Submitted:
09 April 2023
Posted:
11 April 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
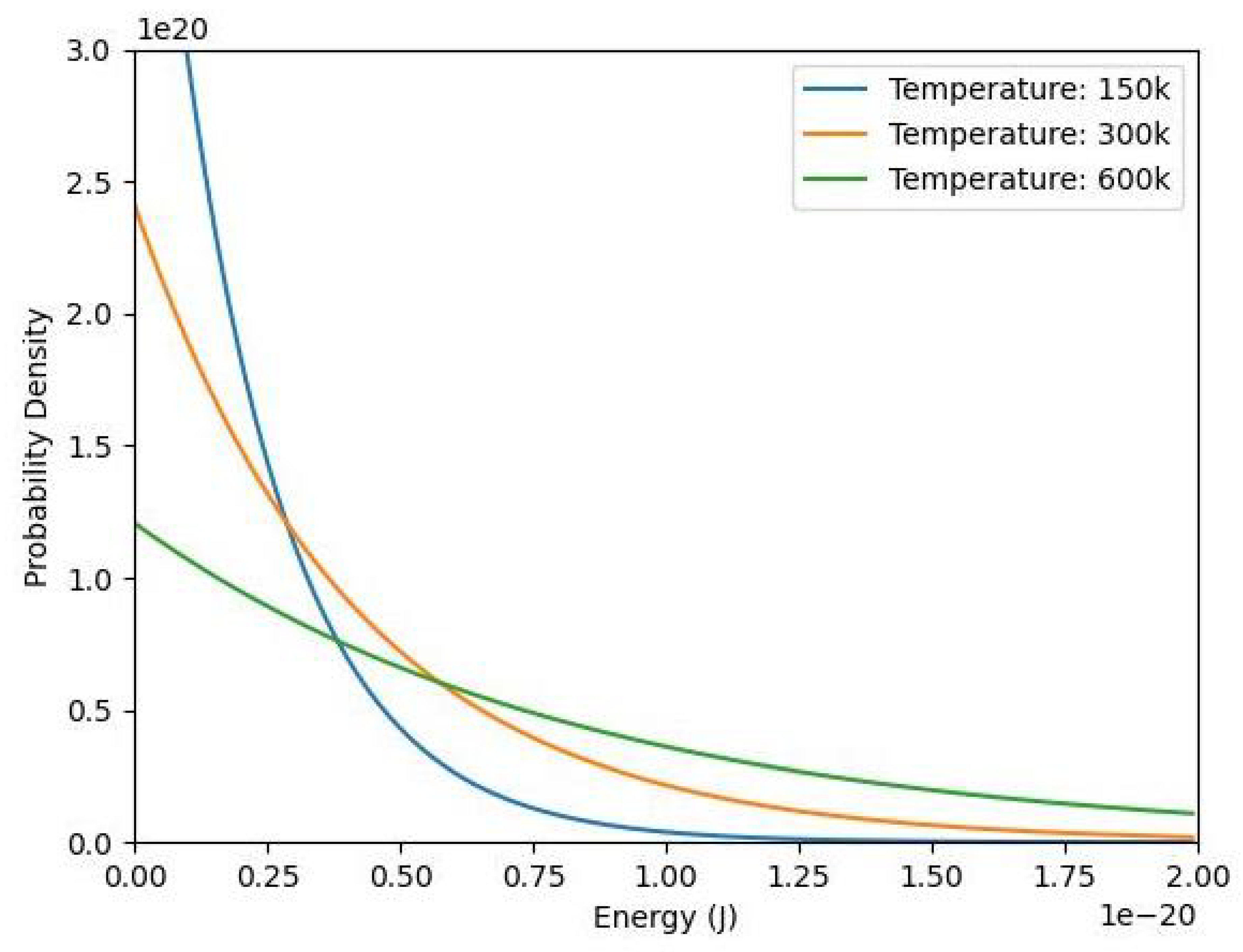
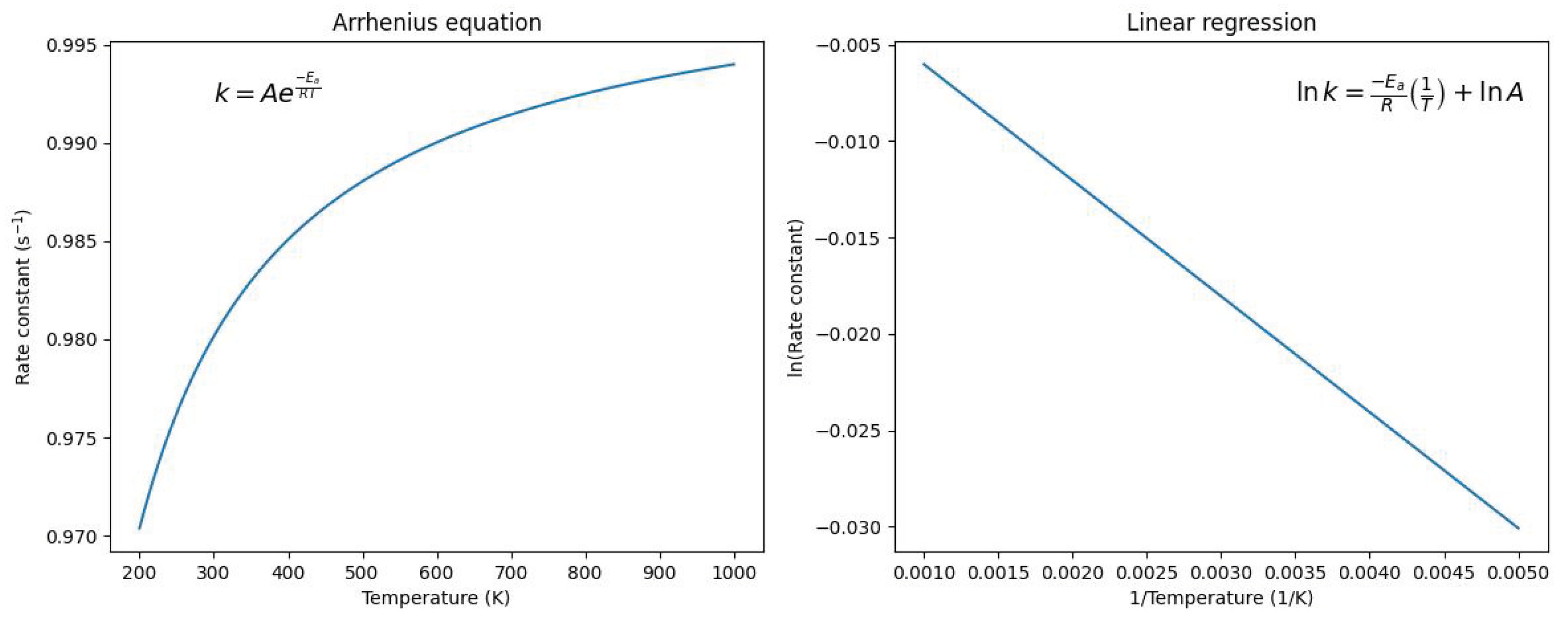
2. Exponential Term

3. Modelling the Pre-exponential Factor
3.1. Collision Frequency
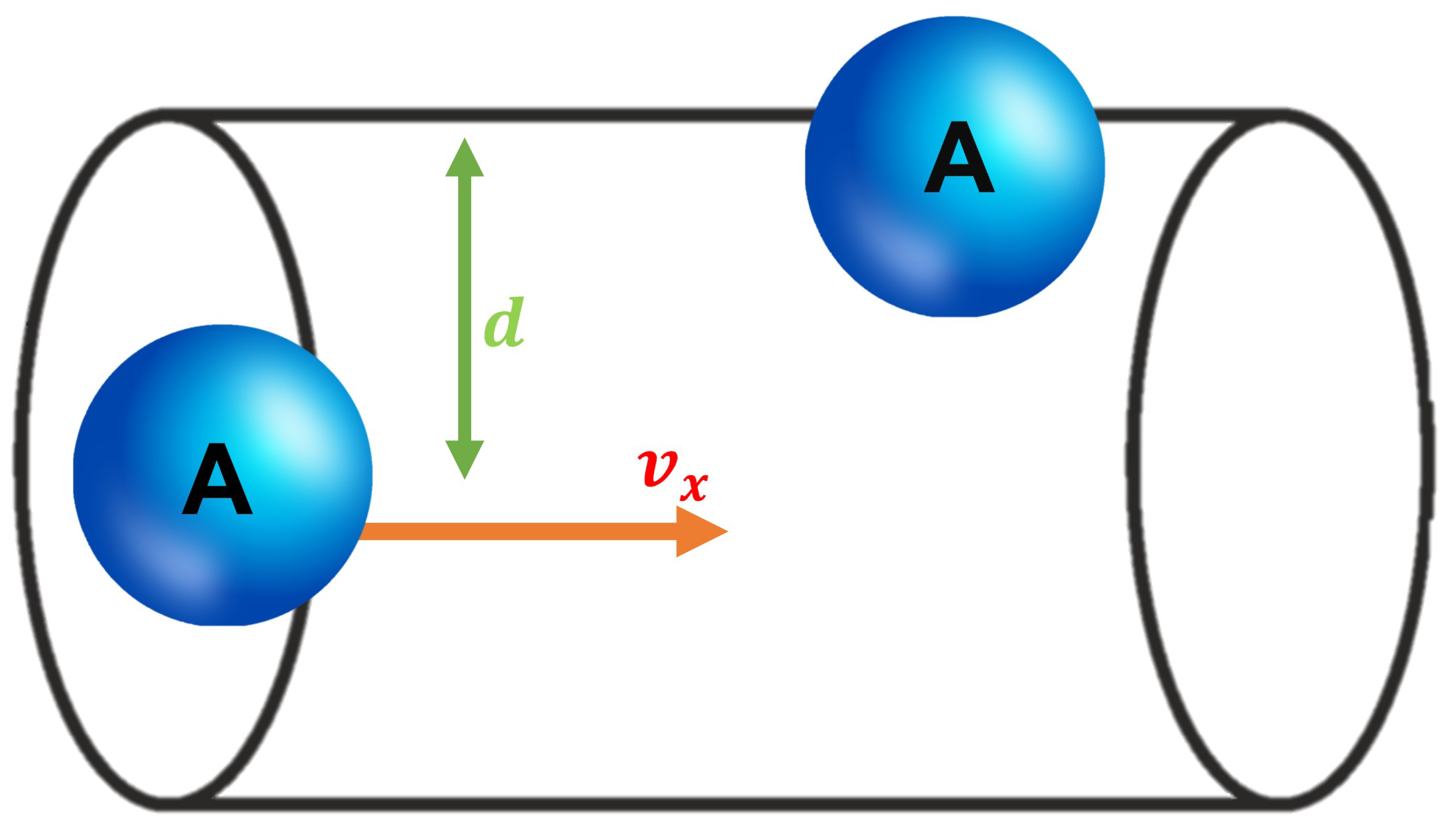
3.1.1. Volume of Collision Cylinder
3.1.2. Collision Frequency for a Single Particle A
3.1.3. Collision Frequency for all Particles A
3.1.4. Collision System Between Different Types of Molecules
- 1.
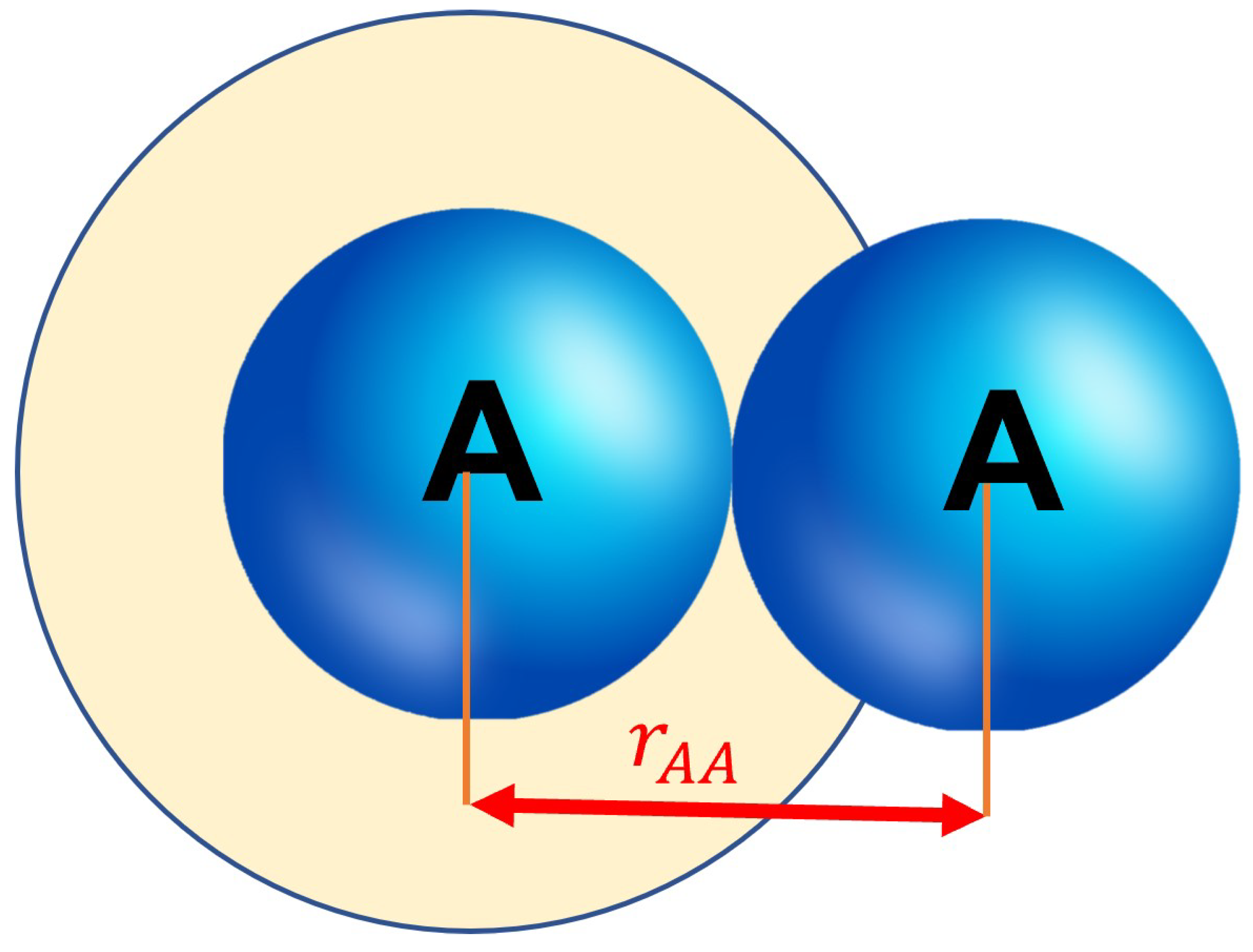
- The collisional cross section should take into account the radius of both particles. Thus should be replaced with .
- 2.
