Submitted:
30 April 2026
Posted:
30 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
Study Design and Ethics
Subjects
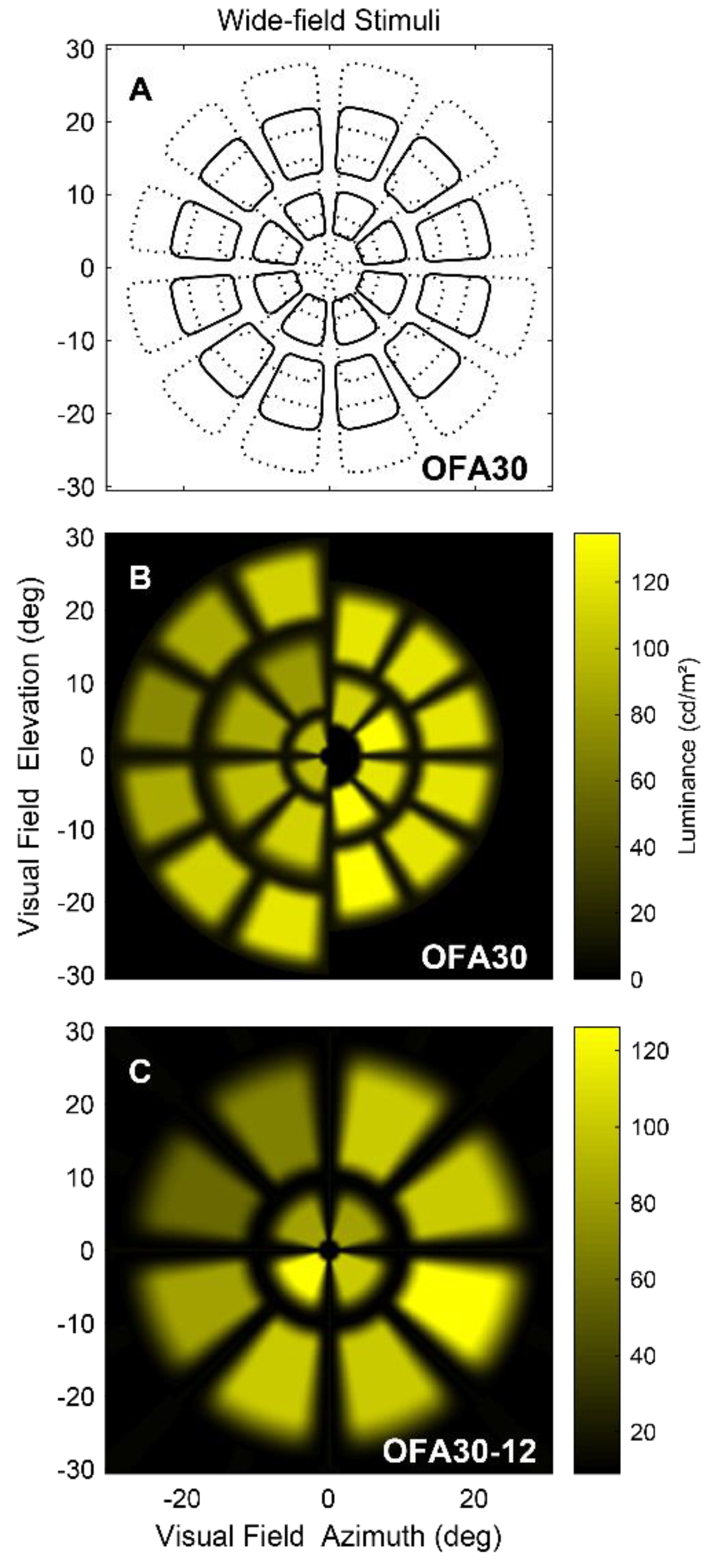
OFA Tests and Stimuli
Ophthalmic Examinations
Analysis
Results
Demographic Characteristics
Diagnostic Power of OFA Tests
Discussion
Conclusions
Author Contributions
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhong, H.; Feng, Y.; Shen, J.; Rao, T.; Dai, H.; Zhong, W.; Zhao, G. Global Burden of Traumatic Brain Injury in 204 Countries and Territories From 1990 to 2021. Am. J. Prev. Med. 2025, 68((4)), 754–763. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Wang, C.; Sun, B. Global, regional, and national burdens of traumatic brain injury from 1990 to 2021. Front Public Health 2025, 13, 1556147. [Google Scholar] [CrossRef]
- Stocchetti, N.; Zanier, E. R. Chronic impact of traumatic brain injury on outcome and quality of life: a narrative review. Crit. Care 2016, 20((1)), 148. [Google Scholar] [CrossRef]
- Maas, A. I. R.; Menon, D. K.; Manley, G. T.; Abrams, M.; Akerlund, C.; Andelic, N. Investigators, Traumatic brain injury: progress and challenges in prevention, clinical care, and research. Lancet Neurol. 2022, 21((11)), 1004–1060. [Google Scholar] [CrossRef]
- Curtin University, Everyday Australians help deliver better treatment for concussions; Curtin University: Perth, Western Australia, 23 September 2022.
