Submitted:
13 January 2023
Posted:
22 January 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials. Subjects
2.2. Drug
2.3. Diets
2.4. To Determine if Binge Eating of a High-Fat Diet (HFD) Induces Conditioned Place Preference or Alter the Rewarding Action Oxycodone, a Mu Opioid Receptor Agonist, and if Sex-Related Differences Exist in These Responses
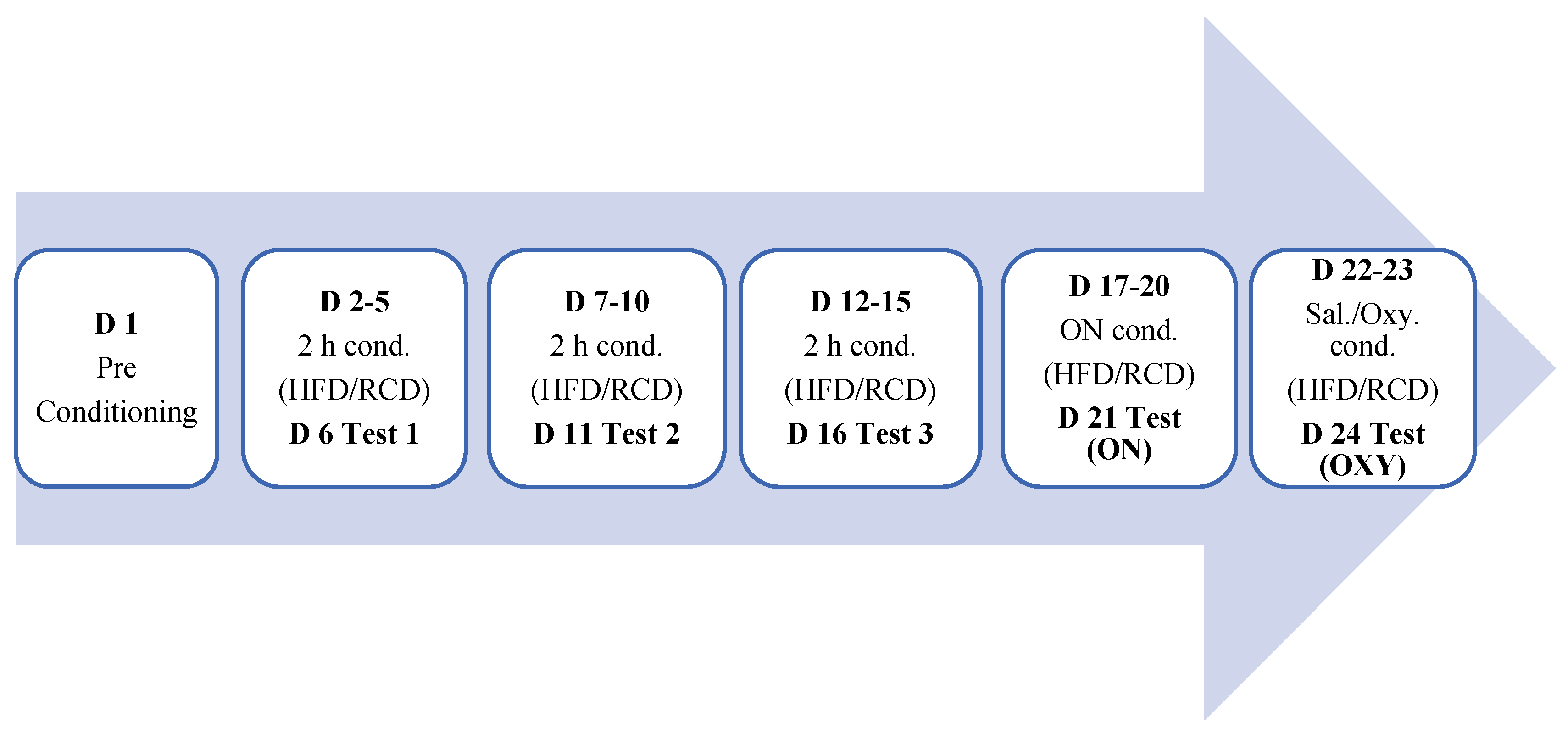
2.5. To Assess the Role of MOR in High-Fat Diet (HFD) Induced Reward and if Sex-Related Differences Exist in These Responses
2.6. Data Analysis
3. Results
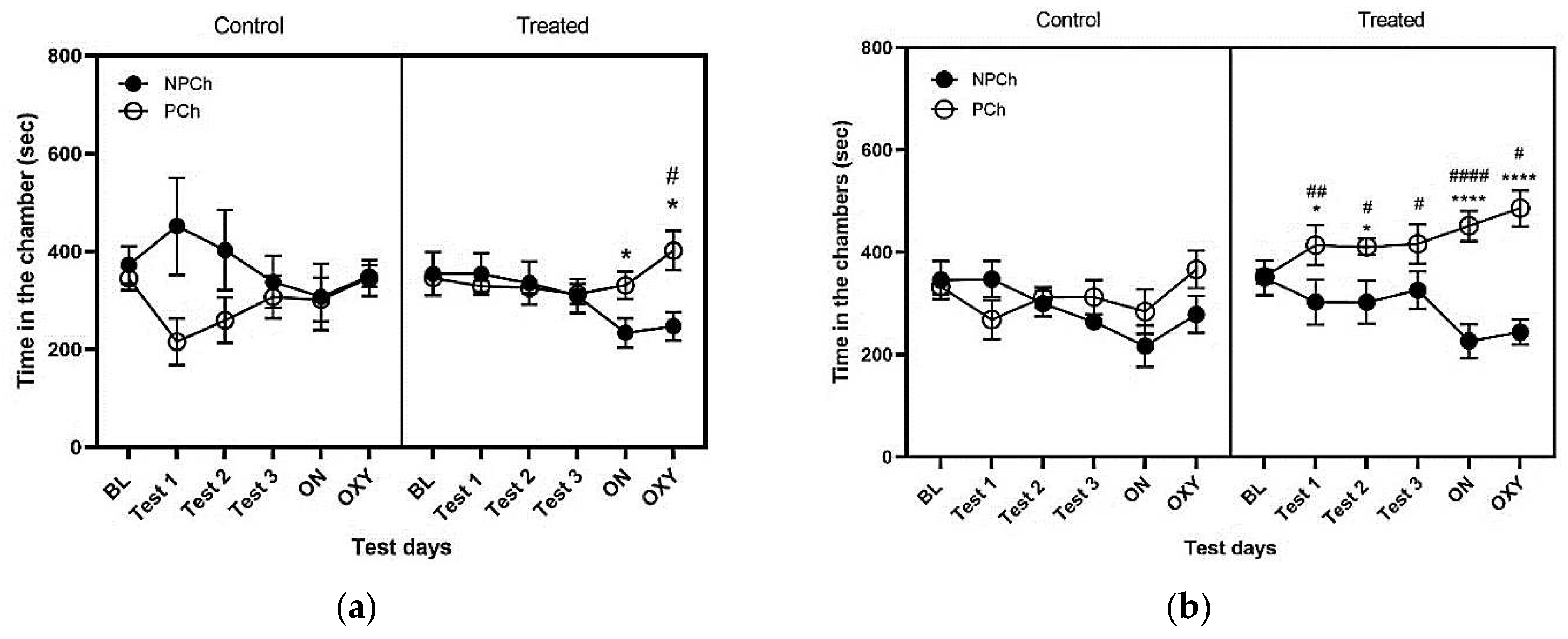
