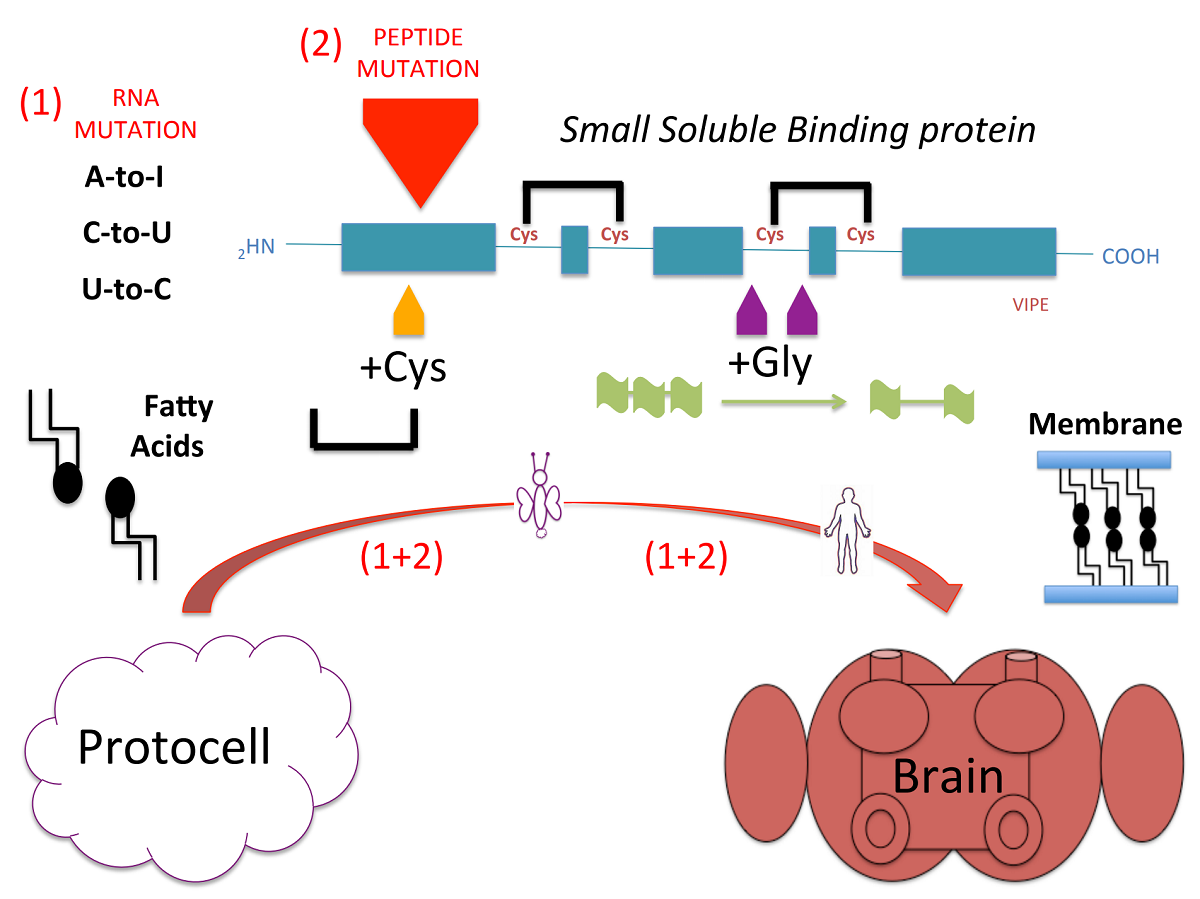
We remind about the dogma initially established with the nucleic acid double helix, i.e. the DNA structure as the primary source of life. However, we bring into the discussion those additional processes that were crucial to enable life and cell evolution. Studying chemosensory proteins (CSPs) and odor binding proteins (OBPs) of insects, we have found a high level of pinpoint mutations on the RNA and peptide sequences. Many of these mutations are found to be tissue-specific and induce subtle changes in the protein structure, leading to a new theory of cell multifunction and life evolution. Here, attention is given to RNA and peptide mutations in small soluble protein families known for carrying lipids and fatty acids as fuel for moth cells. A new phylogenetic analysis of mutations is presented and provides even more support to the pioneer work, i.e. the finding that mutations in binding proteins have spread through moths and various groups of insects. Then, focus is given to specific mechanisms of mutations that are not random, change α-helical profilings and bring new functions at the protein level. In conclusion, RNA and peptide mutations are not seen as representative of a multitude of diseases, but rather as an alternative way by which protocells developed to acquire multifunction and totipotency. This provides a basis for the theory of RNA/peptide mutations for birth and evolution of life on earth’s crust proposed here.