Submitted:
27 April 2026
Posted:
29 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. History of the Disease
3. Significance of Upper Cervical Spine Anatomy
4. Current Treatment Options
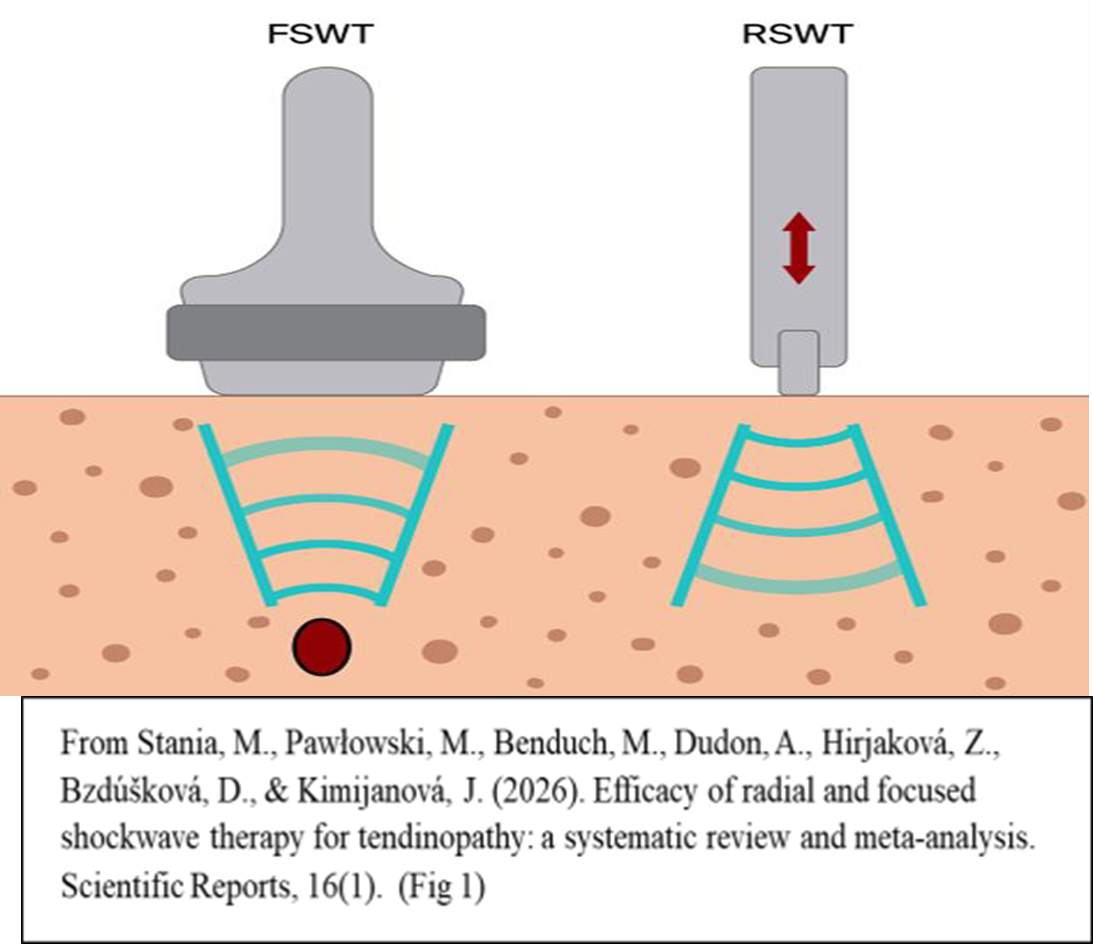
5. A Novel Therapeutic Approach to EDS: Extracorporeal Shock Wave Therapy
References
- Tinkle, B.; Castori, M.; Berglund, B.; Cohen, H.; Grahame, R.; Kazkaz, H.; Levy, H. Hypermobile Ehlers-Danlos syndrome (a.k.a. Ehlers-Danlos syndrome type III and Ehlers-Danlos syndrome hypermobility type): clinical description and nat-ural history. Am. J. Med. Genet C. Semin Med. Genet. 2017, 175, 48–69. [Google Scholar] [CrossRef]
- Malfait, F.; Francomano, C.; Byers, P.; et al. The 2017 international classification of the Ehlers-Danlos syndromes. Am. J. Med. Genet C. Semin Med. Genet. 2017, 175, 8–26. [Google Scholar] [CrossRef]
- Malek, S.; Reinhold, E.J.; Pearce, G.S. The Beighton Score as a measure of generalised joint hypermobility. Rheumatol. Int. 2021, 41(10), 1707–16. [Google Scholar] [CrossRef]
- Childs, Sharon G. Musculoskeletal Manifestations of Ehlers–Danlos Syndrome. Orthop. Nurs. 2010, 29(2), 133–139. [Google Scholar] [CrossRef]
- Celletti, C.; Camerota, F.; Castori, M.; Censi, F.; Gioffrè, L.; Calcagnini, G.; Strano, S. Orthostatic intolerance and postural orthostatic tachycardia syndrome in joint hypermobility syndrome/Ehlers-Danlos syndrome, hypermobility type: neu-rovegetative dysregulation or autonomic failure? Biomed. Res. Int. 2017, 2017, 9161865. [Google Scholar] [CrossRef] [PubMed]
- Hauser, R.A.; Matias, D.; Rawlings, B.R. Cervicovagopathy: ligamentous cervical instability and dysstructure as a poten-tial etiology for vagus nerve dysfunction in the cause of human symptoms and diseases. Front Neurol. 2025, 16, 1572863. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Aziz, Q.; Harris, L.A.; Goodman, B.P.; Simrén, M.; Shin, A. AGA Clinical Practice Update on GI Manifestations and Auto-nomic or Immune Dysfunction in Hypermobile Ehlers-Danlos Syndrome: Expert Review. Clin. Gastroenterol. Hepatol. Epub. 2025, 23(8), 1291–1302. [Google Scholar] [CrossRef] [PubMed]
