Submitted:
28 April 2026
Posted:
29 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Study Design:
2.2. Participants
2.3. Cross-Sectional Study
2.4. Exercise Intervention (Case Study)
2.5. Measurement of Klotho
2.6. Assessment of Exercise Habits
2.7. Statistical Analysis
2.8. Sample Size Calculation
2.9. Rationale for Study Design
2.10. Ethical Approval
3. Results
| Variable | Hemodialysis (n=24) | Healthy Controls (n=18) | p-value |
|---|---|---|---|
| Age (years) | 75.5 ± 6.8 | 50.7 ± 11.4 | 0.001 |
| Male (%) | 25% | 16.7% | 0.64 |
| Prevalence of diabetes | 40% | 0 | N/A |
| Primary disease | ・Diabetic Nephropathy ・IgA Nephropathy ・Nephrosclerosis ・Polycystic kidney disease ・ Idiopathic |
- | N/A |
| α-Klotho (pg/mL) | 446.6±174.6 | 858.8 ±361.2 | 0.0001 |
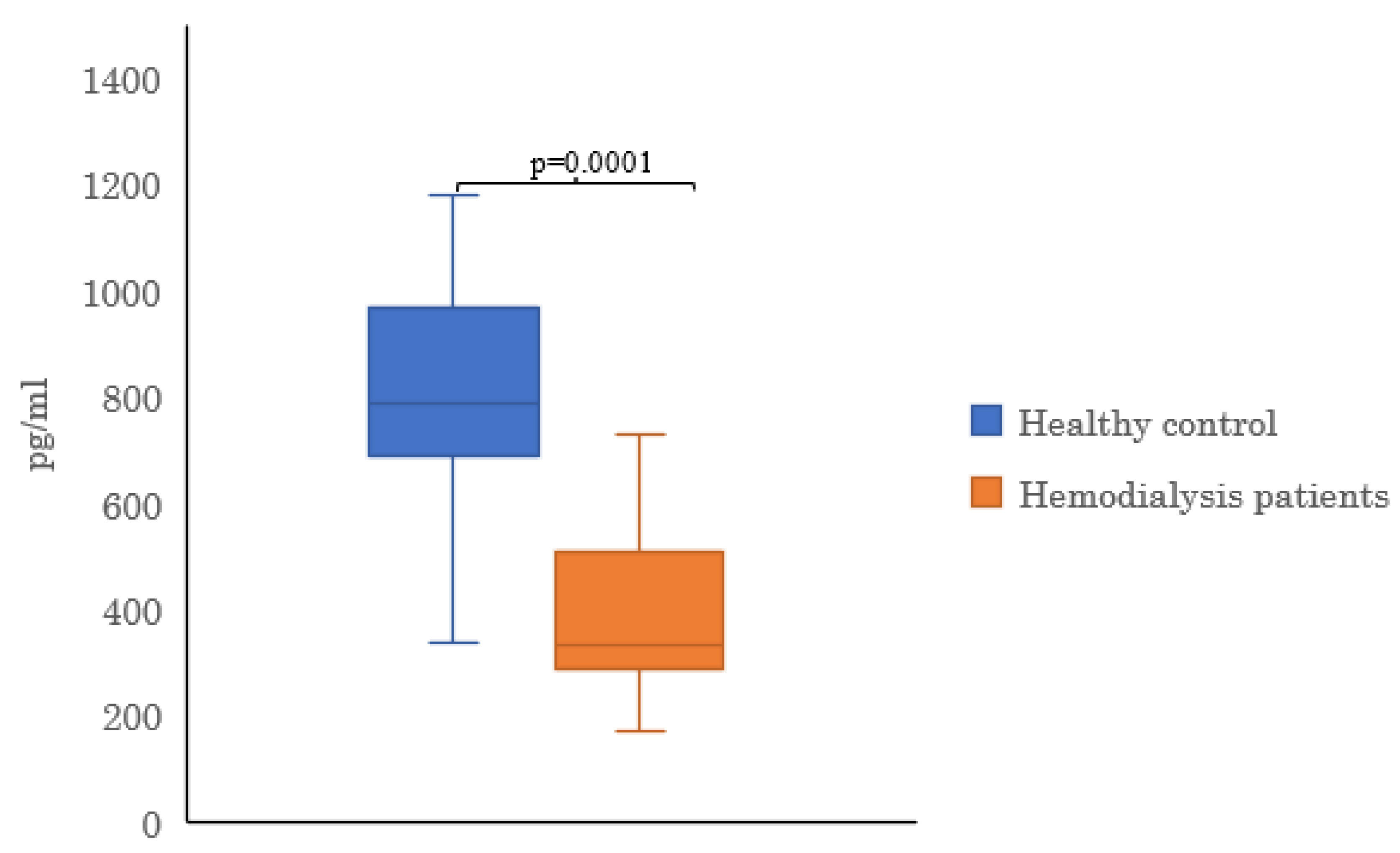
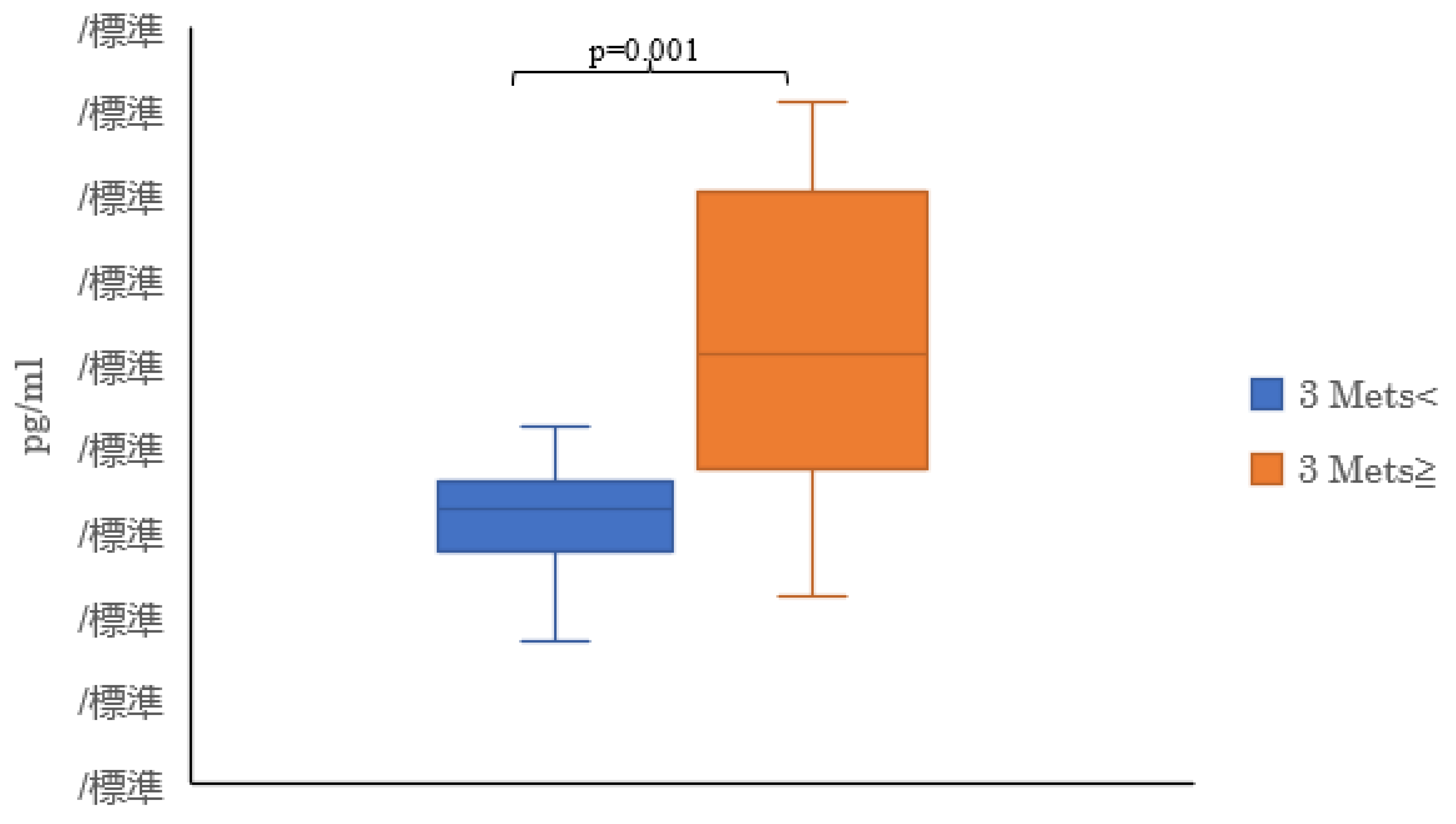
4. Discussion
Conclusion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Imai, E.; Horio, M.; Nitta, K.; Yamagata, K.; Iseki, K.; Hara, S.; Ura, N.; Kiyohara, Y.; Hirakata, H.; Watanabe, T.; et al. Estimation of glomerular filtration rate by the MDRD study equation modified for Japanese patients with chronic kidney disease. Clin. Exp. Nephrol. 2007, 11, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Nitta, K.; Goto, S.; Masakane, I.; Hanafusa, N.; Taniguchi, M.; Hasegawa, T.; Nakai, S.; Wada, A.; Hamano, T.; Hoshino, J.; et al. Annual dialysis data report for 2018, JSDT Renal Data Registry: survey methods, facility data, incidence, prevalence, and mortality. Ren. Replace. Ther. 2020, 6, 41. [Google Scholar] [CrossRef]
- Bündchen, D.C.; Sousa, H.; Afreixo, V.; Frontini, R.; Ribeiro, O.; Figueiredo, D.; Costa, E. Intradialytic exercise in end-stage renal disease: An umbrella review of systematic reviews and/or meta-analytical studies. Clin. Rehabil. 2021, 35, 812–828. [Google Scholar] [CrossRef]
