Submitted:
09 February 2023
Posted:
10 February 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Mitochondria in the Healthy Placenta
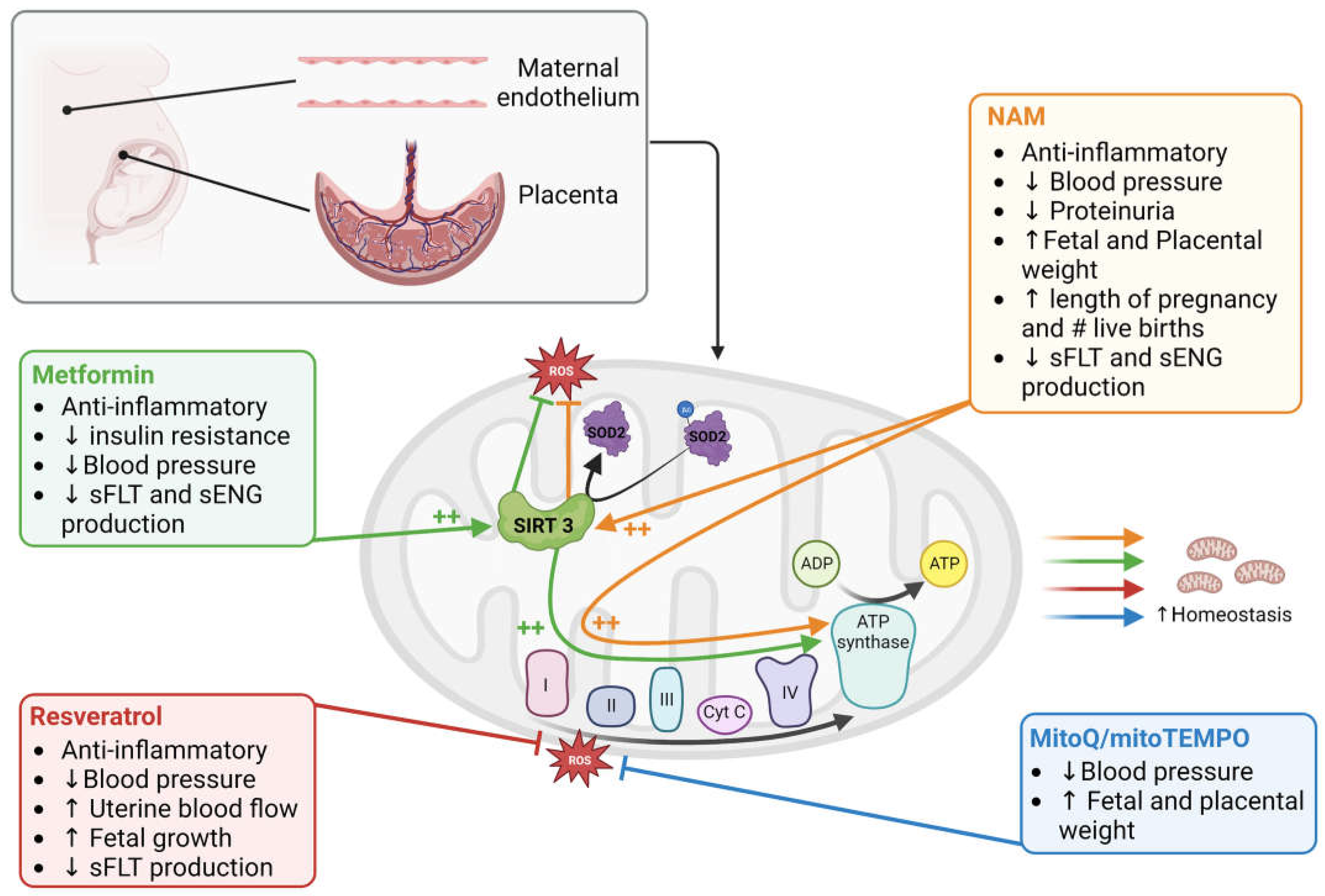
2.1. Placental Mitochondrial Content, Structure, and Function across Pregnancy
2.2. Mitochondrial Content, Structure, and Function in Distinct Placental Cell Types
3. Preeclampsia
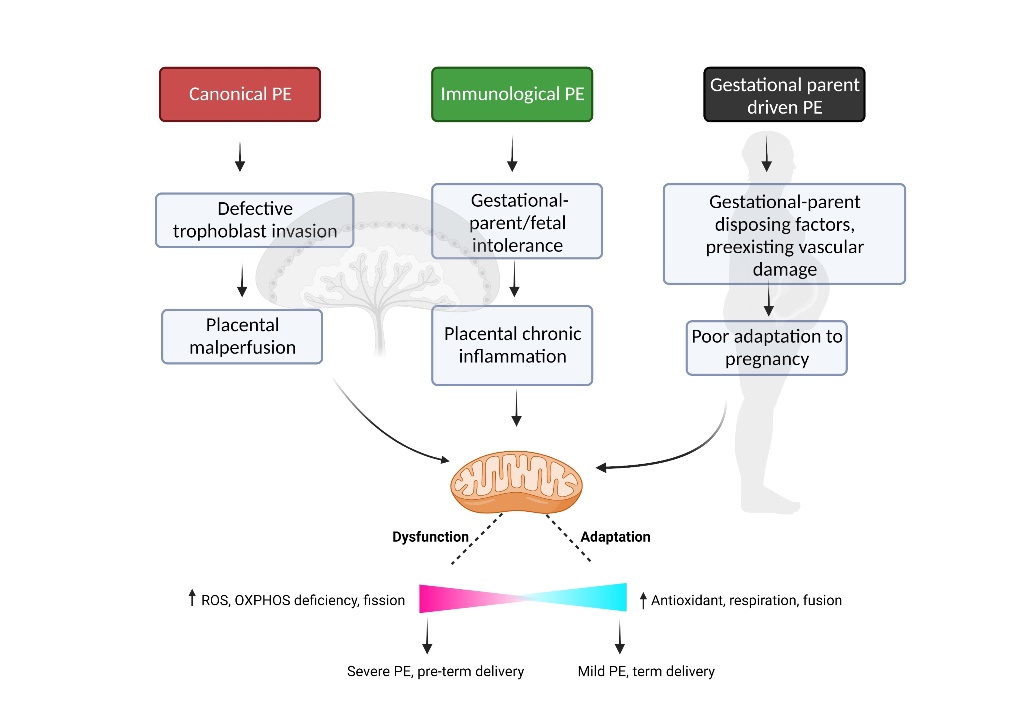
4. Subclasses of Disease in Preeclampsia
4.1. Canonical PE Subclass
4.2. Immunological PE Subclass
4.3. Gestational Parent-Driven PE Subclass
| Canonical PE | Immunological PE | Gestational-parent driven PE |
|
|---|---|---|---|
|
Onset and severity of disease |
|
|
|
| Pathogenesis |
|
|
|
| Genes expressed in placenta |
|
|
|
| Placenta pathology |
|
|
|
| Potential risk factors |
|
|
|
5. Placental Mitochondrial Dysfunction in Preeclampsia
5.1. Mitochondrial Content
5.2. Mitochondrial Structure and Dynamics
5.3. Mitochondrial Function—Respiration and ATP Generation
