Submitted:
24 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
- Presence of ≥16 natural teeth;
- Interdental clinical attachment loss (CAL) of ≥ 2 non-adjacent teeth.
- Buccal or oral CAL ≥ 3 mm with pocketing ≥ 3 mm, detectable at ≥ 2 teeth.
2.1. Laboratory Analyses
2.2. Assessment of Traditional Cardiovascular Risk Factors
2.3. Periodontal Charting
2.4. Statistical Analysis
3. Results
| PPD | CAL | BOP | |
| MMP-8 | −0.08 (0.825) | −0.14 (0.696) | −0.29 (0.413) |
| MPO | −0.15 (0.650) | 0.39 (0.205) | −0.27 (0.402) |
| Lp-PLA2 | 0.06 (0.841) | 0.13 (0.676) | −0.20 (0.504) |
| sCD40L | −0.77 (0.015) | 0.09 (0.828) | −0.61 (0.081) |
4. Discussion
4.1. Carotid Artery Anatomy and Plaque Burden (CpB)
4.2. Imaging Approaches to Carotid Plaque Morphology in Relation to Inflammatory Biomarkers
4.3. Inflammatory Physiopathological Links Between Periodontal Disease and CpB
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CpB | Carotid Plaque Burden |
| CVD | Cardiovascular disease |
| IL-1β | Interleukin-1 beta |
| IL-6 | Interleukin-6 |
| TNF-α | Tumor Necrosis Factor α |
| CRP | C-reactive Protein |
| PPD | Periodontal Pocket Depth |
| CAL | Clinical Attachment Loss |
| BOP | Bleeding on Probing |
| PD | Probing Depth |
| ELISA | Enzyme-Linked Immunosorbent Assay |
| MMP-8 | Matrix Metalloproteinase-8 |
| MMP-9 | Matrix Metalloproteinase-9 |
| MPO | Myeloperoxidase |
| Lp-PLA2 | Lipoprotein-Associated Phospholipase A2 |
| sCD40L | Soluble CD40 Ligand |
| ECM | Extracellular Matrix |
References
- Brinjikji, W.; Huston, J., 3rd; Rabinstein, A.A.; Kim, G.M.; Lerman, A.; Lanzino, G. Contemporary carotid imaging: from degree of stenosis to plaque vulnerability. J. Neurosurg. 2016, 124, 27–42. [Google Scholar] [CrossRef]
- Fuster, V.; García-Álvarez, A.; Devesa, A.; et al. Influence of Subclinical Atherosclerosis Burden and Progression on Mortality. JACC 2024, 84(15), 1391–1403. [Google Scholar] [CrossRef]
- Bentzon, J.F.; Otsuka, F.; Virmani, R.; Falk, E. Mechanisms of plaque formation and rupture. Circ. Res. 2014, 114, 1852–1866. [Google Scholar] [CrossRef] [PubMed]
- De Meyer, G.R.Y.; Zurek, M.; Puylaert, P.; Martinet, W. Programmed death of macrophages in atherosclerosis: mechanisms and therapeutic targets. Nat. Rev. Cardiol. 2024, 21, 312–325. [Google Scholar] [CrossRef] [PubMed]
- Scalise, E.; Costa, D.; Bolboacă, S.D.; Ielapi, N.; Bevacqua, E.; Cristodoro, L.; et al. The role of inflammation biomarkers in carotid artery stenosis procedures. Ann. Vasc. Surg. 2025, 110, 34–46. [Google Scholar] [CrossRef] [PubMed]
