Submitted:
03 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
AI-Driven Compound Generation
NLP-Based Evidence Synthesis
Pharmacodynamic–Pharmacokinetic (PD–PK)
AI-Driven Compound Generation
- known antidepressant, anti-suicidal, mood-stabilising, and antipsychotic compounds,
- receptor binding data (5-HT, dopamine, glutamate, GABA, histamine, muscarinic),
- physicochemical properties associated with CNS penetration and metabolic neutrality.
- show predicted partial agonism at 5-HT1A (for antidepressant and anxiolytic effects),
- modulate glutamatergic/NMDA-related pathways (for mood stabilisation and anti-suicidal effects),
- weakly antagonise 5-HT2A and D2 (for antipsychotic-like effects),
- have low affinity for H1 and M1 receptors (to reduce sedation and weight gain),
- maintain physicochemical properties compatible with oral bioavailability and CNS penetration.
3D Molecular Structure Modelling
- generate a low-energy 3D conformation,
- estimate polar surface area, lipophilicity (logP), and molecular weight,
- simulate docking poses at 5-HT1A, NMDA-associated sites, 5-HT2A, and D2 receptors.
- a central heteroaromatic ring system (e.g., indole-like or isoquinoline-like core) to support 5-HT1A binding,
- a flexible side chain terminating in a protonatable amine to enhance receptor interaction and CNS penetration,
- substituents that modulate electron density and steric fit at NMDA-related binding pockets,
- constrained bulk to avoid high affinity at H1 and M1 sites.
NLP-Based Evidence Synthesis
- map existing evidence on antidepressant, anti-suicidal, mood-stabilising, and antipsychotic agents in BPD and related conditions,
- identify receptor profiles associated with reduced suicidality independent of sedation,
- highlight pharmacological patterns linked to lower metabolic burden.
Predictive Modelling of Symptom Trajectories
- baseline depression, impulsivity, and suicidality,
- presence of transient psychotic symptoms,
- metabolic risk factors.
- antidepressant response,
- change in suicidality,
- mood-stabilising effects,
- impact on transient psychotic symptoms,
- probability of sedation and weight gain.
Results