5.4. Mitochondrial Function—Redox Homeostasis and Apoptosis
6. Mitochondrial Dysfunction—A Point of Convergence across Preeclampsia Subclasses
6.1. Canonical PE Subclass - Ischemia-Reperfusion Induced Mitochondrial Dysfunction
6.2. Immunological PE Subclass - Inflammation Induced Mitochondrial Dysfunction
6.3. Gestational Parent Driven PE - Predisposing Factors and Mitochondrial Dysfunction
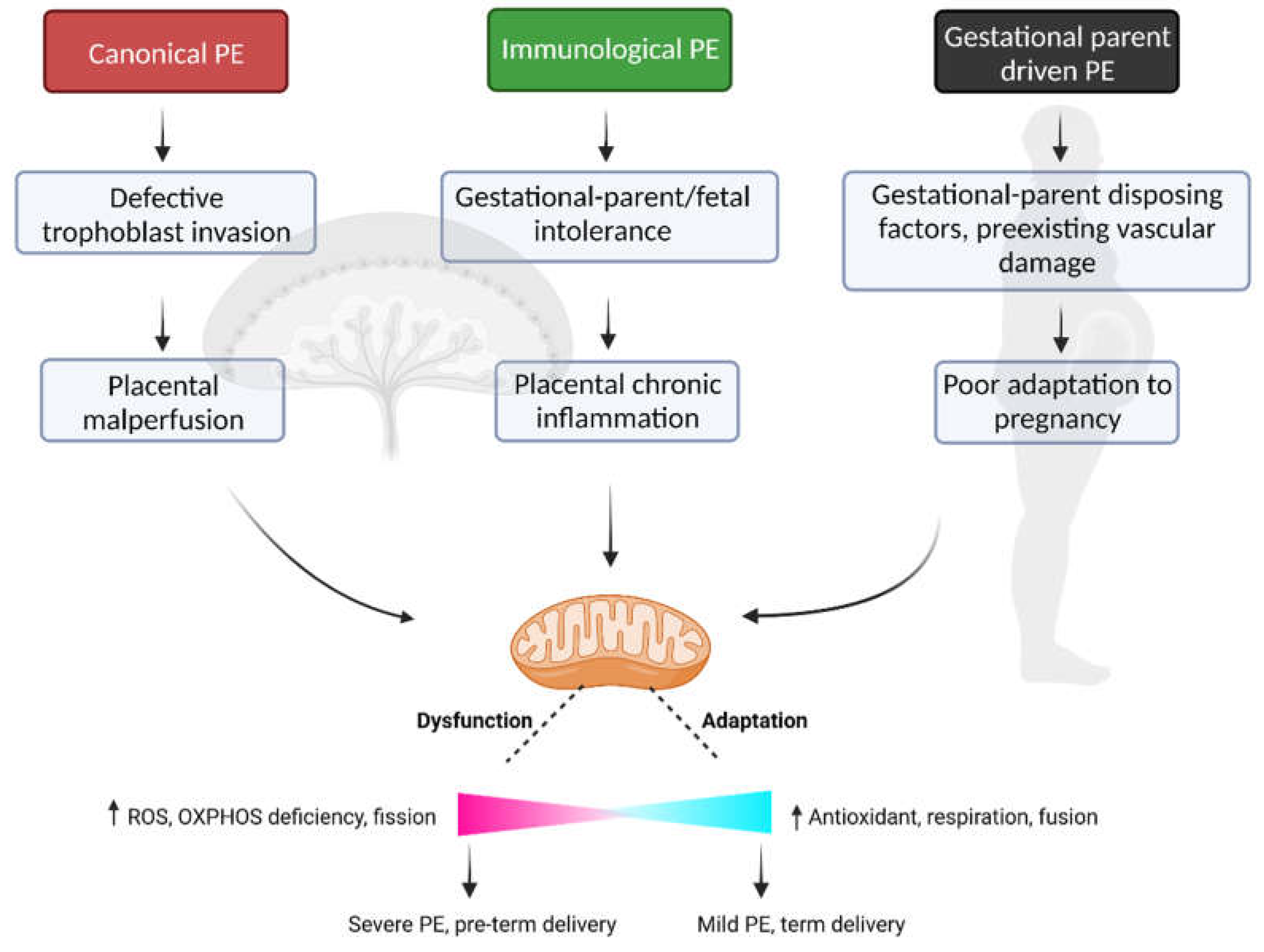
7. Potential for Mitochondrial Targeting Therapies in Preeclampsia
7.1. Resveratrol
7.2. Metformin
7.3. MitoQ and MitoTEMPO
7.4. Nicotinamide (NAM)
Future Perspective
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Bauer, M.K., et al., Fetal growth and placental function. Mol Cell Endocrinol, 1998. 140(1-2): p. 115-20. [CrossRef]
- Gude, N.M., et al., Growth and function of the normal human placenta. Thromb Res, 2004. 114(5-6): p. 397-407. [CrossRef]
- Rana, S., et al., Preeclampsia: Pathophysiology, Challenges, and Perspectives. Circ Res, 2019. 124(7): p. 1094-1112.