- Segura-Ortí, E.; Gordon, P.; Doyle, J.; Johansen, K. Correlates of physical functioning and performance across the spectrum of kidney function. Clin. Nurs. Res. 2018, 27, 579–596. [Google Scholar] [CrossRef]
- Dong, B.; Zheng, Z.; Zhong, S.; Ye, Y.; Wang, Y.; Yang, L.; Xiao, Z.; Fang, Q.; Zhao, H. Integrated Transcriptome and Metabolome Analysis of Color Change and Low-Temperature Response during Flowering of Prunus mume. Int. J. Mol. Sci. 2022, 23, 12831. [Google Scholar] [CrossRef] [PubMed]
- Kuro-o, M.; Matsumura, Y.; Aizawa, H.; Kawaguchi, H.; Suga, T.; Utsugi, T.; Ohyama, Y.; Kurabayashi, M.; Kaname, T.; Kume, E.; et al. Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature 1997, 390, 45–51. [Google Scholar] [CrossRef]
- Hu, M.C.; Shiizaki, K.; Kuro-o, M.; Moe, O.W. Fibroblast growth factor 23 and Klotho: physiology and pathophysiology of an endocrine network of mineral metabolism. Annu Rev. Physiol. 2013, 75, 503–533. [Google Scholar] [CrossRef]
- Kuro-o, M. Aging and FGF23-klotho system. In Vitamins and Hormones; Elsevier, 2021; Volume 115, pp. 317–332. [Google Scholar]
- Corrêa, H.L.; Raab, A.T.O.; Araújo, T.M.; Deus, L.A.; Reis, A.L.; Honorato, F.S.; Rodrigues-Silva, P.L.; Neves, R.V.P.; Brunetta, H.S.; Mori, M.; et al. A systematic review and meta-analysis demonstrating Klotho as an emerging exerkine. Sci. Rep. 2022, 12, 17587. [Google Scholar] [CrossRef]
- Gollie, J.M.; Ryan, A.S.; Sen, S.; Patel, S.S.; Kokkinos, P.F.; Harris-Love, M.O.; Scholten, J.D.; Blackman, M.R. Exercise for patients with chronic kidney disease: from cells to systems to function. Am. J. Physiol. Ren. Physiol. 2024, 326, F420–f437. [Google Scholar] [CrossRef]
- Dun, Y.; Thomas, R.J.; Medina-Inojosa, J.R.; Squires, R.W.; Huang, H.; Smith, J.R.; Liu, S.; Olson, T.P. High-Intensity Interval Training in Cardiac Rehabilitation: Impact on Fat Mass in Patients With Myocardial Infarction. Mayo Clin. Proc. 2019, 94, 1718–1730. [Google Scholar] [CrossRef] [PubMed]