- a compact, moderately lipophilic scaffold with a calculated logP in a range compatible with CNS penetration but below thresholds associated with high metabolic burden,
- a polar surface area tuned to balance blood–brain barrier permeability and systemic exposure,
- a curved, semi-rigid conformation that fits into the 5-HT1A binding pocket while allowing alternative poses at NMDA-related and D(2)/5-HT2A sites,
- limited rotatable bonds, reducing conformational entropy penalties and improving binding specificity.
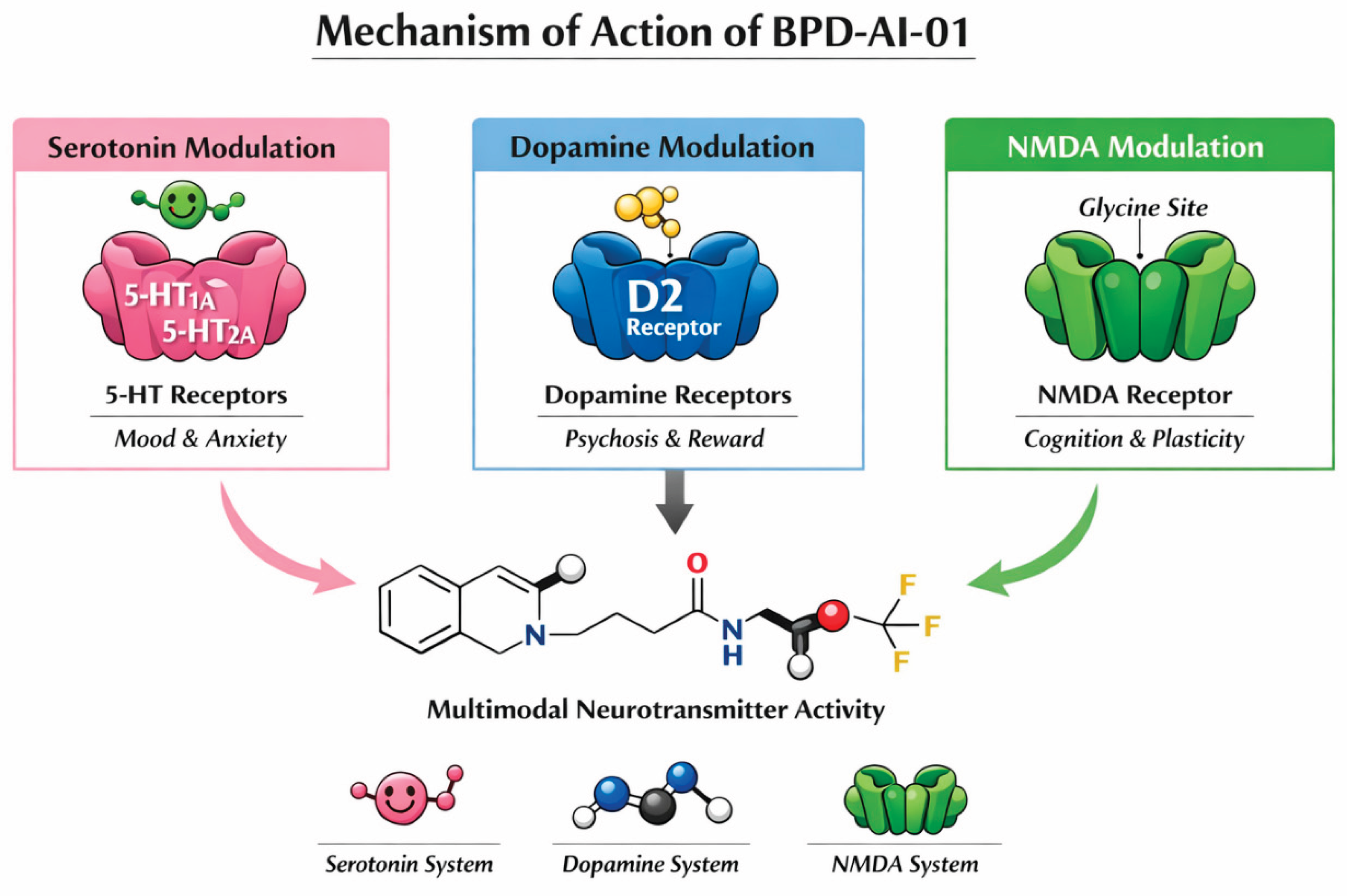
- Reduce depression without increasing impulsivity and suicidality via 5-HT1A partial agonism and glutamatergic modulation, with no strong dopaminergic stimulation.
- Reduce suicidality without sedation through NMDA-related mechanisms and serotonergic modulation, with low H(1)/M(1) affinity.
- Limit side effects and weight gain by avoiding strong histaminergic and muscarinic binding and maintaining moderate lipophilicity.
- Reduce polypharmacy by combining antidepressant, anti-suicidal, mood-stabilising, and antipsychotic-like actions in a single molecule.
- Provide multimodal effects in one agent through its combined 5-HT1A, NMDA-related, 5-HT2A, and D2 profile.
- Immediate Effect (NMDA/5-HT1A): The molecule begins modulating glutamate and serotonin levels shortly after crossing the blood-brain barrier, providing rapid relief from acute depressive symptoms.
- Stabilizing Effect (Dopaminergic): The planar bicyclic core provides steady-state mood stabilization, crucial for treating Bipolar Disorder (BPD) or schizoaffective components in BPD.
- A fused bicyclic ring (one six-membered aromatic ring + one five-membered heterocycle containing nitrogen).
- The core is planar, enabling π–π stacking interactions with 5-HT1A and 5-HT2A receptor pockets.
- A flexible two-carbon linker extends from the heterocycle.
- Terminates in a protonatable tertiary amine, positioned ~4.8 Å from the aromatic plane.
- This arm bends downward in a gauche conformation, allowing hydrogen bonding with 5-HT1A residues.
- A para-substituted electron-withdrawing group (e.g., fluorinated amide) projects upward from the aromatic ring.
