Submitted:
06 April 2023
Posted:
07 April 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
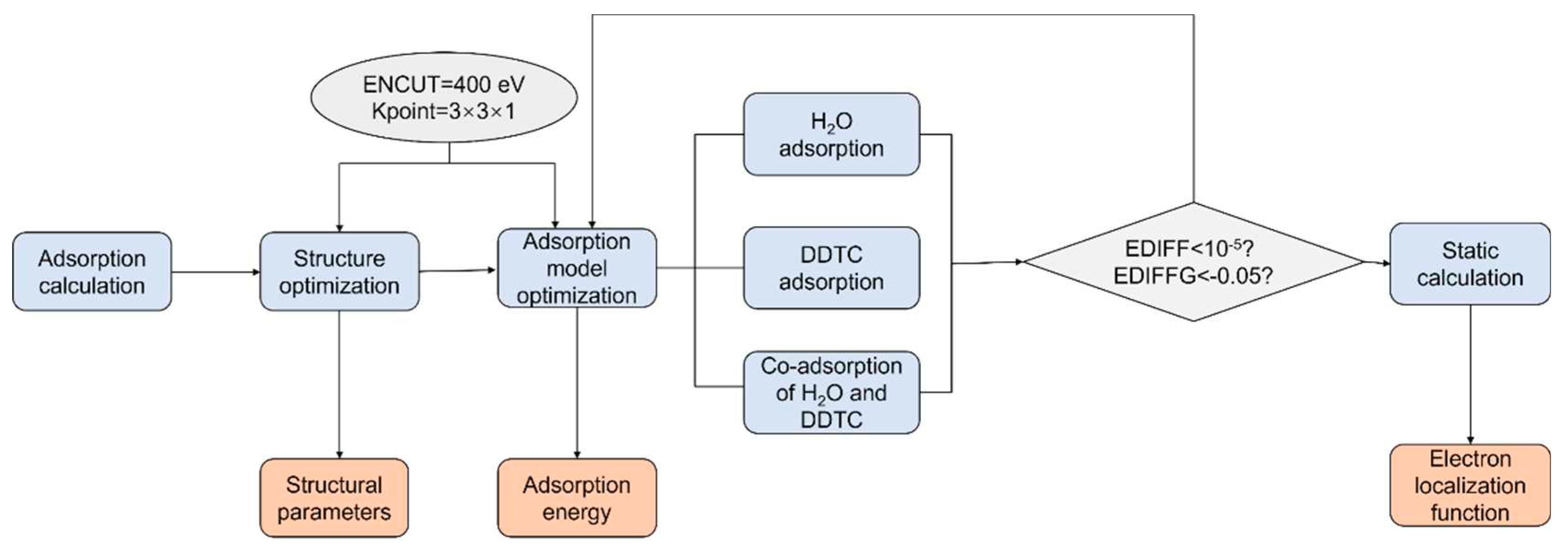
2.1. DFT Calculation
2.2. Material and reagents
2.2.1. High-sulfur residue
2.2.2. Pyrite
2.2.3. Reagents
2.3. Mineral flotation evaluation
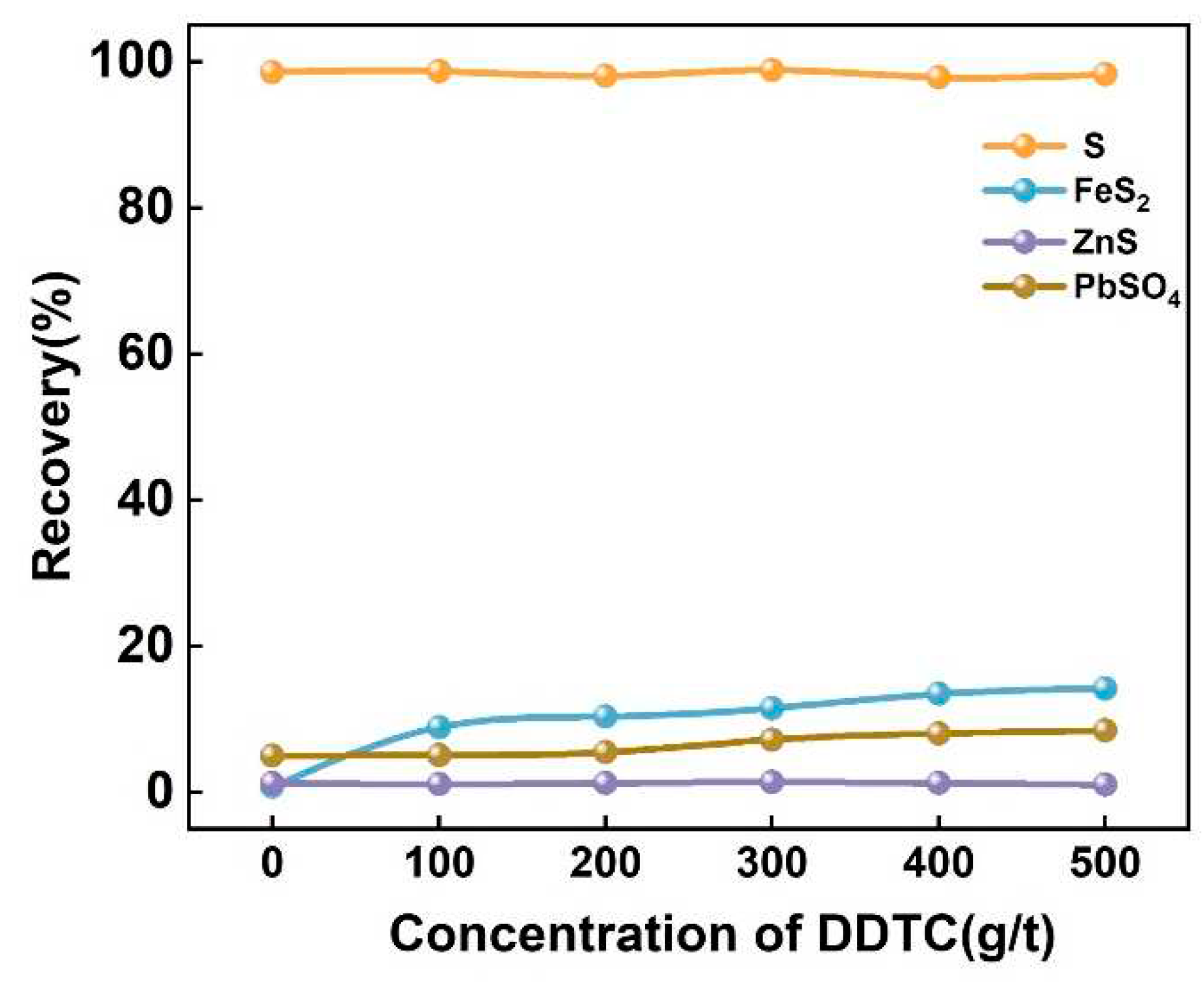
2.3.1. Pure mineral flotation
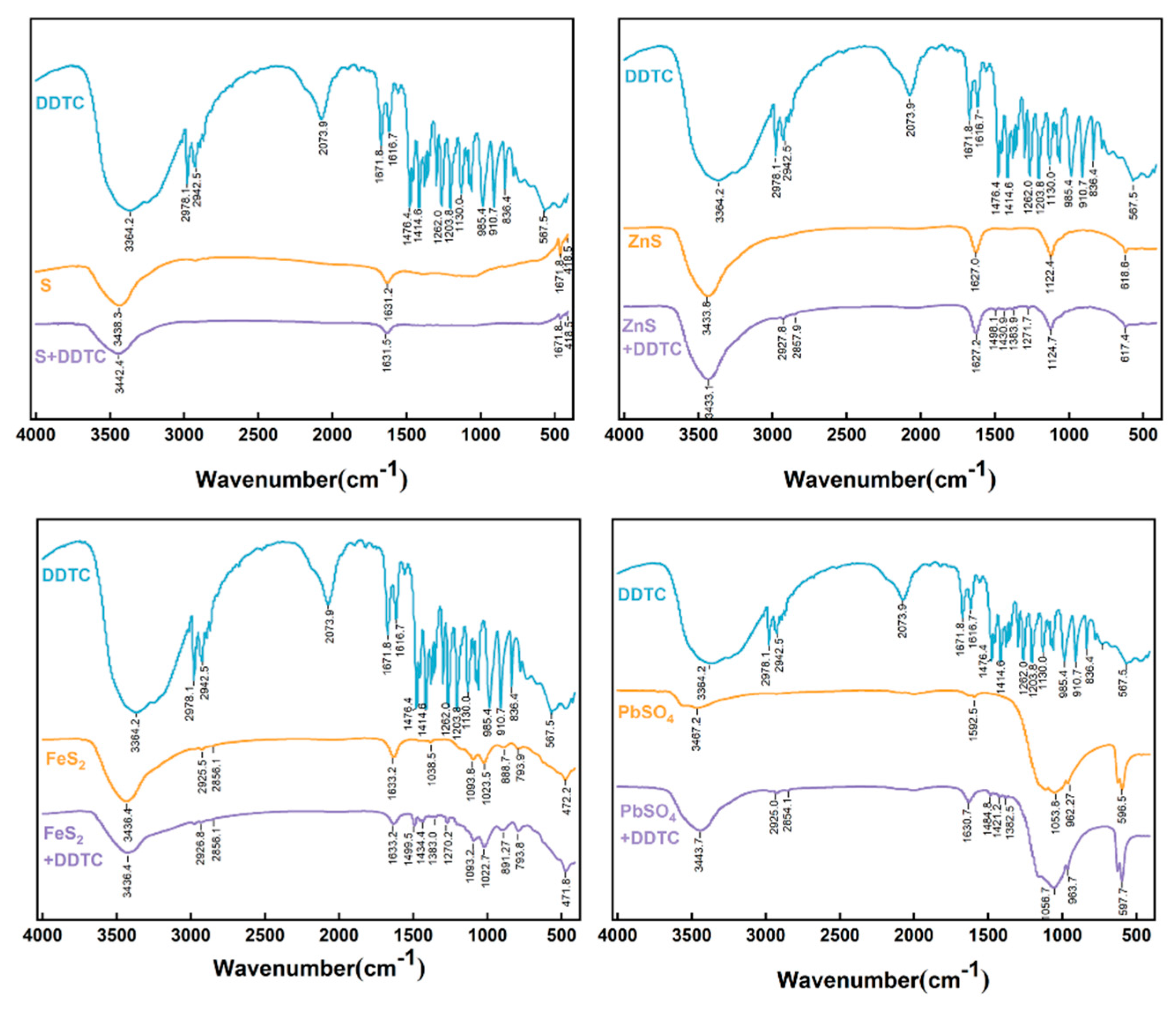
2.3.2. Adsorption behavior of DDTC on pure minerals
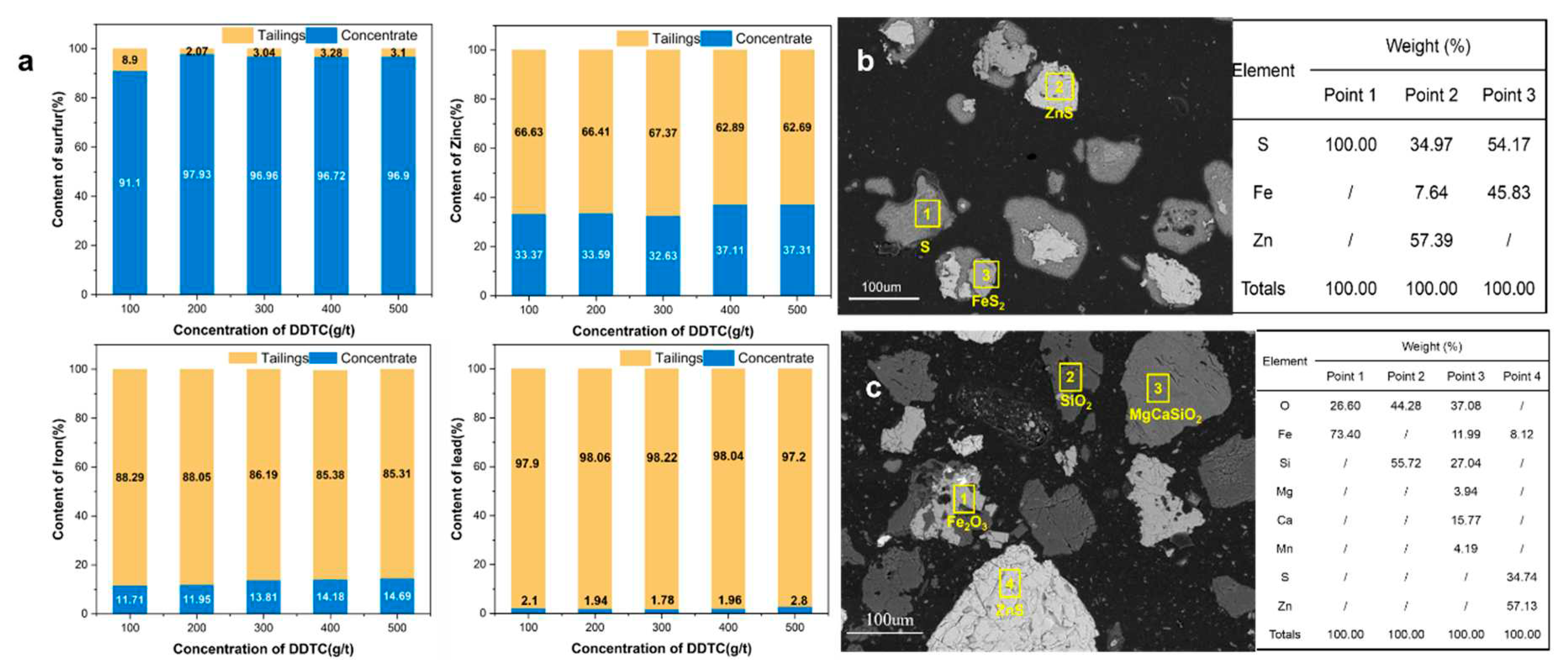
2.3.3. High-sulfur residue flotation
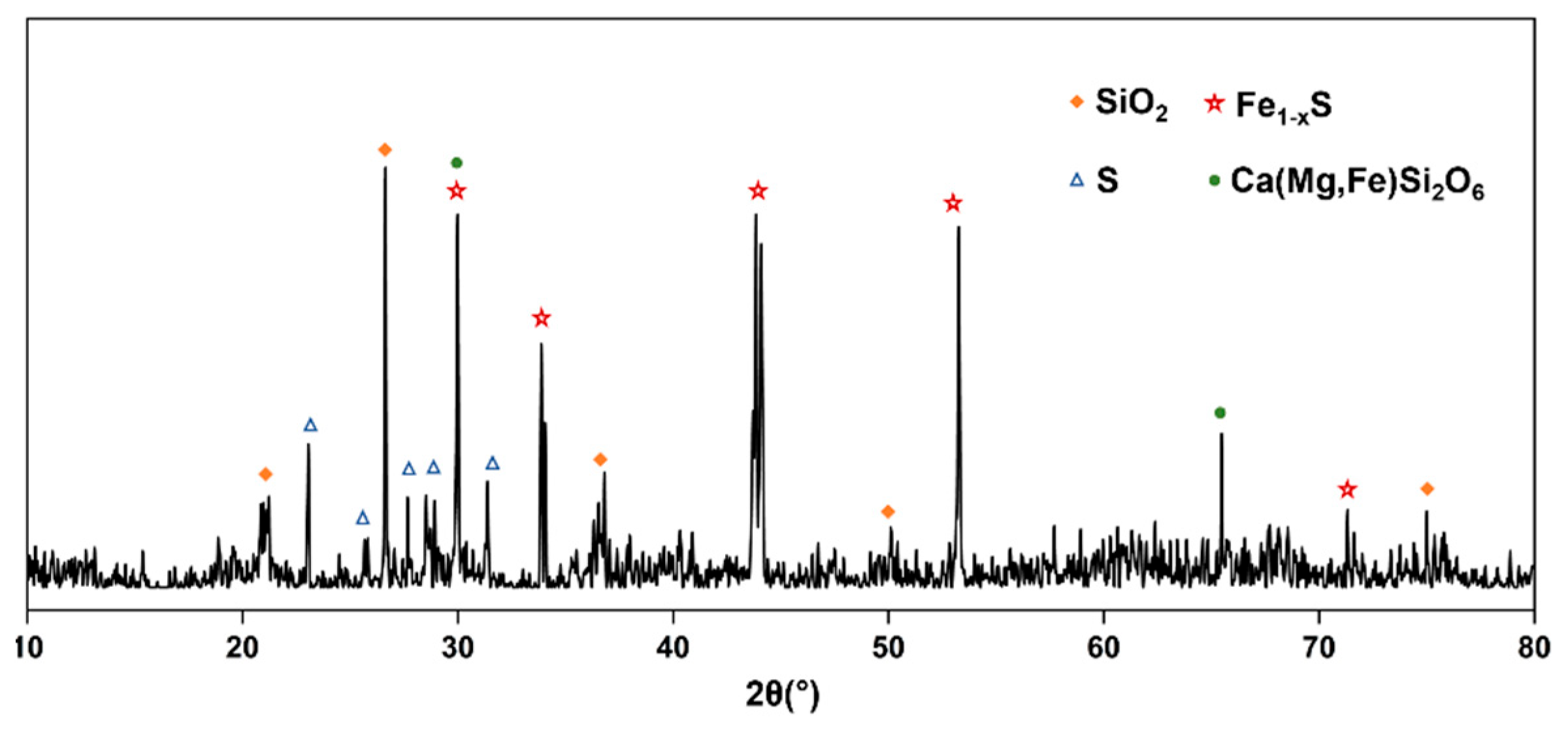
2.3.4. Analysis and characterization
3. Results and discussion
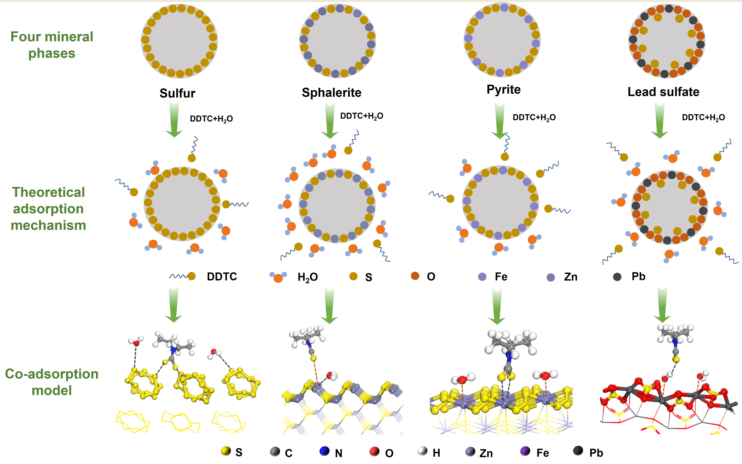
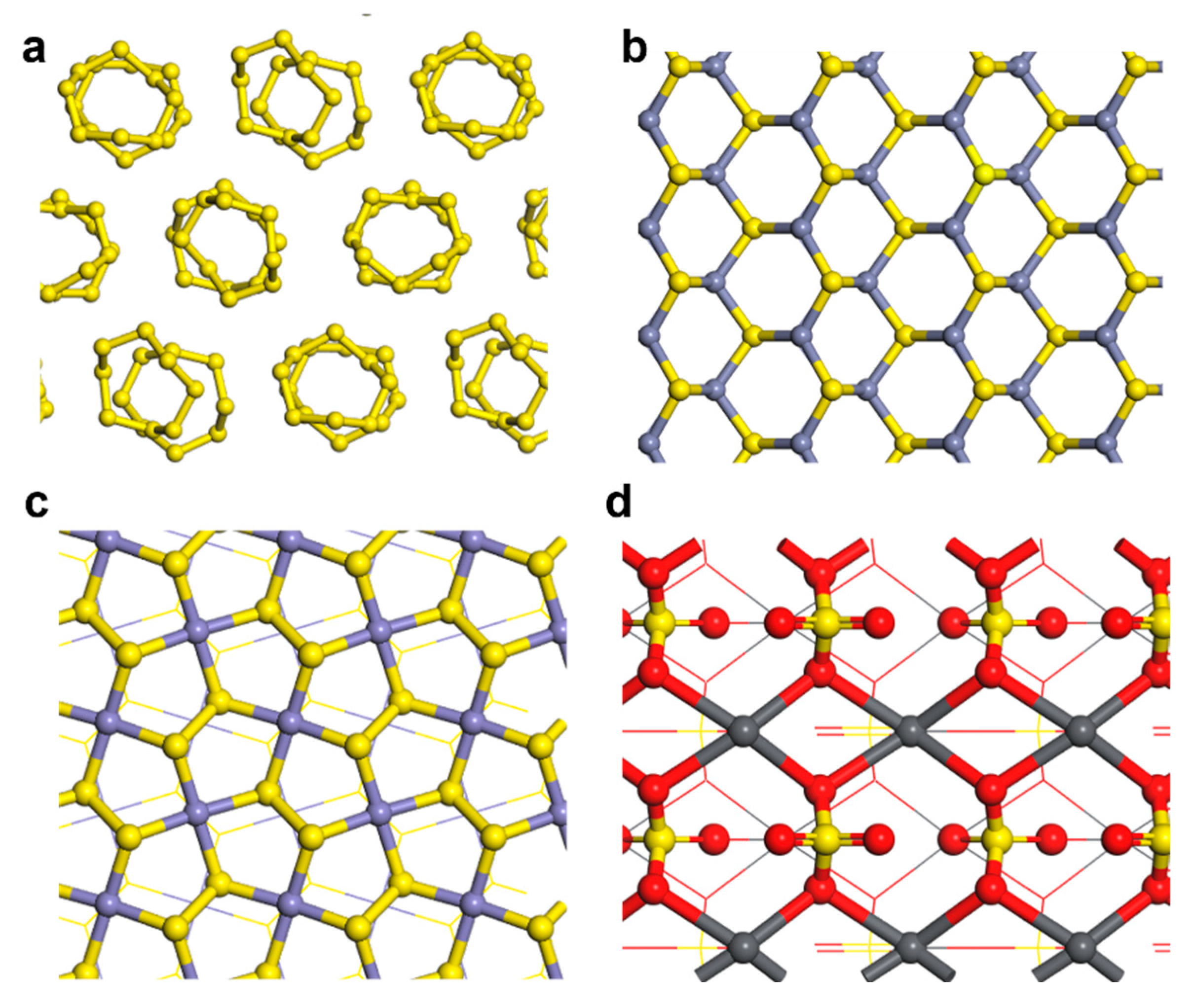
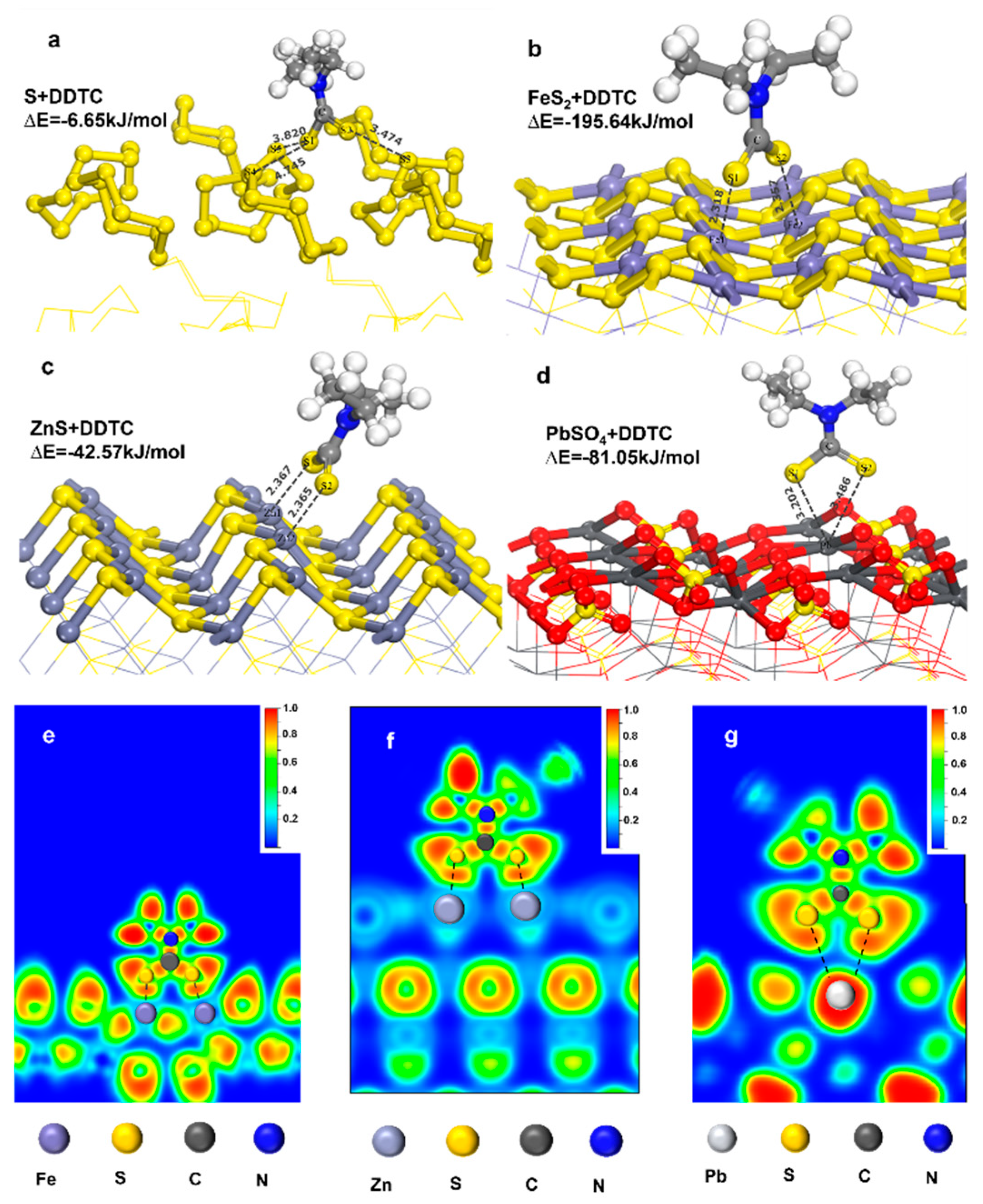
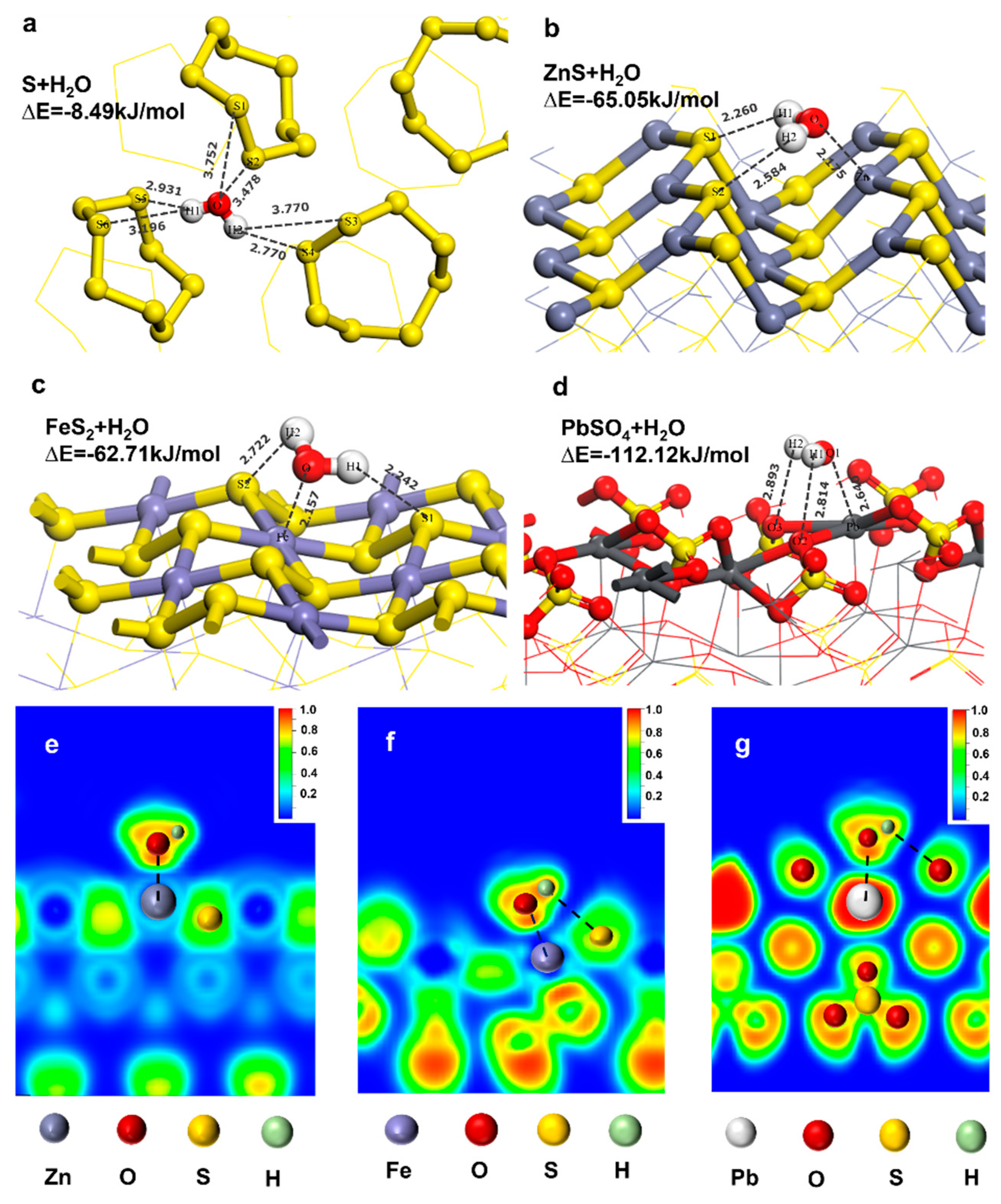
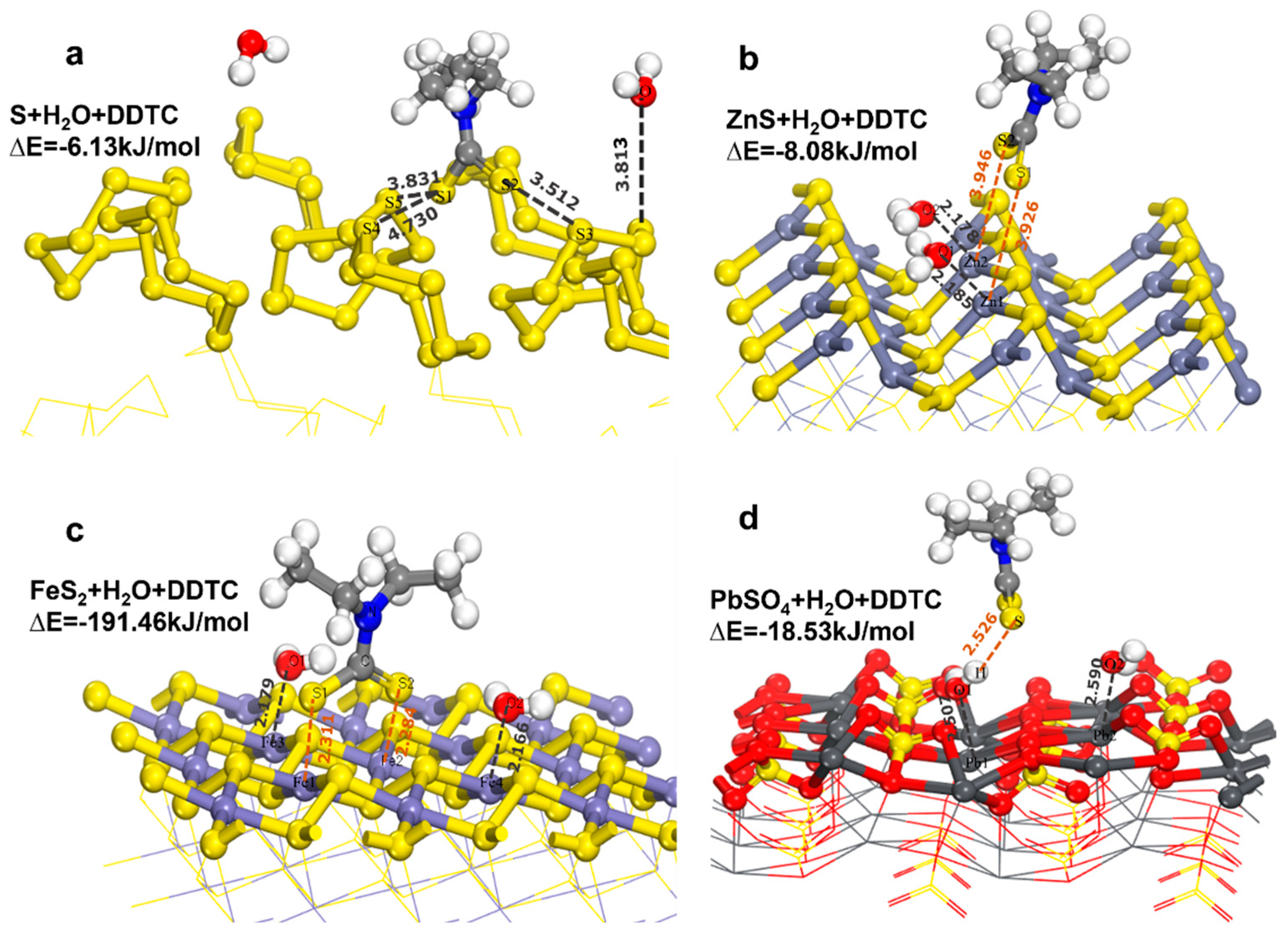
3.1. Co-adsorption model of DDTC and H2O
3.1.1. Adsorption behavior of DDTC
3.1.2. Adsorption behavior of H2O
3.1.3. Co-adsorption model of DDTC and H2O
3.2. Model Validation