- Tonetti, M.S.; Greenwell, H.; Kornman, K.S. Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. J. Periodontol. 2018, 89, S159–S172. [Google Scholar] [CrossRef] [PubMed]
- Ismail, A.; Ravipati, S.; Gonzalez-Hernandez, D.; Mahmood, H.; Imran, A.; Munoz, E.J.; et al. Carotid artery stenosis: diagnostic and management strategies and related complications. Cureus 2023, 15, e38794. [Google Scholar] [CrossRef] [PubMed]
- Widén, C.; Holmer, H.; Sättlin, S.; Renvert, S.; Lernmark, Å.; Persson, G.R. Periodontal conditions, retinopathy, and serum markers in individuals with type 1 diabetes. J. Periodontol. 2020, 91, 1436–1443. [Google Scholar] [CrossRef] [PubMed]
- Polizzi, A.; Santonocito, S.; Vaccaro, M.; Fichera, G.; Torrisi, S.; Ronsivalle, V.; et al. Relationship between periodontitis and psychosocial impact in patients with systemic sclerosis: a clinical study. Mediterr. J. Clin. Psychol. 2020, 8, 2. [Google Scholar]
- Bilgin Çetin, M.; Önder, C.; Orhan, K.; Kumbasar, D.; Serdar, M.A.; Ünsal, E. Relationship of periodontitis and edentulism to angiographically diagnosed coronary artery disease: a cross-sectional study. J. Periodontal Res. 2020, 55, 895–904. [Google Scholar] [CrossRef]
- Schmidt, J.; Strecker, P.; Kreuz, M.; Löffler, M.; Kiess, W.; Hirsch, C.; et al. Stress-related hormones in association with periodontal condition in adolescents—results of the epidemiologic LIFE Child study. Clin. Oral. Investig. 2018, 23, 1793–1802. [Google Scholar] [CrossRef]
- Holmlund, A.; Holm, G.; Lind, L. Number of Teeth as a Predictor of Cardiovascular Mortality in a Cohort of 7,674 Subjects Followed for 12 Years. J. Periodontol. 2010, 81, 870–876. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Lv, Z.; Shi, Z.; Zhu, Y.; Wu, Y.; Li, L.; et al. Periodontal therapy for the management of cardiovascular disease in patients with chronic periodontitis. Cochrane Database Syst. Rev. 2014, 8, CD009197. [Google Scholar] [CrossRef]
- Senini, V.; Amara, U.; Paul, M.; Kim, H. Porphyromonas gingivalis lipopolysaccharide activates platelet Cdc42 and promotes platelet spreading and thrombosis. J. Periodontol. 2019, 90, 1336–1345. [Google Scholar] [CrossRef]
- Sanz, M.; Del Castillo, A.M.; Jepsen, S.; Gonzalez-Juanatey, J.R.; D’Aiuto, F.; Bouchard, P.; et al. Periodontitis and cardiovascular diseases: consensus report. Glob. Heart 2020, 15, 1. [Google Scholar] [CrossRef]
- Schenkein, H.A.; Loos, B.G. Inflammatory mechanisms linking periodontal diseases to cardiovascular diseases. J. Clin. Periodontol. 2013, 40, S51–S69. [Google Scholar] [CrossRef]