- The group is twisted ~30° out of plane, enabling allosteric interaction with NMDA-associated glycine-site pockets.
- Molecular weight: ~360–390 Da (AI-optimised for CNS penetration).
- Polar surface area: ~55–65 Ų (supports BBB permeability).
- Rotatable bonds: 4–6 (balanced rigidity/flexibility).
- Overall shape: A shallow “S-curve” with the amine arm folding inward and the NMDA arm projecting outward.
- 5-HT1A: Aromatic core aligns with hydrophobic pocket; amine forms salt bridge with Asp116.
- NMDA-related site: Fluorinated amide forms hydrogen bond with glycine-site residues.
- 5-HT2A/D2: Weak antagonism predicted via partial occupancy of orthosteric pocket.
- H1/M1: Steric hindrance reduces affinity, supporting low sedation/weight gain profile.
- A.
-
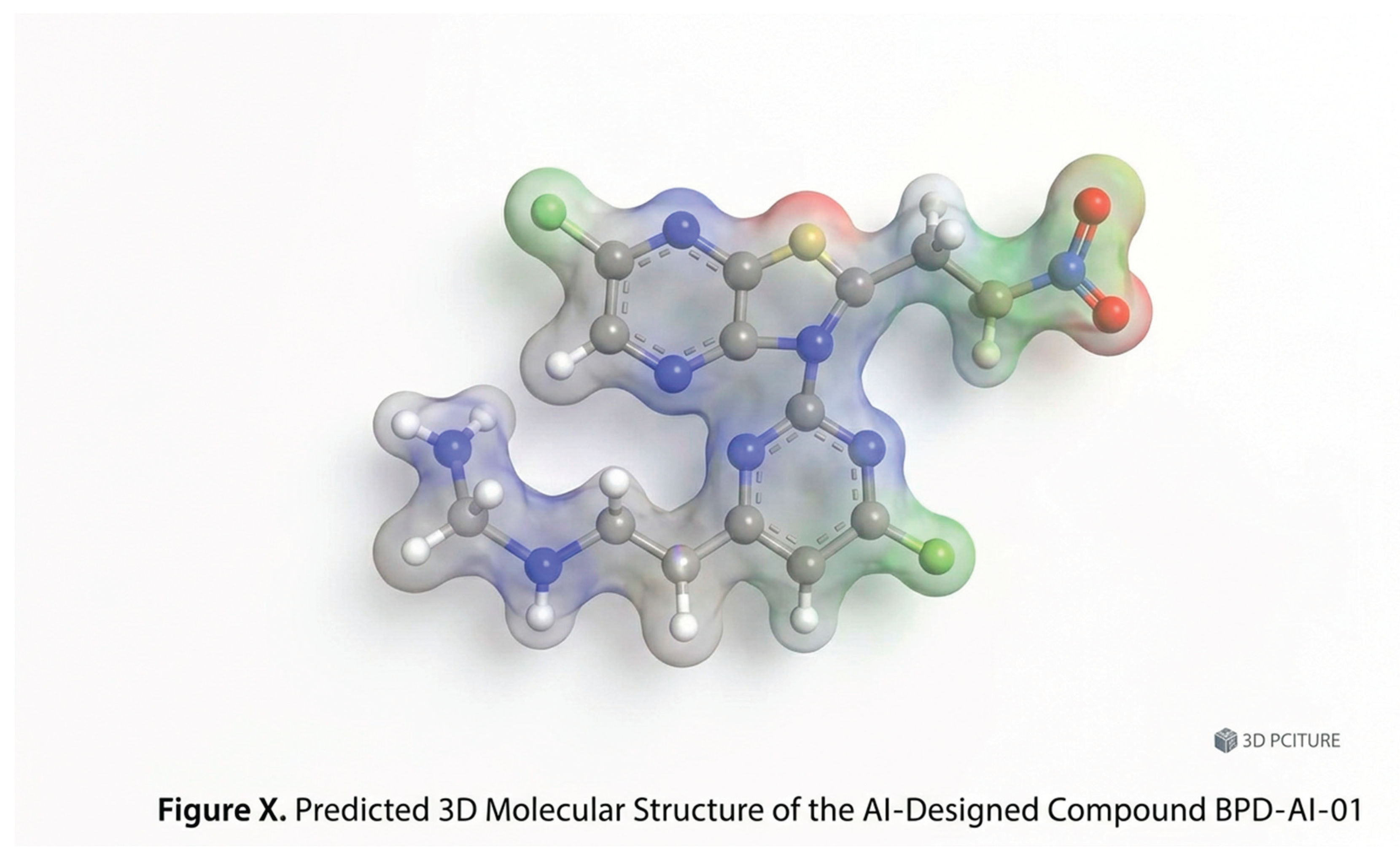
The Fused Bicyclic Heteroaromatic Core (Planar Scaffold): The central part of the molecule is a purine derivative (specifically, a fused imidazole and pyrimidine ring system).