3.3. Practical bench-scale operation of high-sulfur residue flotation
4. Conclusions
Supplementary Materials
Acknowledgement
References
- Liu, G.Q.; Zhang, B.S.; Dong, Z.L.; Zhang, F.; Wan, F.; Jiang, T.; Xu, B. Flotation Performance, Structure-Activity Relationship and Adsorption Mechanism of O-Isopropyl-N-Ethyl Thionocarbamate Collector for Elemental Sulfur in a High-Sulfur Residue. Metals 2021, 11. [Google Scholar] [CrossRef]
- Fan, Y.; Liu, Y.; Niu, L.; Jing, T.; Zhang, T.-a. Separation and purification of elemental sulfur from sphalerite concentrate direct leaching residue by liquid paraffin. Hydrometallurgy 2019, 186, 162–169. [Google Scholar] [CrossRef]
- Zhou, H.; Liu, G.; Zhang, L.; Zhou, C. Mineralogical and morphological factors affecting the separation of copper and arsenic in flash copper smelting slag flotation beneficiation process. Journal of Hazardous Materials 2021, 401, 123293. [Google Scholar] [CrossRef]
- Jorjani, E.; Ghahreman, A. Challenges with elemental sulfur removal during the leaching of copper and zinc sulfides, and from the residues; a review. Hydrometallurgy 2017, 171, 333–343. [Google Scholar] [CrossRef]
- Halfyard, J.E.; Hawboldt, K. Separation of elemental sulfur from hydrometallurgical residue: A review. Hydrometallurgy 2011, 109, 80–89. [Google Scholar] [CrossRef]
- Wang, Z.-y.; Cai, X.-l.; Zhang, Z.-b.; Zhang, L.-b.; Wang, S.-x.; Peng, J.-h. Separation and enrichment of elemental sulfur and mercury from hydrometallurgical zinc residue using sodium sulfide. Transactions of Nonferrous Metals Society of China 2015, 25, 640–646. [Google Scholar] [CrossRef]
- Li, H.; Wu, X.; Wang, M.; Wang, J.; Wu, S.; Yao, X.; Li, L. Separation of elemental sulfur from zinc concentrate direct leaching residue by vacuum distillation. Separation and Purification Technology 2014, 138, 41–46. [Google Scholar] [CrossRef]
- Liu, G.; Yang, X.; Zhong, H. Molecular design of flotation collectors: A recent progress. Advances in Colloid and Interface Science 2017, 246, 181–195. [Google Scholar] [CrossRef]