- The number of particles A and particles B could differ significantly within the container. To find the probability of a collision between these two types of particles, the particle density within the collisional cylinder should be replaced with the number density of the second particle, .
- 3.
- There is no need to divide the collision frequency by two (look at equation 7) since we are now only counting the frequency of an A molecule striking B and not vice versa.
- 4.
- Since we are considering a two bodied system, the mean speed of particle A should be replaced by the relative speed of particles A and B, .
3.2. Pre-Exponential Factor
3.2.1. Reaction Rate According to the Collision Theories
- 1.
- The ratio of particles possessing a sufficient amount of kinetic energy. When colliding, this energy is transformed to potential energy in order to bypass the activation energy barrier, .
- 2.
- The number of collisions within a unit time period.
- 3.
- The particles collide in the exact geometry in such a way that their electrons cloud interact with one another. This coefficient is called the orientation factor. 1

3.3. Limitations of the Model
- 1.
- It is assumed that all particles behave like hard spheres with a defined radius and a defined position. However, in reality, atoms react when their electron clouds come sufficiently close together to interact with one another. The electron cloud is consisted of a probability range, which means there is not a precisely defined distance below which atoms react.
- 2.
- It is assumed that every collision in which the particles possess sufficient kinetic energy will undergo chemical transformation. This statement does not reflect the reality, as molecules must imperatively hit one another in the precise geometry to form an activated complex.
- 3.
- In the collision theory, only the kinetic energy of particles is considered, while rotation and vibration energies are ignored.
4. Conclusion
References
- McQuarrie, D.; Simon, J. Physical Chemistry: A Molecular Approach, Chapter 4; Physical Chemistry: A Molecular Approach, University Science Books, 1997.
- Laidler, K.J. Chemical Kinetics, chapter 4; Chemical Kinetics, Person, 1987.
| 1 | The orientation factor will not be discussed in this demonstration as it is highly dependent on the type of reactant and cannot be generalized in by a simple formula. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



