Submitted:
03 February 2023
Posted:
06 February 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
- The main pathway to produce nitric oxide is through nitric oxide synthase in uncured meats. This enzyme reduces its activity due to low pH, low temperature storage, salting and/or thermal processing operations typical for muscle foods.
- The postmortem loss of calcium can stimulate nitric oxide synthase activity, increasing nitric oxide formation and creating favourable conditions for peroxynitrite production.
2. Materials and Methods
2.1. Readgents
2.1.1. Peroxynitrite synthesis
- Synthesis of 2-ethoxyethylnitrite: 2,78 ml of sulfuric acid 2M are added dropwise to a 12 ml of ethoxyethanol 2.5 M solution with 40 gr sodium nitrite at 0 ºC. The reaction finish when no more nitrogen oxides is released. After one hour, the alkyl nitrite phase is refrigerated in an opaque container with 2 mm molecular sieves.
- Synthesis of peroxynitrite anion: When neaded, peroxynitrite is prepared by mixing 0,2 ml of 2-ethoxyethylnitrite (precursor) with 15 ml of hydrogen peroxide 0,109 M and 15 ml of sodium hydroxide 2 M in 70 ml of H2O. The resulting anion must be stored at -18 °C. Peroxynitrite concentration is determined daily by spectrophotometry at 302 nm.
2.1.2. Other reagents.
2.2. Procedure and measurements
2.3. Kinetic analysis
- Product interference.
- Self-decomposition of reactants.
- Inhibition or autocatalysis effects.
- Presence of competitive reactions.