- Leavey, K., et al., Unsupervised Placental Gene Expression Profiling Identifies Clinically Relevant Subclasses of Human Preeclampsia. Hypertension, 2016. 68(1): p. 137-47. [CrossRef]
- Griffiths, S.K. and J.P. Campbell, Placental structure, function and drug transfer. Continuing Education in Anaesthesia Critical Care & Pain, 2014. 15(2): p. 84-89.
- Aye, I., et al., Placental energy metabolism in health and disease-significance of development and implications for preeclampsia. Am J Obstet Gynecol, 2022. 226(2S): p. S928-S944. [CrossRef]
- Spinelli, J.B. and M.C. Haigis, The multifaceted contributions of mitochondria to cellular metabolism. Nat Cell Biol, 2018. 20(7): p. 745-754. [CrossRef]
- Hebert, J.F. and L. Myatt, Placental mitochondrial dysfunction with metabolic diseases: Therapeutic approaches. Biochim Biophys Acta Mol Basis Dis, 2021. 1867(1): p. 165967. [CrossRef]
- Holland, O., et al., Review: Placental mitochondrial function and structure in gestational disorders. Placenta, 2017. 54: p. 2-9. [CrossRef]
- Burton, G.J., H.W. Yung, and A.J. Murray, Mitochondrial - Endoplasmic reticulum interactions in the trophoblast: Stress and senescence. Placenta, 2017. 52: p. 146-155. [CrossRef]
- Constancia, M., et al., Placental-specific IGF-II is a major modulator of placental and fetal growth. Nature, 2002. 417(6892): p. 945-8. [CrossRef]
- Constancia, M., et al., Adaptation of nutrient supply to fetal demand in the mouse involves interaction between the Igf2 gene and placental transporter systems. Proc Natl Acad Sci U S A, 2005. 102(52): p. 19219-24. [CrossRef]
- Sferruzzi-Perri, A.N., et al., An obesogenic diet during mouse pregnancy modifies maternal nutrient partitioning and the fetal growth trajectory. FASEB J, 2013. 27(10): p. 3928-37. [CrossRef]
- Hay, W.W., Jr., Energy and substrate requirements of the placenta and fetus. Proc Nutr Soc, 1991. 50(2): p. 321-36. [CrossRef]
- Fisher, J.J., et al., Placental mitochondria and reactive oxygen species in the physiology and pathophysiology of pregnancy. Clin Exp Pharmacol Physiol, 2020. 47(1): p. 176-184. [CrossRef]
- Bartho, L.A., et al., Mitochondrial transformations in the aging human placenta. Am J Physiol Endocrinol Metab, 2020. 319(6): p. E981-E994. [CrossRef]
- Correia, Y., et al., Placental mitochondrial function as a driver of angiogenesis and placental dysfunction. Biol Chem, 2021. 402(8): p. 887-909. [CrossRef]
- Burton, G.J. and A.L. Fowden, The placenta: a multifaceted, transient organ. Philos Trans R Soc Lond B Biol Sci, 2015. 370(1663): p. 20140066.
- Jauniaux, E., et al., Polyol concentrations in the fluid compartments of the human conceptus during the first trimester of pregnancy: maintenance of redox potential in a low oxygen environment. J Clin Endocrinol Metab, 2005. 90(2): p. 1171-5. [CrossRef]
- Bloxam, D.L. and P.M. Bobinski, Energy metabolism and glycolysis in the human placenta during ischaemia and in normal labour. Placenta, 1984. 5(5): p. 381-94. [CrossRef]
- Holland, O.J., et al., Changes in mitochondrial respiration in the human placenta over gestation. Placenta, 2017. 57: p. 102-112. [CrossRef]
- Jauniaux, E., et al., Onset of maternal arterial blood flow and placental oxidative stress. A possible factor in human early pregnancy failure. Am J Pathol, 2000. 157(6): p. 2111-22. [CrossRef]
- Drose, S. and U. Brandt, Molecular mechanisms of superoxide production by the mitochondrial respiratory chain. Adv Exp Med Biol, 2012. 748: p. 145-69. [CrossRef]
- Mannaerts, D., et al., Oxidative stress in healthy pregnancy and preeclampsia is linked to chronic inflammation, iron status and vascular function. PLoS One, 2018. 13(9): p. e0202919. [CrossRef]
- Bardin, N., P. Murthi, and N. Alfaidy, Normal and pathological placental angiogenesis. Biomed Res Int, 2015. 2015: p. 354359. [CrossRef]
- Basu, J., et al., Placental Oxidative Status throughout Normal Gestation in Women with Uncomplicated Pregnancies. Obstet Gynecol Int, 2015. 2015: p. 276095. [CrossRef]
- Sferruzzi-Perri, A.N., et al., Placental mitochondria adapt developmentally and in response to hypoxia to support fetal growth. Proc Natl Acad Sci U S A, 2019. 116(5): p. 1621-1626. [CrossRef]
- Sesso, A., et al., Mitochondrial swelling and incipient outer membrane rupture in preapoptotic and apoptotic cells. Anat Rec (Hoboken), 2012. 295(10): p. 1647-59. [CrossRef]
- Huppertz, B., et al., Apoptosis cascade progresses during turnover of human trophoblast: analysis of villous cytotrophoblast and syncytial fragments in vitro. Lab Invest, 1999. 79(12): p. 1687-702.
- Levy, R. and D.M. Nelson, To be, or not to be, that is the question. Apoptosis in human trophoblast. Placenta, 2000. 21(1): p. 1-13. [CrossRef]
- Kolahi, K.S., A.M. Valent, and K.L. Thornburg, Cytotrophoblast, Not Syncytiotrophoblast, Dominates Glycolysis and Oxidative Phosphorylation in Human Term Placenta. Sci Rep, 2017. 7: p. 42941. [CrossRef]
- De los Rios Castillo, D., et al., Atypical cristae morphology of human syncytiotrophoblast mitochondria: role for complex V. J Biol Chem, 2011. 286(27): p. 23911-9.