- Silva, L.A.; Garrot, T.G.; Pereira, A.M.; Correia, J.C.G. Historical perspective and bibliometric analysis of molecular modeling applied in mineral flotation systems. Minerals Engineering 2021, 170, 107062. [Google Scholar] [CrossRef]
- Ngobeni, W.A.; Hangone, G. The effect of using sodium di-methyl-dithiocarbamate as a co-collector with xanthates in the froth flotation of pentlandite containing ore from Nkomati mine in South Africa. Minerals Engineering 2013, 54, 94–99. [Google Scholar] [CrossRef]
- Hu, Y.; Wu, M.; Liu, R.; Sun, W. A review on the electrochemistry of galena flotation. Minerals Engineering 2020, 150, 106272. [Google Scholar] [CrossRef]
- Niu, X.; Ruan, R.; Xia, L.; Li, L.; Sun, H.; Jia, Y.; Tan, Q. Correlation of Surface Adsorption and Oxidation with a Floatability Difference of Galena and Pyrite in High-Alkaline Lime Systems. Langmuir 2018, 34, 2716–2724. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Hu, Y.H.; Xu, J.; Chen, T.J. FTIR spectroscopic study of electrochemical flotation of jamesonite-diethyldithiocarbamate system. Transactions of Nonferrous Metals Society of China 2006, 16, 493–496. [Google Scholar] [CrossRef]
