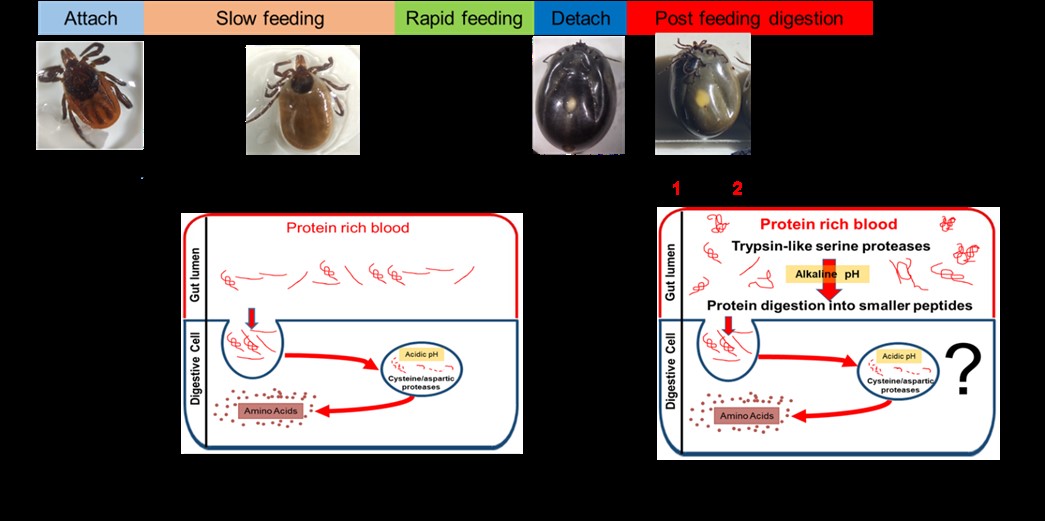
Ixodes scapularis is the major vector of Lyme disease in the eastern United States. Each active life stage (larva, nymph, and adult) takes a blood meal either for developing and molting to the next stage (larvae and nymphs) or for oviposition (adult females). This protein-rich blood meal is the only food taken by Ixodes ticks and therefore blood digestion is very important for tick survival. Most studies on blood digestion in ticks have shown that the initial stages of digestion are carried out by cathepsin proteases within acidic digestive cells. However, most of these studies have focused on partially engorged ticks. In other hematophagous arthropods, the serine proteases play an important role in blood protein degradation. In this study, we determined transcript expression of four I. scapularis serine proteases with previously characterized roles in blood digestion. RNA interference was used for functional analysis and a trypsin-benzoyl-D, L-arginine 4-nitoanilide assay was used to measure active trypsin levels. An in vitro hemoglobinolytic assay was performed with or without serine protease inhibitor. Our data suggest that trypsin levels increase significantly after blood feeding and peaked in larvae, nymphs, and adults at 3, 1, and 1 day post host detachment, respectively. The knockdown of three previously identified serine proteases by RNAi negatively impacted blood intake, survival, fecundity, levels of active trypsin in the gut and resulted in lower hemoglobin degradation in vitro. A trypsin inhibitor, PMSF, blocked the action of trypsin in the gut extract resulting in 65% lower hemoglobin degradation. We provide evidence of the serine proteases as digestive enzymes in fully engorged, replete females. Our data also demonstrated that in addition to blood digestion, these serine proteases might have a role in blood feeding success in I. scapularis.