Submitted:
28 April 2026
Posted:
29 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Study Design
2.3. Clinical Assessment Tools
2.3.1. Symptom Score
2.3.2. Ophthalmological Score
2.3.3. Schirmer Test
2.3.4. Tear Break Up Time (TBUT)
2.3.5. Quality of Life Questionnaire on Rhinoconjunctivitis (RQLQ)
2.4. Treatment
2.4.1. Conventional Treatment
2.4.2. Treatment of the Study
2.5. Evaluation of CD19+ IL-10+ Cells
2.6. Statistical Analysis
2.7. Ethics Statement
3. Results
3.1. Pre-Treatment Evaluation
3.2. Symptom Evaluation
3.3. Ophthalmological Evaluation
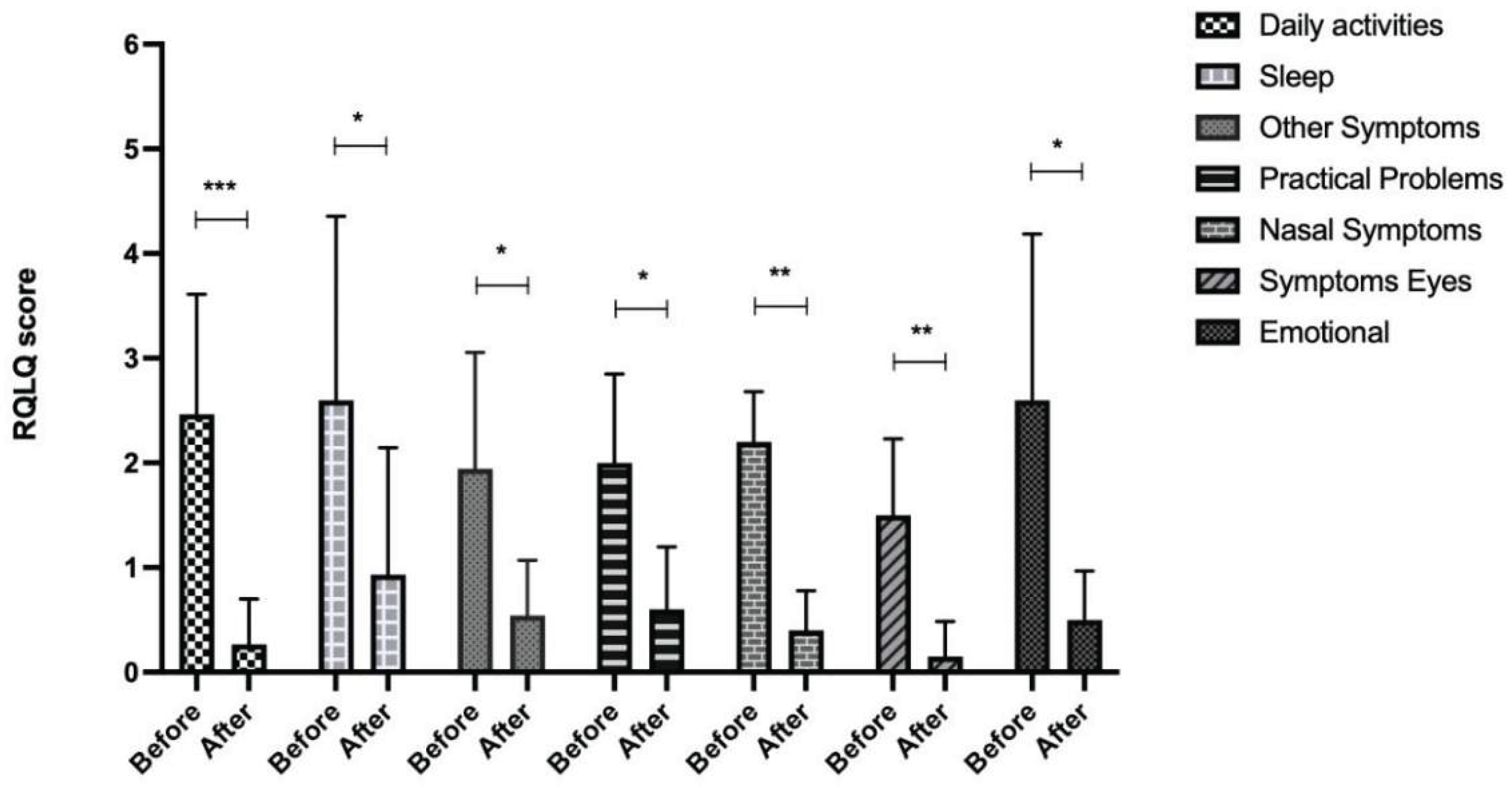
3.5. Quality of Life of Patients with AC Before and After Treatment with BS
3.6. Adverse Reactions Caused by BS
3.7. Changes in the Percentage of CD19+ IL-10+ Cells in Peripheral Blood
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BS | Bacterial Suspension |
| AC | Allergic conjunctivitis |
| IL | Interleukin |
| IQR | Interquartile Rank |
| RQLQ | Quality of Life Questionnaire on Rhinoconjunctivitis. |
| CD | Cluster differentiation. |
| MID | Minimum Importance Difference. |
| DECA | Consensus Document on Allergic Conjunctivitis. |
| TBUT | Tear Break Up Time. |
References
- Tariq, F. Allergic conjunctivitis: Review of current types, treatments, and trends. Life 2024, 14, 650. [CrossRef]
- Rosario, N.; Bielory, L. Epidemiology of allergic conjunctivitis. Curr. Opin. Allergy Clin. Immunol. 2011, 11, 471–476. [CrossRef]
- Gomes, P.J. Trends in prevalence and treatment of ocular allergy. Curr. Opin. Allergy Clin. Immunol. 2014, 14, 451–456. [CrossRef]
- La Rosa, M.; Lionetti, E.; Reibaldi, M.; Russo, A.; Longo, A.; Leonardi, S. Allergic conjunctivitis: A comprehensive review of the literature. Ital. J. Pediatr. 2013, 39, 18. [CrossRef]
- Leonardi, A.; Bogacka, E.; Fauquert, J.L.; Kowalski, M.L.; Groblewska, A.; Jedrzejczak-Czechowicz, M. Ocular allergy: Recognizing and diagnosing hypersensitivity disorders of the ocular surface. Allergy 2012, 67, 1327–1337. [CrossRef]