- Noack, B.; Kipping, T.; Tervahartiala, T.; Sorsa, T.; Hoffmann, T.; Lorenz, K. Association between serum and oral matrix metalloproteinase-8 levels and periodontal health status. J. Periodontal Res. 2017, 52, 824–831. [Google Scholar] [CrossRef]
- Lahdentausta, L.; Paju, S.; Mäntylä, P.; Buhlin, K.; Pietiäinen, M.; Tervahartiala, T.; et al. Smoking confounds the periodontal diagnostics using saliva biomarkers. J. Periodontol. 2019, 90, 475–483. [Google Scholar] [CrossRef]
- Bastos, M.F.; Tucci, M.A.; Siqueira, A.; Faveri, M.; Figueiredo, L.C.; Vallim, P.C.; et al. Diabetes may affect the expression of matrix metalloproteinases and their inhibitors more than smoking in chronic periodontitis. J. Periodontal Res. 2016, 52, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Widén, C.; Holmer, H.; Coleman, M.; Tudor, M.; Ohlsson, O.; Sättlin, S.; et al. Systemic inflamma- tory impact of periodontitis on acute coronary syndrome. J. Clin. Periodontol. 2016, 43, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Yabluchanskiy, A.; Ma, Y.; Iyer, R.P.; Hall, M.E.; Lindsey, M.L. Matrix Metalloproteinase-9: Many Shades of Function in Cardiovascular Disease. Physiology 2013, 28, 391–403. [Google Scholar] [CrossRef]
- Rupprecht, S.; Finn, S.; Hoyer, D.; Guenther, A.; Witte, O.W.; Schultze, T.; et al. Association between systemic inflammation, carotid arteriosclerosis, and autonomic dysfunction. Transl. Stroke Res. 2020, 11, 50–59. [Google Scholar] [CrossRef]
- Nagase, H. Activation mechanisms of matrix metalloproteinases. Biol. Chem. 1997, 378, 151–160. [Google Scholar]
- Boelen, G.-J.; Boute, L.; D’Hoop, J.; Ezeldeen, M.; Lambrichts, I.; Opdenakker, G. Matrix metalloproteinases and inhibitors in dentistry. Clin. Oral. Investig. 2019, 23, 2823–2835. [Google Scholar] [CrossRef]
- Kim, H.D.; Kim, S.; Jeon, S.; Kim, S.J.; Cho, H.J.; Choi, Y.N. Diagnostic and Prognostic ability of salivary MMP-9 and S100A8 for periodontitis. J. Clin. Periodontol. 2020, 47, 1191–1200. [Google Scholar] [CrossRef]
- Chang, Y.-C.; Chu, S.-C.; Yang, S.-F.; Hsieh, Y.-S.; Yang, L.-C.; Huang, F.-M. Examination of the signal transduction pathways leading to activation of gelatinolytic activity by interleukin-1alpha and Porphyromonas gingivalis in human osteosarcoma cells. J. Periodontal Res. 2004, 39, 168–174. [Google Scholar] [CrossRef]
- Ionel, A.; Lucaciu, O.; Tabaran, F.; Berce, C.; Toader, S.; Hurubeanu, L.; et al. Histological and clinical expression of periodontal disease related to the systemic inflammatory response. Histol. Histopathol. 2017, 32, 379–384. [Google Scholar]