- Auricular Vagal Nerve Stimulation for Hypermobile Ehlers-Danlos Syndrome Study Details | NCT05212129 | Au-ricular Vagal Nerve Stimulation for Hypermobile Ehlers-Danlos Syndrome | ClinicalTrials.gov. Accessed. (accessed on 04/12/2025).
- Transcutaneous Auricular Vagus Nerve Stimulation in Postural Tachycardia Syndrome: a Prospective Cross-over Study Study Details | NCT07163130 | Tragus Stimulation for POTS Treatment | ClinicalTrials.gov.
- Parapia, L.A.; Jackson, C. Ehlers-Danlos syndrome--a historical review. Br. J. Haematol. 2008, 141(1), 32–5. [Google Scholar] [CrossRef] [PubMed]
- McKusick, V.A. Heritable disorders of connective tissue: IV. The Ehlers-Danlos syndrome, Journal of Chronic Diseases, 3, (1) 1956, pp 2-24, ISSN 0021-9681, https://doi.org/10.1016/0021-9681(56)90095-9. (https://www.sciencedirect.com/science/article/pii/0021968156900959). [CrossRef]
- Bloom, L.; Byers, P.; Francomano, C.; Tinkle, B.; Malfait, F. Steering Committee of The International Consortium on the Ehlers-Danlos Syndromes. The international consortium on the Ehlers-Danlos syndromes. Am. J. Med. Genet C. Semin Med. Genet 2017, 175(1), 5–7. [Google Scholar] [CrossRef] [PubMed]
- Zaborowska-Sapeta, K.; Tymińska-Wójcik, P.; Sonza, A.; Kluszczyński, M.; Skowrońska, A. The impact of modern lifestyles on spinal health in the pediatric population: a narrative review. Children 2026, 13(3), 341. [Google Scholar] [CrossRef] [PubMed]
- Hauser, R. A.; Griffiths, M.; Watterson, A.; Matias, D.; Rawlings, B. R. Characterizing Initial Cervical Spine and Neurovascular Findings in 84 Consecutive Patients with Hypermobile Ehlers–Danlos Syndrome: A Retrospective Study. J. Clin. Med. 2026, 15(6), 2212. [Google Scholar] [CrossRef]
- Weiner, G. Atrial Fibrillation and its relationship with Thoracic Outlet Syndrome. Asia-Pac Chiropr J. 2025, 6.2. Available online: www.apcj.net/papersissue-6-2/#WeinerAFib.