3.1. Conditioning with a High-Fat Diet (HFD) for 16 h but Not 2 h Induced Conditioned Place Preference and Enhanced the Rewarding Action of Oxycodone in Male and Female C57BL/6J Mice
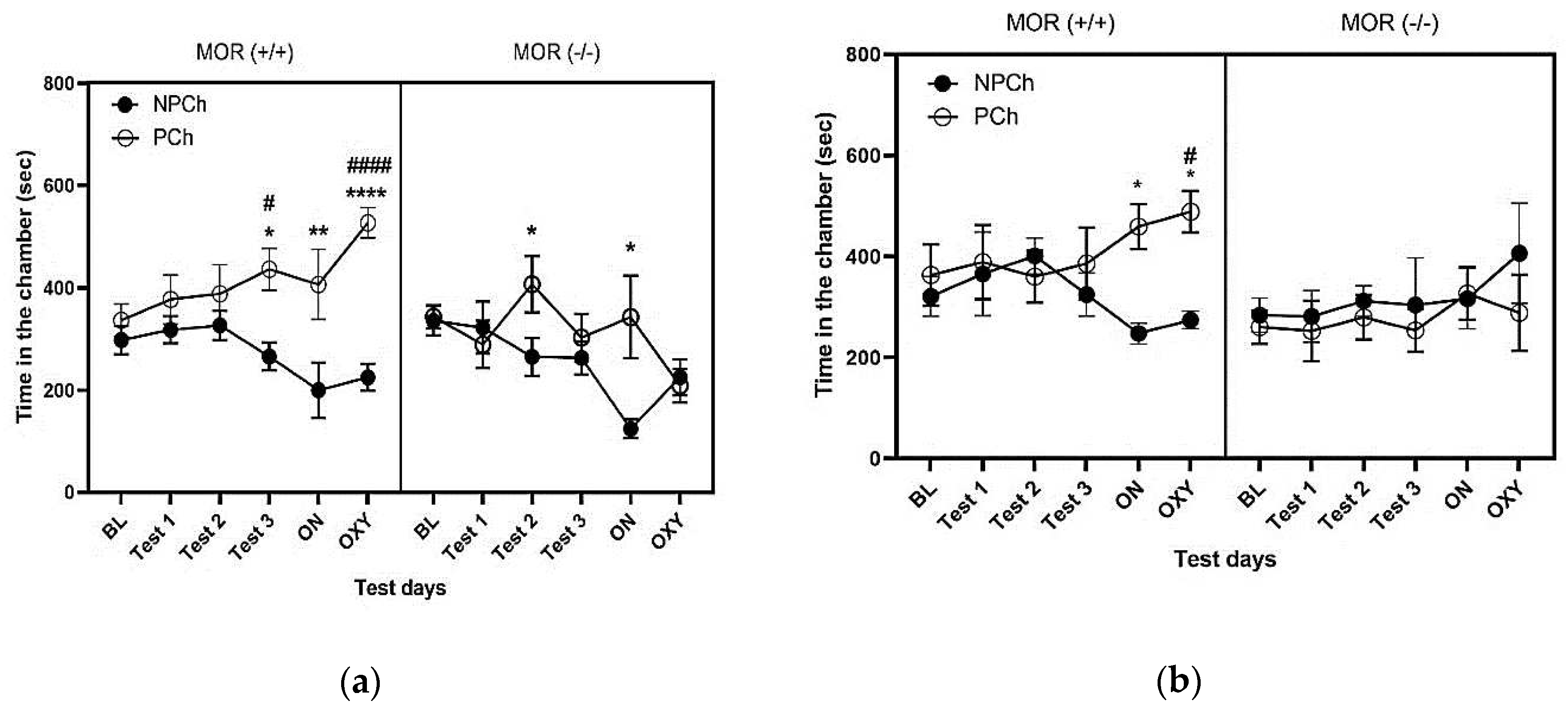
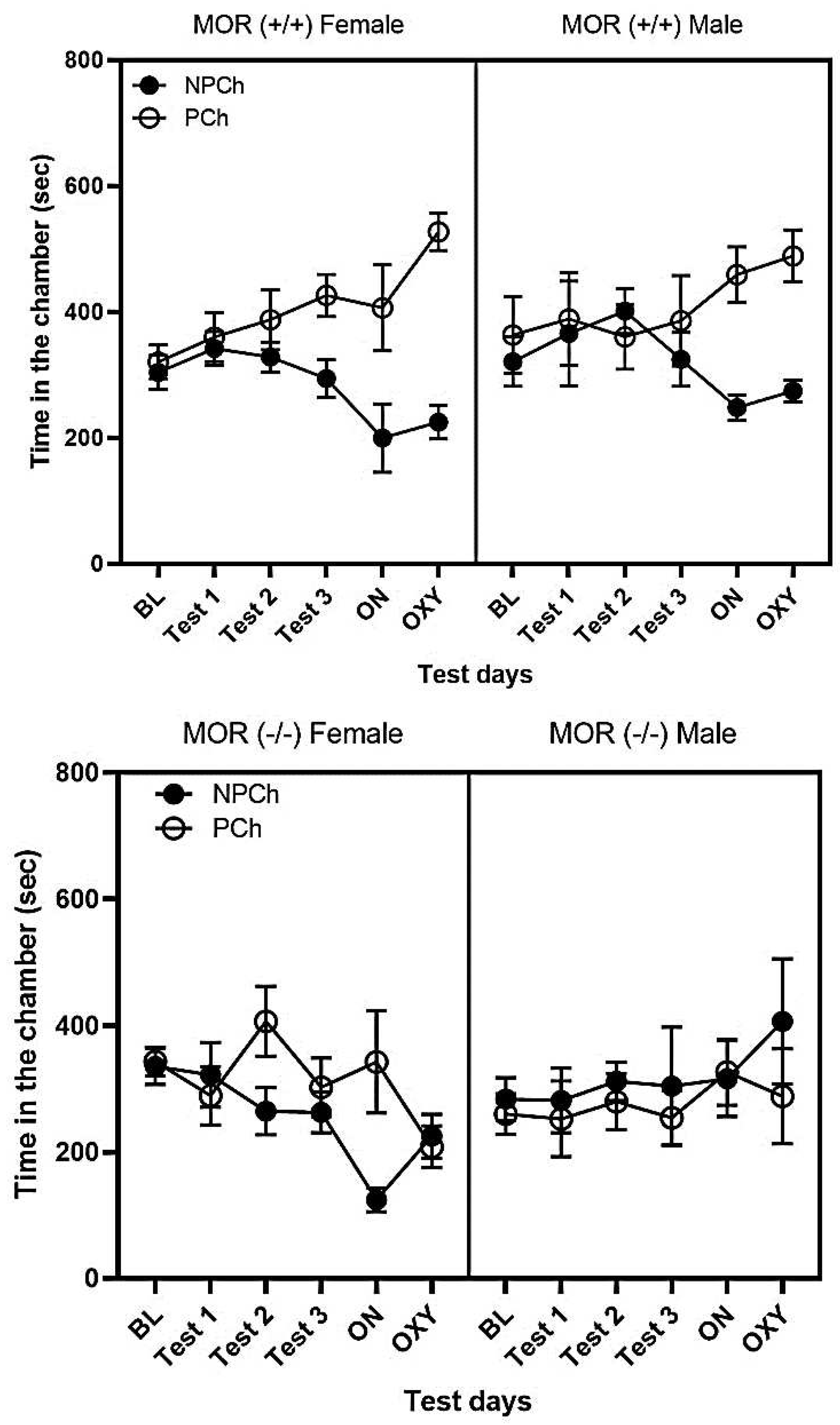
3.2. LAC but Not SAC with HFD Induced CPP and Enhanced the Rewarding Action of Oxycodone in Wild-Type (MOR+/+) but Not MOR Knockout (MOR-/-) Mice
3.3. Comparison of CPP Response between male And Female Control vs. Treated Mice
3.4. Comparison of CPP Response between Male and Female MOR Knockout and Wildtype Mice
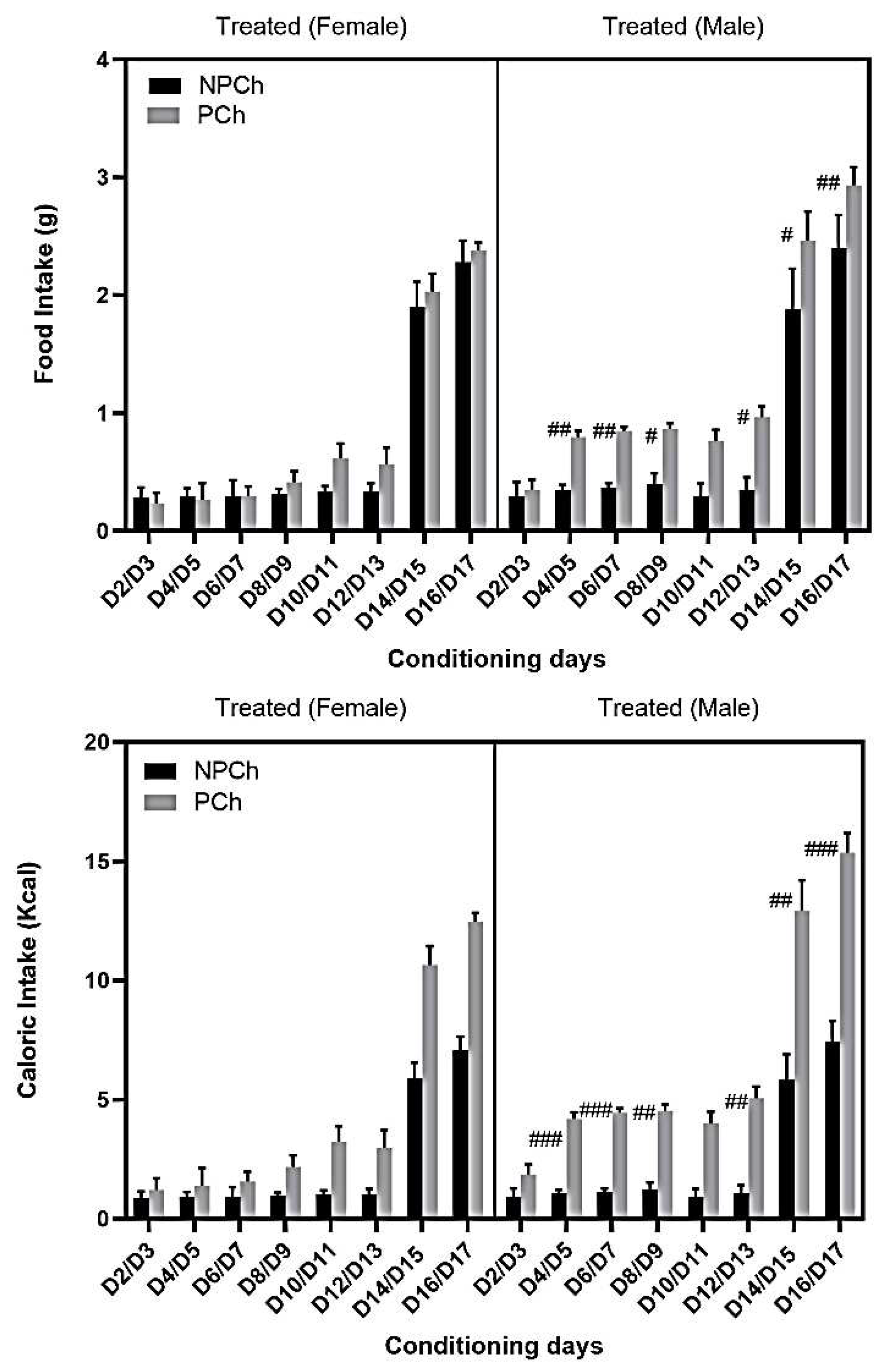