- Beetham, K.S.; Howden, E.J.; Fassett, R.G.; Petersen, A.; Trewin, A.J.; Isbel, N.M.; Coombes, J.S. High-intensity interval training in chronic kidney disease: A randomized pilot study. Scand. J. Med. Sci. Sports 2019, 29, 1197–1204. [Google Scholar] [CrossRef]
- Yamazaki, Y.; Imura, A.; Urakawa, I.; Shimada, T.; Murakami, J.; Aono, Y.; Hasegawa, H.; Yamashita, T.; Nakatani, K.; Saito, Y.; et al. Establishment of sandwich ELISA for soluble alpha-Klotho measurement: Age-dependent change of soluble alpha-Klotho levels in healthy subjects. Biochem Biophys. Res. Commun. 2010, 398, 513–518. [Google Scholar] [CrossRef]
- Heijboer, A.C.; Blankenstein, M.A.; Hoenderop, J.; de Borst, M.H.; Vervloet, M.G. Laboratory aspects of circulating α-Klotho. Nephrol. Dial. Transpl. 2013, 28, 2283–2287. [Google Scholar] [CrossRef] [PubMed]
- Shimamura, Y.; Hamada, K.; Inoue, K.; Ogata, K.; Ishihara, M.; Kagawa, T.; Inoue, M.; Fujimoto, S.; Ikebe, M.; Yuasa, K.; et al. Serum levels of soluble secreted α-Klotho are decreased in the early stages of chronic kidney disease, making it a probable novel biomarker for early diagnosis. Clin. Exp. Nephrol. 2012, 16, 722–729. [Google Scholar] [CrossRef] [PubMed]
- Ainsworth, B.E.; Haskell, W.L.; Herrmann, S.D.; Meckes, N.; Bassett, D.R., Jr.; Tudor-Locke, C.; Greer, J.L.; Vezina, J.; Whitt-Glover, M.C.; Leon, A.S. 2011 Compendium of Physical Activities: a second update of codes and MET values. Med. Sci. Sports Exerc 2011, 43, 1575–1581. [Google Scholar] [CrossRef]
- Sallis, J.F.; Buono, M.J.; Roby, J.J.; Micale, F.G.; Nelson, J.A. Seven-day recall and other physical activity self-reports in children and adolescents. Med. Sci. Sports Exerc 1993, 25, 99–108. [Google Scholar] [CrossRef]
