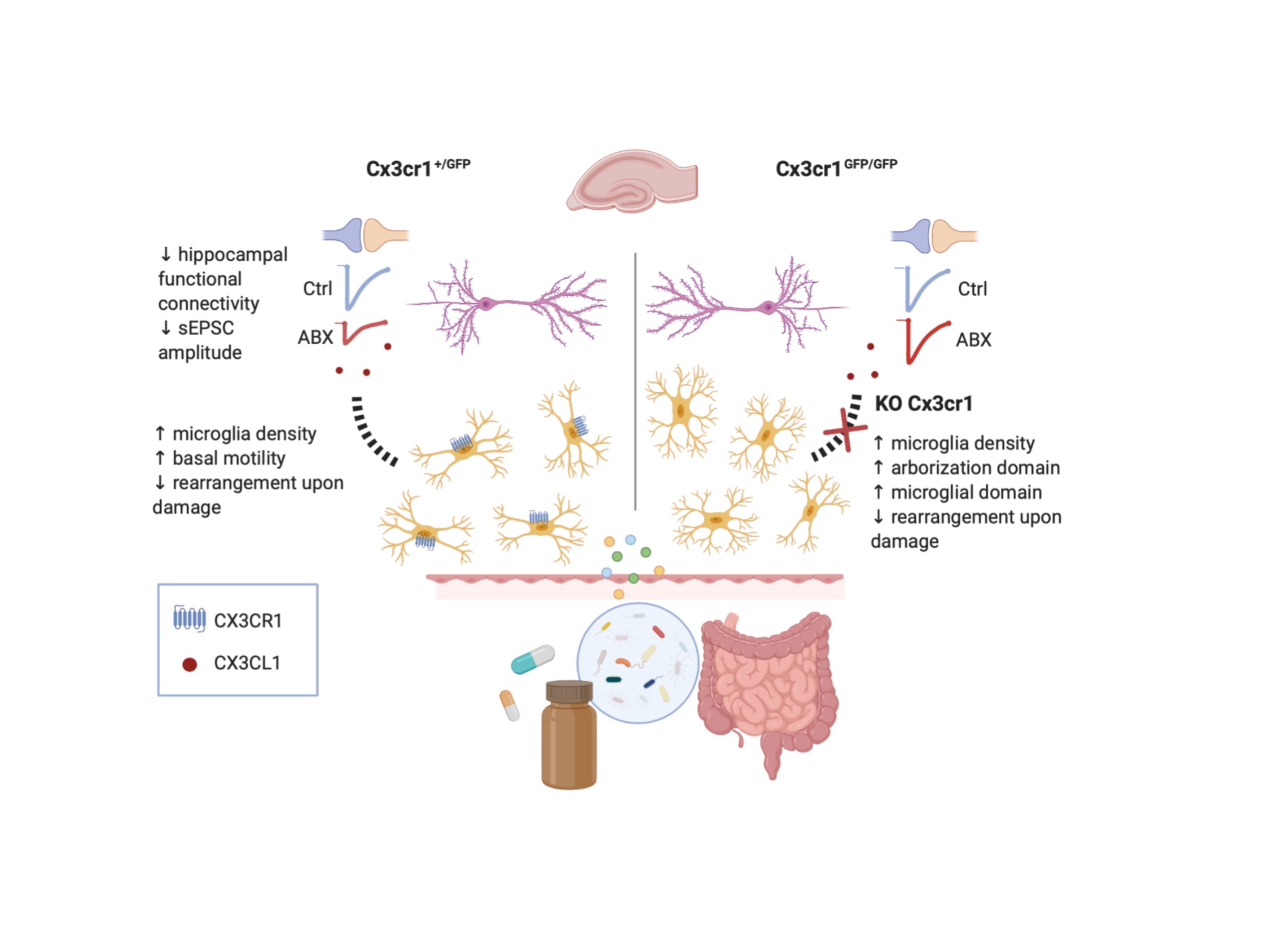
‘Dysbiosis’ of the adult gut microbiota, in response to challenges such as infection, altered diet, stress, and antibiotics treatment has been recently linked to pathological alteration of brain func-tion and behavior. Moreover, gut microbiota composition constantly controls microglia matura-tion as revealed by morphological observations and gene expression analysis. However, it is un-clear whether gut microbiota influences microglia functional properties and crosstalk with neu-rons, known to shape and modulate synaptic development and function. Here, we investigated how antibiotic mediated alteration of the gut microbiota influences microglial and neuronal functions in adult mice hippocampus. Hippocampal microglia from adult mice treated with oral antibiotics exhibited increased microglia density, altered basal patrolling activity, and impaired process rearrangement in response to damage. Patch clamp recordings at CA3-CA1 synapses revealed that antibiotics treatment alters neuronal functions, reducing spontaneous postsynaptic glutamatergic currents and decreasing synaptic connectivity, without reducing dendritic spines density. The effect of dysbiosis on neuronal functions are mediated by microglia-neuron cross-talk through the CX3CL1-CX3CR1 axis, as antibiotics treatment of CX3CR1 deficient mice, mod-ulates microglia density and processes rearrangement leaving unaltered synaptic function. To-gether, our findings show that the antibiotics alteration of gut microbiota impairs synaptic effi-cacy, probably through CX3CL1-CX3CR1 signaling supporting microglia as a major player in in the gut-brain axis, and in particular in the gut microbiota-to-neuron communication pathway.