- Martinez, F., M. Kiriakidou, and J.F. Strauss, 3rd, Structural and functional changes in mitochondria associated with trophoblast differentiation: methods to isolate enriched preparations of syncytiotrophoblast mitochondria. Endocrinology, 1997. 138(5): p. 2172-83. [CrossRef]
- Martinez, F., et al., Multiple functions of syncytiotrophoblast mitochondria. Steroids, 2015. 103: p. 11-22. [CrossRef]
- Watson, A.L., et al., Susceptibility of human placental syncytiotrophoblastic mitochondria to oxygen-mediated damage in relation to gestational age. J Clin Endocrinol Metab, 1998. 83(5): p. 1697-705.
- Seok, J., et al., Mitochondrial Dynamics in Placenta-Derived Mesenchymal Stem Cells Regulate the Invasion Activity of Trophoblast. Int J Mol Sci, 2020. 21(22). [CrossRef]
- Seok, J., et al., Human placenta-derived mesenchymal stem cells induce trophoblast invasion via dynamic effects on mitochondrial function. J Cell Physiol, 2021. 236(9): p. 6678-6690. [CrossRef]
- LeBleu, V.S., et al., PGC-1alpha mediates mitochondrial biogenesis and oxidative phosphorylation in cancer cells to promote metastasis. Nat Cell Biol, 2014. 16(10): p. 992-1003, 1-15. [CrossRef]
- Hogan, M.C., et al., Maternal mortality for 181 countries, 1980-2008: a systematic analysis of progress towards Millennium Development Goal 5. Lancet, 2010. 375(9726): p. 1609-23.
- ACOG Practice Bulletin No. 202: Gestational Hypertension and Preeclampsia. Obstet Gynecol, 2019. 133(1): p. 1.
- Weiler, J., S. Tong, and K.R. Palmer, Is fetal growth restriction associated with a more severe maternal phenotype in the setting of early onset pre-eclampsia? A retrospective study. PLoS One, 2011. 6(10): p. e26937. [CrossRef]
- Mastrolia, S.A., et al., Single vs. Recurrent Episodes of Preeclampsia-population–based Epidemiological and Clinical Characteristics. Maternal-Fetal Medicine, 2021. 3(3): p. 190-196. [CrossRef]
- Hutchinson, E.S., et al., Utero-placental haemodynamics in the pathogenesis of pre-eclampsia. Placenta, 2009. 30(7): p. 634-41. [CrossRef]
- Roberts, J.M. and C.A. Hubel, The two stage model of preeclampsia: variations on the theme. Placenta, 2009. 30 Suppl A(Suppl A): p. S32-7. [CrossRef]
- Lyall, F., S.C. Robson, and J.N. Bulmer, Spiral artery remodeling and trophoblast invasion in preeclampsia and fetal growth restriction: relationship to clinical outcome. Hypertension, 2013. 62(6): p. 1046-54.
- Aardema, M.W., et al., Uterine artery Doppler flow and uteroplacental vascular pathology in normal pregnancies and pregnancies complicated by pre-eclampsia and small for gestational age fetuses. Placenta, 2001. 22(5): p. 405-11. [CrossRef]
- Khong, T.Y., et al., Inadequate maternal vascular response to placentation in pregnancies complicated by pre-eclampsia and by small-for-gestational age infants. Br J Obstet Gynaecol, 1986. 93(10): p. 1049-59. [CrossRef]
- Espinoza, J., et al., Normal and abnormal transformation of the spiral arteries during pregnancy. J Perinat Med, 2006. 34(6): p. 447-58. [CrossRef]
- Jones, C.J. and H. Fox, An ultrastructural and ultrahistochemical study of the human placenta in maternal pre-eclampsia. Placenta, 1980. 1(1): p. 61-76. [CrossRef]
- Longtine, M.S., et al., Villous trophoblast apoptosis is elevated and restricted to cytotrophoblasts in pregnancies complicated by preeclampsia, IUGR, or preeclampsia with IUGR. Placenta, 2012. 33(5): p. 352-9. [CrossRef]
- Redman, C.W., I.L. Sargent, and A.C. Staff, IFPA Senior Award Lecture: making sense of pre-eclampsia - two placental causes of preeclampsia? Placenta, 2014. 35 Suppl: p. S20-5.
- Tannetta, D., et al., Update of syncytiotrophoblast derived extracellular vesicles in normal pregnancy and preeclampsia. J Reprod Immunol, 2017. 119: p. 98-106. [CrossRef]
- Nikuei, P., et al., Accuracy of Soluble Endoglin for Diagnosis of Preeclampsia and its Severity. Iran Biomed J, 2017. 21(5): p. 312-30. [CrossRef]
- von Dadelszen, P., L.A. Magee, and J.M. Roberts, Subclassification of preeclampsia. Hypertens Pregnancy, 2003. 22(2): p. 143-8. [CrossRef]
- McCartney, C.P., Pathological Anatomy of Acute Hypertension of Pregnancy. Circulation, 1964. 30: p. SUPPL 2:37-42. [CrossRef]
- Xiong, X. and W.D. Fraser, Impact of pregnancy-induced hypertension on birthweight by gestational age. Paediatr Perinat Epidemiol, 2004. 18(3): p. 186-91. [CrossRef]
- Redman, C.W. and A.C. Staff, Preeclampsia, biomarkers, syncytiotrophoblast stress, and placental capacity. Am J Obstet Gynecol, 2015. 213(4 Suppl): p. S9 e1, S9-11. [CrossRef]
- Redman, E.K., et al., Clinical Course, Associated Factors, and Blood Pressure Profile of Delayed-Onset Postpartum Preeclampsia. Obstet Gynecol, 2019. 134(5): p. 995-1001. [CrossRef]
- McElrath, T.F., et al., Circulating microparticle proteins obtained in the late first trimester predict spontaneous preterm birth at less than 35 weeks' gestation: a panel validation with specific characterization by parity. Am J Obstet Gynecol, 2019. 220(5): p. 488 e1-488 e11. [CrossRef]
- Powers, R.W., et al., Low placental growth factor across pregnancy identifies a subset of women with preterm preeclampsia: type 1 versus type 2 preeclampsia? Hypertension, 2012. 60(1): p. 239-46.