- Schenkein, H.A.; Loos, B.G. Inflammatory mechanisms linking peri- odontal diseases to cardiovascular diseases. J. Clin. Periodontol. 2013, 40, S51–S69. [Google Scholar] [CrossRef]
- Holtfreter, B.; Empen, K.; Gläser, S.; Lorbeer, R.; Völzke, H.; Ewert, R.; et al. Periodontitis is associated with endothelial dysfunction in a general population. PLoS ONE 2013, 8, e84603. [Google Scholar] [CrossRef]
- Gurav, A.N. The implication of periodontitis in vascular endothelial dysfunction. Eur. J. Clin. Investig. 2014, 44, 1000–1009. [Google Scholar] [CrossRef]
- Herman, M.P.; Sukhova, G.K.; Libby, P.; Gerdes, N.; Tang, N.; Horton, D.B.; et al. Expression of neutrophil collagenase (matrix metalloproteinase-8) in human atheroma. Circulation 2001, 104, 1899–1904. [Google Scholar] [CrossRef]
- Rahman, M.; Zhang, S.; Chew, M.; Syk, I.; Jeppsson, B.; Thorlacius, H. Platelet shedding of CD40L is regulated by matrix metalloproteinase-9 in abdominal sepsis. J. Thromb. Haemost. 2013, 11, 1385–1398. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Qian, Y.; Wan, L.; Zhang, K.; Song, L.; Zhang, X.; et al. Lipoprotein-associated phospholipase A2 (Lp-PLA2): a key hub linking lipid metabolism and immune inflammation. Front Immunol. 2026, 17, 1705738. [Google Scholar] [CrossRef]
- Lin, W.; Chen, H.; Chen, X.; Guo, C. The Roles of Neutrophil-Derived Myeloperoxidase (MPO) in Diseases: The New Progress. Antioxidants 2024, 13, 132. [Google Scholar] [CrossRef]
- Novo, S.; Basili, S.; Tantillo, R.; Falco, A.; Davì, V.; Novo, G.; et al. Soluble CD40L and cardiovascular risk in asymptomatic low-grade carotid stenosis. Stroke 2005, 36, 673–675. [Google Scholar] [CrossRef]
- Zhang, M.; Cai, J.; Cao, Q.; Chen, Z.; Maimaitiaili, S.; Yuan, S.; et al. Radiomic features and carotid stenosis in periodontitis: a multimodal machine learning study. Sci. Rep. 2026, 16, 8177. [Google Scholar] [CrossRef]
- Takasaki, M.; Saito, K.; Fukushima, K.; Yamada, N.; Ishibashi-Ueda, H.; Iihara, K. Plaque imaging of carotid stenosis: an update and future perspective. Brain Nerve (In Japanese) 2011, 63, 963–968. [Google Scholar] [PubMed]
- Wijeratne, T.; Menon, R.; Sales, C.; Karimi, L.; Crewther, S. Carotid artery stenosis and inflammatory biomarkers: the role of inflammation-induced immunological responses affecting the vascular systems. Ann. Transl. Med. 2020, 8, 1276. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ding, S.; Zhang, M.; Zhao, Y.; Chen, W.; Yao, G.; Zhang, C.; et al. The Role of Carotid Plaque Vulnerability and Inflammation in the Pathogenesis of Acute Ischemic Stroke. Am. J. Med. Sci. 2008, 336, 27–31. [Google Scholar] [CrossRef]
