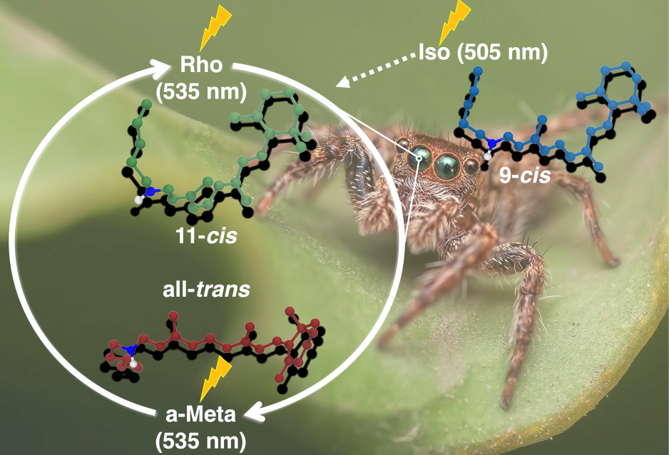
Bistable rhodopsins have two stable forms which can be interconverted by light. Due to their ability to act as photoswitches, these proteins are considered as ideal candidates for applica-tions such as optogenetics. In this work we analyze a recently crystalized bistable rhodopsin, namely the jumping spider rhodopsin-1 (JSR1). This rhodopsin exhibits identical absorption maxima for the parent and the photoproduct form, which impedes its broad application. We have performed hybrid QM/MM simulations to study three isomers of the retinal chromo-phore: the 9-cis, 11-cis and all-trans configurations. The main aim was to gain insight into the specific interactions of each isomer and their impact on the absorption maximum in JSR1. The absorption spectra were computed using sampled snapshots from a QM/MM molecular dy-namics trajectory and compared to experimental counterparts. The chromophore-protein in-teractions were analyzed by visualizing the electrostatic potential of the protein and projecting it onto chromophore. It was found that the distance between a nearby tyrosine (Y126) residue plays a larger role in the predicted absorption maximum than the primary counterion (E194). Geometric differences between the isomers were also noted including a structural change in the polyene chain of the chromophore as well as changes in the nearby hydrogen bond network.