- Benton, S.J., et al., The clinical heterogeneity of preeclampsia is related to both placental gene expression and placental histopathology. Am J Obstet Gynecol, 2018. 219(6): p. 604 e1-604 e25. [CrossRef]
- Wilson, S.L., et al., Mining DNA methylation alterations towards a classification of placental pathologies. Hum Mol Genet, 2018. 27(1): p. 135-146. [CrossRef]
- Leavey, K., S.A. Bainbridge, and B.J. Cox, Large scale aggregate microarray analysis reveals three distinct molecular subclasses of human preeclampsia. PLoS One, 2015. 10(2): p. e0116508. [CrossRef]
- Leavey, K., D. Grynspan, and B.J. Cox, Both "canonical" and "immunological" preeclampsia subtypes demonstrate changes in placental immune cell composition. Placenta, 2019. 83: p. 53-56.
- Roberts, J.M., et al., Subtypes of Preeclampsia: Recognition and Determining Clinical Usefulness. Hypertension, 2021. 77(5): p. 1430-1441. [CrossRef]
- Conrad, K.P. and D.F. Benyo, Placental cytokines and the pathogenesis of preeclampsia. Am J Reprod Immunol, 1997. 37(3): p. 240-9. [CrossRef]
- Harmon, A.C., et al., The role of inflammation in the pathology of preeclampsia. Clin Sci (Lond), 2016. 130(6): p. 409-19. [CrossRef]
- Moffett, A. and F. Colucci, Uterine NK cells: active regulators at the maternal-fetal interface. J Clin Invest, 2014. 124(5): p. 1872-9. [CrossRef]
- Hiby, S.E., et al., Combinations of maternal KIR and fetal HLA-C genes influence the risk of preeclampsia and reproductive success. J Exp Med, 2004. 200(8): p. 957-65. [CrossRef]
- Triche, E.W., et al., Maternal-fetal HLA sharing and preeclampsia: variation in effects by seminal fluid exposure in a case-control study of nulliparous women in Iowa. J Reprod Immunol, 2014. 101-102: p. 111-119. [CrossRef]
- Kenny, L.C. and D.B. Kell, Immunological Tolerance, Pregnancy, and Preeclampsia: The Roles of Semen Microbes and the Father. Front Med (Lausanne), 2017. 4: p. 239. [CrossRef]
- Myatt, L. and J.M. Roberts, Preeclampsia: Syndrome or Disease? Curr Hypertens Rep, 2015. 17(11): p. 83.
- Vangrieken, P., et al., Placental Mitochondrial Abnormalities in Preeclampsia. Reprod Sci, 2021. 28(8): p. 2186-2199. [CrossRef]
- Holland, O.J., et al., Placental mitochondrial adaptations in preeclampsia associated with progression to term delivery. Cell Death Dis, 2018. 9(12): p. 1150. [CrossRef]
- Pandey, D., et al., Mitochondrial DNA copy number variation - A potential biomarker for early onset preeclampsia. Pregnancy Hypertens, 2021. 23: p. 1-4. [CrossRef]
- Cushen, S.C., et al., Reduced Maternal Circulating Cell-Free Mitochondrial DNA Is Associated With the Development of Preeclampsia. J Am Heart Assoc, 2022. 11(2): p. e021726. [CrossRef]
- Marschalek, J., et al., Maternal serum mitochondrial DNA (mtDNA) levels are elevated in preeclampsia - A matched case-control study. Pregnancy Hypertens, 2018. 14: p. 195-199. [CrossRef]
- Qiu, C., et al., A case-control study of maternal blood mitochondrial DNA copy number and preeclampsia risk. Int J Mol Epidemiol Genet, 2012. 3(3): p. 237-44.
- McCarthy, C. and L.C. Kenny, Therapeutically targeting mitochondrial redox signalling alleviates endothelial dysfunction in preeclampsia. Sci Rep, 2016. 6: p. 32683. [CrossRef]
- Westrate, L.M., et al., Mitochondrial morphological features are associated with fission and fusion events. PLoS One, 2014. 9(4): p. e95265. [CrossRef]
- Youle, R.J. and A.M. van der Bliek, Mitochondrial fission, fusion, and stress. Science, 2012. 337(6098): p. 1062-5.
- Yu, J., et al., Downregulation of Mitofusin 2 in Placenta Is Related to Preeclampsia. Biomed Res Int, 2016. 2016: p. 6323086. [CrossRef]
- Ryan, J.J., et al., PGC1alpha-mediated mitofusin-2 deficiency in female rats and humans with pulmonary arterial hypertension. Am J Respir Crit Care Med, 2013. 187(8): p. 865-78. [CrossRef]
- Salgado, S.S. and M.K.R. Salgado, Structural changes in pre-eclamptic and eclamptic placentas--an ultrastructural study. J Coll Physicians Surg Pak, 2011. 21(8): p. 482-6.