3. Results
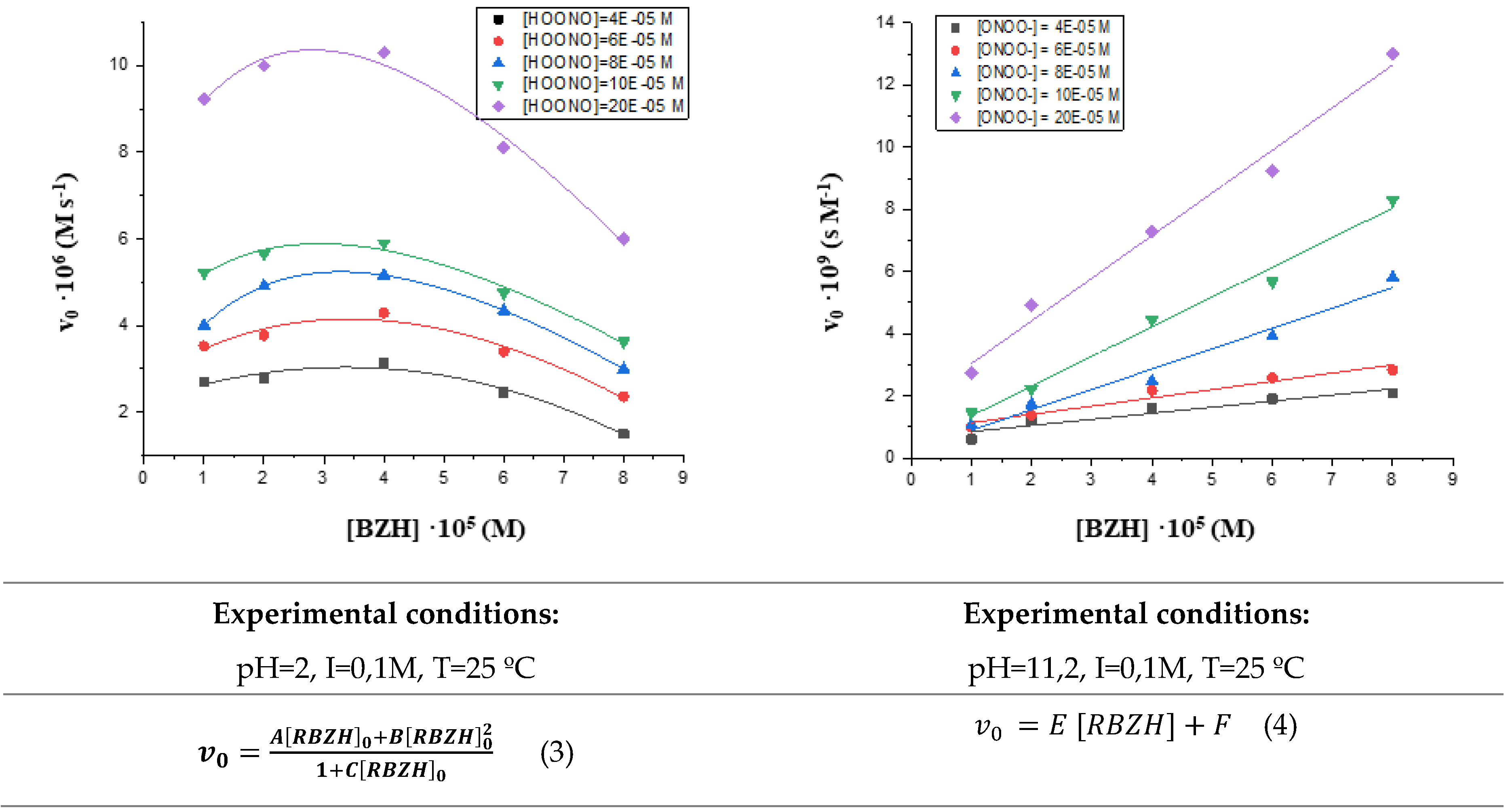
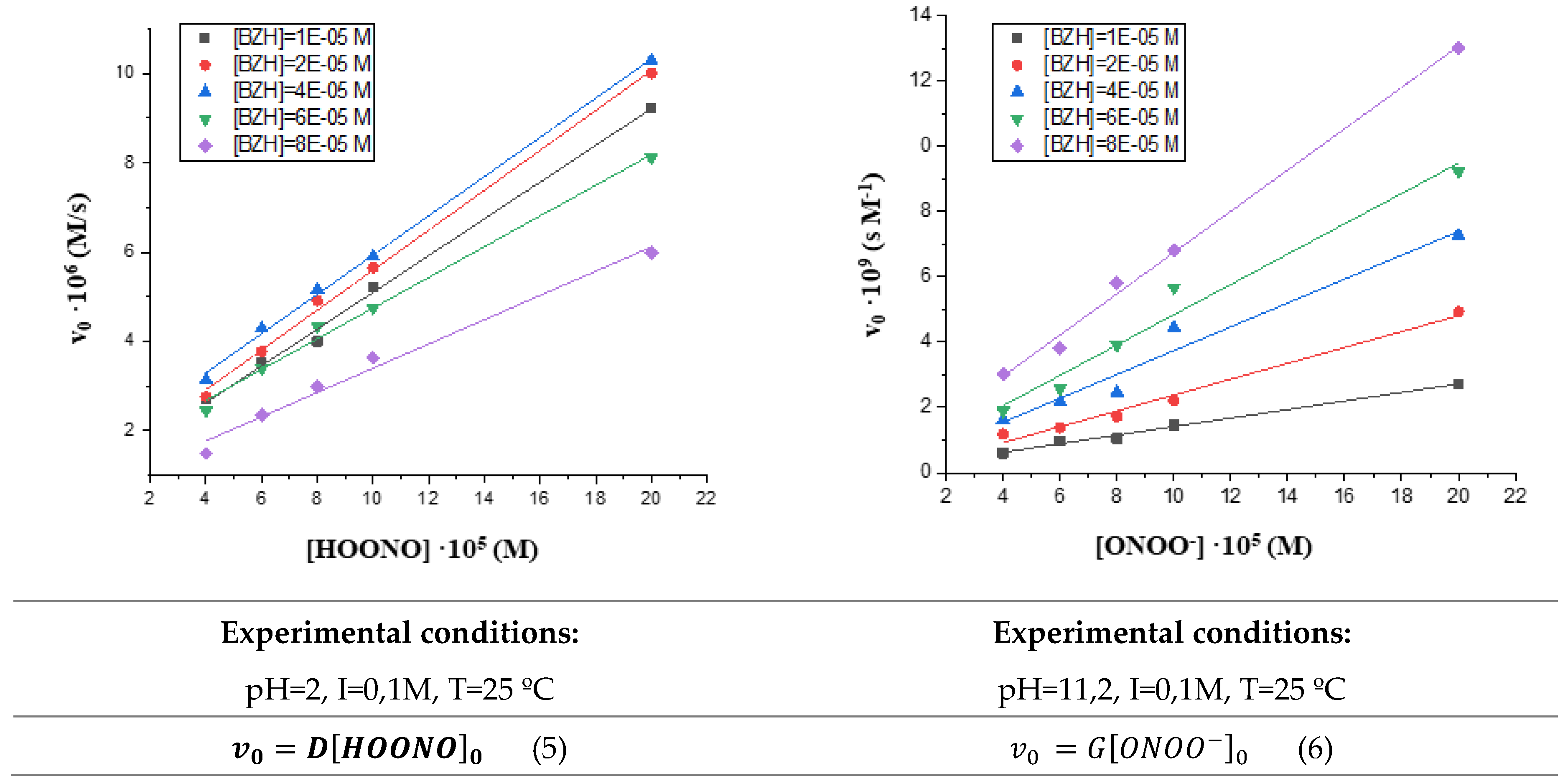
3.1. Influence of substrate and oxidant contentrations on the reaction rate.
3.2. Influence of peroxynirite concentration on the initial reaction rate.
3.3. Influence of pH on the initial reaction rate.
3.4. Influence of the ionic strength on the initial reaction rate.
3.5. Influence of temperature on the initial reaction rate.
3.6. Product determination.
4. Discussion
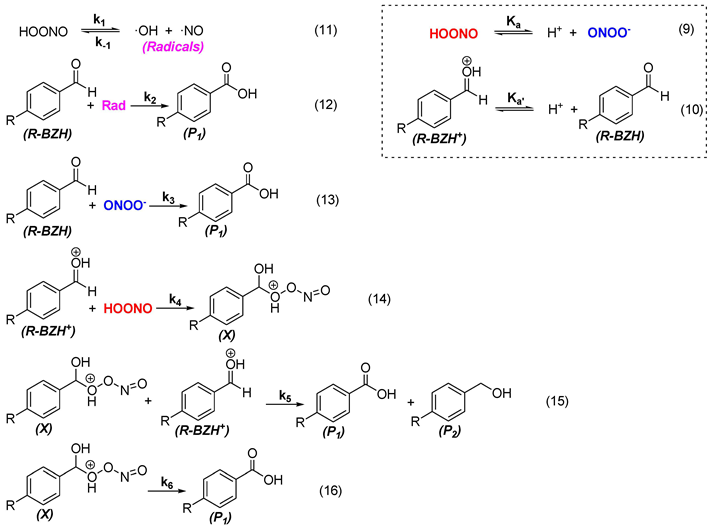
- Firstly, the oxidation of the substrate by the radicals formed by peroxynitrite decomposition (11 and 12).
- The second is the direct oxidation of the aldehyde by the peroxynitrite anion (13).
- The third is the Canizzaro-type reaction in an acid medium (14-16).
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Park, J. B. , Lim, S. H., Sim, H. S., Park, J. H., Kwon, H. J., Nam, H. S., Kim, M. D., Baek, H. H. & Ha, S. J. (2017). Changes in antioxidant activities and volatile compounds of mixed berry juice through fermentation by lactic acid bacteria. Food Science and Biotechnology 26(2), 441–446. [CrossRef]
- Ullah, I. , Khan, A. L., Ali, L., Khan, A. R., Waqas, M., Hussain, J., Lee, I. J. & Shin, J. H. (2015). Benzaldehyde as an insecticidal, antimicrobial, and antioxidant compound produced by Photorhabdus temperata M1021. Journal of Microbiology 53(2), 127–133. [CrossRef]
- Rothe, M. (1991). Volatile Compounds in Foods and Beverages. Herausgegeben von H. Maarse. 764 Seiten, zahlr. Abb. und Tab. Marcel Dekker, Inc., New York, Basel, Hong Kong 1991. Preis 172.50 $. Food / Nahrung, 35, 10, 1080–1080. [CrossRef]
- Krings, U. & Berger, R. G. (1998). Biotechnological production of flavours and fragrances. Applied Microbiology and Biotechnology 49(1), 1–8. [CrossRef]
- Europea, C. (1999). Decisión de la Comisión, de 23 de febrero de 1999, por la que se aprueba un repertorio de sustancias aromatizantes utilizadas en o sobre los productos alimenticios elaborado con arreglo al Reglamento (CE) nº 2232/96 del Parlam.