- Hauser, R. A.; Griffiths, M.; Watterson, A.; Matias, D.; Rawlings, B. Objective test findings in patients with chronic eye symptoms seeking care at an outpatient neck center: ligamentous cervical instability etiology? Front. Neurol. 2025, 16, 1576315. [Google Scholar] [CrossRef]
- Henderson, F. C.; et al. Neurological and spinal manifestations of the Ehlers-Danlos syndromes. Am. J. Med. Genet. Part C. Semin. Med. Genet. 2018, 175(1), 195–211. [Google Scholar] [CrossRef]
- Centenera, M.; et al. Craniocervical instability and vagus nerve compression. Centeno-Schultz Blog. 2021. Available online: https://centenoschultz.com/.
- Kim, D.H.; Vaccaro, A.R.; Dickman, C.A.; Cho, D.; Lee, S.; Kim, I. Surgical Anatomy and Techniques to the Spine; Elsevier: Amsterdam, Netherlands, 2013. [Google Scholar]
- Vernon, L. F.; Benn, A. Documenting Cervical Spine Injuries Following Negative MRI Findings: Clinical and Medico-Legal Overview of Dynamic Imaging. Cureus 2025, 17(7), e88121. [Google Scholar] [CrossRef] [PubMed]
- Rajani, S. Is variant anatomy of atlas clinically important? A review. Basic Sci. Med. 2014, 3, 1–7. [Google Scholar]
- Ivancic, P.C.; Ito, S.; Tominaga, Y.; et al. Whiplash causes increased laxity of cervical capsular ligament. Clin. Biomech. 2008, 23, 159–65. [Google Scholar] [CrossRef]
- Tominaga, Y.; Ndu, A.B.; Coe, M.P.; et al. Neck ligament strength is decreased following whiplash trauma. BMC Muscu-Loskelet Disord. 2006, 7, 103. [Google Scholar] [CrossRef] [PubMed]
- Holdsworth, F.W. Fractures, dislocations and fracture-dislocations of the spine. J. Bone Jt. Surg. Am. 1970, 52, 1534–51. [Google Scholar] [CrossRef]
- Magerl, F.; Aebi, M.; Gertzbein, S.D.; et al. A comprehensive classication of thoracic and lumbar injuries. Eur. Spine J. 1994, 3, 184–201. [Google Scholar] [CrossRef]
- DePace, N. L.; Soloway, S.; Yayac, M.; Colombo, J. Spine and Organ Involvement. In Ehlers-Danlos/Hyper-mobility Syndromes and Other Connective Tissue Disorders: Assessments, Diagnostics, and Therapies; Springer Nature Switzerland: Cham, 2025; pp. 163–190. [Google Scholar]
- Mehta, D.; Simmonds, L.; Hakim, A.J.; Matharu, M. Headache disorders in patients with Ehlers-Danlos syn-dromes and hypermobility spectrum disorders. Front. Neurol. 2024, 15, 1460352. [Google Scholar] [CrossRef]
- Borghei-Razavi, H.; Darvish, O.; Schick, U. Disabling vertigo and tinnitus caused by intrameatal compression of the anterior inferior cerebellar artery on the vestibulocochlear nerve: a case report, surgical considerations, and review of the literature. J. Neurol. Surg. Rep. 2014, 75, e47-51. [Google Scholar] [CrossRef]
- Verma, R.; Junewar, V.; Garg, R.K.; Malhotra, H.S. A rare case of basilar impression. BMJ Case Rep. 2012, 2012. [Google Scholar] [CrossRef]
- Baquiran, M.; Bordoni, B. Anatomy, head and neck: anterior vagus nerve. In StatPearls [Internet]; StatPearls Publishing: Treasure Island (FL), 2025. [Google Scholar]
- Zimmerman, E.E.; Misulis, K.E. Chapter 3.4: brainstem and cranial nerves. In Neurologic Localization and Diagnosis; Zim-merman, E.E., Samuels, M.A., Kirshner, H.S., Eds.; Elsevier: Amsterdam, Netherlands, 2023; pp. 89–106. [Google Scholar] [CrossRef]
- Centenera, M.; et al. Craniocervical instability and vagus nerve compression. Centeno-Schultz Blog. 2021. Available online: https://centenoschultz.com/.