- Muralimanoharan, S., et al., MIR-210 modulates mitochondrial respiration in placenta with preeclampsia. Placenta, 2012. 33(10): p. 816-23. [CrossRef]
- Vishnyakova, P.A., et al., Mitochondrial role in adaptive response to stress conditions in preeclampsia. Sci Rep, 2016. 6: p. 32410. [CrossRef]
- Yung, H.W., et al., Noncanonical mitochondrial unfolded protein response impairs placental oxidative phosphorylation in early-onset preeclampsia. Proc Natl Acad Sci U S A, 2019. 116(36): p. 18109-18118. [CrossRef]
- Ausman, J., et al., Ceramide-induced BOK promotes mitochondrial fission in preeclampsia. Cell Death Dis, 2018. 9(3): p. 298. [CrossRef]
- Acin-Perez, R., et al., A novel approach to measure mitochondrial respiration in frozen biological samples. EMBO J, 2020. 39(13): p. e104073. [CrossRef]
- Cadenas, E. and K.J. Davies, Mitochondrial free radical generation, oxidative stress, and aging. Free Radic Biol Med, 2000. 29(3-4): p. 222-30. [CrossRef]
- Guzy, R.D., et al., Mitochondrial complex III is required for hypoxia-induced ROS production and cellular oxygen sensing. Cell Metab, 2005. 1(6): p. 401-8. [CrossRef]
- Hernansanz-Agustin, P., et al., Mitochondrial complex I deactivation is related to superoxide production in acute hypoxia. Redox Biol, 2017. 12: p. 1040-1051. [CrossRef]
- Gupta, S., A. Agarwal, and R.K. Sharma, The role of placental oxidative stress and lipid peroxidation in preeclampsia. Obstet Gynecol Surv, 2005. 60(12): p. 807-16. [CrossRef]
- Wiktor, H., et al., Oxidative DNA damage in placentas from normal and pre-eclamptic pregnancies. Virchows Arch, 2004. 445(1): p. 74-8. [CrossRef]
- Llurba, E., et al., A comprehensive study of oxidative stress and antioxidant status in preeclampsia and normal pregnancy. Free Radic Biol Med, 2004. 37(4): p. 557-70. [CrossRef]
- Aris, A., et al., Potential biomarkers of preeclampsia: inverse correlation between hydrogen peroxide and nitric oxide early in maternal circulation and at term in placenta of women with preeclampsia. Placenta, 2009. 30(4): p. 342-7. [CrossRef]
- Wang, Y. and S.W. Walsh, Placental mitochondria as a source of oxidative stress in pre-eclampsia. Placenta, 1998. 19(8): p. 581-6. [CrossRef]
- Rafeeinia, A., et al., Serum copper, zinc and lipid peroxidation in pregnant women with preeclampsia in gorgan. Open Biochem J, 2014. 8: p. 83-8. [CrossRef]
- Shaker, O.G. and N.A. Sadik, Pathogenesis of preeclampsia: Implications of apoptotic markers and oxidative stress. Hum Exp Toxicol, 2013. 32(11): p. 1170-8.
- Padmini, E., S. Lavanya, and V. Uthra, Preeclamptic placental stress and over expression of mitochondrial HSP70. Clin Chem Lab Med, 2009. 47(9): p. 1073-80. [CrossRef]
- Wiktor, H. and M. Kankofer, Superoxide dismutase activity in normal and preeclamptic placentas. Ginekol Pol, 1998. 69(12): p. 915-8.
- Wu, F., F.J. Tian, and Y. Lin, Oxidative Stress in Placenta: Health and Diseases. Biomed Res Int, 2015. 2015: p. 293271. [CrossRef]
- Brunelle, J.K. and A. Letai, Control of mitochondrial apoptosis by the Bcl-2 family. J Cell Sci, 2009. 122(Pt 4): p. 437-41. [CrossRef]
- Allaire, A.D., et al., Placental apoptosis in preeclampsia. Obstet Gynecol, 2000. 96(2): p. 271-6.
- Leung, D.N., et al., Increased placental apoptosis in pregnancies complicated by preeclampsia. Am J Obstet Gynecol, 2001. 184(6): p. 1249-50. [CrossRef]
- Fogarty, N.M., A.C. Ferguson-Smith, and G.J. Burton, Syncytial knots (Tenney-Parker changes) in the human placenta: evidence of loss of transcriptional activity and oxidative damage. Am J Pathol, 2013. 183(1): p. 144-52.
- Hung, T.H., et al., Hypoxia-reoxygenation: a potent inducer of apoptotic changes in the human placenta and possible etiological factor in preeclampsia. Circ Res, 2002. 90(12): p. 1274-81.
- Sanchez-Aranguren, L.C., et al., Endothelial dysfunction and preeclampsia: role of oxidative stress. Front Physiol, 2014. 5: p. 372. [CrossRef]
- Vaka, V.R., et al., Role of Mitochondrial Dysfunction and Reactive Oxygen Species in Mediating Hypertension in the Reduced Uterine Perfusion Pressure Rat Model of Preeclampsia. Hypertension, 2018. 72(3): p. 703-711. [CrossRef]
- Covarrubias, A.E., et al., AP39, a Modulator of Mitochondrial Bioenergetics, Reduces Antiangiogenic Response and Oxidative Stress in Hypoxia-Exposed Trophoblasts: Relevance for Preeclampsia Pathogenesis. Am J Pathol, 2019. 189(1): p. 104-114.
- Nagamatsu, T., et al., Cytotrophoblasts up-regulate soluble fms-like tyrosine kinase-1 expression under reduced oxygen: an implication for the placental vascular development and the pathophysiology of preeclampsia. Endocrinology, 2004. 145(11): p. 4838-45. [CrossRef]
- Jiang, Z., et al., A Role of sFlt-1 in Oxidative Stress and Apoptosis in Human and Mouse Pre-Eclamptic Trophoblasts. Biol Reprod, 2015. 93(3): p. 73. [CrossRef]
- Tong, W., et al., Chronic Hypoxia in Ovine Pregnancy Recapitulates Physiological and Molecular Markers of Preeclampsia in the Mother, Placenta, and Offspring. Hypertension, 2022. 79(7): p. 1525-1535. [CrossRef]
- Peracoli, J.C., M.V. Rudge, and M.T. Peracoli, Tumor necrosis factor-alpha in gestation and puerperium of women with gestational hypertension and pre-eclampsia. Am J Reprod Immunol, 2007. 57(3): p. 177-85. [CrossRef]
- Raghupathy, R., Cytokines as key players in the pathophysiology of preeclampsia. Med Princ Pract, 2013. 22 Suppl 1: p. 8-19. [CrossRef]
- Clayton, S.A., et al., Mitochondria as Key Players in the Pathogenesis and Treatment of Rheumatoid Arthritis. Front Immunol, 2021. 12: p. 673916. [CrossRef]
- Green, D.R., L. Galluzzi, and G. Kroemer, Mitochondria and the autophagy-inflammation-cell death axis in organismal aging. Science, 2011. 333(6046): p. 1109-12. [CrossRef]
- Lopez-Armada, M.J., et al., Mitochondrial dysfunction and the inflammatory response. Mitochondrion, 2013. 13(2): p. 106-18. [CrossRef]
- Blaser, H., et al., TNF and ROS Crosstalk in Inflammation. Trends Cell Biol, 2016. 26(4): p. 249-261. [CrossRef]
- Fiers, W., et al., More than one way to die: apoptosis, necrosis and reactive oxygen damage. Oncogene, 1999. 18(54): p. 7719-30. [CrossRef]
- Kim, J.J., et al., TNF-alpha-induced ROS production triggering apoptosis is directly linked to Romo1 and Bcl-X(L). Cell Death Differ, 2010. 17(9): p. 1420-34.
