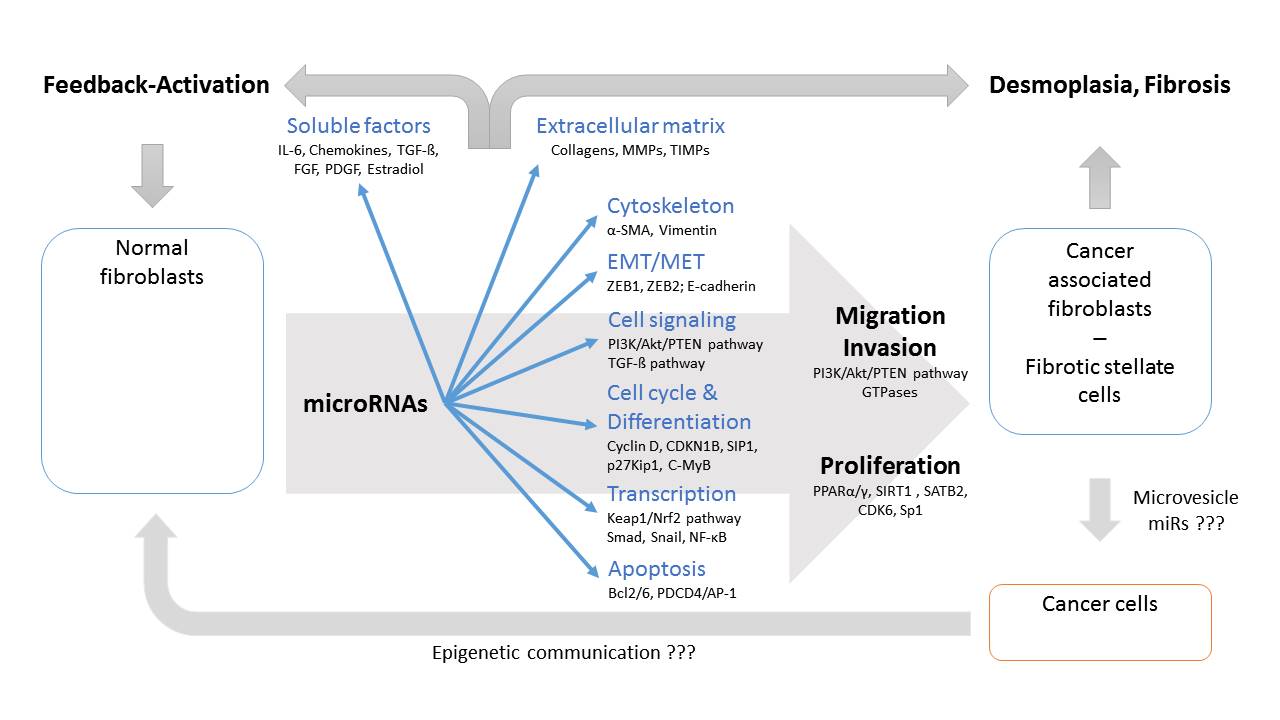
Tumor microenvironment including cancer-associated fibroblasts (CAF) has developed as an important target for understanding tumor progression, clinical prognosis and treatment responses of cancer. Cancer cells appear to transform normal fibroblasts (NF) into CAFs involving direct cell-cell communication and epigenetic regulations. This review summarizes the current understanding on miR involvement in cancer cell – tumor environment/stroma communication, transformation of NFs into CAFs, their involved targets and signaling pathways in these interactions; and clinical relevance of CAF-related miR expression profiles. There is evidence that miRs have very similar roles in activating hepatic (HSC) and pancreatic stellate cells (PSC) as part of precancerous fibrotic diseases. In summary, deregulated miRs affect various intracellular functional complexes, such as transcriptional factors, extracellular matrix, cytoskeleton, EMT/MET regulation, soluble factors, tyrosine kinase and G-protein signaling, apoptosis and cell cycle & differentiation, but also formation and composition of the extracellular microenvironment. These processes result in the clinical appearance of desmoplasia involving CAFs and fibrosis characterized by deregulated stellate cells. In addition, modulated release of soluble factors can act as (auto)activating feedback loop for transition of NFs into their pathological counterparts. Furthermore, epigenetic communication between CAFs and cancer cells may confer to cancer specific functional readouts and transition of NF into their pathological counterparts. MiR related epigenetic regulation with many similarities should be considered as key factor in development of cancer and fibrosis specific environment.