- Guadaño-Ferraz, A.; Obregon, M.J.; St Germain, D.L.; Bernal, J. The type 2 iodothyronine deiodinase is expressed primarily in glial cells in the neonatal rat brain. Proceedings of the National Academy of Sciences of the United States of America 1997, 94, 10391–10396. [Google Scholar] [CrossRef] [PubMed]
- Friesema, E.C.; Ganguly, S.; Abdalla, A.; Manning Fox, J.E.; Halestrap, A.P.; Visser, T.J. Identification of monocarboxylate transporter 8 as a specific thyroid hormone transporter. J Biol Chem 2003, 278, 40128–40135. [Google Scholar] [CrossRef] [PubMed]
- Pizzagalli, F.; Hagenbuch, B.; Stieger, B.; Klenk, U.; Folkers, G.; Meier, P.J. Identification of a novel human organic anion transporting polypeptide as a high affinity thyroxine transporter. Mol Endocrinol 2002, 16, 2283–2296. [Google Scholar] [CrossRef] [PubMed]
- Bernal, J.; Guadaño-Ferraz, A.; Morte, B. Thyroid hormone transporters-functions and clinical implications. Nat Rev Endocrinol 2015, 11, 406–417. [Google Scholar] [CrossRef]
- Friesema, E.C.; Grueters, A.; Biebermann, H.; Krude, H.; von Moers, A.; Reeser, M.; Barrett, T.G.; Mancilla, E.E.; Svensson, J.; Kester, M.H.; et al. Association between mutations in a thyroid hormone transporter and severe X-linked psychomotor retardation. Lancet (London, England) 2004, 364, 1435–1437. [Google Scholar] [CrossRef]
- Dumitrescu, A.M.; Liao, X.H.; Best, T.B.; Brockmann, K.; Refetoff, S. A novel syndrome combining thyroid and neurological abnormalities is associated with mutations in a monocarboxylate transporter gene. Am J Hum Genet 2004, 74, 168–175. [Google Scholar] [CrossRef]
- Tonduti, D.; Vanderver, A.; Berardinelli, A.; Schmidt, J.L.; Collins, C.D.; Novara, F.; Genni, A.D.; Mita, A.; Triulzi, F.; Brunstrom-Hernandez, J.E.; et al. MCT8 deficiency: extrapyramidal symptoms and delayed myelination as prominent features. J Child Neurol 2013, 28, 795–800. [Google Scholar] [CrossRef] [PubMed]
- Masnada, S.; Sarret, C.; Antonello, C.E.; Fadilah, A.; Krude, H.; Mura, E.; Mordekar, S.; Nicita, F.; Olivotto, S.; Orcesi, S.; et al. Movement disorders in MCT8 deficiency/Allan-Herndon-Dudley Syndrome. Mol Genet Metab 2022, 135, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Remerand, G.; Boespflug-Tanguy, O.; Tonduti, D.; Touraine, R.; Rodriguez, D.; Curie, A.; Perreton, N.; Des Portes, V.; Sarret, C.; Group, R.A.S. Expanding the phenotypic spectrum of Allan-Herndon-Dudley syndrome in patients with SLC16A2 mutations. Dev Med Child Neurol 2019, 61, 1439–1447. [Google Scholar] [CrossRef] [PubMed]
- Ono, E.; Ariga, M.; Oshima, S.; Hayakawa, M.; Imai, M.; Ochiai, Y.; Mochizuki, H.; Namba, N.; Ozono, K.; Miyata, I. Three novel mutations of the MCT8 (SLC16A2) gene: individual and temporal variations of endocrinological and radiological features. Clin Pediatr Endocrinol 2016, 25, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Groeneweg, S.; van Geest, F.S.; Abacı, A.; Alcantud, A.; Ambegaonkar, G.P.; Armour, C.M.; Bakhtiani, P.; Barca, D.; Bertini, E.S.; van Beynum, I.M.; et al. Disease characteristics of MCT8 deficiency: an international, retrospective, multicentre cohort study. Lancet Diabetes Endocrinol 2020, 8, 594–605. [Google Scholar] [CrossRef] [PubMed]