- Roca, F.J., et al., Tumor necrosis factor induces pathogenic mitochondrial ROS in tuberculosis through reverse electron transport. Science, 2022. 376(6600): p. eabh2841. [CrossRef]
- Wang, Y. and S.W. Walsh, TNF alpha concentrations and mRNA expression are increased in preeclamptic placentas. J Reprod Immunol, 1996. 32(2): p. 157-69.
- I, C.W., et al., Increased expression of NLRP3 inflammasome in placentas from pregnant women with severe preeclampsia. J Reprod Immunol, 2017. 123: p. 40-47.
- Alexander, B.T., et al., Tumor necrosis factor-alpha-induced hypertension in pregnant rats results in decreased renal neuronal nitric oxide synthase expression. Am J Hypertens, 2002. 15(2 Pt 1): p. 170-5. [CrossRef]
- Guo, X., et al., Upregulation of VEGF by small activating RNA and its implications in preeclampsia. Placenta, 2016. 46: p. 38-44. [CrossRef]
- Xu, B., et al., TNF-alpha inhibits trophoblast integration into endothelial cellular networks. Placenta, 2011. 32(3): p. 241-6. [CrossRef]
- Muralimanoharan, S., et al., Sexual dimorphism in miR-210 expression and mitochondrial dysfunction in the placenta with maternal obesity. Int J Obes (Lond), 2015. 39(8): p. 1274-81. [CrossRef]
- Lien, Y.C., et al., Intrauterine Inflammation Alters the Transcriptome and Metabolome in Placenta. Front Physiol, 2020. 11: p. 592689. [CrossRef]
- Batchuluun, B., et al., Elevated Medium-Chain Acylcarnitines Are Associated With Gestational Diabetes Mellitus and Early Progression to Type 2 Diabetes and Induce Pancreatic beta-Cell Dysfunction. Diabetes, 2018. 67(5): p. 885-897.
- Odegard, R.A., et al., Risk factors and clinical manifestations of pre-eclampsia. BJOG, 2000. 107(11): p. 1410-6. [CrossRef]
- Redman, C., Pre-eclampsia: A complex and variable disease. Pregnancy Hypertens, 2014. 4(3): p. 241-2. [CrossRef]
- Clark, D.E., et al., A vascular endothelial growth factor antagonist is produced by the human placenta and released into the maternal circulation. Biol Reprod, 1998. 59(6): p. 1540-8. [CrossRef]
- Levine, R.J., et al., Circulating angiogenic factors and the risk of preeclampsia. N Engl J Med, 2004. 350(7): p. 672-83. [CrossRef]
- Mor, G., et al., Inflammation and pregnancy: the role of the immune system at the implantation site. Ann N Y Acad Sci, 2011. 1221(1): p. 80-7. [CrossRef]
- Abu-Raya, B., et al., Maternal Immunological Adaptation During Normal Pregnancy. Front Immunol, 2020. 11: p. 575197. [CrossRef]
- Tang, X., et al., Mitochondria, endothelial cell function, and vascular diseases. Front Physiol, 2014. 5: p. 175.
- Kirkman, D.L., et al., Mitochondrial contributions to vascular endothelial dysfunction, arterial stiffness, and cardiovascular diseases. Am J Physiol Heart Circ Physiol, 2021. 320(5): p. H2080-H2100. [CrossRef]
- Ricci, C.A., et al., Mitochondria-mediated Maternal-fetal Interactions and Consequences of Mitochondrial Dysregulation Indicate New Roles for Mitochondria in Hypertensive Pregnancies. medRxiv, 2021: p. 2021.12.18.21268029.
- Sanchez-Aranguren, L.C., et al., Soluble Fms-Like Tyrosine Kinase-1 Alters Cellular Metabolism and Mitochondrial Bioenergetics in Preeclampsia. Front Physiol, 2018. 9: p. 83. [CrossRef]
- Deer, E., et al., Vascular endothelial mitochondrial oxidative stress in response to preeclampsia: a role for angiotension II type 1 autoantibodies. Am J Obstet Gynecol MFM, 2021. 3(1): p. 100275. [CrossRef]
- Duley, L., Pre-eclampsia and the hypertensive disorders of pregnancy. Br Med Bull, 2003. 67: p. 161-76. [CrossRef]
- Uriho, A., et al., Effects of resveratrol on mitochondrial biogenesis and physiological diseases. Advances in Traditional Medicine, 2021. 21(1): p. 1-14. [CrossRef]
- Bonnefont-Rousselot, D., Resveratrol and Cardiovascular Diseases. Nutrients, 2016. 8(5). [CrossRef]
- Poudel, R., et al., Effects of resveratrol in pregnancy using murine models with reduced blood supply to the uterus. PLoS One, 2013. 8(5): p. e64401. [CrossRef]
- Cudmore, M.J., et al., Resveratrol inhibits the release of soluble fms-like tyrosine kinase (sFlt-1) from human placenta. Am J Obstet Gynecol, 2012. 206(3): p. 253 e10-5. [CrossRef]
- Ding, J., et al., Efficacy of resveratrol to supplement oral nifedipine treatment in pregnancy-induced preeclampsia. Endocr Connect, 2017. 6(8): p. 595-600. [CrossRef]
- Giusti, L., et al., The Protective Action of Metformin against Pro-Inflammatory Cytokine-Induced Human Islet Cell Damage and the Mechanisms Involved. Cells, 2022. 11(15). [CrossRef]
- Sung, J.Y., et al., Metformin mitigates stress-induced premature senescence by upregulating AMPKalpha at Ser485 phosphorylation induced SIRT3 expression and inactivating mitochondrial oxidants. Mech Ageing Dev, 2022. 206: p. 111708.