- Cui, W.Y.; Zhang, J.J.; Liu, Z.R.; Chen, J.H. Selective enhancement of jamesonite flotation using Aerophine 3418A/ DDTC mixture. Minerals Engineering 2023, 191. [Google Scholar] [CrossRef]
- Qiu, H.X.; Wu, B.Z.; Deng, J.S.; Sun, X.H.; Hu, M.Z.; Cai, J.Z.; Zheng, C. The effect of collectors on froth stability of frother: Atomic-scale study by experiments and molecular dynamics simulations. Journal of Molecular Liquids 2022, 364. [Google Scholar] [CrossRef]
- Zhang, L.M.; Gao, J.D.; Khoso, S.A.; Wang, L.; Liu, Y.L.; Ge, P.; Tian, M.J.; Sun, W. A reagent scheme for galena/sphalerite flotation separation: Insights from first-principles calculations. Minerals Engineering 2021, 167. [Google Scholar] [CrossRef]
- Huang, X.P.; Jia, Y.; Wang, S.; Ma, X.; Cao, Z.F.; Zhong, H. Novel Sodium O-Benzythioethyl Xanthate Surfactant: Synthesis, DFT Calculation and Adsorption Mechanism on Chalcopyrite Surface. Langmuir 2019, 35, 15106–15113. [Google Scholar] [CrossRef]