- Bueno, A.; March, J.R.; Garcia, P.; Cañibano, C.; Ferruelo, A.; Fernandez-Casado, J.L. Carotid Plaque Inflammation Assessed by 18F-FDG PET/CT and Lp-PLA2 Is Higher in Symptomatic Patients. Angiology 2021, 72, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Duivenvoorden, R.; Mani, V.; Woodward, M.; et al. Relationship of Serum Inflammatory Biomarkers With Plaque Inflammation Assessed by FDG PET/CT: The dal-PLAQUE Study. J. Am. Coll. Cardiol. Img 2013, 6, 1087–1094. [Google Scholar] [CrossRef]
- Wang, L.; Chen, T.; Yuan, C.; Hatsukami, T.; Zhao, X.; Shen, M.; et al. Carotid Plaque Characteristics Combined With Serum Inflammatory Biomarkers Predict Recurrent Ischemic Stroke. Brain Behav. 2025, 15, e70909. [Google Scholar] [CrossRef]
- Bonati, L.H.; Jansen, O.; de Borst, G.J.; Brown, M.M. Management of atherosclerotic extracranial carotid artery stenosis. Lancet Neurol. 2022, 21, 273–283. [Google Scholar] [CrossRef] [PubMed]
- Beck, J.D.; Elter, J.R.; Heiss, G.; Couper, D.; Mauriello, S.M.; Offenbacher, S. Relationship of periodontal disease to carotid artery intima-media wall thickness: the atherosclerosis risk in communities (ARIC) study. Arterioscler. Thromb. Vasc. Biol. 2001, 21, 1816–1822. [Google Scholar] [CrossRef] [PubMed]
- Howell, T.H.; Ridker, P.M.; Ajani, U.A.; Hennekens, C.H.; Christen, W.G. Periodontal disease and risk of subsequent cardiovascular disease in U.S. male physicians. J. Am. Coll. Cardiol. 2001, 37, 445–450. [Google Scholar] [CrossRef]
- Hujoel, P.P.; Drangsholt, M.; Spiekerman, C.; Derouen, T.A. Examining the link between coronary heart disease and the elimination of chronic dental infections. J. Am. Dent. Assoc. 2001, 132, 883–889. [Google Scholar] [CrossRef]
- Desvarieux, M.; Demmer, R.T.; Rundek, T.; Boden-Albala, B.; Jacobs, D.R., Jr.; Papapanou, P.N.; et al. Periodontal disease and carotid plaque. Stroke 2003, 34, 2120–2125. [Google Scholar] [CrossRef]
- Schillinger, T.; Kluger, W.; Exner, M.; Mlekusch, W.; Sabeti, S.; Amighi, J.; et al. Dental status and carotid atherosclerosis progression. Stroke 2006, 37, 2271–2276. [Google Scholar] [CrossRef]
- Debing, E.; Peeters, E.; Demanet, C.; De Waele, M.; Van den Brande, P. Markers of inflammation in patients with symptomatic and asymptomatic carotid artery stenosis: a case-control study. Vasc. Endovasc. Surg. 2008, 42, 122–127. [Google Scholar] [CrossRef]
