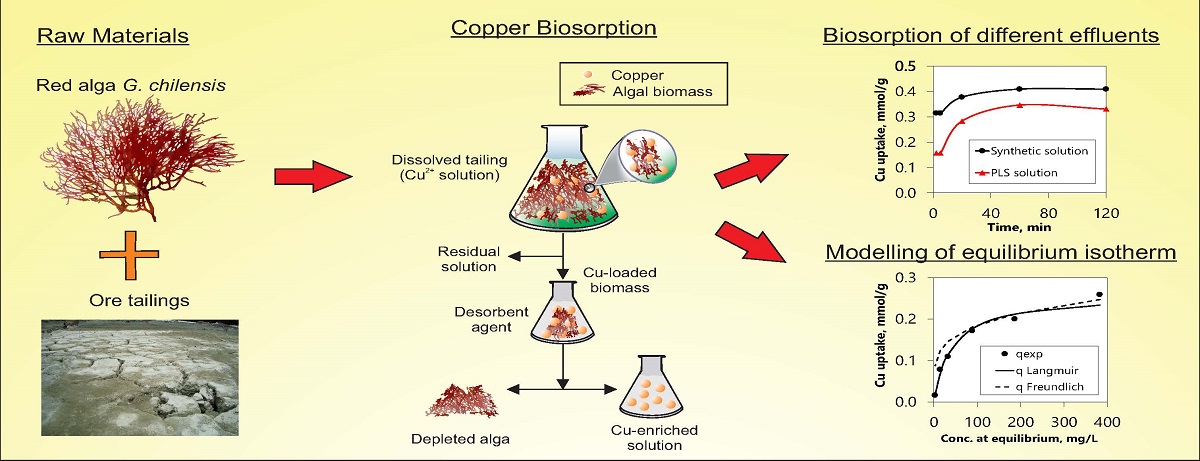
Mining is one of the main economic activities of several developing countries as Chile. Due to the progressive fall of the ore grades and the increasingly refractory composition of minerals, concentrating plants have increased that has led to an increase in the generation of tailings. Tailings, especially those obtained in the past, have remaining copper and other valuable species in quantities that can potentially be recovered, such as gold, silver, vanadium and rare earth elements. This transforms this abundant waste into a potential source of precious or strategic metals for secondary mining. One of the techniques of solid-liquid separation that allows the processing of solutions with low concentrations of metals corresponds to the adsorption, and more recently the biosorption, which is based on the use of biological matrices that do not constitute an environmental liability after application. Biosorption occurs as a consequence of the wide variety of active functional groups present in the different types of biomass. Bacterial, fungal, plant and algal biomasses have been described as biosorbents, mainly for the treatment of diluted and simple solutions. This work aims to recover copper from leached tailings using biomass of the red algae Gracilaria chilensis as a biosorbent. The tailing samples were taken from an abandoned deposit in the north of Chile and after an acid leaching copper was biosorbed, for which the kinetics of adsorption and the equilibrium isotherms were studied, applying the Freundlich and Langmuir models. Operational parameters such as adsorbent dose, pH and initial metal concentration were studied.