- Xin, Q.; Zhao, P.; Chunquan, C.; Chen, J.; Chen, S.; Sheng, Q. Identification of a novel SLC16A2 mutation that causes Allan-Herndon-Dudley syndrome. Tianjin Med J 2020, 048, 132–136. [Google Scholar] [CrossRef]
- Kubota, M.; Yakuwa, A.; Terashima, H.; Hoshino, H. A nationwide survey of monocarboxylate transporter 8 deficiency in Japan: Its incidence, clinical course, MRI and laboratory findings. Brain Dev 2022, 44, 699–705. [Google Scholar] [CrossRef]
- Chen, X.; Liu, L.; Zeng, C. A novel variant in SLC16A2 associated with typical Allan-Herndon-Dudley syndrome: a case report. BMC Pediatr 2022, 22, 180. [Google Scholar] [CrossRef]
- Liu, Z.; Zhao, S.; Chen, J.; Ma, L.; Shi, Q.; Zhou, Y. A novel frameshift mutation in Allan-Herndon-Dudley syndrome. Int J Legal Med 2022, 136, 1181–1187. [Google Scholar] [CrossRef]
- López-Espíndola, D.; Morales-Bastos, C.; Grijota-Martinez, C.; Liao, X.H.; Lev, D.; Sugo, E.; Verge, C.F.; Refetoff, S.; Bernal, J.; Guadaño-Ferraz, A. Mutations of the thyroid hormone transporter MCT8 cause prenatal brain damage and persistent hypomyelination. J Clin Endocrinol Metab 2014, 99, E2799–2804. [Google Scholar] [CrossRef]
- Masnada, S.; Groenweg, S.; Saletti, V.; Chiapparini, L.; Castellotti, B.; Salsano, E.; Visser, W.E.; Tonduti, D. Novel mutations in SLC16A2 associated with a less severe phenotype of MCT8 deficiency. Metab Brain Dis 2019, 34, 1565–1575. [Google Scholar] [CrossRef] [PubMed]
- Kakinuma, H.; Itoh, M.; Takahashi, H. A novel mutation in the monocarboxylate transporter 8 gene in a boy with putamen lesions and low free T4 levels in cerebrospinal fluid. J Pediatr 2005, 147, 552–554. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, O.; Pfarr, N.; Pohlenz, J.; Schmidt, H. Elevated serum triiodothyronine and intellectual and motor disability with paroxysmal dyskinesia caused by a monocarboxylate transporter 8 gene mutation. Dev Med Child Neurol 2009, 51, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Strømme, P.; Groeneweg, S.; Lima de Souza, E.C.; Zevenbergen, C.; Torgersbråten, A.; Holmgren, A.; Gurcan, E.; Meima, M.E.; Peeters, R.P.; Visser, W.E.; et al. Mutated Thyroid Hormone Transporter OATP1C1 Associates with Severe Brain Hypometabolism and Juvenile Neurodegeneration. Thyroid 2018, 28, 1406–1415. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, T.; Montero-Pedrazuela, A.; Guadaño-Ferraz, A.; Rausell, E. Thyroid Hormone Transporters MCT8 and OATP1C1 Are Expressed in Pyramidal Neurons and Interneurons in the Adult Motor Cortex of Human and Macaque Brain. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Lanciego, J.L.; Luquin, N.; Obeso, J.A. Functional neuroanatomy of the basal ganglia. Cold Spring Harb Perspect Med 2012, 2, a009621. [Google Scholar] [CrossRef] [PubMed]
- Albin, R.L.; Young, A.B.; Penney, J.B. The functional anatomy of disorders of the basal ganglia. Trends Neurosci 1995, 18, 63–64. [Google Scholar] [CrossRef]
- Brazis, P.W.; Masdeu, J.C.; Biller, J. Localization in clinical neurology, Sixth edition; Destefano, F., Ed.; Lippincott Williams & Wilkins, 2012; p. 668. [Google Scholar]