- Tamir, S. , Burney, S. & Tannenbaum, S. R. (1996). DNA Damage by Nitric Oxide. Chemical Research in Toxicology, 9(5), 821–827. [CrossRef]
- Goldstein, S. & Czapski, G. (1995). The reaction of NO· with O2·− and HO2·−: A pulse radiolysis study. Free Radical Biology and Medicine, 19, 4, 505–510. [CrossRef]
- Radi, R. , Beckman, J. S., Bush, K. M. & Freeman, B. A. (1991). Peroxynitrite-induced membrane lipid peroxidation: The cytotoxic potential of superoxide and nitric oxide. Archives of Biochemistry and Biophysics, 288, 2, 481–487. [CrossRef]
- Molina, C. , Kissner, R. & Koppenol, W. H. (2013). Decomposition kinetics of peroxynitrite: influence of pH and buffer. Dalton Transactions, 42, 27, 9898. [CrossRef]
- Contreras, R. , Galván, M., Oliva, M., Safont, V. S., Andrés, J., Guerra, D. & Aizman, A. (2008). Two state reactivity mechanism for the rearrangement of hydrogen peroxynitrite to nitric acid. Chemical Physics Letters, 457, 1-3, 216–221. [CrossRef]
- Berski, S. , Latajka, Z. & Gordon, A. J. (2011). Electron localization function and electron localizability indicator applied to study the bonding in the peroxynitrous acid HOONO. Journal of Computational Chemistry 32(8), 1528–1540. [CrossRef]
- Grubb, M. P. , Warter, M. L., Xiao, H., Maeda, S., Morokuma, K. & North, S. W. (2012b). No Straight Path: Roaming in Both Ground- and Excited-State Photolytic Channels of NO 3 → NO + O 2. Science, 335, 6072, 1075–1078. [CrossRef]
- Devasagayam, T. P. , Tilak, J. C., Boloor, K. K., Sane, K. S., Ghaskadbi, S. S. & Lele, R. D. (2004). Free radicals and antioxidants in human health: current status and future prospects. Journal of Association of Physicians of India, 52, 794–804, PMID: 15909857.
- Lobo, V. , Patil, A., Phatak, A. & Chandra, N. (2010). Free radicals, antioxidants and functional foods: Impact on human health. Pharmacognosy Reviews, 4, 8, 118. [CrossRef]
- Brannan, R. G. , Connolly, B. J. & Decker, E. A. (2001). Peroxynitrite: a potential initiator of lipid oxidation in food. Trends in Food Science & Technology, 12, 5-6, 164–173. [CrossRef]
- Velasco, J. , Dobarganes, C. & Márquez-Ruiz, G. (2010). Oxidative rancidity in foods and food quality. ( Chemical Deterioration and Physical Instability of Food and Beverages, 3–32. [CrossRef]
- Uppu, R. M. , Winston, G. W. & Pryor, W. A. (1997). Reactions of Peroxynitrite with Aldehydes as Probes for the Reactive Intermediates Responsible for Biological Nitration. Chemical Research in Toxicology 10(12), 1331–1337. [CrossRef]
- Knudsen, F. S. , Penatti, C. A. A., Royer, L. O., Bidart, K. A., Christoff, M., Ouchi, D. & Bechara, E. J. H. (2000). Chemiluminescent Aldehyde and β-Diketone Reactions Promoted by Peroxynitrite. Chemical Research in Toxicology, 13, 5, 317–326. [CrossRef]
- Ischiropoulos, H. , Nelson, J., Duran, D. & Al-Mehdi, A. (1996). Reactions of nitric oxide and peroxynitrite with organic molecules and ferrihorseradish peroxidase: Interference with the determination of hydrogen peroxide. Free Radical Biology and Medicine, 20, 3, 373–381. [CrossRef]
- Nakao, L. S. , Ouchi, D. & Augusto, O. (1999). Oxidation of Acetaldehyde by Peroxynitrite and Hydrogen Peroxide/Iron(II). Production of Acetate, Formate, and Methyl Radicals. Chemical Research in Toxicology, 12, 10, 1010–1018. [CrossRef]
- Burdock, G. A. (2009). Fenaroli’s Handbook of Flavor Ingredients, (6th ed.). CRC Press.