3.5. Food intake and Calorie Consumption between Treated and Control Male and Female C57BL/6J
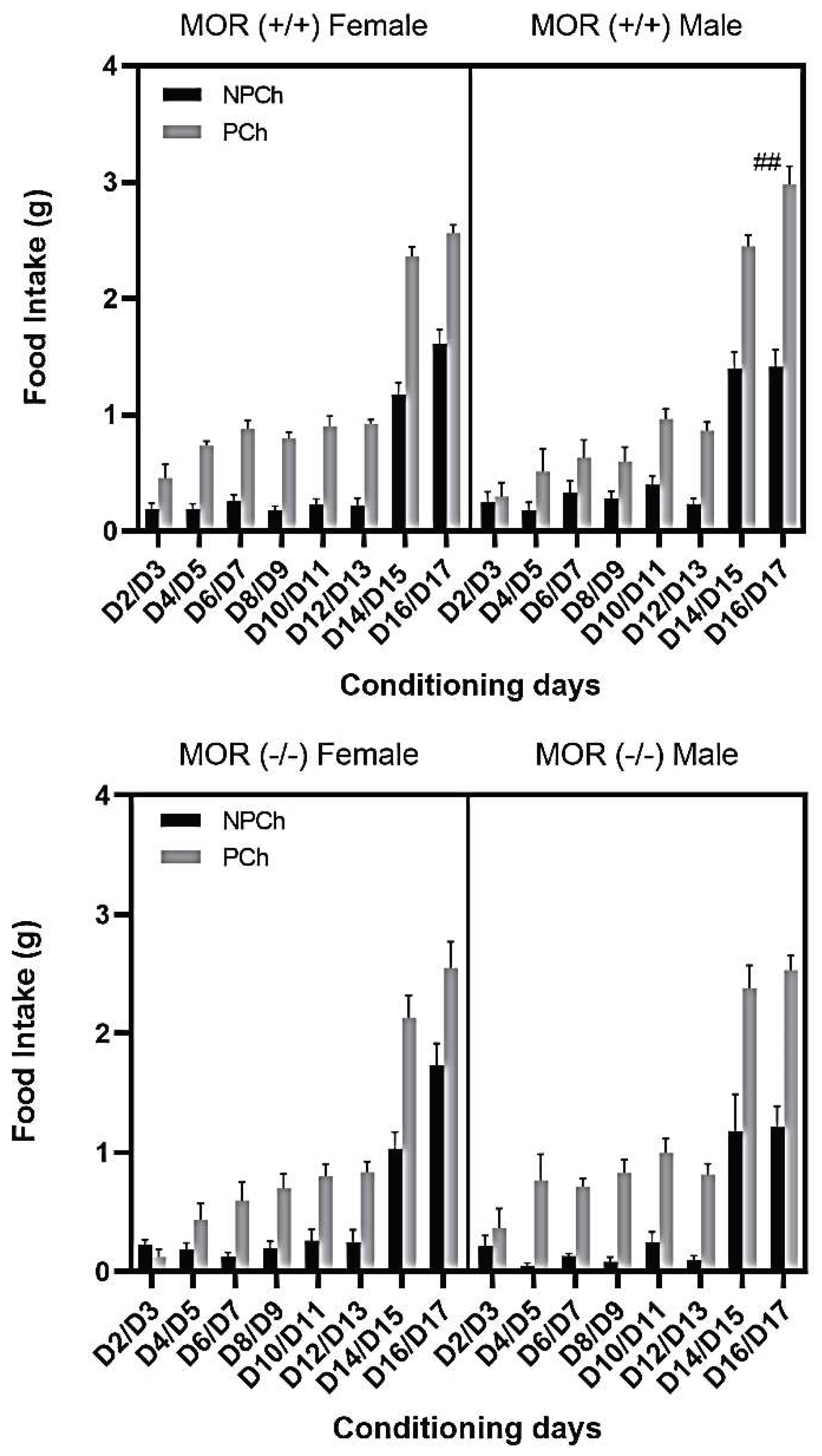
3.6. Comparison in Food Intake between MOR Male and Female Mice (Knockout vs. Wildtype)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nogueiras, R., et al., The opioid system and food intake: homeostatic and hedonic mechanisms. Obes Facts, 2012. 5(2): p. 196-207. [CrossRef]
- Castro, D.C. and K.C. Berridge, Opioid hedonic hotspot in nucleus accumbens shell: mu, delta, and kappa maps for enhancement of sweetness "liking" and "wanting". J Neurosci, 2014. 34(12): p. 4239-50.