- DeLong, M.R. Primate models of movement disorders of basal ganglia origin. Trends Neurosci 1990, 13, 281–285. [Google Scholar] [CrossRef]
- Gerfen, C.R.; Engber, T.M.; Mahan, L.C.; Susel, Z.; Chase, T.N.; Monsma, F.J., Jr.; Sibley, D.R. D1 and D2 dopamine receptor-regulated gene expression of striatonigral and striatopallidal neurons. Science 1990, 250, 1429–1432. [Google Scholar] [CrossRef]
- Benarroch, E.E. Intrinsic circuits of the striatum: Complexity and clinical correlations. Neurology 2016, 86, 1531–1542. [Google Scholar] [CrossRef]
- Prensa, L.; Gimenez-Amaya, J.M.; Parent, A. Chemical heterogeneity of the striosomal compartment in the human striatum. J Comp Neurol 1999, 413, 603–618. [Google Scholar] [CrossRef]
- Kawaguchi, Y.; Wilson, C.J.; Augood, S.J.; Emson, P.C. Striatal interneurones: chemical, physiological and morphological characterization. Trends Neurosci 1995, 18, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Bernacer, J.; Prensa, L.; Gimenez-Amaya, J.M. Morphological features, distribution and compartmental organization of the nicotinamide adenine dinucleotide phosphate reduced-diaphorase interneurons in the human striatum. J Comp Neurol 2005, 489, 311–327. [Google Scholar] [CrossRef] [PubMed]
- Kita, H. Globus pallidus external segment. In Gaba and the Basal Ganglia - From Molecules to Systems; Progress in Brain Research, 2007; pp. 111–133. [Google Scholar]
- Nambu, A. Globus pallidus internal segment. In Gaba and the Basal Ganglia - From Molecules to Systems; Progress in Brain Research, 2007; pp. 135–150. [Google Scholar]
- Gonzalez-Hernandez, T.; Rodriguez, M. Compartmental organization and chemical profile of dopaminergic and GABAergic neurons in the substantia nigra of the rat. J Comp Neurol 2000, 421, 107–135. [Google Scholar] [CrossRef]
- Levesque, J.C.; Parent, A. GABAergic interneurons in human subthalamic nucleus. Mov Disord 2005, 20, 574–584. [Google Scholar] [CrossRef] [PubMed]
- Jones, E.G. The thalamus; Springer Science & Business Media, 2012. [Google Scholar]
- Rausell, E.; Bae, C.S.; Vinuela, A.; Huntley, G.W.; Jones, E.G. Calbindin and parvalbumin cells in monkey VPL thalamic nucleus: distribution, laminar cortical projections, and relations to spinothalamic terminations. J Neurosci 1992, 12, 4088–4111. [Google Scholar] [CrossRef] [PubMed]
- Rausell, E.; Jones, E.G. Chemically distinct compartments of the thalamic VPM nucleus in monkeys relay principal and spinal trigeminal pathways to different layers of the somatosensory cortex. J Neurosci 1991, 11, 226–237. [Google Scholar] [CrossRef]
- Muller, J.; Heuer, H. Expression pattern of thyroid hormone transporters in the postnatal mouse brain. Front Endocrinol (Lausanne) 2014, 5, 92. [Google Scholar] [CrossRef]
- Heuer, H.; Maier, M.K.; Iden, S.; Mittag, J.; Friesema, E.C.; Visser, T.J.; Bauer, K. The monocarboxylate transporter 8 linked to human psychomotor retardation is highly expressed in thyroid hormone-sensitive neuron populations. Endocrinology 2005, 146, 1701–1706. [Google Scholar] [CrossRef]