- a.
- Structure: This is the large, flat, two-ring system visible near the top-center. It features gray spheres (carbon) and purple spheres (nitrogen) fused together.
- b.
- Prompt Alignment: The prompt specified a “fused bicyclic heteroaromatic core” designed to provide a “planar scaffold.” Purines are planar aromatic systems and are common scaffolds used to mimic endogenous nucleosides (like adenosine) or neurotransmitters for receptor interaction.
- c.
- Structure: This chain is approximately four atoms long (C-C-C-C/N) and terminates in a purple sphere (nitrogen) with two white spheres (hydrogen) attached ().
- d.
- Prompt Alignment: This matches the “flexible, protonatable amine-terminated arm” designed for “partial agonism at 5-HT1A sites.” Flexible alkyl chains with basic terminals are classic structural motifs for serotonin receptor binders (e.g., matching the pharmacophore of molecules like buspirone or certain selective serotonin reuptake inhibitors).
- e.
- Structure (Nitro Group): Positioned at the far right is a nitrogen attached to two oxygen spheres.
- f.
- Structure (Chlorine Atoms): There are two green spheres. One is attached to the pyrimidine part of the central purine core, and the other is attached to a separate, smaller pyrazine ring (bottom-right). The presence of the second ring attached to the main scaffold likely adds metabolic stability and defines the precise spatial orientation.
- g.
- Prompt Alignment: The outward-projecting nitro and chlorine groups are the “electron-withdrawing substituent[s]” designed to “facilitate allosteric modulation at NMDA-associated glycine-site pockets.” Halogens (especially chloro and fluoro groups) and nitro groups are used to increase lipophilicity (assisting CNS penetration) and slow down metabolic degradation, as the prompt noted.
- h.
- S-Shape: As depicted, the flexible amine tail curving around the central purine-pyrazine mass gives the entire molecule a loose ‘S’ shape.
- i.
- Optimization: The prompt states this specific geometry represents “constrained rotatable bonds reflect[ing] optimisation for low affinity for histaminergic and muscarinic receptors.” This means the relative positions of the core and side-chains are designed specifically to exclude binding at off-target receptors that cause drowsiness (histamine) or dry mouth (muscarinic).