- Yoo, R. E.; Choi, S. H.; Youn, S. W.; Hwang, M.; Kim, E.; Oh, B. M.; Sohn, C. H. Myelin Content in Mild Traumatic Brain Injury Patients with Post-Concussion Syndrome: Quantitative Assessment with a Multidynamic Multiecho Sequence. Korean J. Radiol. 2022, 23((2)), 226–236. [Google Scholar] [CrossRef]
- Giza, C. C.; Kutcher, J. S. An introduction to sports concussions. Continuum (Minneap Minn) 2014, 20((6 Sports Neurology)), 1545–51. [Google Scholar] [PubMed]
- Giza, C.; Greco, T.; Prins, M. L. Chapter 6 - Concussion: pathophysiology and clinical translation. In Handbook of Clinical Neurology; Hainline, B., Stern, R. A., Eds.; Elsevier, 2018; Vol. 158, pp. 51–61. [Google Scholar]
- Lee, B.; Newberg, A. Neuroimaging in traumatic brain imaging. NeuroRx 2005, 2((2)), 372–83. [Google Scholar] [CrossRef]
- Machamer, J.; Temkin, N. R.; Manley, G. T.; Dikmen, S. Functional Status Examination in Patients with Moderate-to-Severe Traumatic Brain Injuries. J. Neurotrauma 2018, 35((10)), 1132–1137. [Google Scholar] [CrossRef]
- Walsh, D. V.; Capo-Aponte, J. E.; Jorgensen-Wagers, K.; Temme, L. A.; Goodrich, G.; Sosa, J.; Riggs, D. W. Visual field dysfunctions in warfighters during different stages following blast and nonblast mTBI. Mil. Med. 2015, 180((2)), 178–85. [Google Scholar] [CrossRef]
- Sabeti, F.; Carle, C. F.; Jaros, R. K.; Rohan, E. M. F.; Waddington, G.; Lueck, C. J. Maddess, T., Objective Perimetry in Sporting-Related Mild Traumatic Brain Injury. Ophthalmology 2019, 126((7)), 1053–1055. [Google Scholar] [PubMed]
- Maddess, T.; Carle, C. F.; Kolic, M.; Essex, R. W.; Sarac, O.; Rohan, E. M. F.; van Kleef, J. P. Diagnostic power and reproducibility of objective perimetry in glaucoma. J. Glaucoma 2024, 32((12)), 940–950. [Google Scholar] [CrossRef]
- Sabeti, F.; van Kleef, J. P.; Iyer, R. M.; Carle, C. F.; Nolan, C. J.; Chia, R. H.; Maddess, T. Discriminating early-stage diabetic retinopathy with subjective and objective perimetry. Front. Endo 2024, 14((1333826)), 1–12. [Google Scholar] [CrossRef]
- Carle, C. F.; James, A. C.; Kolic, M.; Essex, R. W.; Maddess, T. Blue multifocal pupillographic objective perimetry in glaucoma. Invest Ophthalmol. Vis. Sci. 2015, 56, 6394–6403. [Google Scholar] [CrossRef] [PubMed]
- Sabeti, F.; James, A. C.; Carle, C. F.; Essex, R. W.; Bell, A.; Maddess, T. Comparing multifocal pupillographic objective perimetry (mfPOP) and multifocal visual evoked potentials (mfVEP) in retinal diseases. Sci. Rep. 2017, 7, 45847. [Google Scholar] [CrossRef]
- Rosli, Y.; Carle, C. F.; Ho, Y.; James, A. C.; Kolic, M.; Rohan, E. M. F.; Maddess, T. Retinotopic effects of visual attention revealed by dichoptic multifocal pupillography. Sci. Rep. 2018, 8, 2991. [Google Scholar] [CrossRef]
- Maddess, T.; van Kleef, J. P.; Rohan, E. M. F.; Carle, C. F.; Baird-Gunning, J.; Rai, B. B. Lueck, C. J., Rapid, non-contact multifocal visual assessment in multiple sclerosis. Neurol. Sci. 2023, 44((1)), 273–279. [Google Scholar] [CrossRef]