- Spanagel, R., A. Herz, and T.S. Shippenberg, Opposing tonically active endogenous opioid systems modulate the mesolimbic dopaminergic pathway. Proc Natl Acad Sci U S A, 1992. 89(6): p. 2046-50. [CrossRef]
- Nestler, E.J., Molecular basis of long-term plasticity underlying addiction. Nat Rev Neurosci, 2001. 2(2): p. 119-28. [CrossRef]
- Mendez, I.A., et al., Involvement of Endogenous Enkephalins and β-Endorphin in Feeding and Diet-Induced Obesity. Neuropsychopharmacology, 2015. 40(9): p. 2103-12. [CrossRef]
- Barnes, M.J., et al., Increased expression of mu opioid receptors in animals susceptible to diet-induced obesity. Peptides, 2006. 27(12): p. 3292-8. [CrossRef]
- Reyes, T.M., High-fat diet alters the dopamine and opioid systems: effects across development. Int J Obes Suppl, 2012. 2(Suppl 2): p. S25-8. [CrossRef]
- Ziauddeen, H., et al., Effects of the mu-opioid receptor antagonist GSK1521498 on hedonic and consummatory eating behaviour: a proof of mechanism study in binge-eating obese subjects. Mol Psychiatry, 2013. 18(12): p. 1287-93. [CrossRef]
- Tuulari, J.J., et al., Feeding Releases Endogenous Opioids in Humans. J Neurosci, 2017. 37(34): p. 8284-8291. [CrossRef]
- Glass, M.J., C.J. Billington, and A.S. Levine, Naltrexone administered to central nucleus of amygdala or PVN: neural dissociation of diet and energy. Am J Physiol Regul Integr Comp Physiol, 2000. 279(1): p. R86-92. [CrossRef]
- Lenard, N.R., H. Zheng, and H.R. Berthoud, Chronic suppression of μ-opioid receptor signaling in the nucleus accumbens attenuates development of diet-induced obesity in rats. Int J Obes (Lond), 2010. 34(6): p. 1001-10. [CrossRef]
- Yeomans, M.R. and R.W. Gray, Opioid peptides and the control of human ingestive behaviour. Neurosci Biobehav Rev, 2002. 26(6): p. 713-28. [CrossRef]
- Bodnar, R.J., Endogenous opioids and feeding behavior: a 30-year historical perspective. Peptides, 2004. 25(4): p. 697-725.