- Consortium, G. The GTEx Consortium atlas of genetic regulatory effects across human tissues. Science 2020, 369, 1318–1330. [Google Scholar] [CrossRef]
- Visser, W.E.; Swagemakers, S.M.; Ozgur, Z.; Schot, R.; Verheijen, F.W.; van Ijcken, W.F.; van der Spek, P.J.; Visser, T.J. Transcriptional profiling of fibroblasts from patients with mutations in MCT8 and comparative analysis with the human brain transcriptome. Hum Mol Genet 2010, 19, 4189–4200. [Google Scholar] [CrossRef]
- Grijota-Martínez, C.; Bárez-López, S.; Gómez-Andrés, D.; Guadaño-Ferraz, A. MCT8 Deficiency: The Road to Therapies for a Rare Disease. Front Neurosci 2020, 14, 380. [Google Scholar] [CrossRef] [PubMed]
- Mai, J.K.; Majtanik, M.; Paxinos, G. Atlas of the human brain; Academic Press, 2015. [Google Scholar]
- Winters, W.D.; Kado, R.; Adey, W.R. A stereotaxic brain atlas for Macaca nemestrina; University of California Press, 1969. [Google Scholar]
- Ito, K.; Uchida, Y.; Ohtsuki, S.; Aizawa, S.; Kawakami, H.; Katsukura, Y.; Kamiie, J.; Terasaki, T. Quantitative membrane protein expression at the blood-brain barrier of adult and younger cynomolgus monkeys. J Pharm Sci 2011, 100, 3939–3950. [Google Scholar] [CrossRef] [PubMed]
- <Kim_duke_0066D_15933.pdf>.
- Jones, E.G. Synchrony in the interconnected circuitry of the thalamus and cerebral cortex. Ann N Y Acad Sci 2009, 1157, 10–23. [Google Scholar] [CrossRef] [PubMed]
- Jones, E.G. The thalamic matrix and thalamocortical synchrony. Trends Neurosci 2001, 24, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.K.; Chang, R.C.; Pearce, R.K.; Gentleman, S.M. Nucleus basalis of Meynert revisited: anatomy, history and differential involvement in Alzheimer's and Parkinson's disease. Acta neuropathologica 2015, 129, 527–540. [Google Scholar] [CrossRef] [PubMed]
- Peeters, R.P.; van der Geyten, S.; Wouters, P.J.; Darras, V.M.; van Toor, H.; Kaptein, E.; Visser, T.J.; Van den Berghe, G. Tissue thyroid hormone levels in critical illness. J Clin Endocrinol Metab 2005, 90, 6498–6507. [Google Scholar] [CrossRef]
- Boelen, A.; Kwakkel, J.; Fliers, E. Beyond low plasma T3: local thyroid hormone metabolism during inflammation and infection. Endocr Rev 2011, 32, 670–693. [Google Scholar] [CrossRef]
- López-Espíndola, D.; Garcia-Aldea, A.; Gomez de la Riva, I.; Rodriguez-Garcia, A.M.; Salvatore, D.; Visser, T.J.; Bernal, J.; Guadaño-Ferraz, A. Thyroid hormone availability in the human fetal brain: novel entry pathways and role of radial glia. Brain Struct Funct 2019, 224, 2103–2119. [Google Scholar] [CrossRef]
- Wilpert, N.M.; Krueger, M.; Opitz, R.; Sebinger, D.; Paisdzior, S.; Mages, B.; Schulz, A.; Spranger, J.; Wirth, E.K.; Stachelscheid, H.; et al. Spatiotemporal Changes of Cerebral Monocarboxylate Transporter 8 Expression. Thyroid 2020, 30, 1366–1383. [Google Scholar] [CrossRef]
- Alkemade, A.; Friesema, E.C.; Kalsbeek, A.; Swaab, D.F.; Visser, T.J.; Fliers, E. Expression of thyroid hormone transporters in the human hypothalamus. J Clin Endocrinol Metab 2011, 96, E967–971. [Google Scholar] [CrossRef] [PubMed]