- Maddess, T.; Carle, C. F.; Rohan, E. M. F.; Baird-Gunning, J.; van Kleef, J. P.; Lueck, C. J. Objective perimetry and progression of multiple sclerosis. eNeurol Sci. 2022, 100430, 1–6. [Google Scholar] [CrossRef]
- Ali, E. N.; Carle, C. F.; Lueck, C. J.; Kolic, M.; Maddess, T. Assessing migraine patients with multifocal pupillographic objective perimetry. BMC Neurol. 2021, 21((211)), 1–12. [Google Scholar] [CrossRef]
- Bell, A.; James, A. C.; Kolic, M.; Essex, R. W.; Maddess, T. Dichoptic multifocal pupillography reveals afferent visual field defects in early Type 2 Diabetes. Invest Ophthalmol. Vis. Sci. 2010, 51, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Makdissi, M.; Davis, G.; McCrory, P. Clinical challenges in the diagnosis and assessment of sports-related concussion. Neurol. Clin. Pract. 2015, 5((1)), 2–5. [Google Scholar] [CrossRef] [PubMed]
- McCrory, P.; Meeuwisse, W.; Aubry, M.; Cantu, B.; Dvorak, J.; Echemendia, R. J. Tator, C. H., Consensus statement on concussion in sport--the 4th International Conference on Concussion in Sport held in Zurich, November 2012. Clin. J. Sport Med. 2013, 23((2)), 89–117. [Google Scholar] [CrossRef] [PubMed]
- Silverberg, N. D.; Iaccarino, M. A.; Panenka, W. J.; Iverson, G. L.; McCulloch, K. L.; Dams-O'Connor, K. American Congress of Rehabilitation Medicine Brain Injury Interdisciplinary Special Interest Group Mild, T. B. I. T. F., Management of Concussion and Mild Traumatic Brain Injury: A Synthesis of Practice Guidelines. Arch. Phys. Med. Rehabil. 2020, 101((2)), 382–393. [Google Scholar] [CrossRef]
- McRae, B.; Stay, S. Assessment and management of sport-related concussion in general practice. Aust. J. Gen. Pract. 2024, 53((3)), 121–126. [Google Scholar] [CrossRef]
- Sandel, N.; Reynolds, E.; Cohen, P. E.; Gillie, B. L.; Kontos, A. P. Anxiety and Mood Clinical Profile following Sport-related Concussion: From Risk Factors to Treatment. Sport Exerc Perform. Psychol. 2017, 6((3)), 304–323. [Google Scholar] [CrossRef]
- Signoretti, S.; Lazzarino, G.; Tavazzi, B.; Vagnozzi, R. The pathophysiology of concussion. PM R. 2011, 3((10) Suppl 2, S359–68. [Google Scholar] [CrossRef]
- Gamlin, P. D. The pretectum: connections and oculomotor-related roles. Prog. Brain Res. 2006, 151, 379–405. [Google Scholar] [PubMed]
- Peinkhofer, C.; Knudsen, G. M.; Moretti, R.; Kondziella, D. Cortical modulation of pupillary function: systematic review. Peerj 2019, 7. [Google Scholar]
- Marshall, S. W.; Spencer, R. J. Concussion in Rugby: The Hidden Epidemic. J. Athl. Train. 2001, 36((3)), 334–338. [Google Scholar] [PubMed]
- Wittmer, R.; Buckley, T. A.; Swanik, C. B.; Costantini, K. M.; Ryan, L.; Daly, E. Hunzinger, K. J., Unrecognized and Unreported Concussions Among Community Rugby Players. Sports 2025, 13((8)). [Google Scholar]