- Gilete, J. Craniocervical instability: Causes & treatment. 2025. Available online: https://drgilete.com/specialties/craneocervical-instability-cci/.
- Daylor, V.; Griggs, M.; Weintraub, A.; Byrd, R.; Petrucci, T.; Huff, M.; Norris, R. A. Defining the Clinical Complexity of hEDS and HSD: A Global Survey of Diagnostic Challenge, Comorbidities, and Unmet Needs. medRxiv 2025, 2025–06. Available online: https://www.medrxiv.org/content/medrxiv/early/2025/06/06/2025.06.05.25329074.full.pdf. [CrossRef]
- Henderson, F.C., Sr.; Francomano, C.A.; Koby, M.; Tuchman, K.; Adcock, J.; Patel, S. Cranio-cervical instability in patients with Ehlers–Danlos syndrome: outcomes analysis of 103 surgically treated patients. Front. Surg. 2018, 5, 116. [Google Scholar]
- Henderson, F.C.; Schubart, J.R.; Narayanan, M.V.; et al. Craniocervical instability in patients with Ehlers-Danlos syn-dromes: outcomes analysis following occipito-cervical fusion. Neurosurg. Rev. 2024, 47, 27. [Google Scholar] [CrossRef]
- Eichinger, J.K.; Byrd, R.L.; Bailey, E.P.; Reis, R.J.; Daylor, V.; Schiessl, M.; Gensemer, C.; Friedman, R.J.; Patel, S.J.; Norris, R.A. Or-thopaedic Manifestations in Hypermobile Ehlers-Danlos Syndrome. J. Bone Jt. Surg. Am. 2025, 107(16), 1848–1856. [Google Scholar] [CrossRef] [PubMed]
- Chu, E.C.P.; Lin, A.F.C. Relief of postural orthostatic tachycardia syndrome with chiropractic rehabilitation. J. Fam. Med. Prim. Care 2022, 11(7), 4006–4009. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- La Francis, M.E. A chiropractic perspective on atlantoaxial instability in Down’s syndrome. J. Manip. Physiol. Ther. 1990, 13(3), 157–60. [Google Scholar] [PubMed]
- Katz, E. A.; Katz, S. B.; Freeman, M. D. Non-Surgical Management of Upper Cervical Instability via Im-proved Cervical Lordosis: A Case Series of Adult Patients. J. Clin. Med. 2023, 12(5), 1797. [Google Scholar] [CrossRef] [PubMed]
- Doyle, T.A.; Halverson, C.M.E. Use of complementary and alternative medicine by patients with hypermobile Ehlers-Danlos Syndrome: A qualitative study. Front Med. 2022, 9, 1056438. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Baquiran, M.; Bordoni, B. Anatomy, head and neck: anterior vagus nerve. In StatPearls [Internet]; StatPearls Publishing: Treasure Island (FL), 2025. [Google Scholar]
- Letson, A. K.; Dahners, L. E. The effect of combinations of growth factors on ligament healing. Clin. Orthop. Relat. Res. (1976-2007) 1994, 308, 207–212. [Google Scholar] [CrossRef]
- Fang, J.; Wang, X.; Jiang, W.; Zhu, Y.; Hu, Y.; Zhao, Y.; Wang, Y. Platelet-rich plasma therapy in the treatment of diseases associated with orthopedic injuries. Tissue Eng. Part B Rev. 2020, 26(6), 571–585. [Google Scholar] [CrossRef]
- Centeno, C.J.; Al-Sayegh, H.; Freeman, M.D.; et al. A multi-center analysis of adverse events among two thousand, three hundred and seventy two adult patients undergoing adult autologous stem cell therapy for orthopaedic condi-tions. Int. Orthop. (SICOT) 2016, 40, 1755–1765. [Google Scholar] [CrossRef] [PubMed]
- Hauser, R. A.; Orlofsky, A. Regenerative injection therapy with whole bone marrow aspirate for degenera-tive joint disease: a case series. Clin. Med. Insights Arthritis Musculoskelet. Disord. 2013, 6, CMAMD–S10951. [Google Scholar] [CrossRef] [PubMed]