- Leis, J. R. , Pen̄a, M. E. & Ríos, A. (1993). A novel route to peroxynitrite anion. J. Chem. Soc., Chem. Commun 16, 1298–1299. [CrossRef]
- Fernandez, S. L. (2016). Técnicas de ajuste de las curvas de concentración en cinética química. Tendencias en docencia e investigación en química. 1, 1, 323–329, Obtenido de http://hdl.handle.net/11191/5146.
- Laidler, K. J. (2003). Chemical Kinetics. 3Rd Edition. Pearson India.
- Skoog, D. A. (2021). Fundamentals of Analytical Chemistry (10th ed.). Cengage Learning.
- Shrestha, K. P. , Giri, B. R., Adil, M., Seidel, L., Zeuch, T., Farooq, A. & Mauss, F. (2021). Detailed Chemical Kinetic Study of Acetaldehyde Oxidation and Its Interaction with NOx. Energy & Fuels, 35, 18, 14963–14983. [CrossRef]
- Ingold, K. U. (1961). Inhibition of the Autoxidation of Organic Substances in the Liquid Phase. Chemical Reviews, 61, 6, 563–589. [CrossRef]
- Ritchie, C. D. & Sager, W. F. (2007). An Examination of Structure-Reactivity Relationships. Progress in Physical Organic Chemistry, 323–400. [CrossRef]
- Hammett, L. P. (1937). The Effect of Structure upon the Reactions of Organic Compounds. Benzene Derivatives. Journal of the American Chemical Society, 59, 1, 96–103. [CrossRef]
- Schwab, F. W. & Wichers, E. (1940). Preparation of benzoic acid of high purity. Journal of Research of the National Bureau of Standards, 25(6), 747. [CrossRef]
- Nair, B. (2001). Final report on the safety assessment of Benzyl Alcohol, Benzoic Acid, and Sodium Benzoate. International Journal of Toxicology, 20, 23-50. [CrossRef]
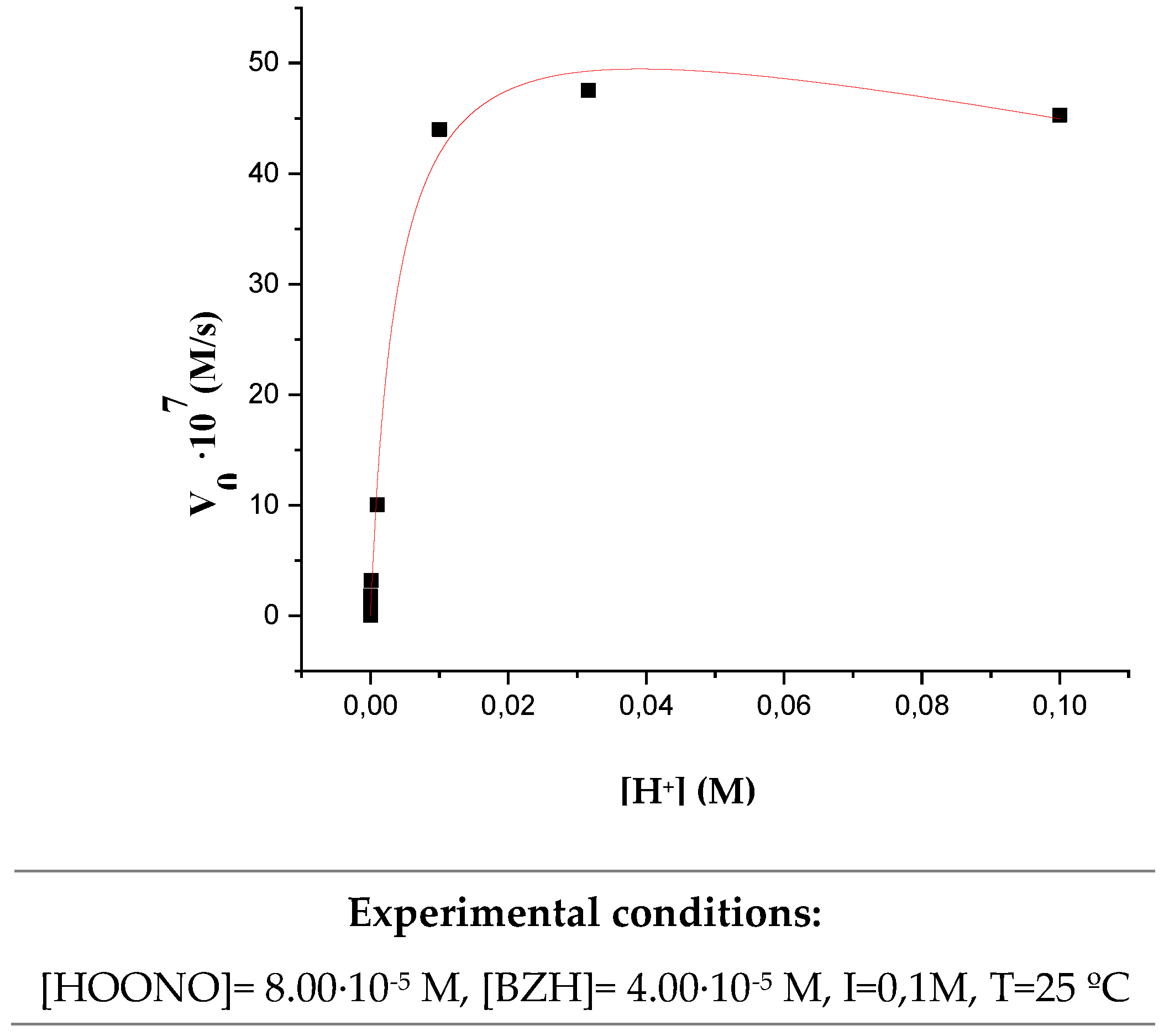
