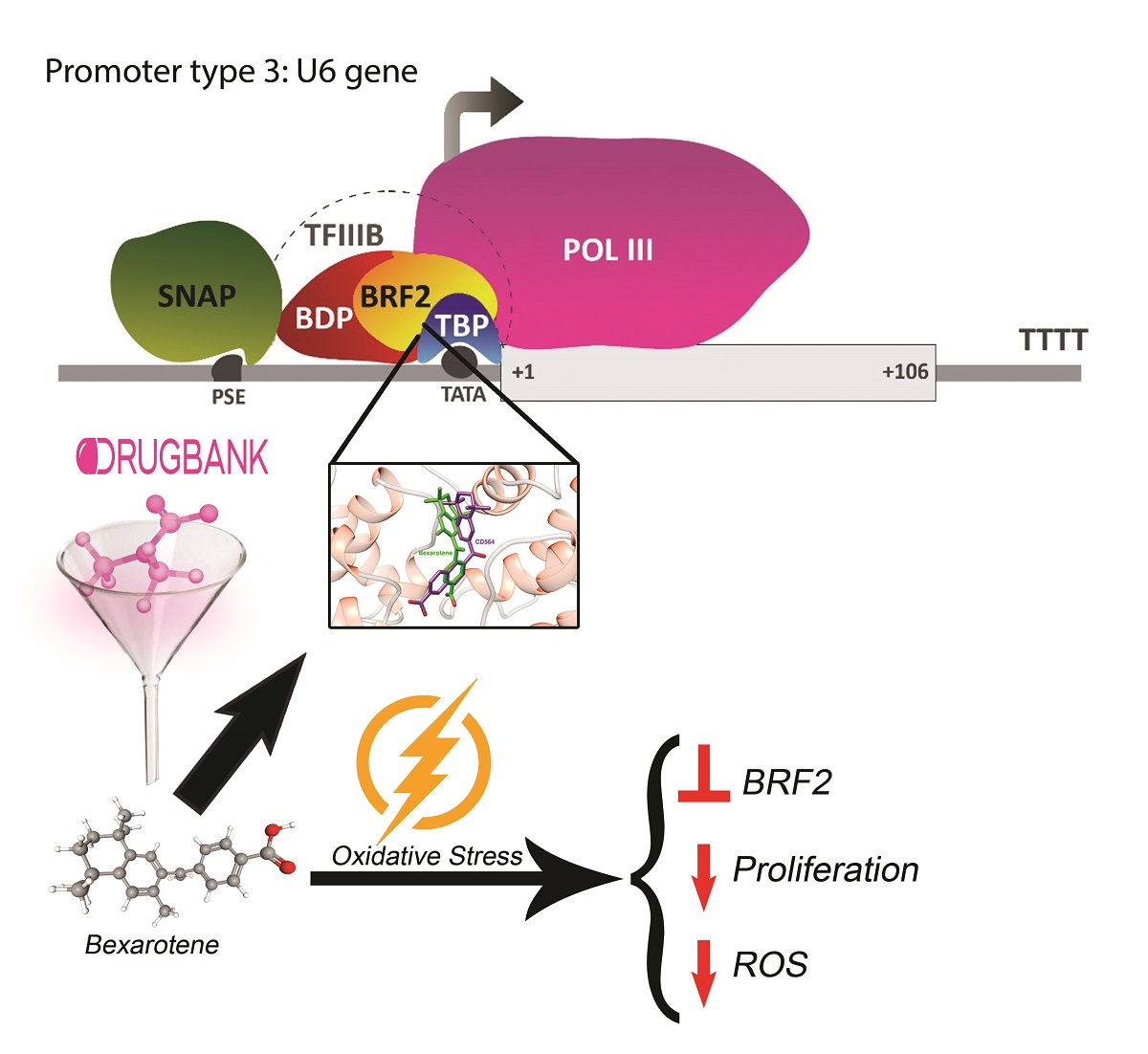
Overexpression of BRF2, a selective subunit of RNA polymerase III, has been shown to be crucial in the development of several types of cancers, including breast cancer and lung squamous cell carcinoma . Predominately, BRF2 acts as a central redox-sensing transcription factor (TF) and is involved in rescuing oxidative stress (OS) -induced apoptosis. Here, we showed a novel link between BRF2 and DNA damage response. Due to the lack of BRF2 specific inhibitors, through virtual and molecular dynamics screening, we identified potential drug candidates that interfere with BRF2-TATA-binding Protein (TBP)-DNA complex interactions based on binding energy, intermolecular, and torsional energy parameters. We experimentally tested Bexarotene as a potential BRF2 inhibitor. We found that Bexarotene (Bex) treatment resulted in a dramatic decline in oxidative stress (Tert-butylhydroquinone (tBHQ))-induced levels of BRF2 and consequently, lead to a decrease in cellular proliferation of cancer cells which may in part be due to drug pretreatment induced reduction of ROS generated by the oxidizing agent. Our data thus, provide the first experimental evidence that BRF2 is a novel player in DNA damage response pathway and Bexarotene can be used as a potential inhibitor to treat cancers with the specific elevation of oxidative stress.