- Alkemade, A.; Friesema, E.C.; Unmehopa, U.A.; Fabriek, B.O.; Kuiper, G.G.; Leonard, J.L.; Wiersinga, W.M.; Swaab, D.F.; Visser, T.J.; Fliers, E. Neuroanatomical pathways for thyroid hormone feedback in the human hypothalamus. J Clin Endocrinol Metab 2005, 90, 4322–4334. [Google Scholar] [CrossRef] [PubMed]
- Friesema, E.C.; Visser, T.J.; Borgers, A.J.; Kalsbeek, A.; Swaab, D.F.; Fliers, E.; Alkemade, A. Thyroid hormone transporters and deiodinases in the developing human hypothalamus. European journal of endocrinology 2012, 167, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Hennemann, G.; Docter, R.; Friesema, E.C.; de Jong, M.; Krenning, E.P.; Visser, T.J. Plasma membrane transport of thyroid hormones and its role in thyroid hormone metabolism and bioavailability. Endocr Rev 2001, 22, 451–476. [Google Scholar] [CrossRef] [PubMed]
- Visser, W.E.; Friesema, E.C.; Jansen, J.; Visser, T.J. Thyroid hormone transport in and out of cells. Trends Endocrinol Metab 2008, 19, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Morreale de Escobar, G.; Obregon, M.J.; Escobar del Rey, F. Role of thyroid hormone during early brain development. Eur J Endocrinol 2004, 151 Suppl 3, U25–37. [Google Scholar] [CrossRef]
- Lu, E.J.; Brown, W.J. An electron microscopic study of the developing caudate nucleus in euthyroid and hypothyroid states. Anat Embryol (Berl) 1977, 150, 335–364. [Google Scholar] [CrossRef]
- Morte, B.; Gil-Ibanez, P.; Heuer, H.; Bernal, J. Brain Gene Expression in Systemic Hypothyroidism and Mouse Models of MCT8 Deficiency: The Mct8-Oatp1c1-Dio2 Triad. Thyroid 2021, 31, 985–993. [Google Scholar] [CrossRef]
- Vargiu, P.; Morte, B.; Manzano, J.; Perez, J.; de Abajo, R.; Gregor Sutcliffe, J.; Bernal, J. Thyroid hormone regulation of rhes, a novel Ras homolog gene expressed in the striatum. Brain Res Mol Brain Res 2001, 94, 1–8. [Google Scholar] [CrossRef]
- Manzano, J.; Morte, B.; Scanlan, T.S.; Bernal, J. Differential effects of triiodothyronine and the thyroid hormone receptor beta-specific agonist GC-1 on thyroid hormone target genes in the brain. Endocrinology 2003, 144, 5480–5487. [Google Scholar] [CrossRef]
- Vallortigara, J.; Chassande, O.; Higueret, P.; Enderlin, V. Thyroid hormone receptor alpha plays an essential role in the normalisation of adult-onset hypothyroidism-related hypoexpression of synaptic plasticity target genes in striatum. J Neuroendocrinol 2009, 21, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Dussault, J.H.; Ruel, J. Thyroid hormones and brain development. Annu Rev Physiol 1987, 49, 321–334. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.D.; Butcher, L.L.; Woolf, N.J. Thyroid hormone modulates the development of cholinergic terminal fields in the rat forebrain: relation to nerve growth factor receptor. Brain Res Dev Brain Res 1991, 59, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.J.; Hayashi, M.; Hunt, A. Role of thyroid hormone and nerve growth factor in the development of choline acetyltransferase and other cell-specific marker enzymes in the basal forebrain of the rat. J Neurochem 1988, 50, 803–811. [Google Scholar] [CrossRef]
- Patel, A.J.; Hayashi, M.; Hunt, A. Selective persistent reduction in choline acetyltransferase activity in basal forebrain of the rat after thyroid deficiency during early life. Brain Res 1987, 422, 182–185. [Google Scholar] [CrossRef] [PubMed]