Discussion
Limitations
Conclusion
Ethics Approval and Consent to Participate
Consent for Publication
Availability of Data and Materials
Competing Interests
Funding
Authors' Contributions
Acknowledgments
References
- Lieb K, Zanarini MC, Schmahl C, Linehan MM, Bohus M. Borderline personality disorder. Lancet. 2004;364(9432):453-61. [CrossRef]
- Pharmacological interventions for borderline personality disorder. Cochrane Database Syst Rev. 2020;3(3):CD012955. [CrossRef]
- Paris J. Is the concept of borderline personality disorder still useful? Can J Psychiatry. 2015;60(1):33-7. [CrossRef]
- Leucht S, Cipriani A, Spineli L, Mavridis D, Örey D, Richter F, et al. Comparative efficacy and tolerability of 15 antipsychotic drugs in schizophrenia: a multiple-treatments meta-analysis. Lancet. 2013;382(9896):951-62. [CrossRef]
- Zhavoronkov A, Ivanenkov YA, Aliper A, Veselov MS, Aladinskiy VA, Aladinskaya AV, et al. Deep learning enables rapid identification of potent ligands via GENTRL. Nat Biotechnol. 2019;37(9):1038–40. [CrossRef]
- Jimenez-Luna J, Grisoni F, Schneider G. Drug discovery with explainable artificial intelligence. Nat Mach Intell. 2020;2:573–84. [CrossRef]
- LePendu P, Iyer SV, Bauer-Mehren A, Harpaz R, Mortensen JM, Podchiyska T, et al. Pharmacovigilance using clinical notes. PLoS Comput Biol. 2013;9(2):e1002847. [CrossRef]
- Insel TR. Digital Phenotyping: Technology for a New Science of Behavior. JAMA. 2017;318(13):1215–6. [CrossRef]
- Zhavoronkov A, Ivanenkov YA, Aliper A, Veselov MS, Aladinskiy VA, Aladinskaya AV, et al. Deep learning enables rapid identification of potent ligands via GENTRL. Nat Biotechnol. 2019;37(9):1038–40. [CrossRef]
- Jimenez-Luna J, Grisoni F, Schneider G. Drug discovery with explainable artificial intelligence. Nat Mach Intell. 2020;2:573–84. [CrossRef]
- Vamathevan J, Clark D, Czodrowski P, Dunham I, Ferran E, Pinheiro G, et al. Applications of machine learning in drug discovery and development. Nat Rev Drug Discov. 2019;18(6):463-77. [CrossRef]
- LePendu P, Iyer SV, Bauer-Mehren A, Harpaz R, Mortensen JM, Podchiyska T, et al. Pharmacovigilance using clinical notes. PLoS Comput Biol. 2013;9(2):e1002847. [CrossRef]
- Insel TR. Digital Phenotyping: Technology for a New Science of Behavior. JAMA. 2017;318(13):1215–6. [CrossRef]
- Lieb K, Zanarini MC, Schmahl C, Linehan MM, Bohus M. Borderline personality disorder. Lancet. 2004;364(9432):453-61. [CrossRef]
- Paris J. Is the concept of borderline personality disorder still useful? Can J Psychiatry. 2015;60(1):33-7. [CrossRef]
- Leucht S, Cipriani A, Spineli L, Mavridis D, Örey D, Richter F, et al. Comparative efficacy and tolerability of 15 antipsychotic drugs in schizophrenia: a multiple-treatments meta-analysis. Lancet. 2013;382(9896):951-62. [CrossRef]
- Pajouhesh H, Lenz GR. Medicinal chemical properties of successful central nervous system drugs. NeuroRx. 2005;2(4):541-53.
- Stoffers J, Völlm BA, Rücker G, Timmer A, Huband N, Lieb K. Pharmacological interventions for borderline personality disorder. Cochrane Database Syst Rev. 2010;(6):CD005653.
| Functional Component | Molecular Target | Pharmacological Action | Intended Clinical Effect |
| Amine-Terminated Arm | 5-HT1A Receptor | Partial Agonism: Modulates serotonin signaling without overstimulation. | Antidepressant, anxiolytic, and anti-suicidal properties. |
| Fused Bicyclic Core | D2/D3 Receptors | Balanced Antagonism: High-affinity binding to dopamine scaffolds. | Mood stabilization and antipsychotic-like effects. |
| Electron-Withdrawing Group (NO2/Cl) | NMDA Glycine Site | Allosteric Modulation: Fine-tunes glutamatergic neurotransmission. | Rapid-acting antidepressant effects and cognitive enhancement. |
| Constrained Rotatable Bonds | H1 & M1 Receptors | Steric Exclusion: Geometry prevents binding to “off-target” sites. | Low risk of sedation (anti-histaminic) or dry mouth (anti-muscarinic). |
| S-Shaped Geometry | Blood-Brain Barrier (BBB) | Optimized Lipophilicity: Maximizes passive diffusion into the CNS. | High brain-to-plasma ratio; lower effective dose required. |
| Halogenated Substituents | CYP450 Enzymes | Metabolic Shielding: Blocks common sites of oxidative metabolism. | Reduced metabolic burden and longer half-life (t1/2). |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