- Gosnell, B.A. and A.S. Levine, Reward systems and food intake: role of opioids. Int J Obes (Lond), 2009. 33 Suppl 2: p. S54-8. [CrossRef]
- Eikemo, M., et al., Sweet taste pleasantness is modulated by morphine and naltrexone. Psychopharmacology (Berl), 2016. 233(21-22): p. 3711-3723. [CrossRef]
- Wang, M., et al., Activation of orexin-1 receptors in the amygdala enhances feeding in the diet-induced obesity rats: Blockade with μ-opioid antagonist. Biochem Biophys Res Commun, 2018. 503(4): p. 3186-3191. [CrossRef]
- Frank, A., L.M. Brown, and D.J. Clegg, The role of hypothalamic estrogen receptors in metabolic regulation. Front Neuroendocrinol, 2014. 35(4): p. 550-7. [CrossRef]
- Becker, J.B. and E. Chartoff, Sex differences in neural mechanisms mediating reward and addiction. Neuropsychopharmacology, 2019. 44(1): p. 166-183. [CrossRef]
- Liu, X. and H. Shi, Regulation of Estrogen Receptor alpha Expression in the Hypothalamus by Sex Steroids: Implication in the Regulation of Energy Homeostasis. Int J Endocrinol, 2015. 2015: p. 949085.
- MacNicol, B., The biology of addiction. Can J Anaesth, 2017. 64(2): p. 141-148. [CrossRef]
- Bardo, M.T. and R.A. Bevins, Conditioned place preference: what does it add to our preclinical understanding of drug reward? Psychopharmacology (Berl), 2000. 153(1): p. 31-43.
- Blanco-Gandía, M.C., et al., The rewarding effects of ethanol are modulated by binge eating of a high-fat diet during adolescence. Neuropharmacology, 2017. 121: p. 219-230. [CrossRef]
- Kenny, P.J., G. Voren, and P.M. Johnson, Dopamine D2 receptors and striatopallidal transmission in addiction and obesity. Curr Opin Neurobiol, 2013. 23(4): p. 535-8. [CrossRef]
- Le Merrer, J., et al., Reward processing by the opioid system in the brain. Physiol Rev, 2009. 89(4): p. 1379-412. [CrossRef]
- Colantuoni, C., et al., Evidence that intermittent, excessive sugar intake causes endogenous opioid dependence. Obes Res, 2002. 10(6): p. 478-88. [CrossRef]
- Kelley, A.E., et al., Restricted daily consumption of a highly palatable food (chocolate Ensure(R)) alters striatal enkephalin gene expression. Eur J Neurosci, 2003. 18(9): p. 2592-8.
- Johnson, P.M. and P.J. Kenny, Dopamine D2 receptors in addiction-like reward dysfunction and compulsive eating in obese rats. Nat Neurosci, 2010. 13(5): p. 635-41.
- Cruz, B., et al., The emergence of insulin resistance following a chronic high-fat diet regimen coincides with an increase in the reinforcing effects of nicotine in a sex-dependent manner. Neuropharmacology, 2021. 200: p. 108787. [CrossRef]
- O'Dell, L.E., et al., Enhanced nicotine self-administration and suppressed dopaminergic systems in a rat model of diabetes. Addict Biol, 2014. 19(6): p. 1006-19. [CrossRef]
- Richardson, J.R., et al., Insulin resistant rats display enhanced rewarding effects of nicotine. Drug Alcohol Depend, 2014. 140: p. 205-7. [CrossRef]
- Volkow, N.D., et al., Obesity and addiction: neurobiological overlaps. Obes Rev, 2013. 14(1): p. 2-18. [CrossRef]
- Vucetic, Z., J. Kimmel, and T.M. Reyes, Chronic high-fat diet drives postnatal epigenetic regulation of μ-opioid receptor in the brain. Neuropsychopharmacology, 2011. 36(6): p. 1199-206. [CrossRef]
- Benarroch, E.E., Endogenous opioid systems: current concepts and clinical correlations. Neurology, 2012. 79(8): p. 807-14.
- Maggi, R., et al., Binding characteristics of hypothalamic mu opioid receptors throughout the estrous cycle in the rat. Neuroendocrinology, 1993. 58(3): p. 366-72. [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