- Gould, E.; Butcher, L.L. Developing cholinergic basal forebrain neurons are sensitive to thyroid hormone. J Neurosci 1989, 9, 3347–3358. [Google Scholar] [CrossRef]
- Calza, L.; Giardino, L.; Aloe, L. Thyroid hormone regulates NGF content and p75LNGFR expression in the basal forebrain of adult rats. Exp Neurol 1997, 143, 196–206. [Google Scholar] [CrossRef]
- Sarkar, P.K.; Ray, A.K. Involvement of L-triiodothyronine in acetylcholine metabolism in adult rat cerebrocortical synaptosomes. Horm Metab Res 2001, 33, 270–275. [Google Scholar] [CrossRef]
- Moskovkin, G.N.; Kalman, M.; Kardos, J.; Yarigin, K.N.; Hajos, F. Effect of triiodothyronine on the muscarinic receptors and acetylcholinesterase activity of developing rat brain. Int J Neurosci 1989, 44, 83–89. [Google Scholar] [CrossRef]
- Lebel, J.M.; Dussault, J.H.; Puymirat, J. Overexpression of the beta 1 thyroid receptor induces differentiation in neuro-2a cells. Proceedings of the National Academy of Sciences of the United States of America 1994, 91, 2644–2648. [Google Scholar] [CrossRef]
- Hasselmo, M.E.; Bower, J.M. Acetylcholine and memory. Trends Neurosci 1993, 16, 218–222. [Google Scholar] [CrossRef] [PubMed]
- Sarter, M.; Bruno, J.P. Cognitive functions of cortical acetylcholine: toward a unifying hypothesis. Brain Res Brain Res Rev 1997, 23, 28–46. [Google Scholar] [CrossRef] [PubMed]
- Hasselmo, M.E. The role of acetylcholine in learning and memory. Curr Opin Neurobiol 2006, 16, 710–715. [Google Scholar] [CrossRef]
- Quirin-Stricker, C.; Nappey, V.; Simoni, P.; Toussaint, J.L.; Schmitt, M. Trans-activation by thyroid hormone receptors of the 5' flanking region of the human ChAT gene. Brain Res Mol Brain Res 1994, 23, 253–265. [Google Scholar] [CrossRef]
- Juarez de Ku, L.M.; Sharma-Stokkermans, M.; Meserve, L.A. Thyroxine normalizes polychlorinated biphenyl (PCB) dose-related depression of choline acetyltransferase (ChAT) activity in hippocampus and basal forebrain of 15-day-old rats. Toxicology 1994, 94, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Sawin, S.; Brodish, P.; Carter, C.S.; Stanton, M.E.; Lau, C. Development of cholinergic neurons in rat brain regions: dose-dependent effects of propylthiouracil-induced hypothyroidism. Neurotoxicol Teratol 1998, 20, 627–635. [Google Scholar] [CrossRef]
- Provost, T.L.; Juarez de Ku, L.M.; Zender, C.; Meserve, L.A. Dose- and age-dependent alterations in choline acetyltransferase (ChAT) activity, learning and memory, and thyroid hormones in 15- and 30-day old rats exposed to 1.25 or 12.5 PPM polychlorinated biphenyl (PCB) beginning at conception. Prog Neuropsychopharmacol Biol Psychiatry 1999, 23, 915–928. [Google Scholar] [CrossRef]
- Wilson, C.J. The generation of natural firing patterns in neostriatal neurons. Prog Brain Res 1993, 99, 277–297. [Google Scholar] [CrossRef]
- Berke, J.D.; Okatan, M.; Skurski, J.; Eichenbaum, H.B. Oscillatory entrainment of striatal neurons in freely moving rats. Neuron 2004, 43, 883–896. [Google Scholar] [CrossRef]