- Kohn, J.; Spicher, S.; Bursch, M.; Grimme, S. Quickstart guide to model structures and interactions of artificial molecular muscles with efficient computational methods. Chemical Communications 2021, 58, 258–261. [Google Scholar] [CrossRef]
- Pracht, P.; Bohle, F.; Grimme, S. Automated exploration of the low-energy chemical space with fast quantum chemical methods. Physical Chemistry Chemical Physics 2020, 22, 7169–7192. [Google Scholar] [CrossRef]
- Lu, T.; Chen, F.W. Multiwfn: A multifunctional wavefunction analyzer. Journal of Computational Chemistry 2012, 33, 580–592. [Google Scholar] [CrossRef]
- Mkhonto, P.P.; Zhang, X.R.; Lu, L.; Xiong, W.; Zhu, Y.G.; Han, L.; Ngoepe, P.E. Adsorption mechanisms and effects of thiocarbamate collectors in the separation of chalcopyrite from pyrite minerals: DFT and experimental studies. Minerals Engineering 2022, 176. [Google Scholar] [CrossRef]
- Sahraei, A.A.; Larachi, F. How Do Surface Defects Change Local Wettability of the Hydrophilic ZnS Surface? Insights into Sphalerite Flotation from Density Functional Theory Calculations. Journal of Physical Chemistry C 2021, 125, 998–1009. [Google Scholar] [CrossRef]
- Lu, T.; Chen, F.W. Meaning and Functional Form of the Electron Localization Function. Acta Physico-Chimica Sinica 2011, 27, 2786–2792. [Google Scholar]
- Momma, K.; Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. Journal of Applied Crystallography 2011, 44, 1272–1276. [Google Scholar] [CrossRef]
- Sit, P.H.L.; Cohen, M.H.; Selloni, A. Interaction of Oxygen and Water with the (100) Surface of Pyrite: Mechanism of Sulfur Oxidation. Journal of Physical Chemistry Letters 2012, 3, 2409–2414. [Google Scholar] [CrossRef]
- Pollet, R.; Boehme, C.; Marx, D. Ab initio simulations of desorption and reactivity of glycine at a water-pyrite interface at “iron-sulfur world” prebiotic conditions. Origins of Life and Evolution of Biospheres 2006, 36, 363–379. [Google Scholar] [CrossRef]