- Arita, A.; Tobita, M. Adverse events related to platelet-rich plasma therapy and future issues to be resolved. Regen. Ther. 2024, 26, 496–501. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Eymard, F.; Louati, K.; Noel, É.; Abouqal, R.; et al. Indications and contraindications to platelet-rich plasma injections in musculoskeletal diseases in case of infectious, oncological and haematological comorbidities: A 2025 formal consensus from the GRIIP (International Research Group on Platelet Injections). Knee Surg Sports Traumatol Arthrosc. 2025 Jun;33(6):2293-2306. Epub 2025 Apr 22. [CrossRef] [PubMed] [PubMed Central]
- Artoni, A.; Bassotti, A.; Abbattista, M. Hemostatic abnormalities in patients with Ehlers–Danlos syndrome. J. Thromb. Haemost. 16, 2425–2431. [CrossRef]
- Hunt, W. E.; Baird, W. C. Complications following injection of sclerosing agent to precipitate fibro-osseous proliferation. J. Neurosurg. 1961, 18(4), 461–465. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Huang, X.; Wang, H.; Liu, X.; Zhang, T.; Wang, Y.; Hu, D. The challenges and promises of allogeneic mesenchymal stem cells for use as a cell-based therapy. Stem Cell. Res. Ther. 2015, 6, 234. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Schmitz, C.; Császár, N.B.; Milz, S.; Schieker, M.; Maffulli, N.; mpe, J.D.; et al. Efficacy and safety of extracorporeal shock wave therapy for orthopedic conditions: a systematic review on studies listed in the PEDro database. Br. Med. Bull. 2015, 116(1), 115–38. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chaussy, C.; Eisenberger, F.; Forssmann, B. Extracorporeal shockwave lithotripsy (ESWL): a chronology. J. Endourol. 2007, 21(11), 1249–53. [Google Scholar] [CrossRef] [PubMed]
- Konjen, N.; Napnark, T.; Janchai, S. A comparison of the effectiveness of radial extracorporeal shock wave therapy and ultrasound therapy in the treatment of chronic plantar fasciitis: a randomized controlled trial. J. Med. Assoc. Thai 2015, 98 Suppl 1, S49–56. [Google Scholar] [PubMed]
- https://provider.univerahealthcare.com/documents/d/global/uni-prv-extracorporeal-shock-wave-therapy-eswt-for-musculoskeletal-conditions-and-soft-tissue-wounds#:~:text=The%20OssaTron%20device%20(HealthTron-ics%2C%20Inc,describes%20a%20low%2Ddose%20protocol.
- https://www.medscape.com/viewarticle/890703?form=fpf.
- Cao, B.; Tang, X.; Liu, C.; Xu, G.; Lei, M.; Wu, F.; Chen, W.; Ni, H.; Zhang, F. Unlocking new Frontiers: The cellular and molec-ular impact of extracorporeal shock wave therapy (ESWT) on central nervous system (CNS) disorders and peripheral nerve injuries (PNI). Exp. Neurol. 2025, 384, 115052. [Google Scholar] [CrossRef] [PubMed]
- Leuk, J.S.P.; Low, L.L.N.; Teo, W.-P. An Overview of Acoustic-Based Interventions to Improve Motor Symptoms in Parkinson’s Disease. Front. Aging Neurosci. 2020, 12, 243. [Google Scholar] [CrossRef] [PubMed]
- Simplicio, C.L.; Jácomo, A.A.E.; de Barros. GAM | Treatment with Shockwave Therapy in a Patient with Joint Hypermo-bility and Temporomandibular Dysfunction | Journal of Regenerative Science. 2024, 4(1), 09–15. [Google Scholar]
- Kou, D.; Chen, Q.; Wang, Y.; et al. The application of extracorporeal shock wave therapy on stem cells therapy to treat various diseases. Stem Cell. Res. Ther. 2024, 15, 271. [Google Scholar] [CrossRef]
- Zhai, L.; Sun, N.; Zhang, B.; Liu, S.T.; Zhao, Z.; Jin, H.C.; Ma, X.L.; Xing, G.Y. Effects of Focused Extracorporeal Shock Waves on Bone Marrow Mesenchymal Stem Cells in Patients with Avascular Necrosis of the Femoral Head. Ultrasound Med. Biol. 2016, 42(3), 753–62. [Google Scholar] [CrossRef] [PubMed]