- Adler, A.; Katabi, S.; Finkes, I.; Israel, Z.; Prut, Y.; Bergman, H. Temporal convergence of dynamic cell assemblies in the striato-pallidal network. J Neurosci 2012, 32, 2473–2484. [Google Scholar] [CrossRef]
- Purves, D.; Augustine, G.J.; Fitzpatrick, D.; Hall, W.C.; LaManita, A.-S.; White, L.E. Neuroscience, (5th ed.); Sinauer Associates, 2012; p. 759. [Google Scholar]
- Iglesias, J.E.; Insausti, R.; Lerma-Usabiaga, G.; Bocchetta, M.; Van Leemput, K.; Greve, D.N.; van der Kouwe, A.; Alzheimer's Disease Neuroimaging, I.; Fischl, B.; Caballero-Gaudes, C.; et al. A probabilistic atlas of the human thalamic nuclei combining ex vivo MRI and histology. Neuroimage 2018, 183, 314–326. [Google Scholar] [CrossRef] [PubMed]
- Uroz, V.; Prensa, L.; Gimenez-Amaya, J.M. Chemical anatomy of the human paraventricular thalamic nucleus. Synapse 2004, 51, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Guadaño-Ferraz, A.; Vinuela, A.; Oeding, G.; Bernal, J.; Rausell, E. RC3/neurogranin is expressed in pyramidal neurons of motor and somatosensory cortex in normal and denervated monkeys. J Comp Neurol 2005, 493, 554–570. [Google Scholar] [CrossRef] [PubMed]
- Emmers, R.; Akert, K. A stereotaxic atlas of the brain of the squirrel monkey (Saimiri sciureus); The University of Wisconsin Press, 1963. [Google Scholar]
- Smyth, L.C.D.; Rustenhoven, J.; Scotter, E.L.; Schweder, P.; Faull, R.L.M.; Park, T.I.H.; Dragunow, M. Markers for human brain pericytes and smooth muscle cells. J Chem Neuroanat 2018, 92, 48–60. [Google Scholar] [CrossRef]
- Kadota, E.; Tanji, K.; Nishida, S.; Takahashi, M.; Maeda, M.; Hiruma, S.; Enomoto, Y.; Hashimoto, S.; Akai, F. Lectin (UEA-1) reaction of capillary endothelium with reference to permeability in autopsied cases of cerebral infarction. Histol Histopathol 1986, 1, 219–226. [Google Scholar]
- Rausell, E.; Avendano, C. Thalamocortical neurons projecting to superficial and to deep layers in parietal, frontal and prefrontal regions in the cat. Brain Res 1985, 347, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Gerfen, C.R.; Baimbridge, K.G.; Miller, J.J. The neostriatal mosaic: compartmental distribution of calcium-binding protein and parvalbumin in the basal ganglia of the rat and monkey. Proceedings of the National Academy of Sciences of the United States of America 1985, 82, 8780–8784. [Google Scholar] [CrossRef]


| Sex | Age (y) | Postmortem interval (h) | Brain weight (g) | Cause of death |
|---|---|---|---|---|
| Male | 29 | 4 | 1500 | Lung tumor |
| Male | 32 | 3 | 1420 | Hemorrhagic gastroenteritis |
| Male | 54 | 12 | 1350 | Aortic aneurysm |
| Male | 59 | <24 | 1020 | Pneumonia |
| Male | 86 | <24 | - | - |
| Male | 97 | 9 | 1238 | Septic shock |
| Female | 98 | 6 | 1168 | - |
| Species | Age (y) | Sex |
|---|---|---|
| M. fascicularis | 3 | Female |
| Saimiri sciureus | 3 | Female |
| M. fascicularis | 5 | Male |
| M. fascicularis | 5 | Male |
| M. fascicularis | 5 | Female |
| M. fascicularis | 5 | Male |
| M. fascicularis | 5 | Male |
| M. fascicularis | 5 | Female |
| M. fascicularis | 5 | Male |
| M. mulatta | 7 | Male |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