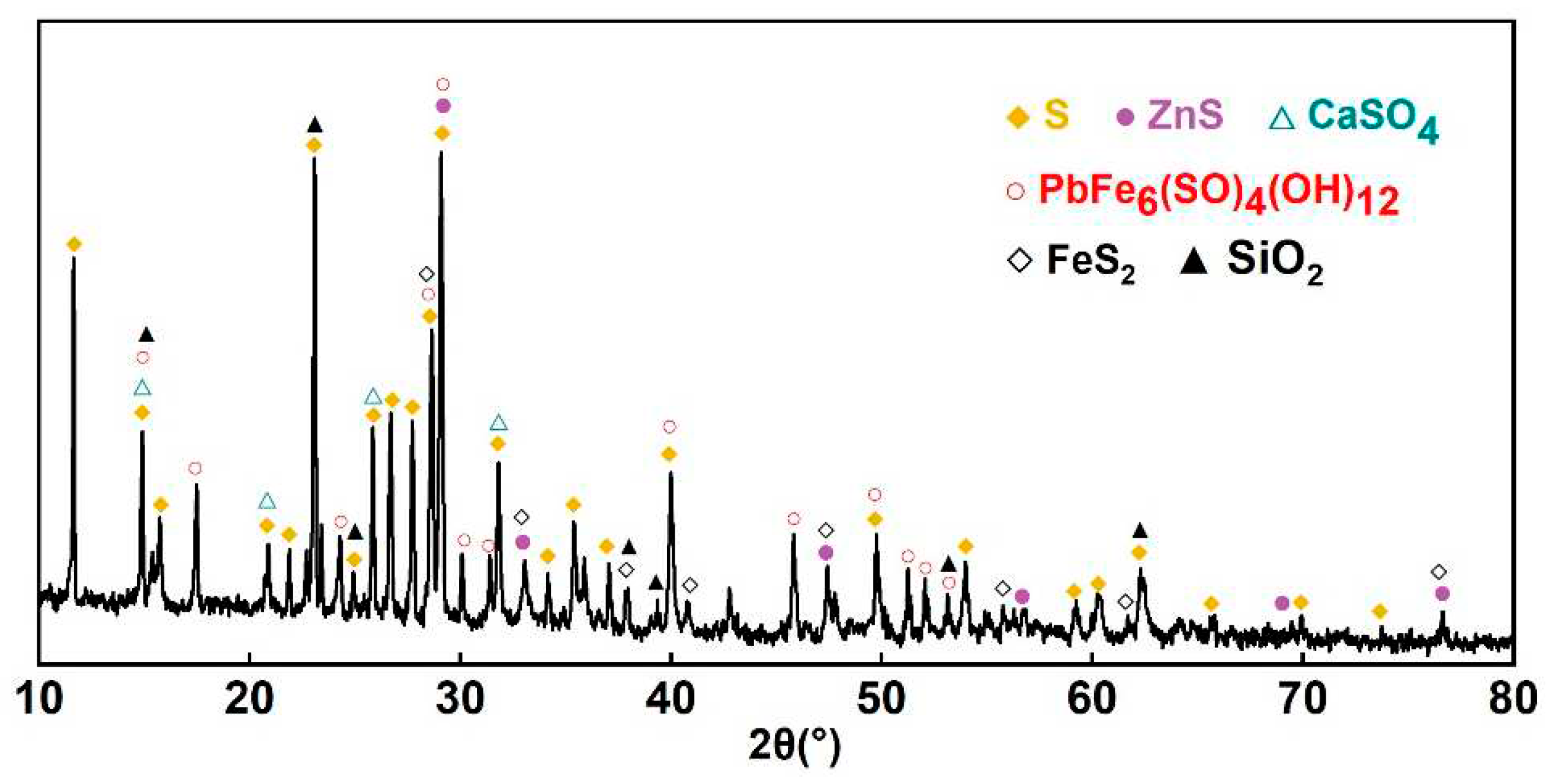
| element | S | O | Zn | Si | Ba | Fe | Pb | Al | Ca |
|---|---|---|---|---|---|---|---|---|---|
| content | 28.81 | 30.3 | 3.02 | 3.79 | 0.27 | 15.48 | 2.18 | 1.21 | 2.30 |
| element | Cr | Sr | Se | Cu | Ag | Hg | Ti | S* | S※ |
| content | 0.018 | 0.19 | 0.004 | 0.09 | 0.02 | 0.066 | 0.043 | 38.20 | 32.98 |
| element | Fe | O | S | Si | Al | Ca | Zn | K | Mg |
|---|---|---|---|---|---|---|---|---|---|
| content | 38.49 | 34.7 | 15.91 | 4.812 | 1.93 | 1.749 | 0.5794 | 0.411 | 0.269 |
| Reagents | Molecular formula | Standard | Manufacturer |
|---|---|---|---|
| DDTC | C4H12NCSSNa | AR | Tianjin Kemiou Chemical Reagent Co., Ltd. |
| sulfur | S | AR | Tianjin Kemiou Chemical Reagent Co., Ltd. |
| Lead sulfate | PbSO4 | AR | Tianjin Kemiou Chemical Reagent Co., Ltd. |
| Zinc sulfide | ZnS | AR | Tianjin Kemiou Chemical Reagent Co., Ltd. |
| Terpineol | C10H11OH | Industrial | Tianjin Kemiou Chemical Reagent Co., Ltd. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