- Bielory, L.; Delgado, L.; Katelaris, C.; et al. ICON: Diagnosis and management of allergic conjunctivitis. Ann. Allergy Asthma Immunol. 2019, 124, 118–134. [CrossRef]
- Juniper, E.F.; Thompson, A.K.; Ferrie, P.J.; Roberts, J.N. Development and validation of the mini Rhinoconjunctivitis Quality of Life Questionnaire. Clin. Exp. Allergy 2000, 30, 132–140. [CrossRef]
- Anish, D.; Biswas, R.; R.H., S.R.; Komarla, N.P.; K., P.; Ruhana, S.; S., S.; L., A. A study on therapeutic management of allergic conjunctivitis and assessment of quality of life in patients. Int. J. Basic Clin. Pharmacol. 2025, 14, 797–802. [CrossRef]
- Dhami, S.; Nurmatov, U.; Arasi, S.; Khan, T.; Asaria, M.; Zaman, H.; Agarwal, A.; Netuveli, G.; Roberts, G.; Pfaar, O.; Muraro, A.; Ansotegui, I.J.; Calderon, M.; Cingi, C.; Durham, S.; van Wijk, R.G.; Halken, S.; Hamelmann, E.; Hellings, P.; Jacobsen, L.; Sheikh, A. Allergen immunotherapy for allergic rhinoconjunctivitis: A systematic review and meta-analysis. Allergy 2017, 72, 1597–1631. [CrossRef]
- Velázquez-Soto, H.; Jiménez Martínez, M. Fundamentals of Molecular Allergy: From Bench to Bedside. In Allergic Disease - New Developments in Diagnosis and Therapy; IntechOpen: London, UK, 2023. [CrossRef]
- Li, C.; Zhou, H.; Zhang, W.; Che, D. Bacterial lysate treatment in allergic disease: A systematic review and meta-analysis. Pediatr. Allergy Immunol. 2021, 32, 1813–1823. [CrossRef]
- Bodemer C, Guillet G, Cambazard F, Boralevi F, Ballarini S, et al. (2017) Adjuvant treatment with the bacterial lysate (OM-85) improves management of atopic dermatitis: A randomized study. PLOS ONE 12(3): e0161555. [CrossRef]
- Li C, Zhou H, Zhang W, Che D. Bacterial lysate treatment in allergic disease: A systematic review and meta-analysis. Pediatr Allergy Immunol. 2021;32:1813–1823. [CrossRef]
- Licari, A.; Manti, S.; Castagnoli, R.; Marseglia, A.; Foiadelli, T.; Brambilla, I.; Marseglia, G.L. Immunomodulation in Pediatric Asthma. Front. Pediatr. 2019, 7, 289. [CrossRef]