- Prince, S.A.; Cardilli, L.; Reed, J.L.; Saunders, T.J.; Kite, C.; Douillette, K.; Fournier, K.; Buckley, J.P. A comparison of self-reported and device measured sedentary behaviour in adults: a systematic review and meta-analysis. Int. J. Behav. Nutr. Phys. Act. 2020, 17, 31. [Google Scholar] [CrossRef]
- Altman, D.; Machin, D.; Bryant, T.; Gardner, M. Statistics with confidence: confidence intervals and statistical guidelines; John Wiley & Sons, 2013. [Google Scholar]
- Hanley, J.A.; McNeil, B.J. The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology 1982, 143, 29–36. [Google Scholar] [CrossRef]
- Hertzog, M.A. Considerations in determining sample size for pilot studies. Res. Nurs. Health 2008, 31, 180–191. [Google Scholar] [CrossRef]
- Pavik, I.; Jaeger, P.; Ebner, L.; Poster, D.; Krauer, F.; Kistler, A.D.; Rentsch, K.; Andreisek, G.; Wagner, C.A.; Devuyst, O.; et al. Soluble klotho and autosomal dominant polycystic kidney disease. Clin. J. Am. Soc. Nephrol. 2012, 7, 248–257. [Google Scholar] [CrossRef]
- Cohen, J. Statistical power analysis for the behavioral sciences; routledge, 2013. [Google Scholar]
- Hulley, S.B.; Cummings, S.R.; Browner, W.S.; Grady, D.G.; Newman, T.B. Designing Clinical Research, 4e; Lippincott Williams & Wilkins, 2013. [Google Scholar]
- Vandenbroucke, J.P. In defense of case reports and case series. Ann. Intern Med. 2001, 134, 330–334. [Google Scholar] [CrossRef]
- Japanese Society of Renal Re. Guideline for Renal Rehabilitation, 2nd ed.; Ishiyaku Publishers: Tokyo, 2026. [Google Scholar]
- World Medical Association. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. Jama 2013, 310, 2191–2194. [Google Scholar] [CrossRef]