- Ansari, A., et al., Function of the SIRT3 mitochondrial deacetylase in cellular physiology, cancer, and neurodegenerative disease. Aging Cell, 2017. 16(1): p. 4-16.
- Feng, J., et al., Mitochondria as an important target of metformin: The mechanism of action, toxic and side effects, and new therapeutic applications. Pharmacol Res, 2022. 177: p. 106114. [CrossRef]
- Herzig, S. and R.J. Shaw, AMPK: guardian of metabolism and mitochondrial homeostasis. Nat Rev Mol Cell Biol, 2018. 19(2): p. 121-135. [CrossRef]
- Brownfoot, F.C., et al., Metformin as a prevention and treatment for preeclampsia: effects on soluble fms-like tyrosine kinase 1 and soluble endoglin secretion and endothelial dysfunction. Am J Obstet Gynecol, 2016. 214(3): p. 356 e1-356 e15. [CrossRef]
- Wang, F., et al., Effect of Metformin on a Preeclampsia-Like Mouse Model Induced by High-Fat Diet. Biomed Res Int, 2019. 2019: p. 6547019. [CrossRef]
- Alqudah, A., et al., Risk of pre-eclampsia in women taking metformin: a systematic review and meta-analysis. Diabet Med, 2018. 35(2): p. 160-172. [CrossRef]
- Glueck, C.J., et al., Height, weight, and motor-social development during the first 18 months of life in 126 infants born to 109 mothers with polycystic ovary syndrome who conceived on and continued metformin through pregnancy. Hum Reprod, 2004. 19(6): p. 1323-30. [CrossRef]
- Glueck, C.J., et al., Effects of metformin-diet intervention before and throughout pregnancy on obstetric and neonatal outcomes in patients with polycystic ovary syndrome. Curr Med Res Opin, 2013. 29(1): p. 55-62. [CrossRef]
- De Leo, V., et al., The administration of metformin during pregnancy reduces polycystic ovary syndrome related gestational complications. Eur J Obstet Gynecol Reprod Biol, 2011. 157(1): p. 63-6. [CrossRef]
- Cluver, C.A., et al., Use of metformin to prolong gestation in preterm pre-eclampsia: randomised, double blind, placebo controlled trial. BMJ, 2021. 374: p. n2103. [CrossRef]
- Thompson, J.D. and D. Diers, Management of nursing intensity. Nurs Clin North Am, 1988. 23(3): p. 473-92. [CrossRef]
- Long, J., et al., Mitochondria Targeted Antioxidant Significantly Alleviates Preeclampsia Caused by 11beta-HSD2 Dysfunction via OPA1 and MtDNA Maintenance. Antioxidants (Basel), 2022. 11(8).
- Gane, E.J., et al., The mitochondria-targeted anti-oxidant mitoquinone decreases liver damage in a phase II study of hepatitis C patients. Liver Int, 2010. 30(7): p. 1019-26. [CrossRef]
- Snow, B.J., et al., A double-blind, placebo-controlled study to assess the mitochondria-targeted antioxidant MitoQ as a disease-modifying therapy in Parkinson's disease. Mov Disord, 2010. 25(11): p. 1670-4.
- Kirkman, D.L., et al., Role of mitochondria-derived reactive oxygen species in microvascular dysfunction in chronic kidney disease. Am J Physiol Renal Physiol, 2018. 314(3): p. F423-F429. [CrossRef]
- Mason, S.A., et al., Effect of mitochondrial-targeted antioxidants on glycaemic control, cardiovascular health, and oxidative stress in humans: A systematic review and meta-analysis of randomized controlled trials. Diabetes Obes Metab, 2022. 24(6): p. 1047-1060.
- Yang, Y., et al., The Potent Antioxidant MitoQ Protects Against Preeclampsia During Late Gestation but Increases the Risk of Preeclampsia When Administered in Early Pregnancy. Antioxid Redox Signal, 2021. 34(2): p. 118-136. [CrossRef]
- Ayla, S., et al., Doxorubicin induced nephrotoxicity: protective effect of nicotinamide. Int J Cell Biol, 2011. 2011: p. 390238. [CrossRef]
- Wahlberg, G., et al., Protective effect of nicotinamide against nephropathy in diabetic rats. Diabetes Res, 1985. 2(6): p. 307-12.
- Rongvaux, A., et al., Reconstructing eukaryotic NAD metabolism. Bioessays, 2003. 25(7): p. 683-90. [CrossRef]
- Canto, C., K.J. Menzies, and J. Auwerx, NAD(+) Metabolism and the Control of Energy Homeostasis: A Balancing Act between Mitochondria and the Nucleus. Cell Metab, 2015. 22(1): p. 31-53. [CrossRef]
- Li, F., et al., Nicotinamide benefits both mothers and pups in two contrasting mouse models of preeclampsia. Proc Natl Acad Sci U S A, 2016. 113(47): p. 13450-13455. [CrossRef]
- Li, D., et al., Nicotinamide supplementation induces detrimental metabolic and epigenetic changes in developing rats. Br J Nutr, 2013. 110(12): p. 2156-64. [CrossRef]
- Mahmoud, Y.I. and A.A. Mahmoud, Role of nicotinamide (vitamin B3) in acetaminophen-induced changes in rat liver: Nicotinamide effect in acetaminophen-damged liver. Exp Toxicol Pathol, 2016. 68(6): p. 345-54.
- Tian, Y.J., et al., Maternal nicotinamide supplementation causes global DNA hypomethylation, uracil hypo-incorporation and gene expression changes in fetal rats. Br J Nutr, 2014. 111(9): p. 1594-601. [CrossRef]
- Sun, W.P., et al., Comparison of the effects of nicotinic acid and nicotinamide degradation on plasma betaine and choline levels. Clin Nutr, 2017. 36(4): p. 1136-1142. [CrossRef]
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