- Hedges, L. V. Distribution Theory for Glass's Estimator of Effect Size and Related Estimators. J. Educ. Stat. 1981, 6((2)), 107–128. [Google Scholar] [CrossRef]
- Rai, B. B.; van Kleef, J. P.; Sabeti, F.; Vlieger, R.; Suominen, H.; Maddess, T. Early diabetic eye damage: Comparing detection methods using diagnostic power. Surv. Ophthalmol. 2024, 69, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Rai, B. B.; Maddess, T.; Carle, C. F.; Rohan, E. M. F.; van Kleef, J. P.; Barry, R. C.; Sabeti, F. Comparing objective perimetry, matrix perimetry, and regional retinal thickness in mild diabetic macular edema. Transl. Vis. Sci. Technol. 2021, 10((13)), 32. [Google Scholar] [CrossRef]
- Rai, B. B.; Essex, R. W.; Sabeti, F.; Maddess, T.; Rohan, E. M. F.; van Kleef, J. P.; Carle, C. F. An objective perimetry study of central versus. peripheral sensitivities and delays in age-related macular degeneration. Trans. Vis. Sci. Tech. 2021, 10((14(24))), 1–14. [Google Scholar] [CrossRef] [PubMed]
- Rai, B. B.; Sabeti, F.; Carle, C. F.; Maddess, T. Visual Field Tests: A Narrative Review of Different Perimetric Methods. J. Clin. Med. 2024, 13, 9. [Google Scholar] [CrossRef] [PubMed]
- Rai, B. B.; Sabeti, F.; Carle, C. F.; Rohan, E. M. F.; van Kleef, J. P.; Essex, R. W. Maddess, T., Rapid objective testing of visual function matched to the ETDRS-grid, and its diagnostic power in AMD. Ophthalmol. Sci. 2022, 2((2)), 1–9. [Google Scholar] [CrossRef]
- Bixenmann, B.; Bigsby, K.; Hasselfeld, K.; Khoury, J.; Mangine, R.; Pyne-Geithman, G.; Clark, J. Retinal and balance changes based on concussion history: a study of division 1 football players. Int. J. Phys. Med. Rehabil. 2014, 2. [Google Scholar]
- Kumar Das, N.; Das, M. Structural changes in retina (Retinal nerve fiber layer) following mild traumatic brain injury and its association with development of visual field defects. Clin. Neurol. Neurosurg. 2022, 212, 107080. [Google Scholar] [CrossRef]
- Kelman, J. C.; Hodge, C.; Stanwell, P.; Mustafic, N.; Fraser, C. L. Retinal nerve fibre changes in sports-related repetitive traumatic brain injury. Clin. Exp. Ophthalmol. 2020, 48((2)), 204–211. [Google Scholar] [CrossRef]
| Effect-size | Hedge’s g |
| Small | 0.2 |
| Medium | 0.5 |
| Large | 0.8 |
| Very large | 1.2 |
| Huge | 2.0 |
| Athlete Controls |
AUROC+ (N=6) |
AUROC+ (N=9) |
Hedge’s g (N=6) |
Hedge’s g N=9 |
| OFA30-12 - ACUTE | 73.5 ± 6.14 | 74.0 ± 6.17 | 0.82 | 0.79 |
| OFA30-12 - CHRONIC | 65.1 ± 6.98 | 63.2 ± 7.06 | 0.32 | 0.27 |
| OFA30- ACUTE | 48.9 ± 4.11 | 49.4 ± 4.17 | 0.15 | 0.14 |
| OFA30 - CHRONIC | 57.4 ± 7.11 | 57.5 ± 7.17 | 0.22 | 0.21 |
| Non- Athlete Controls | ||||
| OFA30-12 - ACUTE | 79.4 ± 4.11 | 77.9 ± 4.47 | 1.22 | 1.11 |
| OFA30-12 - CHRONIC | 82.6 ± 5.19 | 82.9 ± 4.99 | 1. 45 | 1.44 |
| OFA30- ACUTE | 76.0 ± 5.53 | 75.6 ± 5.59 | 0.93 | 0.94 |
| OFA30 - CHRONIC | 78.1 ± 5.28 | 78.4 ± 5.25 | 1.00 | 1.00 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