- Sánchez-Hernández, M.C.; Montero, J.; Rondon, C.; Benitez del Castillo, J.M.; Velázquez, E.; Herreras, J.M.; Fernández-Parra, B.; Merayo-Lloves, J.; Del Cuvillo, A.; Vega, F.; Valero, A.; Panizo, C.; Montoro, J.; Matheu, V.; Lluch-Bernal, M.; González, M.L.; González, R.; Dordal, M.T.; Dávila, I.; Colás, C.; Spanish Group Ocular Surface-GESOC. Consensus document on allergic conjunctivitis (DECA). J. Investig. Allergol. Clin. Immunol. 2015, 25, 94–106.
- Juniper, E.F.; Thompson, A.K.; Ferrie, P.J. Validation of the standardized version of the rhinoconjunctivitis quality of life questionnaire. J. Allergy Clin. Immunol. 1999, 104, 364–369. [CrossRef]
- Juniper, E.F.; Guyatt, G.H.; Griffith, L.E. Interpretation of quality of life questionnaire data in rhinoconjunctivitis. J. Allergy Clin. Immunol. 1996, 98, 843–845. [CrossRef]
- Nieto, A.; Mazón, A.; Calderón, R. Bacterial mucosal immunotherapy with MV130 prevents recurrent wheezing in children: A randomized, double-blind, placebo-controlled clinical trial. Am. J. Respir. Crit. Care Med. 2021, 204, 462–472. [CrossRef]
- Iovieno, A.; Lambiase, A.; Sacchetti, M.; Stampachiacchiere, B.; Micera, A.; Bonini, S. Preliminary evidence of the efficacy of probiotic eye-drop treatment in patients with vernal keratoconjunctivitis. Graefes Arch. Clin. Exp. Ophthalmol. 2008, 246, 435–441. [CrossRef]
- Li, Y.; Li, R.; Liu, Z.-C.; et al. Analysis of the correlation between tear film changes of allergic conjunctivitis and dry eye. Guoji Yanke Zazhi (Int. Eye Sci.) 2014, 14, 1272–1274. [CrossRef]
- Mahesh, P.A.; Samajdar, S.S.; Nagarajan, S.A.; Murthy, G.M.V.; Moitra, S. Immunotherapy: Current indications and recommendations in the management of ocular allergy. Indian J. Ophthalmol. 2025, 73, 526–536. [CrossRef]
- Peng, G.C.; Hsu, C.H. The efficacy and safety of heat-killed Lactobacillus paracasei for treatment of allergic rhinitis: a randomized controlled trial. Allergy Asthma Proc. 2005, 26, 433–438. [CrossRef]
- Bore, M. Managing ocular allergy in resource-poor settings. Community Eye Health 2016, 29, 47–49.