| Temperature (K) | 283 | 288 | 293 | 298 | 303 |
|---|---|---|---|---|---|
| v0·106 (M·s-1) | 0,82±0,02 | 1,52±0,02 | 3,41±0,02 | 5,16±0,02 | 8,27±0,02 |
| Experimental Conditions: [HOONO] = 8.00·10-5 M, [BZH] = 4.00·10-5 M, I= 0,1M, pH = 2 | |||||
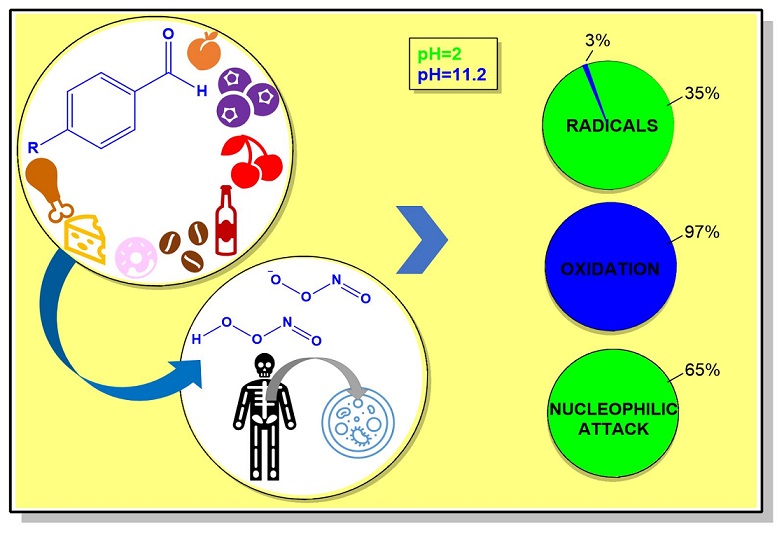
| pH=2 | pH=11,2 | |
| Radicals | 35% | 3% |
| Oxidation | - | 97% |
| Nucleophilic attack (Cannizzaro) | 65% | - |
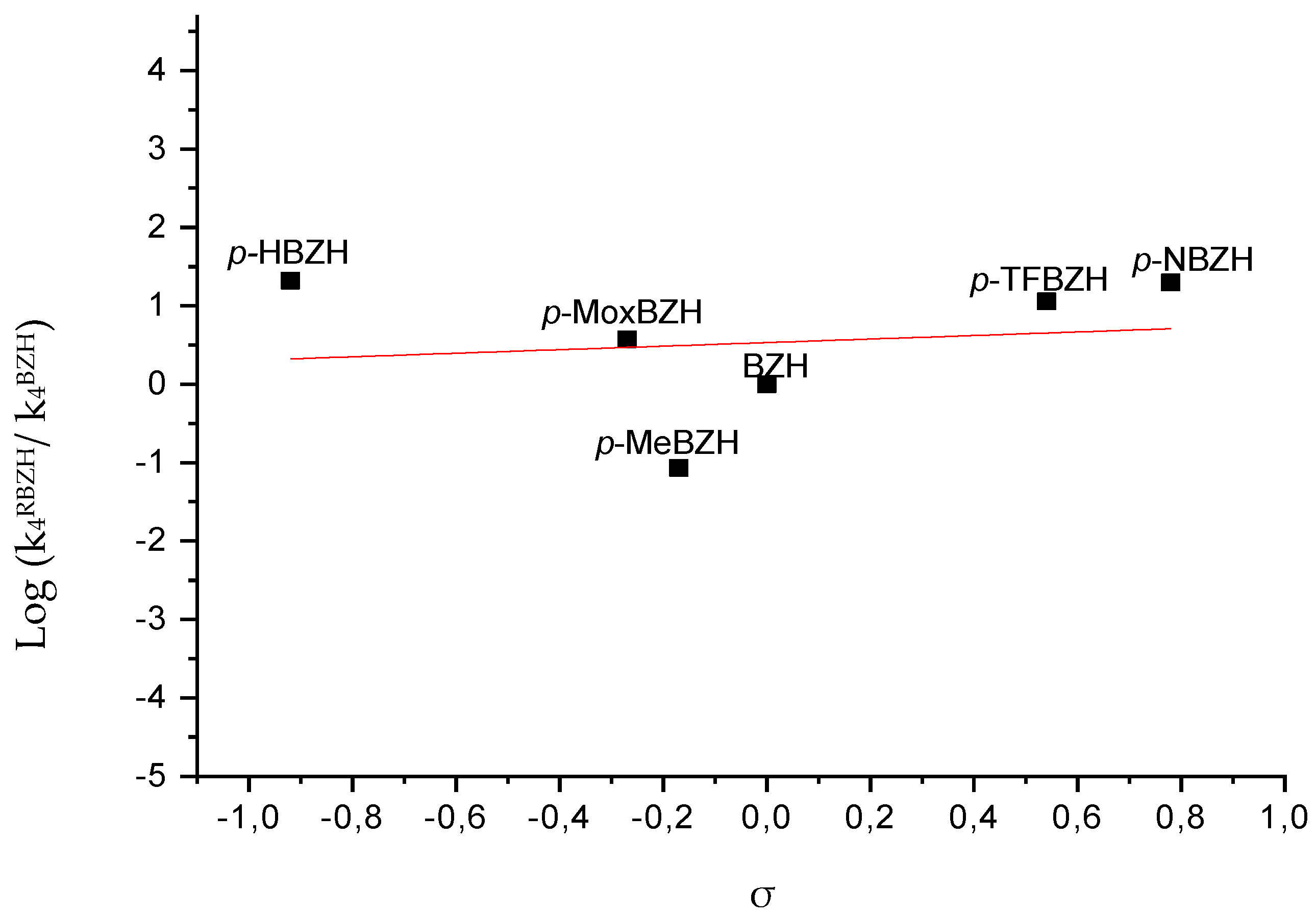
| Aldehyde | k4·1014 (M-1s-1) | k5/k6·1025 | σ |
|---|---|---|---|
| BZH | 0,39±0.02 | 0,69±0.01 | 0.00 |
| pHBZH | 8,13±0.08 | 21,5±0.01 | -0,92 |
| pMeBZH | 0,03±0.08 | 0,06±0.02 | -0,17 |
| pMoxBZH | 1,45±0.02 | 9,04±0.01 | -0,27 |
| pNBZH | 7,73±0.06 | 15,6±0.02 | 0,78 |
| pTFBZH | 4,45±0.03 | 9,66±0.02 | 0,54 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