- Mendoza, M.F.; Anzelmo, M.A.; Suan, N.M.; Cuccia, C.S.; Lavie, C.J. Inflammatory mechanisms linking periodontal and cardiovascular disease. Biomedicines 2025, 13, 1512. [Google Scholar] [CrossRef]
- Xu, B.; Jiang, X.; Li, H.; Wei, K.; Ma, H.; Jiang, S.; et al. Impact of biomarkers on carotid artery disease and stroke. Sci. Rep. 2025, 15, 27821. [Google Scholar] [CrossRef]
- Djurić, T.; Stanković, A.; Končar, I.; Radak, D.; Davidović, L.; Alavantić, D.; et al. MMP-8 polymorphisms and carotid atherosclerosis. Atherosclerosis 2011, 219, 673–678. [Google Scholar] [CrossRef] [PubMed]
| No. | Age | Sex | Total Cholesterol (mg/dL) | HDL | LDL | Triglycerides | CRP | ESR | Weight (kg) | Height (cm) | BMI (kg/m²) | Diabetes mellitus | Smoking |
| 1 | 52 | F | 152 | 43.3 | 93 | 138 | 12.4 | 7 | 82 | 161 | 31.63 | no | no |
| 2 | 71 | M | 228 | 40.2 | 160 | 210 | 11.3 | 6 | 90 | 180 | 27.8 | no | no |
| 3 | 76 | M | 120 | 38.9 | 182 | 270 | 16.5 | 10 | 95 | 182 | 28.7 | no | no |
| 4 | 67 | M | 215 | 35.7 | 162 | 267 | 9.7 | 15 | 89 | 174 | 29.4 | no | no |
| 5 | 67 | M | 233 | 60.1 | 176 | 211 | 6.9 | 14 | 97 | 175 | 31.7 | no | no |
| 6 | 57 | M | 260 | 58.2 | 188 | 167 | 12.5 | 14 | 93 | 169 | 32.6 | yes | no |
| 7 | 33 | M | 254 | 62.1 | 132 | 180 | 13.5 | 18 | 125 | 180 | 38.6 | no | no |
| 8 | 76 | F | 182 | 55.2 | 148.8 | 189 | 6.2 | 14 | 70 | 167 | 25.1 | no | no |
| 9 | 59 | M | 284 | 36.6 | 87.9 | 220 | 7.6 | 16 | 88 | 180 | 26.5 | no | no |
| 10 | 73 | M | 320 | 38.6 | 166.2 | 272 | 24.6 | 47 | 110 | 175 | 35.9 | no | yes |
| 11 | 44 | M | 241 | 66.2 | 102 | 150 | 38.7 | 45 | 84 | 174 | 27.7 | yes | no |
| 12 | 74 | M | 221 | 52.6 | 115 | 151 | 18.2 | 17 | 93 | 169 | 32.6 | no | no |
| 13 | 52 | M | 205 | 66.2 | 98 | 156 | 13.8 | 17 | 82 | 176 | 26.5 | yes | yes |
| Variable | Mean | SD | Minimum | Maximum |
| PPD (mm) | 5.54 | 2.03 | 3 | 8 |
| CAL (mm) | 6.46 | 3.67 | 2 | 12 |
| BOP (%) | 66.92 | 21.69 | 38 | 98 |
| Variable | Value | Range |
| MMP-8 | 25.64 ± 20.31 | 3.00–78.00 |
| MPO | 46.00 (median) | 6.00–197.00 |
| Lp-PLA2 | 46.60 (median) | 62.00–402.00 |
| sCD40L | 3.91 (median) | 1.20–6.80 |
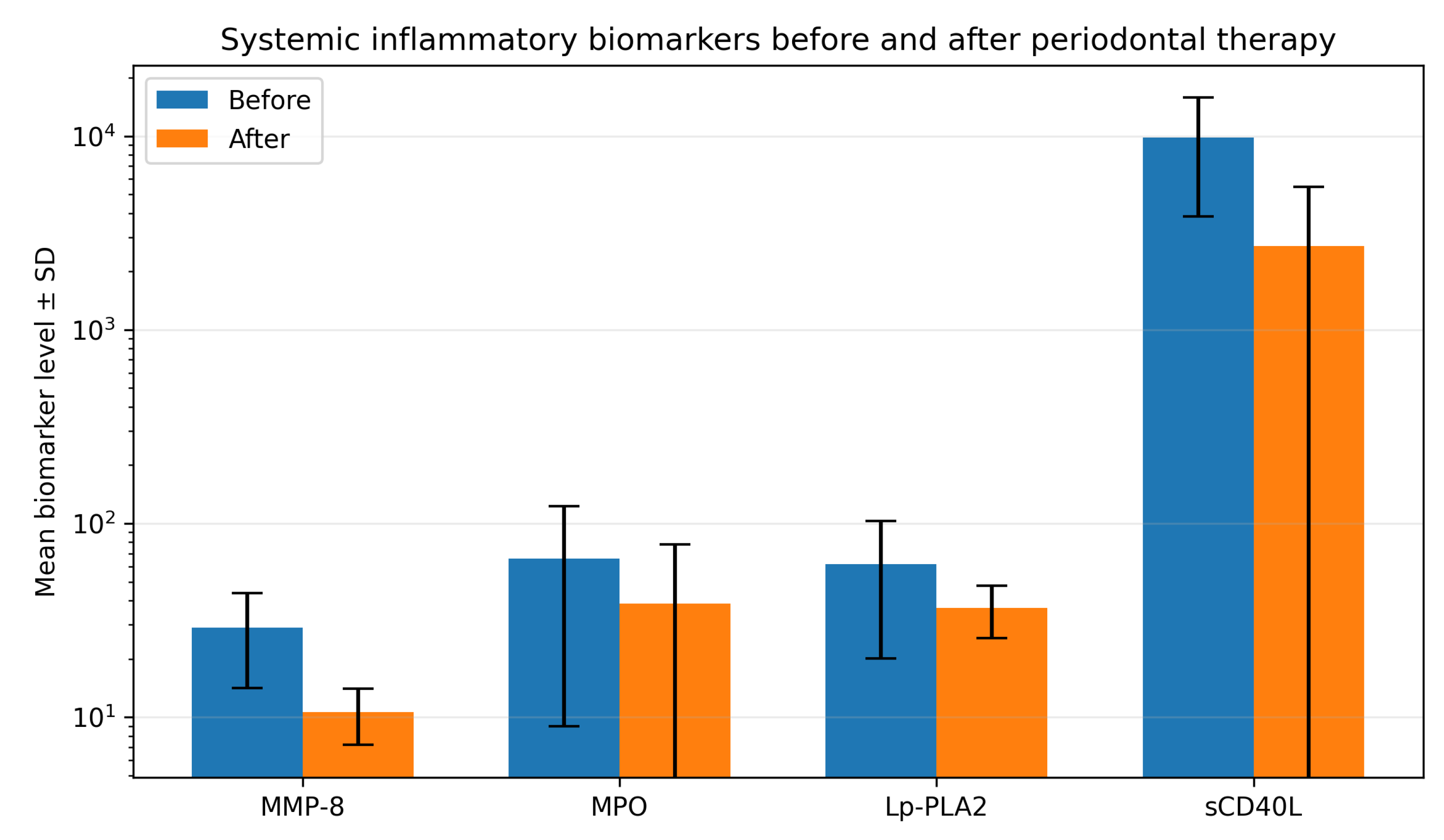
| Biomarker | Baseline | Post-treatment | p-value |
| MMP-8 | 25.64 ± 20.31 | 10.60 ± (SD) | 0.010 |
| MPO | 45.50 (median) | 23.50 (median) | 0.097 |
| Lp-PLA2 | 46.80 (median) | 35.30 (median) | 0.020 |
| sCD40L | 9538 (median) | 1642 (median) | 0.156 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