- Greenwood, S.A.; Koufaki, P.; Mercer, T.H.; MacLaughlin, H.L.; Rush, R.; Lindup, H.; O'Connor, E.; Jones, C.; Hendry, B.M.; Macdougall, I.C.; et al. Effect of exercise training on estimated GFR, vascular health, and cardiorespiratory fitness in patients with CKD: a pilot randomized controlled trial. Am. J. Kidney Dis. 2015, 65, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Myers, J.; Chan, K.; Chen, Y.; Lit, Y.; Patti, A.; Massaband, P.; Kiratli, B.J.; Tamura, M.; Chertow, G.M.; Rabkin, R. Effect of a home-based exercise program on indices of physical function and quality of life in elderly maintenance hemodialysis patients. Kidney Blood Press. Res. 2021, 46, 196–206. [Google Scholar] [CrossRef]
- McGarvey, C.; Doyle, K.; Roche, A.; Mockler, D.; Knight, S.; Kenny, R.A.; Sexton, D.J. Age-related physiological and molecular biomarkers associated with kidney function: a systematic review and meta-analysis. QJM An. Int. J. Med. 2025, 118, 731–742. [Google Scholar] [CrossRef]
- Pedersen, B.K. Muscle as a secretory organ. Compr. Physiol. 2013, 3, 1337–1362. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, K.; Nishida, M.; Taneike, M.; Yamamoto, R.; Moriyama, T.; Yamauchi-Takihara, K. Serum Klotho levels contribute to the prevention of disease progression. Int. J. General. Med. 2021, 229–236. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Febbraio, M.A. Muscle as an endocrine organ: focus on muscle-derived interleukin-6. Physiol. Rev. 2008, 88, 1379–1406. [Google Scholar] [CrossRef] [PubMed]
- Kuro, O.M. The Klotho proteins in health and disease. Nat. Rev. Nephrol. 2019, 15, 27–44. [Google Scholar] [CrossRef]
| Variable | Pre-intervention | Post-intervention (HIIT) |
|---|---|---|
| Barthel Index (BI) | 90/100 | 100/100 |
| SPPB score | 10/12 | 12/12 (Full score) |
| Max Exercise Intensity | 2.0 METs | 5.5 METs |
| MMT (Lower extremity) | 4 | 4~5 |
| Lower back pain (NRS) | 5~6/10 | 0/10 (Disappeared) |
| Blood Pressure | Stable | Stable (No change) |
| α-Klotho (pg/mL) | 342.2 | 517.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