- Wang, C. J. Extracorporeal shockwave therapy in musculoskeletal disorders. J. Orthop. Surg. Res. 2012, 7(1), 11. Available online: https://pmc.ncbi.nlm.nih.gov/articles/PMC3666498/. [CrossRef]
- Moya, D.; et al. The biological effects of extracorporeal shock wave therapy on tendon injury. Interna-Tional J. Mol. Sci. 2018, 19(11), 3567. [Google Scholar]
- Venosa, M.; Vespasiani, A.; Romanini, E.; Nigri, M.; Fidanza, A.; Logroscino, G. Radial Extracorporeal Shockwave Therapy for the Treatment of Chronic Refractory Coccygodynia. J. Med. Cases 2025, 16(12), 467–474. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- De la Corte-Rodriguez, H.; Roman-Belmonte, J.M.; Rodriguez-Damiani, B.A.; Vazquez-Sasot, A.; Rodriguez-Merchan, E.C. Extracor-poreal shock wave therapy for the treatment of musculoskeletal pain: a narrative review. Healthcare 2023, 11(21), 2830. [Google Scholar] [CrossRef]
- Novak, P. Physics: F-SW and R-SW. Basic information on focused and radial shock wave physics. In Multidisciplinary Medical Applications; Lohrer, H., Gerdesmeyer, L., Eds.; Level 10 Buchverlag Daniela Bamberg, 2015; pp. 28–49. [Google Scholar]
- Ogden, J. A.; Tóth-Kischkat, A.; Schultheiss, R. Principles of shock wave therapy. Clin. Orthop. Relat. Res. 2001. [Google Scholar] [CrossRef]
- Al-Abbad, H.; et al. The effects of shockwave therapy on musculoskeletal conditions based on changes in imaging: A systematic review and meta-analysis with meta-regression. BMC Musculoskelet. Disord. 2020, 21, 1–26. [Google Scholar] [CrossRef]
- Şah, V.; Elasan, S.; Kaplan, Ş. Comparative effects of radial and focused extracorporeal shock wave therapies in coccydynia. Turk. J. Phys. Med. Rehabil. 2023, 69(1), 97–104. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Şenlikci, H. B.; Yücealp, Ö.; Borman, P.; Ince Keskin, Z.; Gümüş Atalay, S.; Yaşar, E. Comparison of the efficacy of radial and focused extracorporeal shock-wave therapy (ESWT) in myofascial pain syndrome: a randomized, sham-controlled study. Lasers Med. Sci. 2025, 40(1), 404. [Google Scholar] [CrossRef] [PubMed]
- https://shockwavetherapy.org/wp-content/uploads/2024/01/ISMST-Guidelines-for-ESWT-_-engl-20240103.pdf.
- d’Agostino, M. C.; et al. Shock wave as biological therapeutic tool: From mechanical stimulation to recovery and healing. Perspect. Med. 2015, 4(1), 183–197. [Google Scholar]
- Chung, B.; Wiley, J. P. Effectiveness of extracorporeal shock wave therapy in the treatment of previ-ously untreated lateral epicondylitis. Clin. Sport Med. 2002, 12(5), 266–270. [Google Scholar]
- Lin, T.-Y.; Chen, J.-T.; Chen, Y.-Y.; Chen, T.-W.; Lee, C.-L.; Chen, C.-H.; Huang, M.-H. The efficacy of ultrasound-guided extracorporeal shockwave therapy in patients with cervical spondylosis and nuchal ligament calci-fication. Kaohsiung J. Med. Sci. 2015, 31, 337–343. [Google Scholar] [CrossRef]
- Fiani, B.; Davati, C.; Griepp, D.W.; Lee, J.; Pennington, E.; Moawad, C.M. Enhanced Spinal Therapy: Extracorporeal Shock Wave Therapy for the Spine. Cureus 2020, 12(10), e11200. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Li, S.; Liu, J.; Wang, Y.; Zhu, C.; Tang, Y.; Gu, M. Clinical evaluation of the efficacy of focused extracorporeal shock-wave therapy in patients with cervical spondylosis: A randomized control trial. Medicine 2024, 103(46), e40573. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