- Hempel, S.; Newberry, S.; Ruelaz, A.; Wang, Z.; Miles, J.N.; Suttorp, M.J.; Johnsen, B.; Shanman, R.; Slusser, W.; Fu, N.; Smith, A.; Roth, B.; Polak, J.; Motala, A.; Perry, T.; Shekelle, P.G. Safety of probiotics used to reduce risk and prevent or treat disease. Evid. Rep. Technol. Assess. 2011, 200, 1–645.
- Whelan, K.; Myers, C.E. Safety of probiotics in patients receiving nutritional support: A systematic review of case reports, randomized controlled trials, and nonrandomized trials. Am. J. Clin. Nutr. 2010, 91, 687–703. [CrossRef]
- Kang, M.G.; Han, S.W.; Kang, H.R.; Hong, S.J.; Kim, D.H. Probiotic NVP-1703 alleviates allergic rhinitis by inducing IL-10 expression: A four-week clinical trial. Nutrients 2020, 12, 1427. [CrossRef]
- Akdis, C.A.; Akdis, M. Mechanisms of immune tolerance to allergens: Role of IL-10 and Tregs. J. Clin. Invest. 2014, 124, 4678–4680. [CrossRef]
- Rosser, E.C.; Mauri, C. Regulatory B cells: Origin, phenotype, and function. Immunity 2015, 42, 607–612. [CrossRef]
- Salazar, A.; Nieto, J.E.; Velazquez-Soto, H.; Jiménez-Martínez, M.C. Activation of IL-10+ B cells: A novel immunomodulatory mechanism for therapeutic bacterial suspensions. SAGE Open Med. 2020, 8, 2050312120901547.
| Variable | Median (IQR) |
|---|---|
| Age (years) | 8 (7–9) |
| Peripheral blood eosinophils (%) | 2 (1–11.2) |
| Serum IgE (IU/ mL) | 73.2 (43.3–127.5) |
| Serial Stool Examination (3 samples) |
Negative in all patients |
| Skin Prick Test (3 samples) |
Negative in all patients |
| Symptom | Severity Before Value (Medians, IQR) |
Severity After Value (Medians, IQR) |
p Value Comparisons (Wilcoxon) |
|---|---|---|---|
| Itching | 2 (1-3) | 0 (0-0.5) | 0.012 |
| Tearing | 2 (0-4) | 0 (0-1) | 0.143 |
| Light Sensitivity | 2 (0-4) | 0 (0-0.5) | 0.044 |
| Gritty Sensation | 1 (1-2) | 0 (0-0.5) | 0.020 |
| Burning Sensation | 2 (0-4) | 0 (0-0.5) | 0.020 |
| Value before Median (IQR) |
Value after Median (IQR) |
P-value Comparisons (Wilcoxon) |
|
|---|---|---|---|
| TBUT (sec) | 5 (4.0-6.5) | 9 (7-10) | 0.002 |
| Schirmer I (mm) | 29 (17.5-35) | 12 (11-14) | 0.004 |
| Description | Relationship | Start date | Severity | Resolution | Aftermath |
|---|---|---|---|---|---|
| Diarrhea 10012727 |
Possible | 3 | Mild | Resolved | No |
| Nausea 10028817 |
Possible | 3 | Mild | Resolved | No |
| Abdominal pain 10000087 | Possible | 1 | Mild | Resolved | No |
| Vomit 10028817 |
Possible | 30 | Mild | Resolved | No |
| Diarrhea 10012727 |
Possible | 63 | Mild | Resolved | No |
| Nausea 10028817 |
Possible | 1 | Mild | Resolved | No |
| Abdominal pain 10000087 | Possible | 1 | Mild | Resolved | No |
| Dysgeusia 10013911 |
Possible | 3 | Mild | Resolved | No |
| Weight loss 10047900 |
Unlikely | 30 | Mild | Resolved | No |
| Abdominal pain 10000087 | Possible | 1 | Mild | Resolved | No |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




